Rev test 1 atomic structure and bonding
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
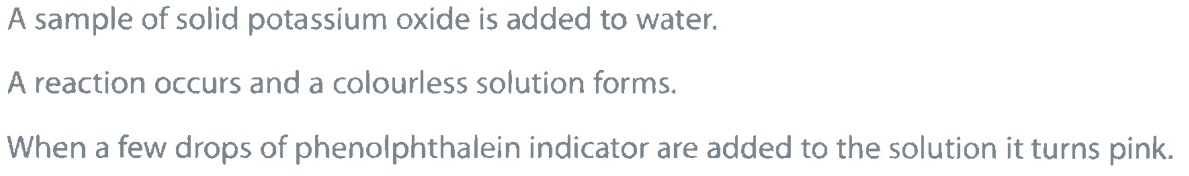
A sample of solid potassium oxide is added to water.
Identify the ion responsible for the colour change when phenolphthalein is added to the solution. (1)
M1: hydroxide ion / OH-
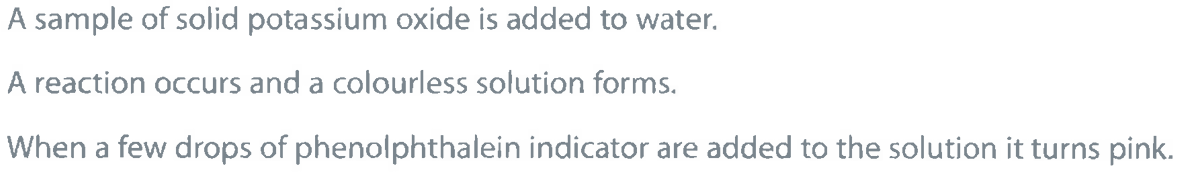
Give a chemical equation for the reaction between potassium oxide and water. (1)
M1: K_2O + H_2O \rightarrow 2KOH (Allow multiples/submultiples)
Explain why ionic compounds conduct electricity when molten or in aqueous solution, but not when in the solid state. (2)
M1: (When) molten / (in) aqueous (solution) the ions are mobile / free to move
M2: (In) solid state the ions are locked in (fixed) lattice positions / (ions) are not free to move
A molecule of silicon tetrafluoride (SiF_4) contains covalent bonds. Draw a dot and cross diagram to show the outer electrons in this molecule. (2)
M1: (for) BP (bonding pairs) (being) correct > [The diagram must show four pairs of shared electrons, one between the Si atom and each of the four F atoms]
M2: (for) LP (lone pairs) (being) correct on each F atom - [Each Fluorine atom must have 6 non-bonding electrons to complete its octet]
Note: M2 (is) dependent on M1
![<ul><li><p><strong>M1:</strong> (for) <strong>BP (bonding pairs)</strong> (being) <strong>correct</strong> > [The diagram must show four pairs of shared electrons, one between the Si atom and each of the four F atoms]</p></li><li><p><strong>M2:</strong> (for) <strong>LP (lone pairs)</strong> (being) <strong>correct on each F atom - </strong><span style="background-color: transparent; font-size: 18px;"><em>[Each Fluorine atom must have 6 non-bonding electrons to complete its octet]</em></span></p></li></ul><p><strong><em>Note:</em></strong><em> </em><strong><em>M2</em></strong><em> (is) </em><strong><em>dependent on M1</em></strong></p>](https://assets.knowt.com/user-attachments/306015fe-c6af-4685-aa14-121194010cd1.png)
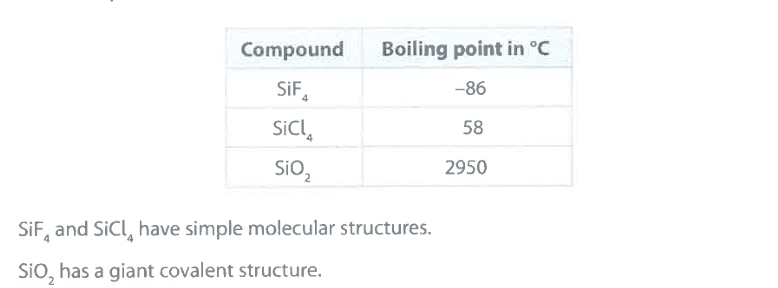
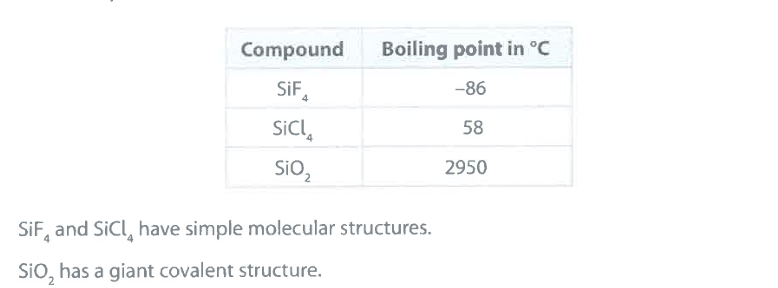
Explain why the boiling point of SiCl_4 is greater than the boiling point of SiF_4 . (2)
M1: intermolecular forces (of attraction)
M2: (are) stronger in
SiCl_4
M2 dependent on M1
accept: "more energy needed to overcome IM forces in SiCl_4 for M2
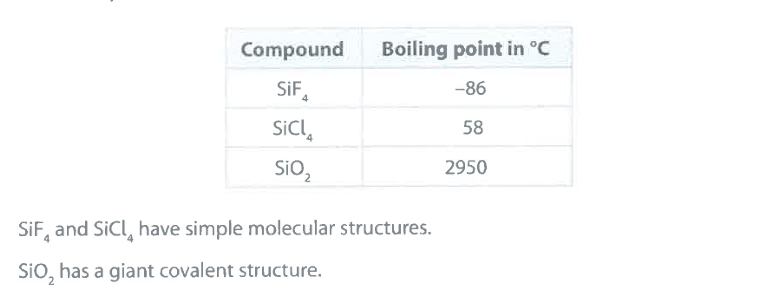
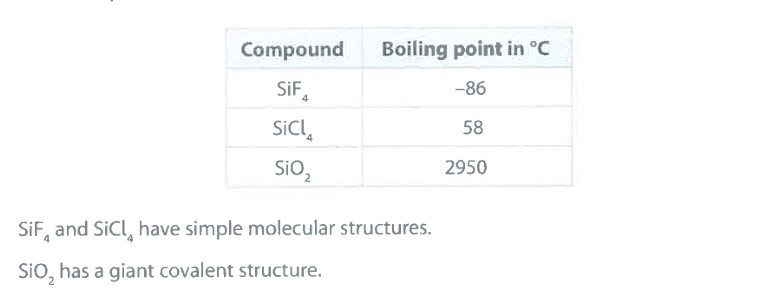
Explain why the boiling point of SiO_2 is very much greater than the boiling point of SiCl_4 . (2)
M1: SiO_2 has (many) strong internal (covalent) bonds between atoms
M2: (whereas) SiCl_4 has weak intermolecular forces of attraction (between particles)
accept: for M1+M2 — "more energy needed to break internal bonds in SiO_2 than overcome intermolecular attractions in SiCl_4"
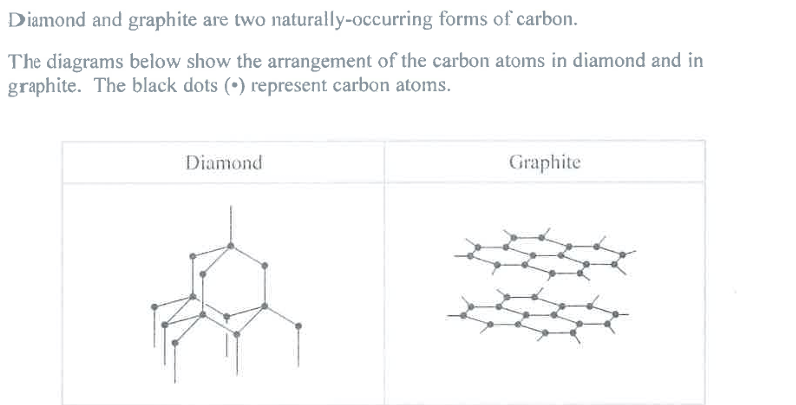
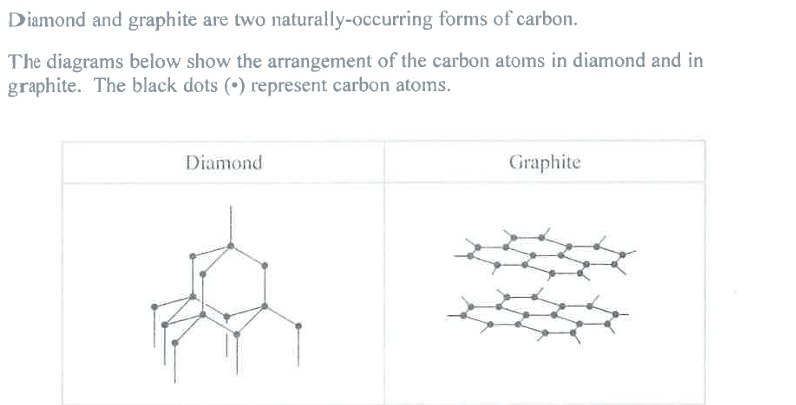
Name the type of structure in diamond and explain, in terms of its bonding, why diamond has a high melting point. (4)
M1: Diamond (is)
M3: Giant Covalent
M2: (many) strong (internal) bonds must be broken to melt diamond
M3: requires lots of energy
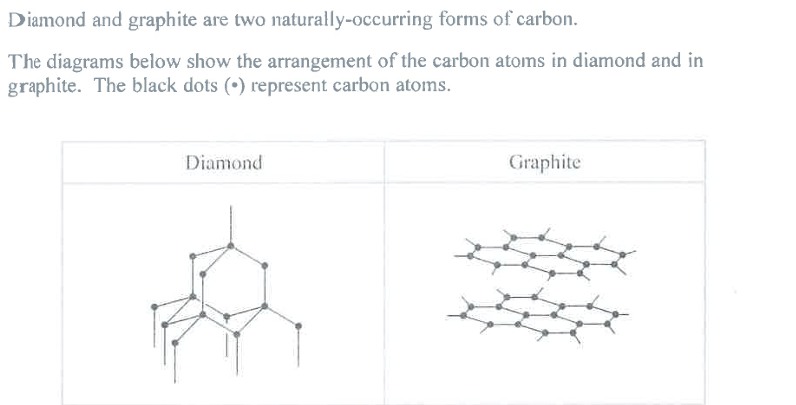
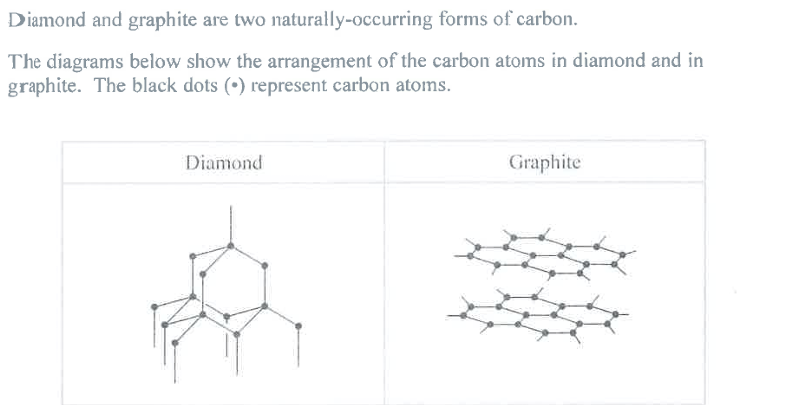
Explain, in terms of its structure, why graphite can act as a lubricant. (2)
M1: layers of atoms can slide past each other
M2: due to the weak (~~intermolecular~~) attractions between layers
Reject: mention of ions/molecules
Reject: mention of bonds between layers
Reject: "intermolecular", see June 25 paper 1, q8c
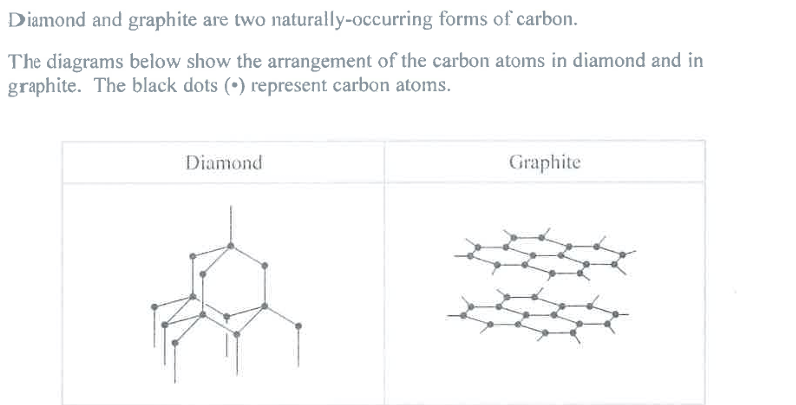
The structure of graphite has one feature in common with that of metals. This feature allows graphite to conduct electricity.
Suggest what this feature is and why it allows graphite to conduct electricity. (2)
M1: delocalised electrons (within each layer)
M2: (are) MOBILE
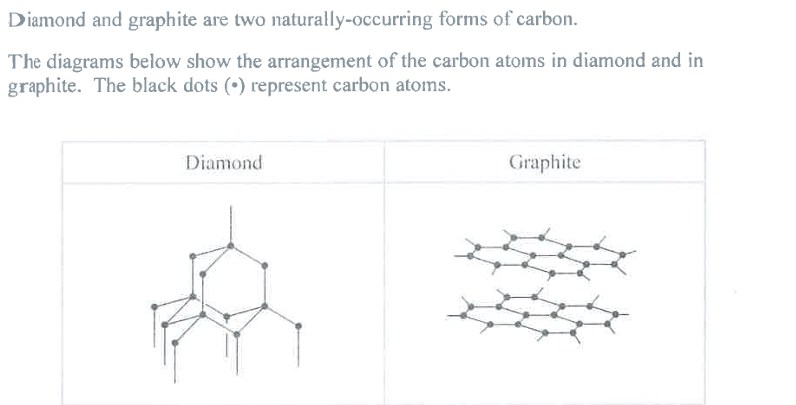
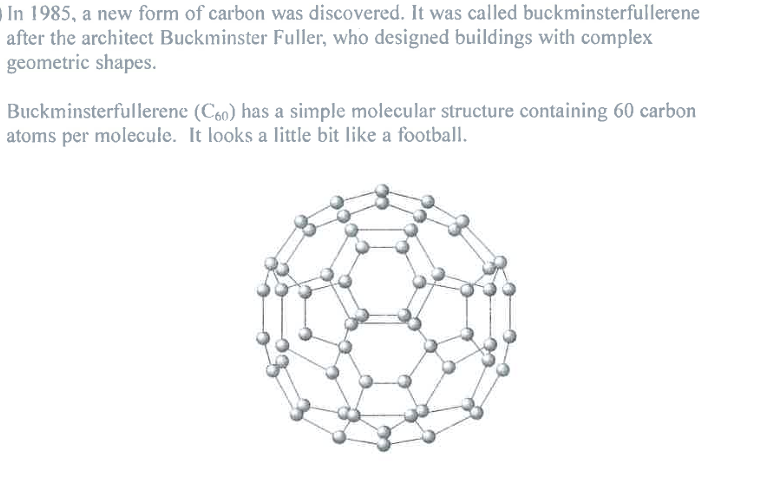
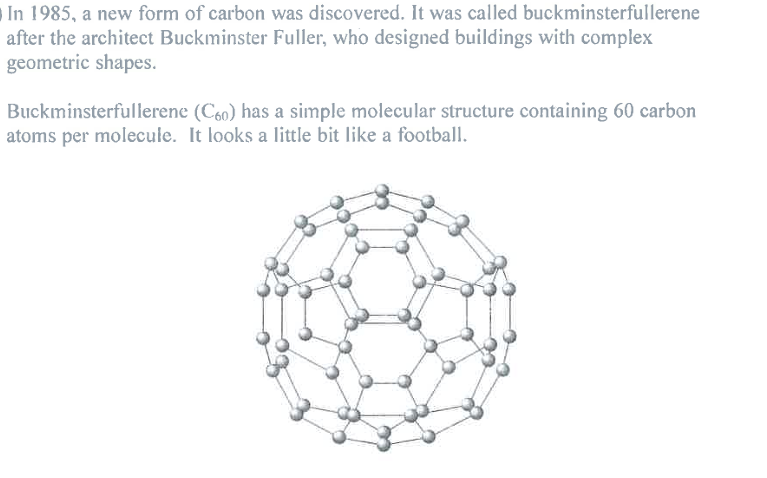
Suggest why buckminsterfullerene (C60) has a much lower melting point than diamond. (2)
M1: not a giant structure
M2: weak intermolecular attractions (between C60 particles) are easily overcome
Ignore: "simple molecular" as given in Qu (Question)
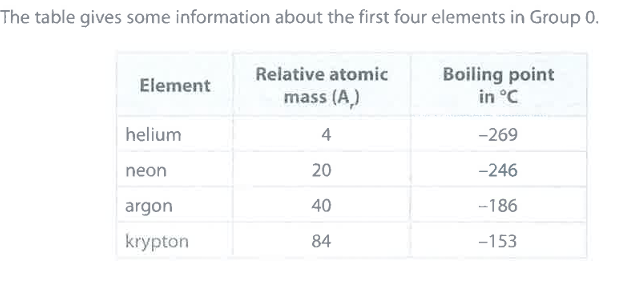
State the relationship between the relative atomic mass and the boiling point of these elements. (1)
M1: as A_r increases, so does boiling point
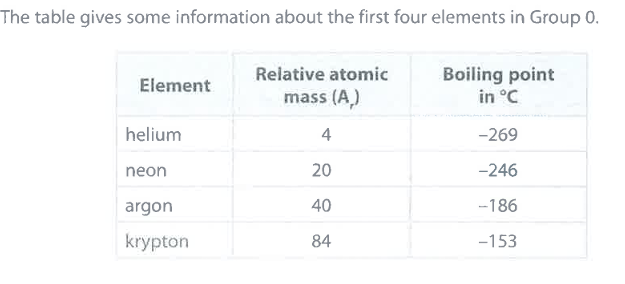
State why the elements in Group 0 are unreactive. (2)
M1: they have full outer shells of electrons
M2: therefore, they do not readily gain or lose electrons
tate why the halogens have similar chemical properties. Refer to electronic configurations in your answer. (1)
M1: same no. of outer shell electrons (7)
When chlorine is added to sodium bromide solution, chlorine displaces bromine. Write a chemical equation for this reaction. (1)
M1: Cl_2 + 2NaBr \rightarrow 2NaCl + Br_2
chlorine displaces bromine because smaller atomic radius allows its nucleus to attract electrons more strongly
State the colour of the solution formed in this reaction. (1)
M1: orange
Chlorine is a stronger oxidising agent than bromine. Explain this statement in terms of atomic structure. (4)
M1: (an) oxidising agent is itself reduced
M2: Cl gains electrons more readily than Br
M3: Cl (2,8,7) has 1 less shell of electrons than Br / (Cl) is smaller than Br
M4: (the) incoming electron is more strongly attracted to (the) Cl nucleus (as a result)