Chapter 25- Macromolecules
- Macromolecule: A long chain molecule containing hundreds of atoms joined together by covalent bonds. It is also called a polymer.
- Monomers: Small repeating units that join to form a macromolecule.
- Polymerisation: the process of joining together a large number of monomers to make a polymer.
- Addition polymeristion: the type of polymeristion where no atoms are lost so just one product is formed.
- Condensation polymeristion: the type of polymerisation where a small molecule, usually water, is also produced as a byproduct.
ADDITION POLYMERISATION
- At high temperature and pressure, thousands of alkenes join together to form an addition polymer.
- The double bonds in monomers break at such high temperature and pressure, forming single bonds with two other monomers.
- Ethene is used to manufacture the polymer, poly(ethene), commonly known as polythene.
- Addition polymers are used to make disposable containers, PVC pipes, raincoats and gloves etc.
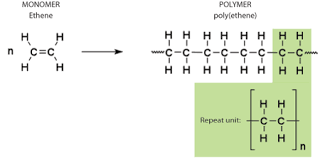
\
CONDENSATION POLYMERISATION
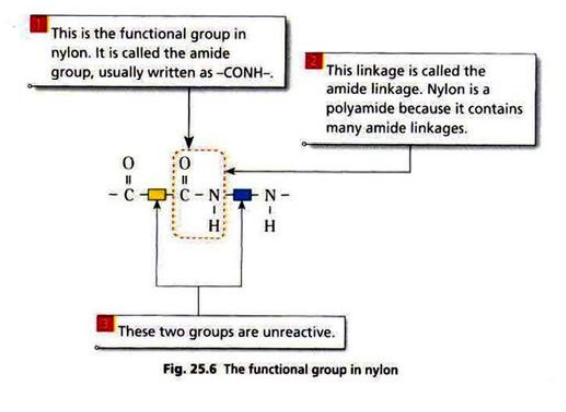
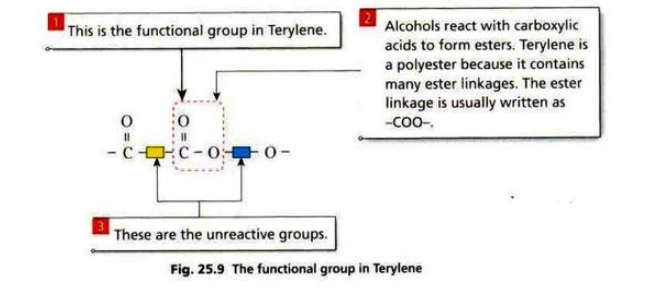
- There are two groups of condensation polymers: polyamides and polyesters.
Nylon is made from diamine and dicarboxylic acid.
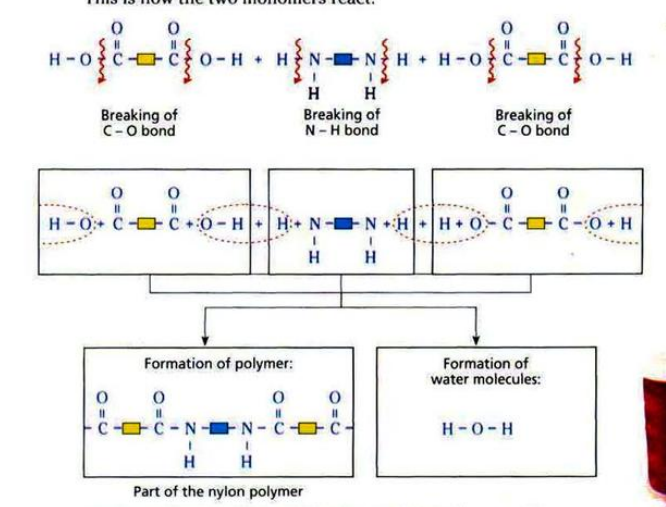
\
\
\ \ Terylene is made from diol and dicarboxylic acid.
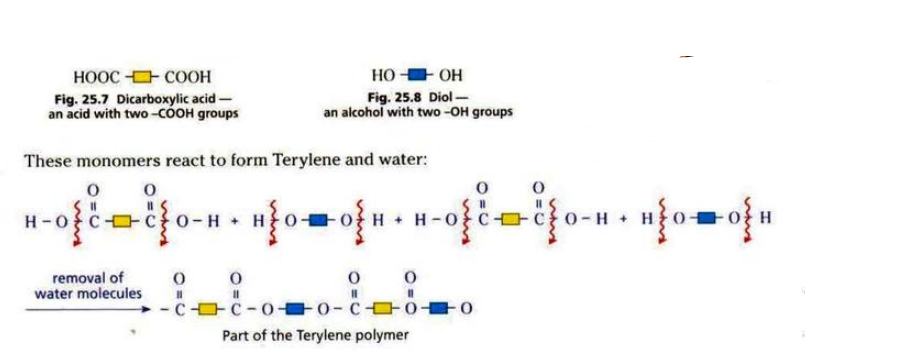
\
\
\
- Plastics and polymers are non-biodegradable and cause pollution problems.
\