Physical Science Exam
1/124
Earn XP
Description and Tags
Good Luck!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
125 Terms
Matter in which particles are arranged in repeating geometric patterns, and has a definite volume and a definite shape
Solid
A gas-like mixture with no definite volume or shape that is made up of positively and negatively charged particles
Plasma
Matter that has definite volume but no shape
Liquid
Matter in which the particles are free to move in all directions until they have spread evenly throughout their container
Gas
Most matter ____ when heated
expands
Liquids which keep some ordered structure
Liquid crystals
Who was the SI unit of pressure named for
Blasie Pascal
The amount of energy needed to change a material from the solid state to the liquid state is the heat of
Fusion
The amount of energy needed to change a material from a liquid to a gas is the heat of
Vaporization
The pressure of a gas will increase as the volume of the container decreases provided temperature does not change
Boyle’s Law
The idea that the volume of a gas increases with increasing temperature provided the pressure does not change
Charles’s Law
The buoyant force on an object in a fluid is equal to the weight of the fluid displaced by the object
Archimedes’ Principle
Pressure applied to a fluid is transmitted unchanged throughout the fluid
Pascal’s Principle
Whether an object will sink or float in a fluid
Archimedes’ Principle
As a sample of matter is heated, its particles ___
move more quickly
The equation P1V1 = constant P2V2
Boyle’s Law
The idea stating matter is made up of small particles that are in constant motion
The kinetic theory of matter
The particles that make up a solid move ___ than do the particles that make up a gas
more slowly
In applying Charles’s law, temperature must be on the ___ scale
Kelvin
When water is frozen it ____
Expands
As the velocity of a fluid increases, the pressure exerted by the fluid decreases
Bernoulli’s Principle
If the buoyant force of a fluid is less than the weight of the object floating in it, the object will ___
Sink
A hydraulic lift is used to lift a heavy machine that is pushing on a 7.6 m2 piston with a force of 1450 N. What force needs to be exerted on a 0.0048 m2 piston to lift the machine
1 N
A balloon has a volume of of 10 L at a pressure of 110 kPa. What will be the new volume when the pressure drops to 50 kPa?
22.0 L
A computer is sitting on a desk. The computer contacts the desk over an area of 0.07 m2. If the computer weighs 25 N, what is the pressure the computer exerts on the desk?
357 Pa
The force which supports objects in fluids
Buoyant force
Solids that gradually soften and turn into a liquid over a temperature range
Amorphous solids
Random spreading of molecules
Diffusion
A fluid’s resistance to flow
Viscosity
The mass per unit volume
Density
Where is the one place you might find matter in the state of plasma?
Sun, stars, auroras
According to Archimedes’ principle, what will happen if the buoyant force of a fluid is more than or equal to the weight of an object placed in the fluid?
The object will float
Smoke is an example of a ____
Colloid
When two or more substances are combined so that each substance maintains its original properties
Mixture
The scattering of light by colloids
The Tyndall Effect
_____ is another name for homogeneous mixture
Solution
Three examples of physical change
Boiling water, bursting a balloon, and sharpening a pencil
______ is NOT a homogeneous
Suspension
Is the best indication that a chemical reaction has occurred
Producing a new substance
Distillation can separate dissolved substances through ______
Physical changes
Can be separated into simpler parts through differences in physical properties
Mixture
When two or more elements are combined so that each element is bonded to the other element forming a completely new substance with new properties
Compound
The law that describes the relationship between the mass of the substances entering into a chemical reaction and the mass of the substances resulting from that reaction
Law of Conservation of Mass
Matter is classified as ______ and mixtures
Substances
The substance calcium carbonate (CaCO3) is an example of a
Compound
Any characteristic of a material that you can observe without changing the identity of the substances that make up the material
Physical Property
Compounds are made from the atoms of two or more ______
Elements
A solution is a type of _______ mixture
Homogeneous
A mixture in which different materials are easily identified
Heterogeneous
Is a mixture that has large particles that do not settle out of the liquid
Colloid
Is a heterogeneous mixture in which visible particles settle
Suspension
Is a process that can separate two liquids using their boiling points
Distillation
Muddy water
Heterogeneous
Blood
Homogeneous
Distilled water
Homogeneous
Chocolate Chip ice cream
Heterogeneous
Saline Solution
Homogeneous
Size of an object
Physical Property
Sharpening a pencil
Physical Change
Flammability of a substance
Chemical Property
Boiling water
Physical change
Freezing point
Physical Property
Formation of water when hydrogen burns
Chemical change
Each energy level of an atom has a maximum number of ____ it can hold
Electrons
Dot diagrams are used to represent _____
Outer electrons
Particles of matter that make up protons and neutrons
Quarks
A chemical symbol represents the ____ of an element
Name
The horizontal rows of elements
Periods
Vertical columns of elements
Groups
The number of protons in an atom
Atomic number
The center of the atom where protons and neutrons are located
Nucleus
A total count of the neutrons and protons in an atom
Mass number
Atoms of the same element but with different numbers of neutrons
Isotopes
Elements that have some properties of both metals and nonmetals
Metalliod
Particles that move around the nucleus and have a negative charge
Electrons
A chart that shows the classification of elements
Periodic table
Is Neon a metal, nonmetal, or metalliod?
Nonmetal
What is the atomic number for the element Barium?
56
What element is in group 7 and period 4
Manganese
What is the element symbol for the element Iron?
Fe
How many electron levels are present in an atom of fluorine?
2
How many electrons are present in an atom of fluorine?
9
How many electrons are in the outer energy level of fluorine
7
Why are the elements in group 18 stable?
They have 8 valence electrons so it won’t react
The element Boron has two isotopes, boron-10 and boron-11. Which isotope is more abundant?
Boron-11 is more abundant because it is closer to the average atomic mass
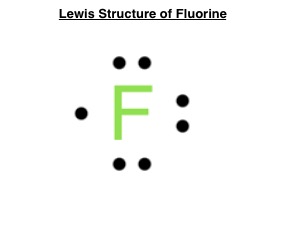
(On the Exam) Draw the electron dot diagram for fluorine
The elements in Groups 3 through 12 of the periodic table
Transition elements
All synthetic elements ____
Are not typically found on Earth
In the gaseous state ____ form diatomic covalent molecules
Halogens
Metals can be used as a wire because they are ____
Ductile
Any element with an atomic number greater than 92
Transuranium element
Elements that lie along the stair-step line of the periodic table
Metalliods
Hydrogen is grouped with the alkali metals because it ______
has one electron in its outer energy level
A group of elements that has two electrons in its outer energy level
Alkali metals
Bromine is the only halogen and nonmetal that is ____ at room temperature
Liquid
Molecule that consists of two atoms of the same element
Diatomic
Elements in Group 2 are Alkali ______ metals
Earth
Element in Group 1
Alkali
Different forms of same element with different molecular structures
Allotropes
Able to be hammered or rolled into sheets
Malleable