biomedical chem week 1 + 2
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Where are electrons confined in an atom?
Electrons do not move freely in the space around a nucleus, but rather are confined to regions of space called principal energy levels or shells.
How many electrons can each shell hold?
Each shell can hold 2n^2 electrons (n = 1, 2, 3…) where n is the number of shells.
How is each shell further divided?
Each shell is divided into subshells, which are designated by the letter s, p, d… and further divided to orbitals.
How many orbitals and electrons does each subshell hold?
S subshell has one orbital therefore 2 electrons. P subshell has three orbitals therefore 6 electrons. D subshell has five orbitals therefore 10 electrons. F subshell has seven orbitals therefore 14 electrons.
What is an orbital?
An orbital is a region of space that can hold 2 electrons.
Which subshell has the lowest and which has the highest energy level?
S has the lowest energy level and f has the highest energy level.
What is the ground state electron configuration?
The ground state electron configuration is the electron configuration of lowest energy.
What are valence electrons?
Valence electrons are the electrons in the outer shell that bond with other atoms. The group (column) number equals the number of valence shell electrons.
What is the valence shell?
The valence shell is the energy level in which the valence electrons are found. Electrons in this shell are the ones involved in the formation of chemical bonds and in chemical reactions.
What are core electrons?
Core electrons are other electrons closer to the nucleus.
Why do elements in the same group have similar chemical properties?
Elements in the same group have similar chemical properties because each group member has the same number of valence electrons.
What is the hierarchy of electron organisation?
Shell > Subshell > Orbital > Electrons.
When does a covalent bond form?
A covalent bond forms when electron pairs are shared between two atoms whose difference in electronegativity is 1.9 or less.
How is a covalent bond formed?
A covalent bond is formed when a portion of an atomic orbital of one atom overlaps a portion of an atomic orbital of another atom in order to share an electron pair.
What is ionic bonding?
Ionic bonding is the transfer of one or more electrons from an atom with lower electronegativity (usually a metal) to an atom with higher electronegativity (usually a non-metal), forming oppositely charged ions that are held together by electrostatic attraction, and electronegativity difference is more than 1.9.
What determines whether bonding is covalent or ionic?
Covalent or ionic bonding is decided by electronegativity. If we want to know if an atom will bond ionically or covalently we must know the electronegativity difference between the atoms. The greater the difference, the more likely it is an ionic bond.
What is electronegativity?
Electronegativity is a measure of the force of an atom's attraction for electrons that it shares in a chemical bond with another atom.
Card 18
Card 20
What happens to the more and less electronegative atom during ionic bonding? Back:
The more electronegative atom gains one or more valence electrons and becomes an anion; the less electronegative atom loses one or more valence electrons and becomes a cation.
Why does electronegativity increase from left to right across a period?
The electronegativity value increases from left to right because of the increasing positive charge on the nucleus due to an increase in the atomic number, which leads to a stronger attraction for electrons in the valence shell.
Why does electronegativity increase going up a column?
The electronegativity value increases going up a column because of the decreasing distance of the valence electrons from the nucleus and decrease in the number of electron shells, which leads to stronger attraction between a nucleus and its valence electrons.
What is the difference between a nonpolar and polar covalent bond?
In a nonpolar covalent bond, electrons are shared equally and there electronegativity differences less than 0.5. In a polar covalent bond, they are shared unequally and there electronegativity differences is between 0.5 and 1.9.
Why does the less electronegative atom acquire a partial positive charge in a polar covalent bond?
The less electronegative atom has a lesser fraction of the shared electrons and acquires a partial positive charge due to it being less likely to attract an electron therefore positively charged.
Hybridization
Hybridization = mixing of atomic orbitals on the same atom to create new, equivalent orbitals arranged for maximum separation
Hybrid orbitals arrange themselves to:
Maximize separation
Minimize electron–electron repulsion
This is done by fusing the valence orbitals.
The 90° issue arises only when pure p orbitals on the same atom try to form multiple bonds, which is why hybridization is needed.
Example: Carbon sp³ hybridization
Carbon mixes one s orbital and three p orbitals from the same shell to form four new equivalent orbitals through the fusing of valence orbitals
These new orbitals are directional and point toward the corners of a tetrahedron.
Explains why methane (CH₄) has 109.5° bond angles instead of 90°.
Weak Interactions
Hydrogen bonding and dipole–dipole interactions occur because electrons are unevenly shared in polar bonds, creating partial positive and negative regions. These polar regions cause molecules to orient themselves so that oppositely charged areas attract each other, thus attracting other molecules
Water is liquid because of hydrogen bonding, if not it would be a gas.
Formal Charge Equation
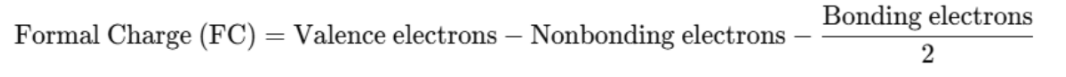
Resonance Structures
Resonance contributing structures are representations of a molecule or ion that differ only in the distribution of valence electrons which occurs when delocalised electrons shift around in order to reach octet creating different structures
three resonance contributing structures are all equivalent, meaning that they have identical patterns of covalent bonding (each contributing structure has one double bond and two single bonds) and are of equal energy.
Resonance Hybrid
Resonance hybrid A molecule or ion that is best described as a composite of a number of contributing structures.
According to this theory, many molecules and ions are best described by writing two or more Lewis structures and considering the real molecule or ion to be a composite of these structures. We show that the real mole- cule or ion is a resonance hybrid of the various contributing structures by interconnecting them with double‐headed arrows.
The resonance method is a way to describe the real structure and at the same time retain Lewis structures with electron‐pair bonds.
Valence Shell Electron Pair Repulsion (VSEPR)
Valence Shell Electron Pair Repulsion (VSEPR) is a model used to predict the shape of molecules. The basic premise is: “Each group of valence electrons around a central atom is located as far away from the others as possible in order to minimise repulsions
How bonding partners affect shape
2 bonds, 0 lone pairs → Linear
3 bonds, 0 lone pairs → Trigonal Planar
2 bonds, 1 lone pair → Bent
4 bonds, 0 lone pairs → Tetrahedral
3 bonds, 1 lone pair → Pyramidal
2 bonds, 2 lone pairs → Bent
Polar and Non Polar symmetry
Symmetrical shape = non polar because charge is evenly distributed
If there are lone pair it is not symmetrical because it creates partial charges
Symmetrical shapes that are nonpolar (when all outer atoms are identical with no long pairs) are:
Linear
Trigonal planar
Tetrahedral
Trigonal bipyramidal
Octahedral
Equilibrium Equation
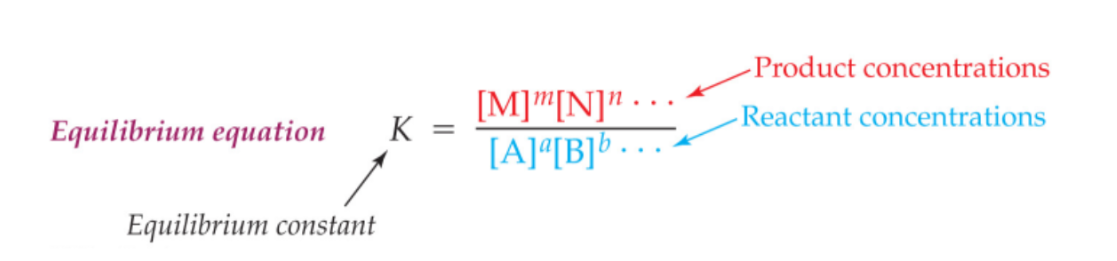
How does K affect concertation of products/reactants
K much smaller than 0.001: Only reactants are present at equilibrium; essentially no reaction occurs.
K between 0.001 and 1: More reactants than products are present at equilibrium.
K between 1 and 1000: More products than reactants are present at equilibrium.
K much larger than 1000: Only products are present at equilibrium; reaction goes essentially to completion.
So if K is really big more products therefore forward reaction
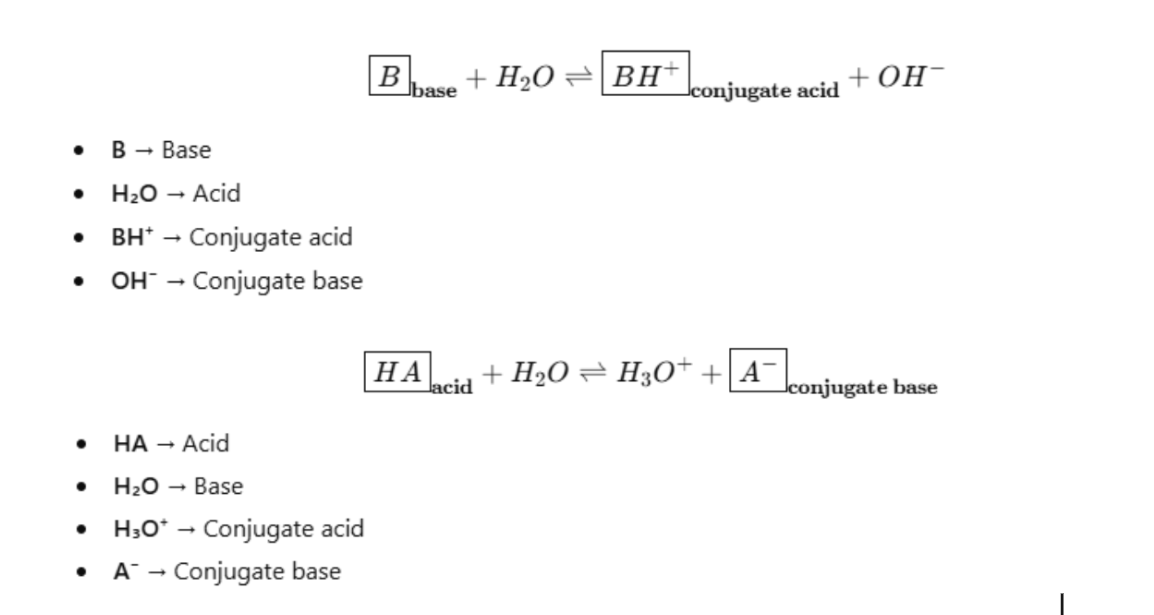
Acids and Bases
An acid is a proton donor
It must contain H in its formula, e.g. HNO3, H2PO4 - , H2SO4, HCl.
An acid acts as an reactant and donate its proton to create a product and the remaining becomes a conjugate base
A base is a proton acceptor
Must contain a lone pair of electrons to bind the H+ ion, e.g. NH3, CO3 2- , OH- .
Strong Acid Example
HCl = strong acid
Completely ionises in water.
Cl⁻ = conjugate base
Extremely weak base
Due to its weak basic strength it has almost no attraction for H⁺ and no tendency to accept H⁺
Reaction goes almost fully forward (no meaningful reverse reaction).
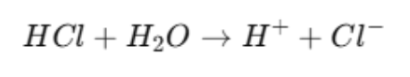
Weak Acid Example
CH₃COOH = weak acid
Only partially ionises.
CH₃COO⁻ = conjugate base
Stronger basic strength than Cl⁻
Due to its stronger basic strength it has more attraction for H⁺ thus greater tendency to to accept H⁺
Reaction is then reversible
The reaction is reversible and reaches equilibrium rather than going fully forward.
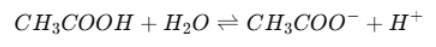
Strong Base Example
NaOH = strong base
Fully dissociates.
Na⁺ = conjugate acid (very weak)
Has no acidic behavior in water.
Due Na+ weaker acidic strength it has less ability to donate H⁺.
Due to OH⁻ does not give the H⁺ back easily because it is such a strong base
Reaction goes fully forward.
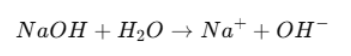
Weak Base Example
NH₃ = weak base
Partially dissociates
Only partially accepts H⁺.
NH₄⁺ = conjugate acid
Stronger acid than Na⁺ (which has none).
Due to its stronger acidic strength it has greater ability to donate H⁺ back
Reaction is reversible.
Therefore the reaction is reversible and reaches equilibrium.
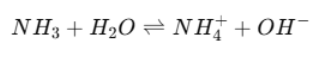
Acid and Bases examples
Strong Acid | Strong Base | Weak Acid | Weak Base |
HCl, HBr, HNO3, H3PO3 , H2SO4, | NaOH, KOH, CaO, Mg(OH)2 | CH3COOH, C6H5COOH, HNO2, H3PO4 | NH3, C5H5N |
pH relationships
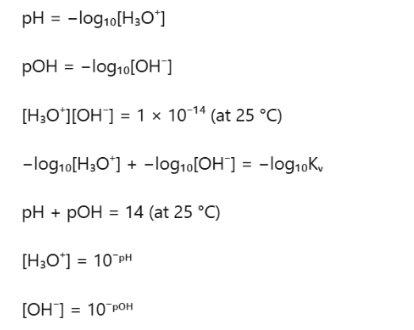
Kw
Kw stands for the ionic product of water which tells you how much water naturally splits into hydronium and hydroxide ions at a given temperature
at 25 degrees Kw=1×10−14
Comparing Equilibrium and Strength of Acidity
Equilibrium constant (Ka) measures how far the reaction proceeds toward forming products
A higher Ka means more products are formed → more H3O+ ions → lower pH, so the solution is more acidic.
pKa is the negative logarithm of Ka
Therefore, a larger Ka corresponds to a smaller pKa, indicating a stronger acid.

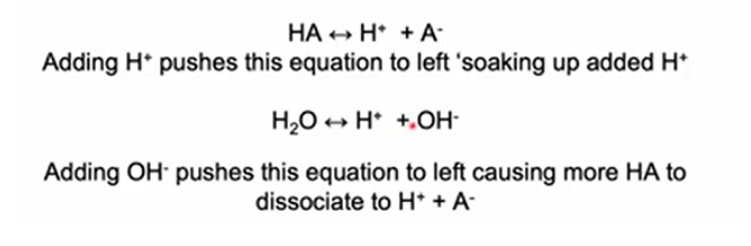
Buffer
Buffers are a mixture of a weak acid and it’s conjugate base that show maximum buffering capacity when both species are present in equal amounts. They resist change in pH caused by addition of H+/H3O+ or OH-, due to a balancing act and equilbirum between the two processes
Buffering results from two reversible reaction equilibria occurring in a solution of nearly equal concentrations of a proton donor and its conjugate proton acceptor.
Whenever H+ or OH− is added to a buffer, the result is a small change in the ratio of the relative concentrations of the weak acid and its anion and thus a small change in pH. The decrease in concentration of one component of the system is balanced exactly by an increase in the other.
Each conjugate acid-base pair has a characteristic pH zone in which it is an effective buffer
formulas
Alkanes
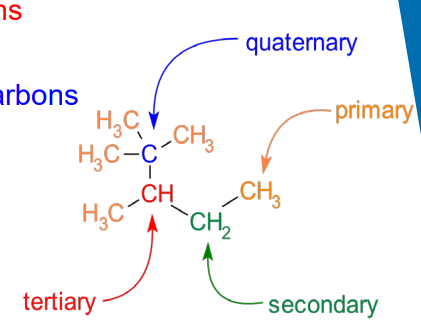
Primary (1o) : a carbon bonded to 1 other carbon
Secondary (2o): a carbon bonded to 2 other carbons
Tertiary (3o): a carbon bonded to 3 other carbons
Quaternary (4o): a carbon bonded to 4 other carbons
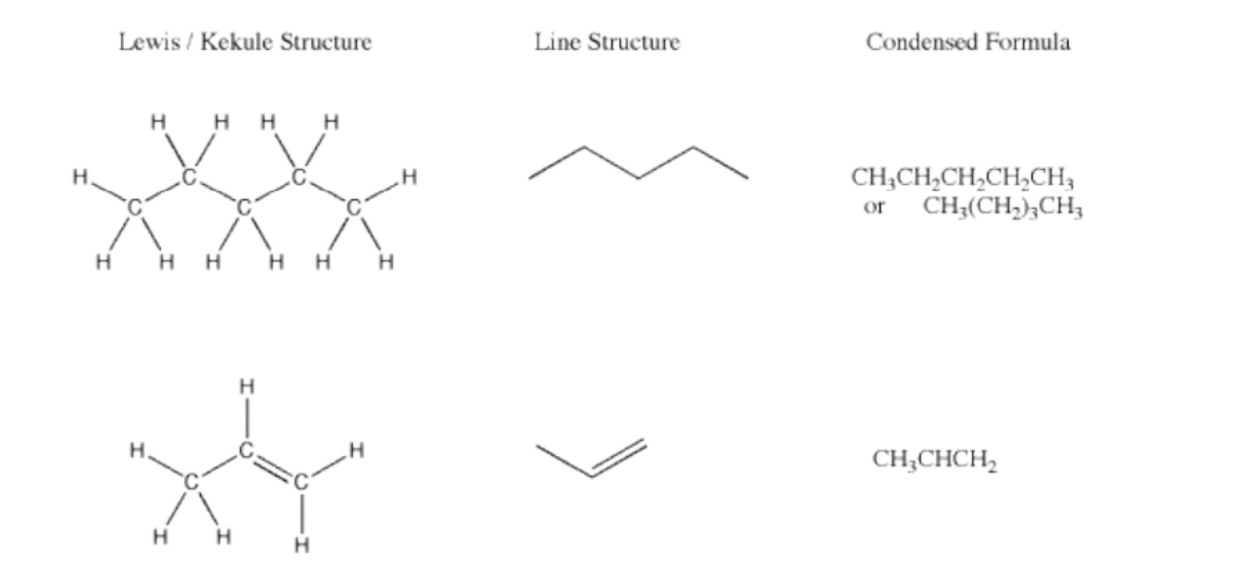
Alkanes are saturated hydrocarbons whose carbons are arranged in a chain (aliphatic) or in a ring (cycloalkane)
A saturated molecule has only single bonds between carbon atoms (or other atoms in the chain). “Saturated” means it contains the maximum number of hydrogen atoms possible for that number of carbons.
Shape = tetrahedral carbon, all bond angles are approximately 109.5°.
Alkanes have the general formula CnH2n+2
Cycloalkanes have the general formula CnH2n
Alkene
Alkenes are unsaturated hydrocarbons containing one or more carbon-carbon double bonds.
Carbons can be arranged in a chain (aliphatic) or in a ring (cycloalkene)
Shape = trigonal planar at carbon, all bond angles are approximately 120° .
Alkenes have the general formula CnH2n
Alcohol
Alcohols are polar compounds
both the C-O and O-H bonds are polar covalent.
Dipole-dipole interaction: the attraction between the positive end of one dipole and the negative end of another.
Hydrogen bonding: when the positive end of one dipole is an H bonded to F, O, or N (atoms of high electronegativity)
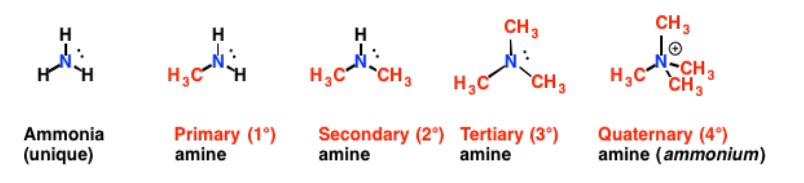
Amines
Primary (1o) : a nitrogen bonded to only 1 other carbon
Secondary (2o): a nitrogen bonded to 2 other carbons
Tertiary (3o): a nitrogen bonded to 3 other carbons
Quaternary (4o): a nitrogen bonded to 4 other carbons
Amines are polar compounds
Both 1° and 2° amines form hydrogen bonds
All amines are weak bases
Carboxylic Acid
In liquid and solid states, carboxylic acids are associated by hydrogen bonding into dimer structures
Carbonyl Derivatives
Aldehydes – C=O at the end of a carbon chain, bonded to H (R–CHO)
Ketones – C=O within the chain, bonded to two carbons (R–CO–R′)
Carboxylic acids – C=O bonded to –OH (R–COOH)
Esters – C=O bonded to –OR (R–COOR′)
Amides – C=O bonded to –NR₂ (R–CONH₂, R–CONHR′, R–CONR′₂)
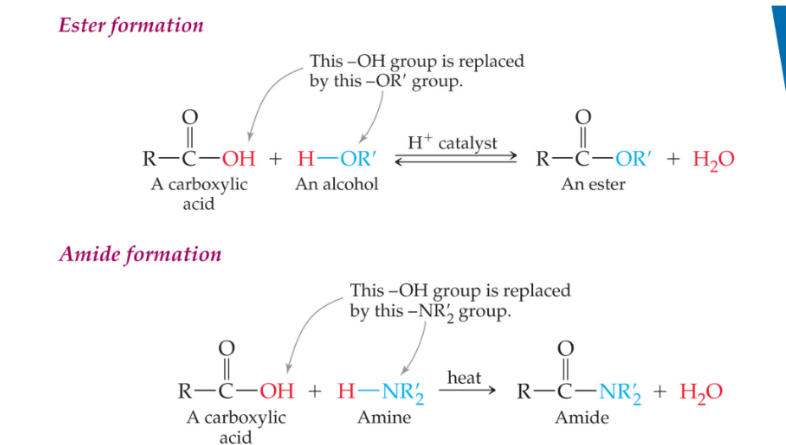
ester and amide formation
How do you convert an alkane to a haloalkane?
Alkane + X₂ with UV light → Haloalkane Conditions: UV light Reaction Type: Substitution
If you have an alkane with Cl₂ and UV light, what do you get?
Haloalkane Reaction Type: Substitution
How do you convert a haloalkane to an alcohol?
Haloalkane + NaOH → Alcohol Conditions: Aqueous NaOH Reaction Type: Substitution
If you have a haloalkane with NaOH, what do you get?
Alcohol Reaction Type: Substitution
How do you convert a haloalkane to an amine?
Haloalkane + NH₃ → Amine Conditions: Excess NH₃ Reaction Type: Substitution
If you have a haloalkane with NH₃, what do you get?
Amine Reaction Type: Substitution
How do you convert an alkene to an alkane?
Alkene + H₂ → Alkane Conditions: H₂, Pt catalyst Reaction Type: Addition
If you have an alkene with H₂ and Pt catalyst, what do you get?
Alkane Reaction Type: Addition
How do you convert an alkene to an alcohol?
Alkene + H₂O → Alcohol Conditions: H₃PO₄, 300°C, steam Reaction Type: Addition
If you have an alkene with H₃PO₄ at 300°C and steam, what do you get?
Alcohol Reaction Type: Addition
How do you convert a secondary alcohol to a ketone?
Secondary alcohol + [O] → Ketone Conditions: Cr₂O₇²⁻/H⁺ Reaction Type: Oxidation
If you have a secondary alcohol with Cr₂O₇²⁻/H⁺, what do you get?
Ketone Reaction Type: Oxidation
How do you convert a primary alcohol to an aldehyde?
Primary alcohol + [O] → Aldehyde Conditions: Cr₂O₇²⁻/H⁺, distillation Reaction Type: Oxidation
If you have a primary alcohol with Cr₂O₇²⁻/H⁺ and distillation, what do you get?
Aldehyde Reaction Type: Oxidation
How do you convert an aldehyde to a carboxylic acid?
Aldehyde + [O] → Carboxylic acid Conditions: Cr₂O₇²⁻/H⁺, reflux Reaction Type: Oxidation
If you have an aldehyde with Cr₂O₇²⁻/H⁺ and reflux, what do you get?
Carboxylic acid Reaction Type: Oxidation
How do you convert a carboxylic acid to an ester?
Carboxylic acid + Alcohol → Ester + H₂O Conditions: H₂SO₄ catalyst, heat Reaction Type: Condensation
If you have a carboxylic acid with an alcohol and H₂SO₄ catalyst with heat, what do you get?
Ester Reaction Type: Condensation
How do you convert an ester to a carboxylic acid and alcohol?
Ester + H₂O → Carboxylic acid + Alcohol Conditions: H₂SO₄ catalyst, heat Reaction Type: Hydrolysis
If you have an ester with H₂O and H₂SO₄ catalyst with heat, what do you get?
Carboxylic acid + Alcohol Reaction Type: Hydrolysis
member of the same homologus group
similar chem properties
different physical properties
Constitutional isomers and Stereoisomers
Constitutional isomers
In structural (constitutional) isomerism, the atoms are arranged in a different order.
They have different physical and chemical properties
Stereoisomers isomers
In stereoisomerism, the atoms are arranged in the same order, but in different 3-dimensional orientation
They have similar physical and chemical properties
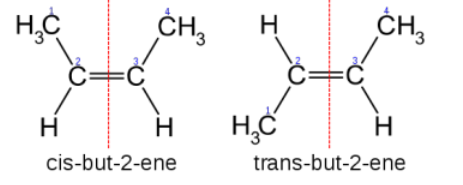
Cis-trans isomers
Depending on whether the substituents of highest priority at each end of the double bond are next to or opposite each other
same side (cis) or opposite sides (trans)
Priority based on atomic number THAT IMMEDIATELY CONNECTS TO TEH CHIRLA CENTRE
If two groups have an atom common, then u move on to the next atom connected
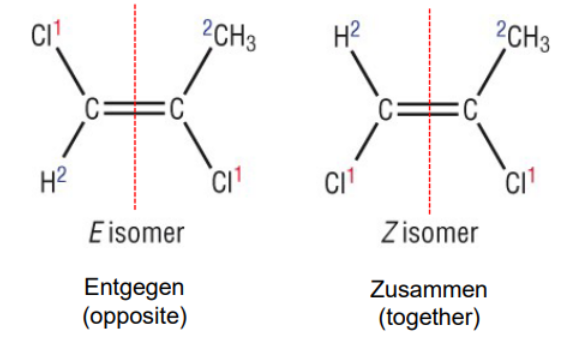
E & Z Isomers
To assign an E, Z configuration, first assign a priority to the substituents on each side of the double bond
Priority based on atomic number THAT IMMEDIATELY CONNECTS TO TEH CHIRLA CENTRE
If two groups have an atom common, then u move on to the next atom connected
Z is if the groups with the same priority are on the same side
E is if the groups with the same priority are on opposite sides
Chirality
A molecule (or object) that is not superposable on its mirror image is described as being chiral.
Many chiral molecules contain an asymmetric carbon centre, a so-called stereogenic centre.
This is easily identified as a carbon atom attached to four different groups.
Stereoisomers
Stereoisomers that are mirror images of each other are enantiomers
MUST be chiral
Enantiomers do not differ in their physical characteristics, yet interact differently with polarized light and with stereospecific environments!
If a molecule has enantiomers → it is chiral
If a molecule is achiral → it cannot have enantiomers
Achiral compounds
If an object and its mirror image are superimposable, they are identical, they are called achiral (without chirality).
An object or molecule is achiral if it has a plane of symmetry.
A plane of symmetry is an imaginary plane passing through an object dividing it such that one half is the mirror image of the other half.
Naming Stereoisomers
Look at the molecule so that group 4 point away from you
So it just basically disappears and you only see group 1-3
Read the groups from 1→ 3.
If the reading is clockwise, the stereochemistry is designated as R (Latin: Rectus, right)
If the reading is anticlockwise, the stereochemistry is designated as S (Latin: Sinister, left)
Calculating the number of stereoisomers
For a molecule with n stereocenters, a maximum of 2n stereoisomers are possible