BIOS 301 - Fermentation & Pentose Phosphate Pathway
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
2-deoxyglucose
Glycolysis occurs at elevated rates in tumor cells
2-deoxyglucose can be administered to cancer patients - enters glycolysis pathway but cannot proceed past second step
Limits energy production in cancer cells
But not a good drug - has to be given in toxically high doses to compete with glucose in glycolysis pathway
Pet scanning
Administer 18F-fluorodeoxyglucose to patient
Can be converted to FDG-6P but not metabolized further
Builds up quickly cells in with high metabolic activity (e.g. tumor cells)
FDG-6P can be detected in PET scans to show location of cancer metastases
Fates of pyruvate (in animals)
Heart muscle and brain: pyruvate is always subject to the TCA cycle and complete oxidation to ensure a constant supply of ATP reaches vital tissues
Skeletal muscle: when this tissue goes anaerobic, lactic acid fermentation occurs
Liver: can conduct lactic acid fermentation in reverse (gluconeogenesis) to ensure energy derived from glucose can be maximized
Purpose of fermentation
Generates ATP under anaerobic conditions
Recycles NADH → NAD+ for further glycolysis (i.e. energy production)
Lactic acid fermentation
2 pyruvate + 2 NADH → 2 lactate + 2 NAD+
Catalyzed by lactate dehydrogenase
Occurs in an oxidation-reduction reaction
Reduction potential
High reduction potential corresponds to high affinity for electrons
Electrons are transferred from lower to higher E
Delta E = Eacceptor -Edonor
Delta E must be positive to be thermodynamically favorable (negative delta G)
To find delta E use half reactions of pyruvate (acceptor) and NADH (donor)
Isozymes of lactate dehydrogenase
Muscle LDH has higher affinity for pyruvate than heart LDH
Allows for shorts bursts of energy by anaerobic glycolysis in muscle
Forces aerobic, complete oxidation of glucose in the heart
Heart LDH is also subject to excess substrate inhibition
There are two genes for LDH monomers (H and M) which combine randomly to form tetramer enzyme molecules
Blood plasma tests can reveal identity of injured/necrotic tissue based on LDH subunit representation
Ethanol fermentation (step 1)
Pyruvate → Acetaldehyde + CO2
Enzyme: pyruvate decarboxylase
Cofactors: Mg2+, TPP
Ethanol fermentation (step 2)
Acetaldehyde + NADH → Ethanol + NAD+
Enzyme: alcohol dehydrogenase
TPP
Thiamine pyrophosphate
Contains thioazolium ring: allows for electron delocalization → generates carbanion that attacks substrate → facilitates C-C bond cleavage and decarboxylation
Pentose Phosphate Pathway (overall result)
Substrate is glucose-6-phosphate
Main products are NADPH and ribose-5-phosphate
NADPH purpose
Reductive biosynthesis of fatty acids and steroids
Repair of oxidative damage
Reduction of glutathione
Ribose-5-phosphate purpose
Used in synthesis of DNA/RNA or certain coenzymes
Pentose Phosphate Pathway (substrates, products, enzymes)
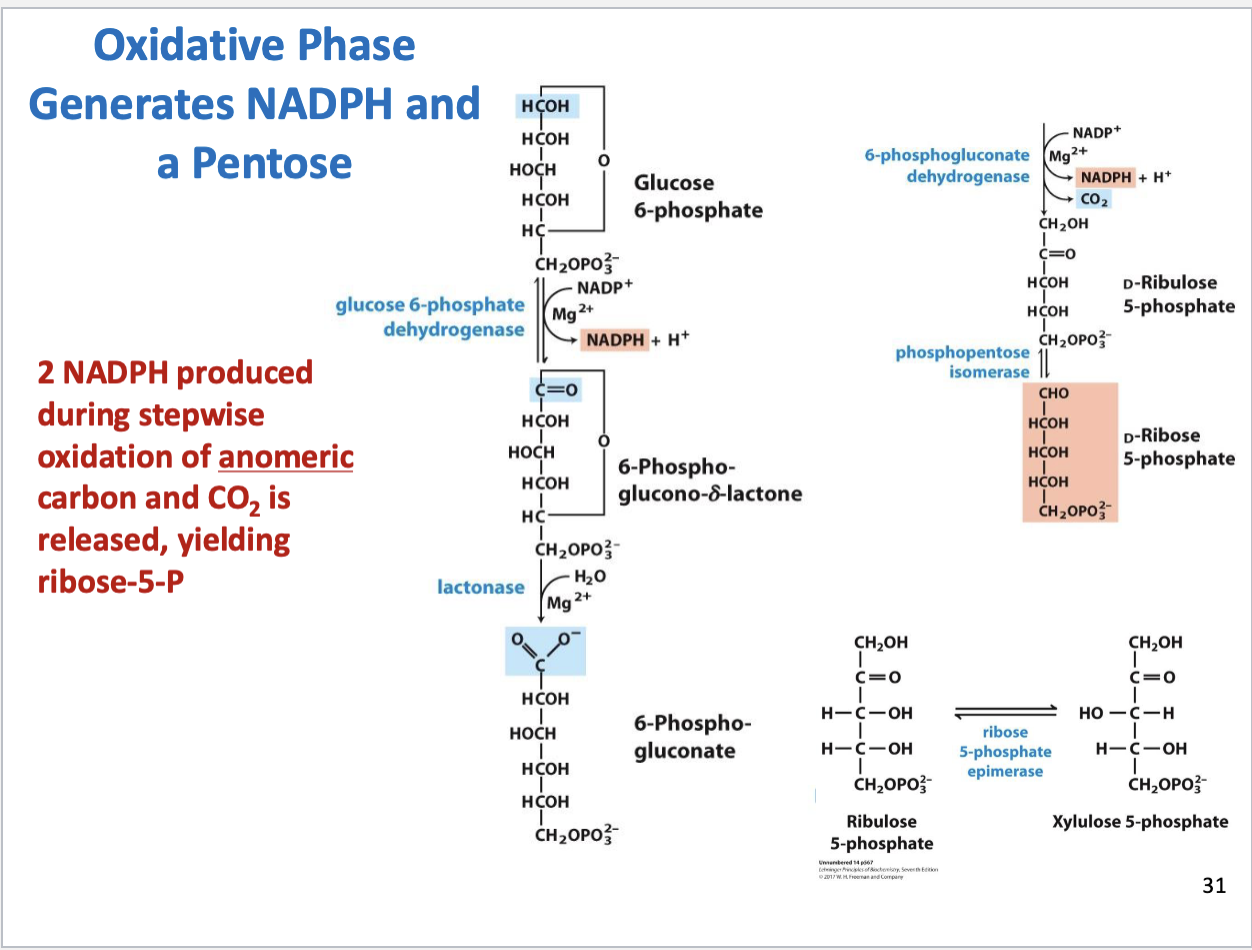
Oxidative vs. Nonoxidative phase of PPP
Oxidative phase yields NADPH and ribulose-5-phosphate
Nonoxidative phase: if ribulose-5-phosphate is already abundant, can be converted to xylulose-5-phosphate and back to G-6-P
Generates NADPH while conserving glucose
Transketolases
Assist in nonoxidative phase of PPP
Transfer 2C units from ketose donor → aldose acceptor
Reaction intermediate stabilized by TPP
Transaldolases
Assist in nonoxidative phase of PPP
Transfer 3C units from ketose donor → aldose acceptor
Reaction intermediate is a protonated Schiff’s base
Partitioning into glycolysis vs. PPP
If NADPH is sufficiently high, PPP is inhibited, and glycolysis proceeds instead
G-6-P Dehydrogenase Deficiency
G-6-P Dehydrogenase is responsible for recycling NADP+ into NADPH
NADPH is required by glutathione reductase to convert oxidized glutathione → reduced form
Glutathione is necessary to neutralize reactive oxygen species
PPP in tumor cells
G-6-P Dehydrogenase, 6-phosphogluconolactonase, and 6-phosphogluconate dehydrogenase are all upregulated in tumor cells