C2.2 - Bonding (Ai generated with instructions by me)
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What are the two broad types of elements?
Metals and non-metals — most elements are metals, a small number are non-metals, and some are called metalloids/semi-metals
What is an ion?
An atom or molecule that has become electrically charged through the loss or gain of one or more electrons — the number of protons does NOT change
How do metals form ions?
Metals LOSE outer shell electrons → become positively charged ions (cations) — "CATions are PAWsitive"
How do non-metals form ions?
Non-metals GAIN electrons → become negatively charged ions (anions) — "ANIons are A Negative ion"
How does metallic character change across a period?
Decreases from left to right across a period
How does metallic character change down a group?
Increases going down a group — outer shell electrons are further from nucleus so more easily lost
Why is a metal further down a group MORE reactive?
Outer shell electrons are further from the nucleus (more shielding) → weaker attraction → electrons lost more easily
Why is a non-metal further down a group LESS reactive?
Harder to attract extra electrons to gain a full outer shell as the atom is larger with more shielding
What are the general properties of METALS?
1–3 outer shell electrons. Metallic bonding. Good conductors of electricity. Form basic oxides. Many react with acids. Lustrous, solid at room temp (except mercury), malleable, high mp/bp
What are the general properties of NON-METALS?
4–7 outer shell electrons. Covalent bonding. Poor conductors of electricity. Form acidic oxides (some neutral). Usually don't react with acids. Dull, various states at room temp, flaky/brittle, low mp/bp [DRAW: metals vs non-metals properties table]
What is electronic structure?
The arrangement of electrons in shells (energy levels) around the nucleus of an atom
What are the rules for filling electron shells?
First shell holds max 2 electrons. Second shell holds max 8. Third shell holds max 8 (for first 20 elements). Shells fill from inside out
What does the GROUP number tell you about electronic structure?
The number of electrons in the outer shell (e.g. Group 7 → 7 outer electrons)
What does the PERIOD number tell you about electronic structure?
The number of electron shells the atom has (e.g. Period 3 → 3 shells)
Write the electronic configuration of chlorine (atomic number 17)
2, 8, 7 — 2 in first shell, 8 in second, 7 in third [DRAW: electron shell diagram for Cl showing 2,8,7]
What is ionic bonding?
The strong electrostatic force of attraction between oppositely charged ions (formed by transfer of electrons from metal to non-metal)
How does ionic bonding form?
Metal atom LOSES outer shell electrons → positive ion. Non-metal atom GAINS those electrons → negative ion. Opposite charges attract → ionic bond
Describe the formation of NaCl (sodium chloride)
Na is Group 1 → loses 1 electron → Na⁺ (2,8). Cl is Group 7 → gains 1 electron → Cl⁻ (2,8,8). Electrostatic attraction between Na⁺ and Cl⁻ forms ionic bond [DRAW: dot-and-cross diagram of NaCl formation]
![<p>Na is Group 1 → loses 1 electron → Na⁺ (2,8). Cl is Group 7 → gains 1 electron → Cl⁻ (2,8,8). Electrostatic attraction between Na⁺ and Cl⁻ forms ionic bond [DRAW: dot-and-cross diagram of NaCl formation]</p>](https://assets.knowt.com/user-attachments/6eaa1472-ec4e-4e88-af17-c1ff2b0b0364.png)
Describe the formation of MgO (magnesium oxide)
Mg is Group 2 → loses 2 electrons → Mg²⁺ (2,8). O is Group 6 → gains 2 electrons → O²⁻ (2,8). Electrostatic attraction between Mg²⁺ and O²⁻ forms ionic bond [DRAW: dot-and-cross diagram of MgO formation]
![<p>Mg is Group 2 → loses 2 electrons → Mg²⁺ (2,8). O is Group 6 → gains 2 electrons → O²⁻ (2,8). Electrostatic attraction between Mg²⁺ and O²⁻ forms ionic bond [DRAW: dot-and-cross diagram of MgO formation]</p>](https://assets.knowt.com/user-attachments/b59835cc-7de0-4cad-bf6d-8595f4b5510c.png)
What are the advantages and disadvantages of dot-and-cross diagrams for ionic compounds?
Advantage: clearly shows electron transfer and which atom electrons came from. Disadvantage: fails to show 3D arrangement of atoms, can't illustrate the shape of the ionic lattice
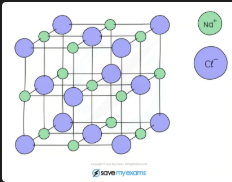
What are the advantages and disadvantages of ball-and-stick models for ionic compounds?
Advantage: shows 3D arrangement of ions in space, good for visualising ionic lattice. Disadvantage: fails to show electron movement, atoms appear too far apart, doesn't show forces of attraction
What is covalent bonding?
When two non-metal atoms share a pair of electrons so both achieve a full outer shell
What is a single covalent bond?
One shared pair of electrons between two atoms
What are simple covalent molecules?
Small discrete molecules held together by strong covalent bonds within the molecule but only weak forces between molecules (e.g. H₂, Cl₂, O₂, N₂, HCl, H₂O, NH₃, CH₄)
Why do simple covalent molecules have low melting and boiling points?
The intermolecular forces between molecules are very weak — little energy needed to overcome them. The covalent bonds within the molecule are NOT broken
Why do simple covalent molecules NOT conduct electricity?
They have no free electrons and no ions — there are no charge carriers
Name six examples of simple covalent molecules
H₂ (hydrogen), Cl₂ (chlorine), O₂ (oxygen), N₂ (nitrogen), H₂O (water), NH₃ (ammonia), CH₄ (methane), HCl (hydrogen chloride)
What are the advantages and disadvantages of dot-and-cross diagrams for covalent molecules?
Advantage: clearly shows the sharing of electrons and bonding. Disadvantage: fails to show the 3D arrangement of atoms or the shape of the molecule
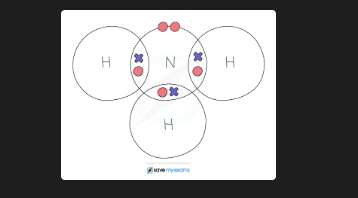
What are the advantages and disadvantages of ball-and-stick models for covalent molecules?
Advantage: shows 3D arrangement of atoms and shape. Disadvantage: doesn't show electron movement, atoms placed too far apart (gaps are much smaller in reality)
What are giant covalent structures?
Substances where very large numbers of non-metal atoms are covalently bonded together in a continuous 3D lattice — also called macromolecular structures
Why do giant covalent structures have very high melting and boiling points?
They have a very large number of strong covalent bonds — enormous amounts of energy needed to break them all
Describe the structure and properties of diamond
Each carbon atom is covalently bonded to 4 others in a tetrahedral arrangement. Very hard. Very high mp/bp. Does NOT conduct electricity (no free electrons or ions) [DRAW: diamond tetrahedral structure]
Describe the structure and properties of graphite
Each carbon atom is bonded to 3 others in flat hexagonal layers. One delocalised electron per carbon atom → conducts electricity. Layers held by weak forces → can slide over each other → soft and slippery (good lubricant) [DRAW: graphite layered structure]
Why can graphite conduct electricity but diamond cannot?
Graphite has one delocalised (free) electron per carbon atom that can carry charge. Diamond has all electrons in covalent bonds — none are free to move
Describe the structure and properties of silicon dioxide (SiO₂)
Each silicon atom is bonded to 2 oxygen atoms in a giant 3D lattice. Very hard. Very high mp/bp. Does NOT conduct electricity
What is a polymer?
A very large molecule made by joining together many small monomer units — has a repeating unit and a very high molecular mass
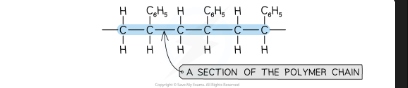
How are polymers formed?
Small monomer molecules join together (polymerisation) to form long polymer chains with a repeating unit
What is metallic bonding?
The strong electrostatic attraction between a lattice of positive metal ions and a sea of delocalised (free) electrons surrounding them
Why do metals conduct electricity and heat?
The delocalised electrons are free to move throughout the structure — they carry charge (electricity) and transfer energy (heat)
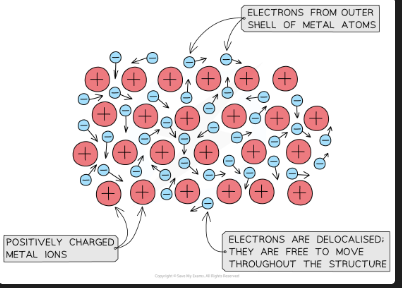
Why do metals have high melting and boiling points?
The electrostatic attraction between the positive metal ions and the delocalised electrons is very strong — large amounts of energy needed to break it [DRAW: metallic bonding diagram showing positive ions in sea of electrons]
Why are metals malleable (can be bent and shaped)?
The layers of positive ions can slide over each other while still being surrounded by the sea of delocalised electrons — the bonding is maintained
What did Mendeleev do to create his periodic table?
Arranged elements in order of atomic mass into groups based on chemical properties. Left gaps for undiscovered elements and predicted their properties. Some elements placed out of atomic mass order to fit chemical behaviour
What is the key difference between Mendeleev's table and the modern periodic table?
Modern periodic table arranges elements by atomic number (proton number) NOT atomic mass
What does the group number tell you in the modern periodic table?
The number of electrons in the outer shell → elements in the same group have similar chemical properties
What does the period number tell you in the modern periodic table?
The number of electron shells the atom has
Why do elements in the same group have similar chemical properties?
They have the same number of outer shell electrons — it is the outer electrons that determine how an element reacts