MIDTERM - REVIEWER FOR CHEMISTRY
1/104
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
105 Terms
The solids featuring highly ordered arrangements of their particles (atoms, ions, and molecules) in microscopic structures.
These ordered microscopic structures make up a crystal lattice that accounts for the structure of the solid at any given point. Examples of crystalline solids include salt (sodium chloride), diamond, and sodium nitrate.
Crystalline Solids
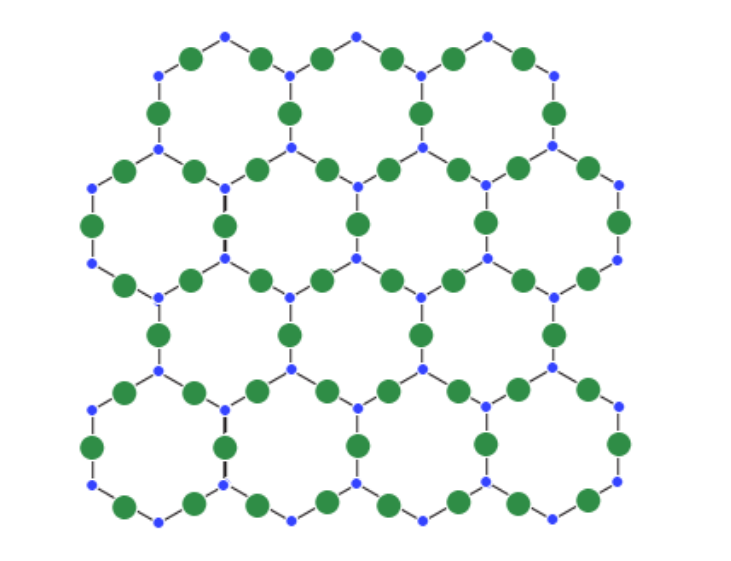
Crystalline Solids
is the smallest portion of a crystal lattice that shows the three-dimensional pattern of the entire crystal.
Unit Cell
is a point in space where atoms, ions, or molecules are positioned in a repeating pattern to form a crystal lattice. These points represent the locations where the particles in the crystal structure are positioned, and the pattern of these points determines the overall structure of the crystal.
Lattice Point
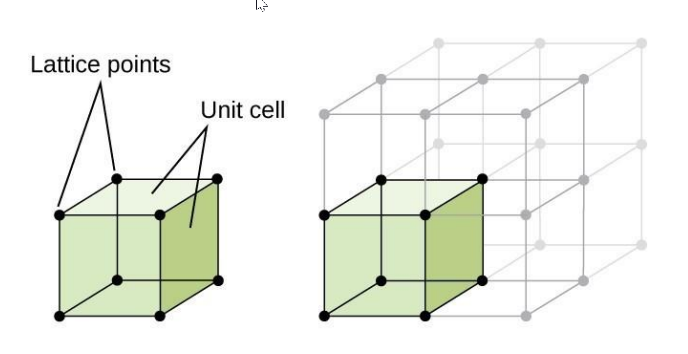
PARTS

Simple Cubic
Tetragonal
Orthorhombic
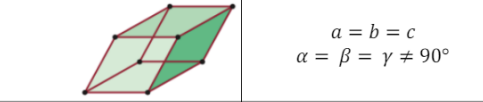
Rhombohedral
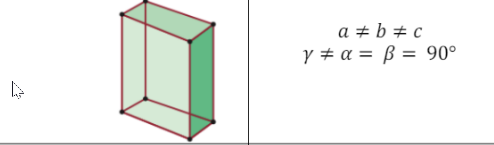
Monoclinic
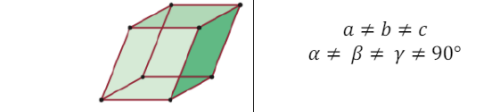
Tricilinic
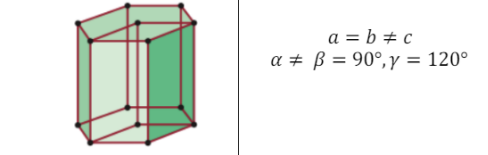
Hexagonal
Cubic
Tetragonal
Orthorhombic
Rhombohedral
Monoclinic
Tricilinic
Hexagonal
Seven Crystal Systems
consists of atoms arranged in a cube where each corner of the cube has a fraction of an atom with six additional full atoms positioned at the center of each cube face.
Face Centered Cubic (FCC)
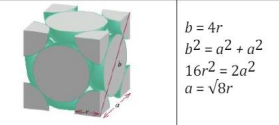
Face Centered Cubic (Figure)
is a crystal structure that consists of a cube-shaped unit cell with atoms located at the corners and in the center of the cube. In this structure, each atom is surrounded by eight nearest neighbors located at the corners of the cube.
Body Centered Cubic (BCC)
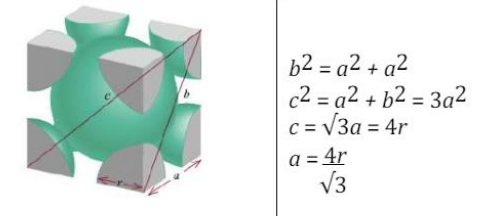
Body Centered Cubic (BCC) (Figure)
is a crystal structure that consists of three layers of atoms. The bottom layer has six corner atoms and one face centered atom. The middle layer has three full atoms. The upper layer has six corner atoms and one face centered atom.
Hexagonal Close-Packed (HCP)
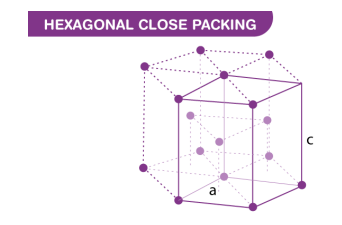
Hexagonal Close-Packed (HCP) (Figure)
are crystalline structures that grow from ionic bonds and are held together by electrostatic attraction. Ionic bonds are atomic bonds created by the attraction of two differently charged ions. The bond is typically between a metal and a non-metal.
Ionic Crystals
It is one of the properties of ionic crystals. The way that ionic crystals structure themselves when they bond is called a _____. This is because the ions organize themselves into a regular lattice shape.
Crystal Lattice (Properties of Ionic Crystals)
It is one of the properties of ionic crystals. Ionic crystals melt or boil only at very high temperatures. The enthalpy of these crystals (the amount of fusion and vaporization necessary to melt one mole) is as much as 100 times that of other molecular compounds.
High Melting Point (Properties of Ionic Crystals)
It is one of the properties of ionic crystals. Ionic crystals are very hard. They are so brittle because if one layer of ions must move past another, the entire arrangement is disturbed.
Hard Structures (Properties of Ionic Crystals)
also called atomic crystals are the molecular solids in which the same or different atoms are joined together by covalent bonding. Atoms of the same or different kinds with no/small changes in electronegativity form the structural units of such crystals.
Covalent Crystals
As the atoms are held together by the covalent bonding via sharing of electron between atoms, it possesses strong forces, and hence high amount of energy is required to break this bond. Thus, Covalent crystals have ________
Melting and Boiling Point (Properties of Covalent Crystals)
Because of the directional properties of the covalent bonds, the atoms are strongly bounded in the lattice and cannot be easily displaced or compressed. This is the reason for the ___ of covalent crystals.
Hardness (Properties of Covalent Crystals)
Covalent crystals are insoluble in polar solvent, but soluble in non-polar solvent.
Solubility (Properties of Covalent Crystals)
Since the electrons are tightly held by the atoms and cannot move through the lattice, covalent crystals possesses poor electrical ________
Conductivity (Properties of Covalent Crystals)
When a crystal is subjected to sufficient force, covalent bonds are broken as the lattice is distorted, and as a result of which shattering occurs in the crystal. Thus, covalent crystals like diamond are highly _____ in nature.
Brittleness (Properties of Covalent Crystals)
consist of molecules at the lattice points of the crystal, held together by relatively weak intermolecular forces. The intermolecular forces may be dispersion forces in the case of nonpolar crystals, or dipole-dipole forces in the case of polar crystals. Some molecular crystals, such as ice, have molecules held together by hydrogen bonds.
Molecular Crystals
Low Melting Points
Flexibility
Poor Conductors
Properties of Molecular Crystals
are structures made entirely of metal elements. The atoms in these structures are bonded through metallic bonding – this is a type of bonding in which an electrostatic force between a cloud of delocalized electrons is attracted to the positively-charged metal atoms.
Metallic Crystals
They have a very high melting point and high boiling point.
They both conduct electric currents and thermal energy (heat).
They are ductile - they are capable of being ‘stretched out’ to form thin wires.
They are malleable - they are capable of being ‘hammered out’ to form thin sheets without breaking.
Properties of Metallic Crystals
the solids in which the particles are not arranged in any specific order or the solids that lack the overall order of a crystal lattice.
The term ‘amorphous’, when broken down into its Greek roots, can be roughly translated to “without form”. Many polymers are amorphous solids. Other examples of such solids include glass, gels, and nanostructured materials.
it is that in which the constituent particles do not possess a regular three-dimensional arrangement.
Amorphous Solids
It lacks the three-dimensional long-range order of a crystalline material, possess a more random arrangement of molecules, exhibit short-range order over a few molecular dimensions, and have physical properties quite different from those of their corresponding crystalline states.
Amorphous Solids
does not have a long-range order of arrangement of their constituent particles. However, they may possess small regions of orderly arrangement. These crystalline parts of an otherwise amorphous solid are known as crystallites.
Lack of Long-Range Order (Properties of Amorphous Solids)
does not have a sharp melting point but melts over a range of temperatures. For example, glass on heating first softens and then melts over a temperature range. Glass, therefore, can be moulded or blown into various shapes. Amorphous solid does not possess the characteristic heat of fusion.
No Sharp Melting Point (Properties of Amorphous Solids)
when heated and then cooled slowly by annealing, becomes crystalline at some temperature. That is why glass objects of ancient time look milky because of some crystallization having taken place.
Conversion into Crystalline Form (Properties of Amorphous Solids)
it can be defined as materials possessing, at minimum, one external dimension measuring 1-100nm. The definition given by the European Commission states that the particle size of at least half of the particles in the number size distribution must measure 100nm or below.
Nanomaterials
Size, shape, specific surface area, aspect ratio
Agglomeration/aggregation state
Size distribution
Structure, including crystallinity and defect structure
Solubility
Physical Properties of Nanomaterials
Structural formula/molecular structure
Composition of nanomaterial (including degree of purity, known impurities or additives)
Phase identity
Surface chemistry (composition, charge, tension, reactive sites, physical structure, photocatalytic properties, zeta potential)
Chemical Properties of Nanomaterials
It brought about a great change in electronic commodities
Blessing for the medical field
Manufacturing advantages
Energy generation
Disease treatment is possible to a great extent
Diagnostic equipment has improved
Advantages of Nanomaterial
Negative Impact on Environment
Unemployment may prevail
Economical imbalance
Hazardous weapons accessible
Costly
Negative impact on people’s health
Disadvantages of Nanomaterial
Nanocomposites: Reinforcing traditional materials like carbon fiber composites with nanoparticles can make aircraft components lighter, stronger, and more resistant to wear and tear.
Example: Boeing's 787 Dreamliner uses carbon nanotubereinforced composites, reducing weight and increasing fuel efficiency.
Lightweight and Strong Structural Components
Aerodynamics: Nanocoating on aircraft surfaces can reduce drag, improving fuel efficiency.
Example: Nanostructured coatings on wings and fuselages reduce friction, making aircraft more aerodynamic.
Enhanced Fuel Efficiency
Nanocoating: Using nanomaterials as coatings on engine components can increase durability and reduce wear and tear, leading to more efficient and longerlasting engines.
Example: Coating turbine blades with nanomaterials improves their performance in high-temperature environments.
Improved Engine Efficiency
Nanosensors: Nanomaterial-based sensors can detect minute changes in aircraft conditions, helping to monitor structural integrity, temperature, and pressure in real-time.
Example: Nanosensors embedded in composite materials can provide early warnings of potential structural issues.
Advanced Sensing and Monitoring
Nanomaterials like carbon nanotubes and graphene are being explored for lightweight and high-capacity energy storage solutions, which can power aircraft systems more efficiently.
Lightweight Energy Storage
Nanomaterials can be used for fire-resistant and self-cleaning interior materials, reducing maintenance and enhancing passenger safety and comfort.
Improved Materials for Cabin and Interior
Anti-icing coatings with nanoparticles can prevent ice buildup on the aircraft's surfaces, reducing the risk of accidents due to ice accumulation.
Enhanced Coatings
Nanomaterials can be used to create sound-absorbing materials that reduce cabin noise and improve the overall flying experience.
Noise Reduction
Nanomaterials can improve fuel combustion efficiency and reduce emissions, contributing to greener aviation. Nanocatalysts in engines can help break down pollutants in exhaust gases.
Environmental Impact Reduction
Nanotechnology-enabled manufacturing techniques can streamline production processes, reduce waste, and improve the precision of aircraft components.
Advanced Manufacturing
refers to the ability of a metal to resist abrasion, penetration, cutting action, or permanent distortion
Hardness
It is the property of a metal which allows little bending or deformation without shattering.
brittleness
it is the property of a metal which permits it to permanently drawn, bent, or twisted into various shapes without breaking.
ductility
is that property which enables a metal to return to its original shape when the force which causes the change in shape is removed.
elasticity
It will withstand tearing or shearing and may be stretched or otherwise deformed without breaking.
toughness
It is the weight of a unit volume of a material
density
It is the ability of a metal to become liquid by the application of heat.
fusibility
It is the property which enables a metal to carry heat or electricity
conductivity
are reactions produced in metals as the result of heating or cooling
Contraction and expansion
its resistance to a force which tends to pull it apart.
Tensile strength
its resistance to a crushing force which is the opposite of tensile strength
Compression strength
It is the tendency on the part of parallel members to slide in opposite directions.
shear
It is described as the deflection or curving of a member due to forces acting upon it.
bending
It is a twisting force
torsion
It is the eating away or pitting of the surface or the internal structure of metals.
Corrosion
The term “ferrous” applies to the group of metals having iron as their principal constituent.
Iron is the base metal
Approximately 1% of carbon is added to iron
Carbon Steel forms the base of those alloy steels produced by combining carbon steel with other elements known to improve the properties of steel
A base metal (such as iron) to which small quantities of other metals have been added is called an alloy
Ferrous Metals
Society of Automotive Engineers
SAE
American Iron and Steel Institute
AISI
is used to designate the plain carbon and alloy steels
Four-numeral series
are used to designate certain types of alloy steels
five numerals
first two digits indicate the type of steel
second digit also generally (but not always) gives the approximate amount of the major alloying element
the last two (or three) digits are intended to indicate the approximate middle of the carbon range

The term “nonferrous” refers to all metals which have elements other than iron as their base or principal constituent.
“aluminum or alloys are the best example of nonferrous metals.
Aluminum melts at the comparatively low temperature of 1,250 °F.
Non-Ferrous Metals
Formed by physically working the metal into shape
Wrought
1xxx Pure Aluminum
2xxx Copper
3xxx Manganese
4xxx Silicon
5xxx Magnesium
6xxx Magnesium and Silicon
7xxx Zinc
8xxx Other Element
Alloying Agents (Wrought)
The first digit of a designation identifies the alloy type.
The second digit indicates specific alloy modifications.
The last two digits of the 1xxx group are used to indicate the hundredths of 1 percent above the original 99 percent designated by the first digit
Non-Ferrous Metals
Formed by pouring molten metal into molds
Cast
1xxx Pure Aluminum
2xxx Copper
3xxx Silicon and Copper
4xxx Silicon
5xxx Magnesium
6xxx Unused
7xxx Zinc
8xxx Tin
9xxx Other Elements
Alloying Agents (Cast)
It occurs when a metal such as aluminum is exposed to a gas containing oxygen
OXIDATION CORROSION
It is caused by the reaction of metallic surfaces with atmospheric contaminants
UNIFORM SURFACE CORROSION
It is A likely result of uniform surface corrosion left untreated
Pitting corrosion
It Occurs any time two dissimilar metals make electrical contact in the presence of an electrolyte
Galvanic corrosion
It can occur in humid conditions and is often initiated in a defect or a local damage in the coating.
Filiform corrosion
It is An attack along the grain boundaries of a material
Intergranular corrosion
It is An extreme case of intergranular corrosion
Exfoliation corrosion
It Occurs when metal is subjected to a tensile stress in the presence of a corrosive environment
Stress corrosion
metal deterioration caused by repetitive slope at the interface between two surfaces in contact that were not intended to move in that fashion.
fretting corrosion
It is a large molecule or a macromolecule, which essentially is a combination of many subunits. The term polymer in Greek means ‘many parts’
It can be found all around us, from the strand of our DNA, which is a naturally occurring biopolymer, to polypropylene which is used throughout the world as plastic.
polymers
They occur naturally and are found in plants and animals.
Natural Polymers
They are derived from naturally occurring polymers and undergo further chemical modification.
Semi-synthetic Polymers
These are human-made polymers. Plastic is the most common and widely used synthetic polymer.
Synthetic Polymers
The structure of polymers containing long and straight chains falls into this category.
Linear Polymers
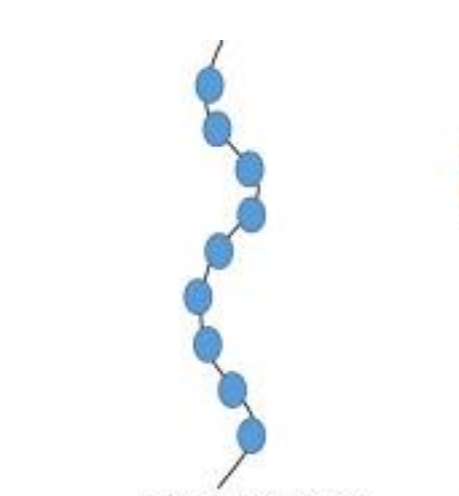
When linear chains of a polymer form branches, then such polymers are categorized as branched chain polymers.
Branched-chain Polymers
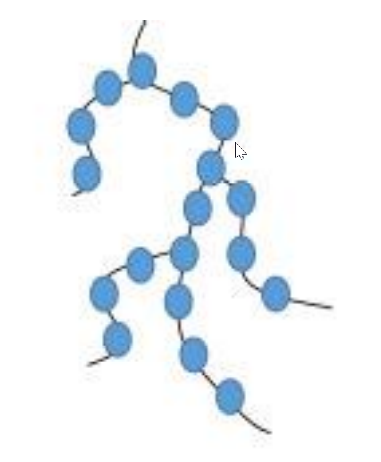
They are composed of bifunctional and trifunctional monomers. They have a stronger covalent bond in comparison to other linear polymers.
Cross-linked Polymers
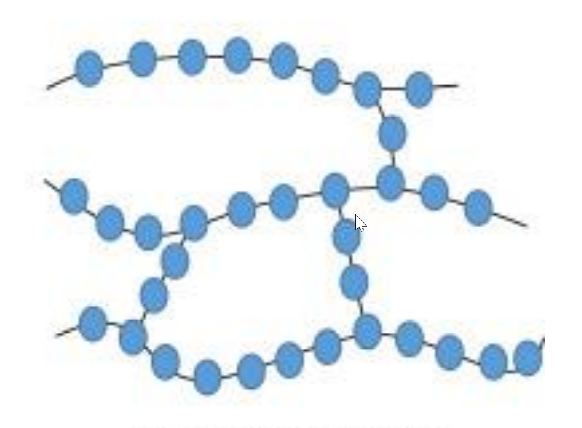
Polymers are commonly used in composite materials, such as carbon fiber-reinforced polymers (CFRP) and glass fiber-reinforced polymers (GFRP). These composites are used in the construction of aircraft structures, including wings, fuselages, and tail sections, to reduce weight while maintaining strength and durability.
Composite materials
Polymers are used in the interior of aircraft for various components, including seats, cabin walls, overhead bins, and tray tables. They offer a balance between weight savings and aesthetics.
Interior components
Polymers are used as sealants and adhesives in aircraft manufacturing and maintenance. They help join various components, create airtight seals, and prevent leaks. These adhesives are critical for composite structures.
Sealants and adhesives
Polymers are used as wire insulation and cable jackets to protect electrical systems in the aircraft. They offer lightweight and electrically insulating properties while being resistant to harsh environmental conditions.
Wiring and insulation