chem paper 1 flashcards (exlcuding triple)
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
What is an element? What is a molecule?
A substance made up of one type of atom. A molecule is atoms bonded together.
What is a compound?
A compound is two or more different atoms chemically bonded together.
How are compounds separated?
By chemical reaction.
What is a mixture?
A mixture is different atoms not chemically bonded.
How are mixtures separated?
Physical separation techniques (e.g. filtration) - these involve no chemical reactions and no new substances are made
Describe filtration.
Filtration is used to separate an insoluble solid from a liquid.
Place filter paper into a funnel. Pour the mixture through this to a conical flask. The filtrate (liquid) will pass through.
Describe crystallisation.
Crystallisation is used to separate a soluble solid from a liquid.
Gently heat your solution to evaporate the liquid. This can be done in an evaporating basin over a bunsen burner - however, heat slowly and carefully to leave crystals.
Describe simple distillation.
Simple distillation is used to separate a soluble solid from a liquid, whilst keeping the liquid.
Place the solution into a flask with a thermometer, which is connected to a continuous glass tube. This has a condenser on it which keeps the glass tube cold.
First, heat the solution to evaporate the liquid, turning it into a vapour. The vapour will pass into the cold condenser and condense back into a liquid, collecting in a beaker. Crystals of solid should be left in the flask.
Describe paper chromatography.
Chromatography is used to separate substances based on different solubilities.
Draw a pencil (pencil as it’s insoluble). line near the bottom of chromatography paper. Put dots of ink along this line. Place the bottom of the paper into a solvent, BELOW the line. A more soluble substance travels further.
Give a brief timeline of the model of an atom.
Dalton - tiny indivisible spheres
Scientists discovered electrons
JJ Thompson - Plum Pudding model - atoms are balls of positive charge with electrons embedded in it
Rutherford - alpha scattering experiment
Rutherford - Nuclear model - mass of an atom concentrated at a positive nucleus centre, with electrons around the edge of the atom
Protons discovered
Bohr - electron shell model - electrons orbit the nucleus on electron shells
20 years later, Chadwick discovered neutrons, also contained in nucleus
Describe the alpha scattering experiment
Scientists fired tiny alpha particles (+) at gold foil. Most passed straight through, but sometimes the particle was deflected, and sometimes it bounced off the foil entirely.
What did the alpha scattering experiment show?
Most went straight through: atoms mainly empty space
Some were deflected: centre of atom must be positive
Some bounced straight back: centre of atom must contain most of the mass
How big is an atom and its nucleus?
Radius is 0.1nm (1 × 10-10m)
Radius of nucleus is less than 1/10000 of the atom (1 × 10-14m)
What are the relative masses and charges of subatomic particles? What is the charge of an atom?
An atom has no overall charge as the number of electrons is equal to the number of protons.

What is the atomic number and what is the atomic mass of an element?
Atomic number = number of protons (and therefore electrons)
Same if same element
Atomic mass = number of protons + neutrons in total
Atomic mass - atomic number = number of neutrons
Define isotope.
Isotopes are atoms of the same element with different numbers of neutrons.
Define ion
Atoms with a charge - due to losing / gaining electrons
How are atoms drawn / electronic configuration?
2, 8, 8 rule - 2 electrons on the first energy level, then 8 on the other energy levels
See symbol of an element for number of electrons.
What do groups and periods show in the periodic table?
Groups - number of electrons in outer shell, have similar properties
Periods - number of electron shells
Give a brief timeline of the periodic table.
Mendeleev - arranged elements in order of atomic weight, but sometimes switched order to fit patterns (as isotopes could mess this up). He also left gaps for undiscovered elements.
Modern periodic table - elements arranged in order of atomic number, meaning every element is in the correct group
What is the equation for relative atomic mass?
(mass x abundance) + (mass x abundance) /100
Mass is the number, for example, 35 in chlorine-35. The abundance will be a percentage.
Where are the metals found on the periodic table? Which ones are highly reactive?
Metals are found on the left and centre. The metals in groups 1 and 2 are highly reactive.
What happens when metals react?
They lose their outer electrons to achieve a full outer electron shell. They become positive ions.
What are the key characteristics of group 0? (Noble gases)
Unreactive / do not easily form molecules because they all have a full outer electron shell
As you go down, boiling point increases
What are the key characteristics of group 1? (Alkali metals)
All have one electron on their outer shell
As you go down, reactivity increases
React rapidly with oxygen and / or chlorine
How do alkali metals react with water? What is produced?
A metal hydroxide and hydrogen gas is produced
Effervescence is seen (fizzing / production of a gas)
For example: sodium + water —> sodium hydroxide + hydrogen
What is electrostatic attraction and how does it affect reactivity?
The more electron shells there are, the more reactive. This is because there is weaker electrostatic attraction between the positive nucleus and negative outermost electron. This means that an outer electron could get lost more easily, causing a chemical reaction.
In halogens, more electron shells, less reactive as harder to gain an electron
What are the key characteristics of group 7? (Halogens)
All have 7 electrons in their outer shell
Every group 7 element forms a molecule consisting of two atoms joined by a covalent bond
As you go down, melting point and boiling point increases. Reactivity deceases.
As you go down, relative molecular mass increases (molecule is bigger)
What is formed when group 7 elements react with other non-metal elements?
Non-metals - covalent compounds. They join so they both have full outer shells.
Metals - ionic compounds. The halogen atom gains one electron and forms an ion with a -1 charge
What are the state symbols?
Solid - (s)
Liquid - (l)
Gas - (g)
Aqueous solutions - (aq)
What are the key traits of ionic bonding?
Metal and non-metal
Produces ions with full outer shells
Electrons transferred
Strong electrostatic attraction between + and - ion = high melting point
What does a dot and cross diagram look like for ionic bonding?
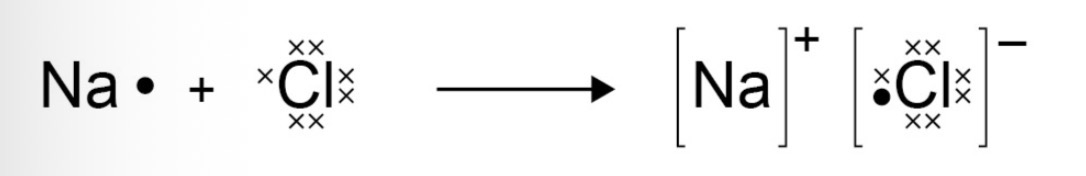
What is a giant ionic lattice and its properties?
A structure of many ions, where oppositely charged ions are held together by strong electrostatic forces of attraction (ionic bonds).
High melting and boiling points - the strong electrostatic forces require lots of heat energy to break.
Only conducts electricity in liquid state - in liquids, ions are free to flow and move meaning they can carry the electrical charge
What are the key traits of covalent bonding?
Two non-metals
Produces neutral molecules
Electrons shared
Weak intermolecular forces = low melting point
Stick diagrams show covalent bonds
What does a dot and cross diagram look like for covalent bonding?

What are properties of small covalent molecules (simple covalent structures)?
Few atoms
Low melting + boiling points due to weak intermolecular forces (between molecules) that are easily broken in melting / boiling
Liquid / gas at room temperature
Cannot conduct electricity because there is no overall charge
What are the key traits of metallic bonding?
Two metals
Metals consist of giant positive ions arranged in regular structures, with a sea of delocalised electrons (from the outer shell of each ion)
Strong electrostatic attraction between positive ions and negative electrons = strong metallic bonds
What are properties of metals?
High melting and boiling points due to strong metallic bonds requiring lots of energy
Conduct electricity well because delocalised electrons carry electrical charge
Soft so can be bent and shaped, as the layers can slide over each other easily
Why are alloys stronger than pure metals?
An alloy is a mixture of metals. The different sizes of atoms distorts the layers, so they cannot slide over eachother.
What are properties of giant covalent structures?
Many atoms
High melting + boiling points due to strong covalent bonds which require lots of energy to overcome
Solid at room temp
What are the properties of diamond (a giant covalent structure)?
Allotrope of carbon
Every carbon atom bonds to 4 others
Cannot conduct electricity because it has no delocalised electrons to carry charge
Very hard because of strong covalent bonds which and tetrahedral structure
Very high melting point because it has strong covalent bonds which require lots of energy to break
What are the properties of graphite (a giant covalent structure)?
Allotrope of carbon
Every carbon atom bonds to 3 others
Can conduct electricity because it has delocalised electrons to carry charge (similar to metals)
Soft / slippery because of weak intermolecular forces and hexagonal ring layers which slide over each other easily
Very high melting point because it has strong covalent bonds which require lots of energy to break
What is graphene and what are its uses?
One layer of graphite; only one atom thick
Conducts electricity because it has delocalised electrons
Useful in electronics + composite materials
Very strong with high melting + boiling points due to many strong covalent bonds
What are fullerenes and what are their uses?
Carbon molecules with hollow shapes
Based on hexagonal rings of carbon but may also contain rings with 5 or 7 carbon atoms
Buckminsterfullerene is a hollow sphere made of 60 carbon atoms. Useful as lubricants or catalysts.
Carbon nanotubes are cylindrical fullerenes with high length to diameter ratios. Useful to strengthen materials or in electronics.
What is the law of conservation of mass?
No atoms are lost or made during a chemical reaction so the mass of the products equals the mass of the reactants.
How do you balance an equation?

What is relative formula mass (Mr) of a compound and how can it be calculated?
It is the sum of the relative atomic masses of the atoms involved.

What is the formula for calculating percentage by mass?

What is 1 mole (Avogadro’s constant) and what is the unit for moles?
6.022 X 1023
1 mole of an element is its relative atomic mass in grams.
The unit for a mole is mol.
What is the equation that links moles, mass and relative formula mass?
Mass = Mr x Mole
Why do some reactions appear to involve a change in mass?
A reactant / product may be a gas.
How can you calculate unknown masses?

What is a limiting reactant?
The reactant that is completely used up and so limits the amount of products.
How can you calculate what is the limiting reactant?

How to convert from dm3 to cm3?
Multiply by 1000
What is the equation for concentration?
Concentration = mass / volume
How do metals react with oxygen? What do they form?
It is an oxidation reaction (gain oxygen) and they form oxides.
How are metals found in the earth?
Unreactive metals usually exist as pure metals so are just mined; most metals are found in compounds / ores and so require extraction.
Which metals can be reduced (oxygen lost) by carbon?
Zinc, iron, copper
Describe oxidation and reduction in terms of electrons.
OILRIG
Oxidation
Is
Loss
Reduction
Is
Gain
Write an ionic equation for potassium + sodium chloride —> potassium chloride + sodium
K + NaCl —> KCl + Na
K + Na+ + Cl- —> K+ + Cl- Na
Cl remains the same so is a spectator ion and removed
FINAL EQUATION:
K + Na+ —> K+ + Na
What do acids contain?
Hydrogen ions (H+)
What do alkalis and bases contain?
Hydroxide ions (OH-)
What is the difference between alkalis and bases?
Both can neutralise acid, however bases are insoluble, and alkalis are soluble in water.
What happens when metals and acids react?
MASH - Metal + Acid —> Salt (metal name + acid ending) + Hydrogen
These are REDOX reactions, meaning both reduction and oxidation happen (the acid is reduced, and metal is oxidised)
What salts do hydrochloric and sulfuric acids produce?
Hydrochloric —> -chloride
Sulfuric —> -sulfate
Nitric —> -nitrate
What happens when acids and bases react?
Acid + Base —> Salt + Water
Acid + Carbonate Base —> Salt + Water + Carbon Dioxide
This is a neutralisation reaction.
How can you find the formulae of an ionic compound?
Crossing over method.
Use periodic table to find charges (or if a transition metal, Roman numerals represent positive charge number), then cross lines to find bottom numbers.
Example: Magnesium chloride

Required Practical: soluble salts
Measure a fixed volume of sulfuric acid into a beaker.
Heat the acid gently until almost boiling, using a bunsen burner.
Use a spatula to add small amounts of copper oxide powder, stirring with a glass rod.
Stop adding it when some black powder remains after stirring. This ensures all the acid has been neutralised.
Filter into a conical flask and pour into an evaporating basin.
Evaporate gently in a water bath until half the solution remains, then leave for 24 hours in a cool place.
What is the difference between a strong and a weak acid with examples?
Strong acids fully dissociate when placed in solution (all hydrogen ions and negative ions split up). Examples: hydrochloric, nitric and sulfuric acids.
Weak acids partially dissociate when placed in solution (only some hydrogen ions and negative ions split up). Examples: ethanol, citric and carbonic acid.
Which has a lower pH: strong or weak acids?
Strong acids. As pH decreases by 1, hydrogen ions concentration increases by a factor of 10 (x10, x100, etc)
What is the difference between a concentrated and a dilute acid?
Concentrated acids have more acid molecules in a given volume.
Dilute acids have less acid molecules in a given volume.
Why can ions only conduct electricity when molten or dissolved in water?
The ions are free to move and carry charge.
In electrolysis, where do the ions go? What are the charges of the electrodes?
Cations (pawsitive) go to the cathode. Anions (negative) go to the anode.
PANIC:
Positive
Anode
Negative
Is
Cathode
Why is electrolysis used?
To extract metals that are more reactive than carbon.
How is aluminium oxide extracted?
With electrolysis.
It is first mixed with cryolite which lowers the melting point (so saves money). The electrodes are made of graphite which is a good conductor and has a high melting point.
Why does the anode need to be replaced regularly in the electrolysis of aluminium oxide?
Oxygen react with the graphite to form carbon dioxide gas.
Why is electrolysis expensive?
Melting the compounds requires lots of energy
Lots of energy is needed to produce the electric current
What is produced at the cathode in aqueous electrolysis?
If metal is more reactive than hydrogen —> hydrogen gas
If metal is less reactive than hydrogen (copper, silver, gold) —> metal
What is produced at the anode in aqueous electrolysis?
If the solution contains group 7 ions —> halogen gas
If the solution doesn’t contain group 7 ions —> oxygen gas + water
Required Practical: electrolysis of copper chloride
Pour 50ml of copper chloride solution into a beaker.
Insert two graphite electrodes into the beaker. They should not touch.
Attach crocodile leads to the electrodes and connect to a power pack via wires.
Turn on powerpack to 6V and leave for 5 minutes.
How is a half equation for a cathode written?
Reduction half equation.

How is the half equation for an anode written?
Oxidation half equation.

Give the formulae for the following ionic compounds: ammonium, hydroxide, sulphate, carbonate, and nitrate.

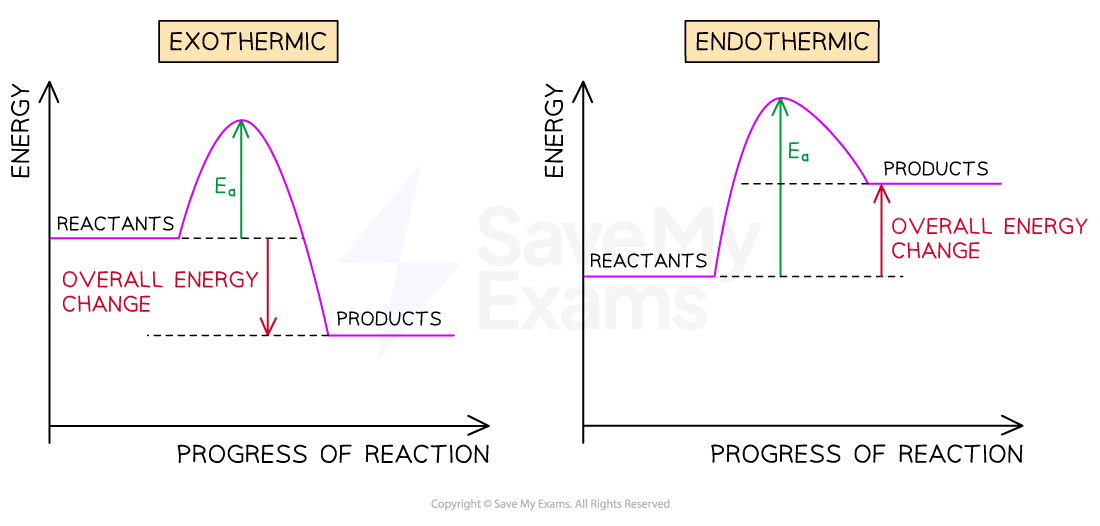
What is an exothermic reaction? Give examples.
They release energy to the surroundings, so the temperature of the surroundings increases.
EXAMPLES: combustion, many oxidation reactions, neutralisation, self-heating cans, hand warmers
What is an endothermic reaction? Give examples.
They take in energy from the surroundings, so the temperature of the surroundings decreases.
EXAMPLES: thermal decompositions, citric acid + sodium hydrogencarbonate reaction, some sport injury packs
When it comes to endo and exothermic reactions, what does the thermometer measure?
Temperature of the surroundings
What do reaction profiles for endo and exothermic reactions look like?
Ea meaning activation energy.
Be able to do bond energy calculations.