The mole and a bit of nuclear physics ahhhhhhhhhhhhhhhhhhhhh
1/51
Earn XP
Description and Tags
ahhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhh
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
1 Angström
10^-10 m
Diameter of an atom:
Diameter of an atom: ≈ 60 – 550 pm (1 pm ≙10-12 m) ≙ 0.60 – 5.50 Å
atomic number (Z)
Thus the number of protons is also called atomic number (Z) (Ordnungszahl, OZ).
If necessary (which is rare in chemistry, because element symbols are unambiguous), it is stated as a subscript to the left of the element symbol:
• Every carbon atom has 6 protons in the nucleus: 6C
• Every oxygen atom has 8 protons in the nucleus: 8O
isotopes. •There are ____ different isotopes for hydrogen. • There are ____ isotopes of helium etc
Atoms of the same kind (same atomic number) but a different number of neutrons are called

Different isotopes have different _____.
Different isotopes have different mass numbers (A). The mass number (Massenzahl) is the number of protons (= Z) and protons (P) present in the respective nucleus: A = Z + #N (The mass of the electrons is so low it can be ignored.) Protons and neutrons are often grouped as nucleons, so: A = number of nucleons.

The atomic mass is stated relative to the ___ isotope ___:
The atomic mass is stated relative to the carbon isotope 12C: if A = 12, and Z(C) = 6, #N = 6. The atomic mass of one atom of 12C is defined to be
The atomic mass of one atom of 12C is defined to be
M(12C):=12.000 000 atomic mass units (a.m.u.), often abbreviated amu or just u
1 u =
1.660 54 ∙ 10-24 g
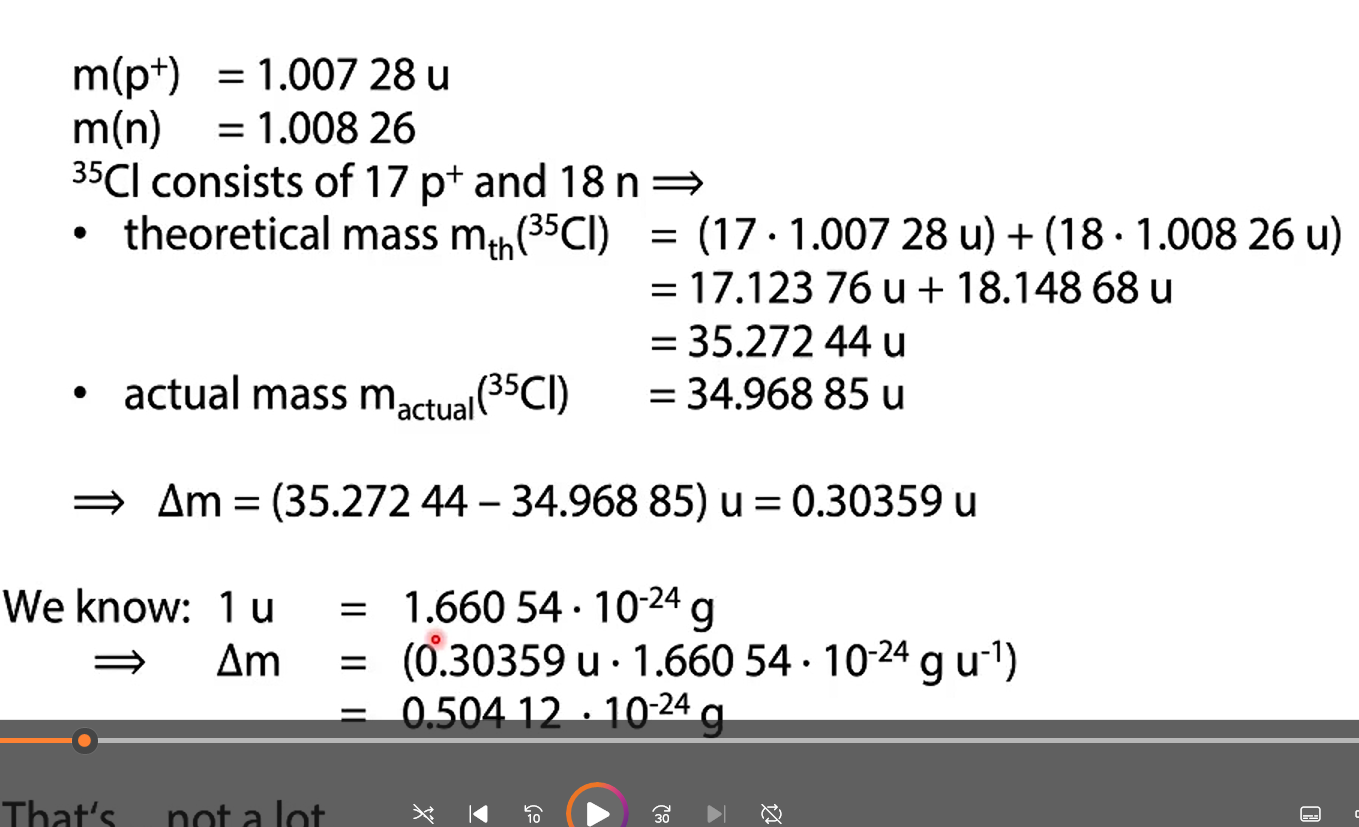
Mass numbers (obviously) must always be integer. The actual mass of single protons and neutrons in fact are not integer: m(p+) = m(n) =
Mass numbers (obviously) must always be integer. The actual mass of single protons and neutrons in fact are not integer: m(p+) = 1.00728 u m(n) = 1.00866 u
relative atomic mass
Relative atomic mass (symbol: Ar) is defined as the ratio of the average mass of atoms of a chemical element in a given sample
mass defect
Mass defect occurs because the mass of a nucleus is always less than the sum of the masses of its constituent protons and neutrons. This difference arises because some of the mass is converted into binding energy, which holds the nucleus together

calculate 35C theoretical mass and mass defect in gram
with the actual mass of 35,27244
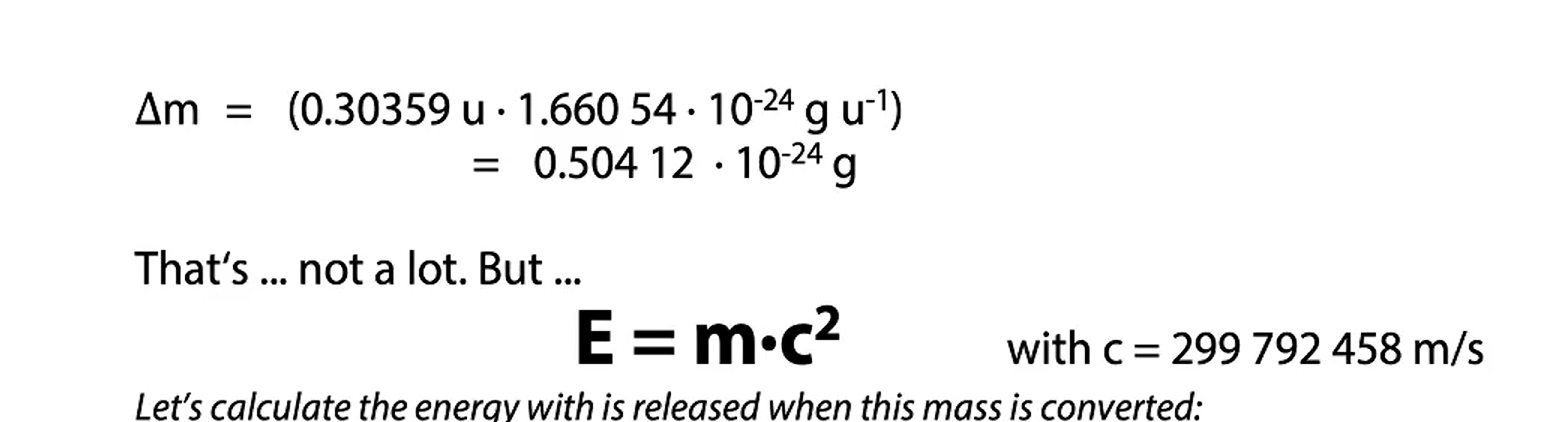
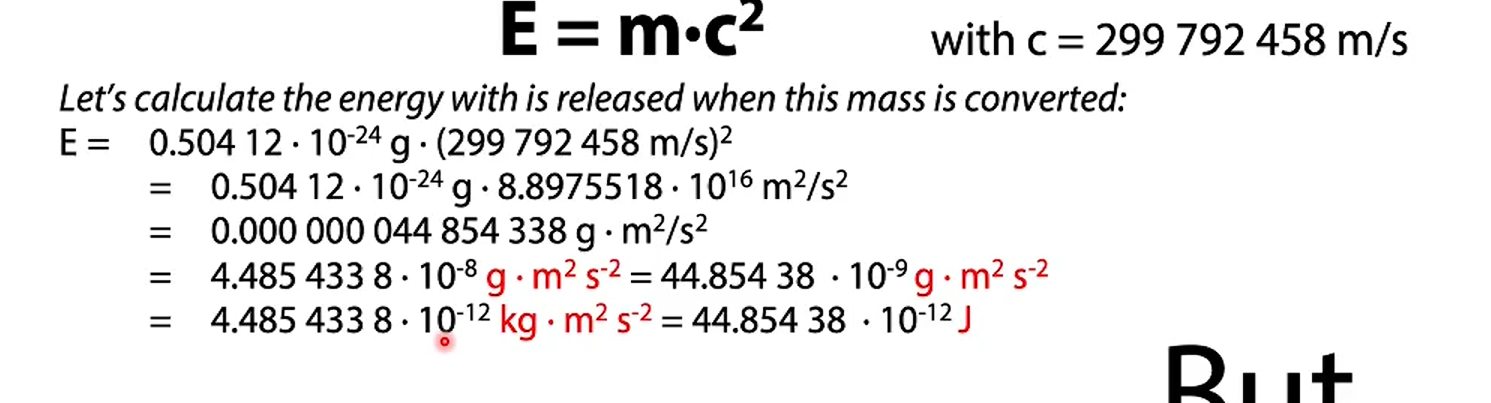
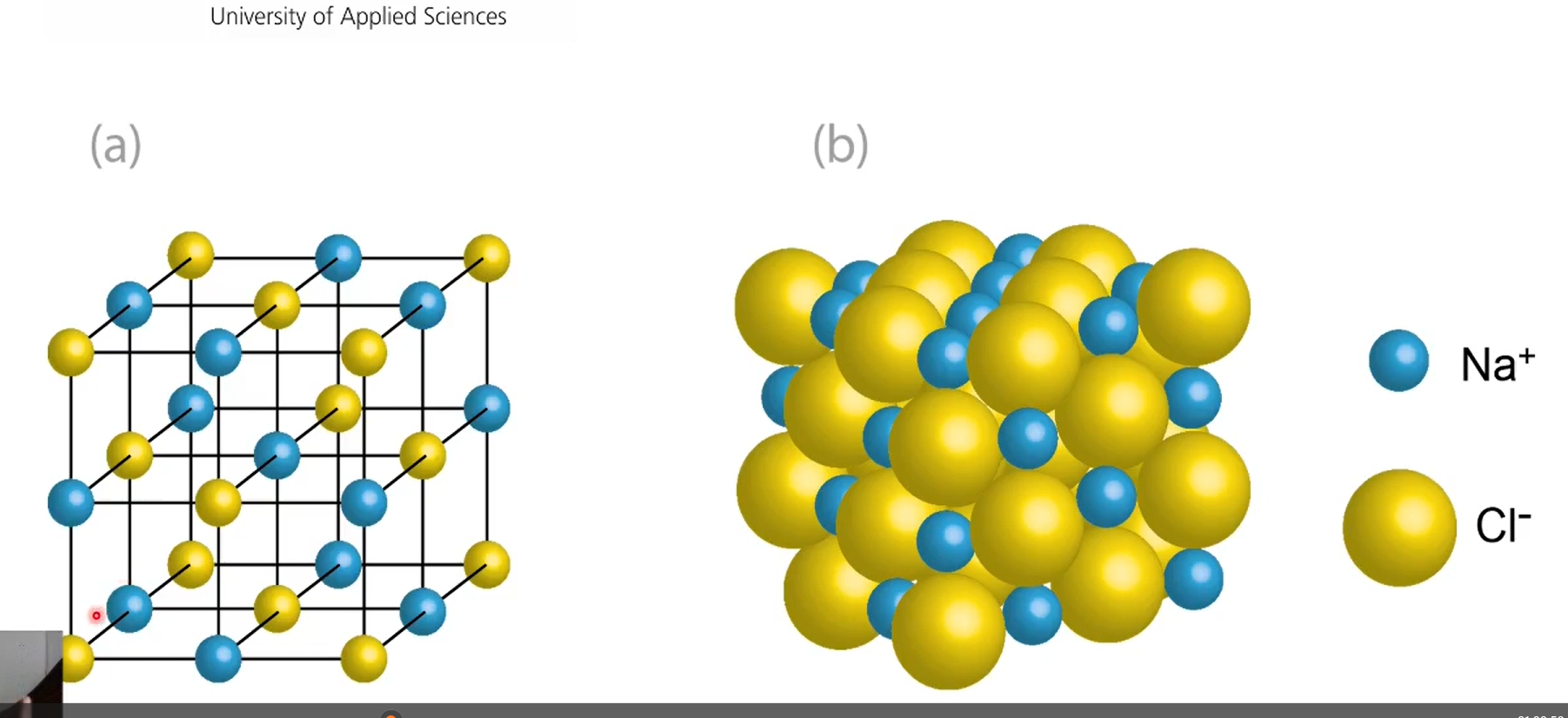
what does octahedrally surrounded mean?
A central atom (often a metal ion) is surrounded by 6 atoms or ligands, arranged at the corners of an octahedron.
Octahedron = a shape with 8 triangular faces
The 6 surrounding atoms are positioned symmetrically around the central atom
Ex: a chlorine atom is octahedrally surrounded by 6 sodium ions
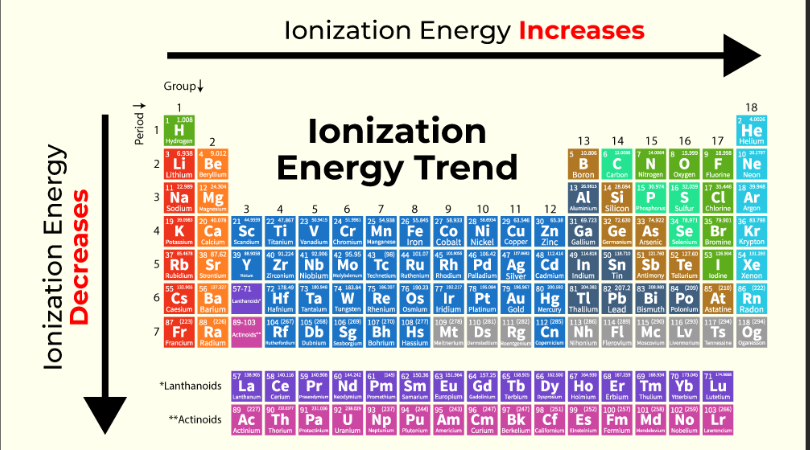
ionization energy in the periodic table
the nobel gases have the highest ionization energy
which are the electrons most easily removed?
valence electrons
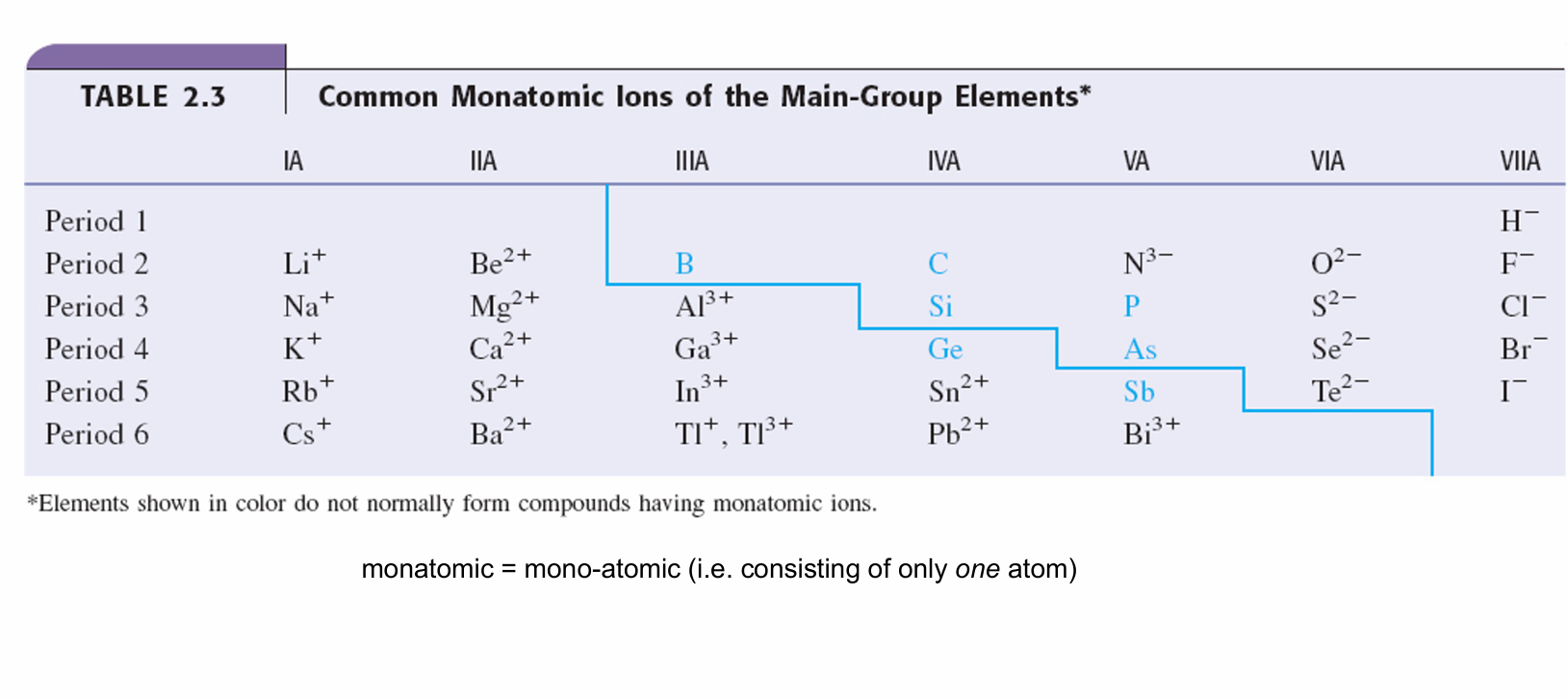
monatomic atoms and their charge
lattice energy
What would have a higher lattice energy NaCl or RbCl
MgF2 than NaCl?
There is an optimal distance between the cations and anions attracting each other
Energy necassary to pull ions apart in a lattice
NaCl since Na^+ is smaller than Cl^- so higher charge density more lattice energy required to pull apart
MgF2 because F^- is smaller than Cl and Mg²+ has a larger charge
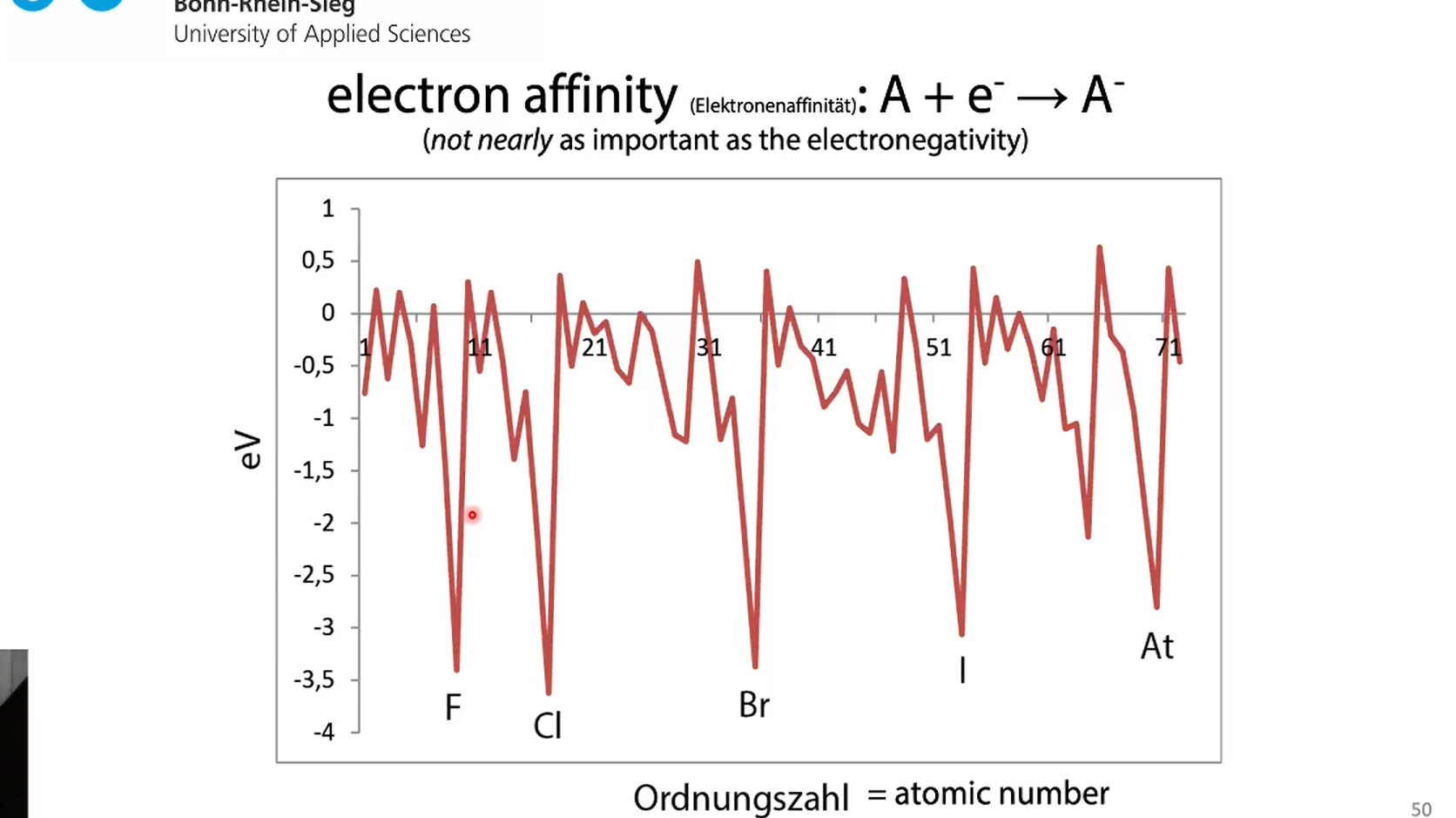
electron affinity
=) energy to force an atom to pick up a electron
He is high because 1s2 so it needs to open a 2s orbital
nobal gases dont want a electron
Nitrogen has three p orbitals with positive spin each (energetically favorable) so adding one is not optimal
thats also why Carbon has such a low electron affinity
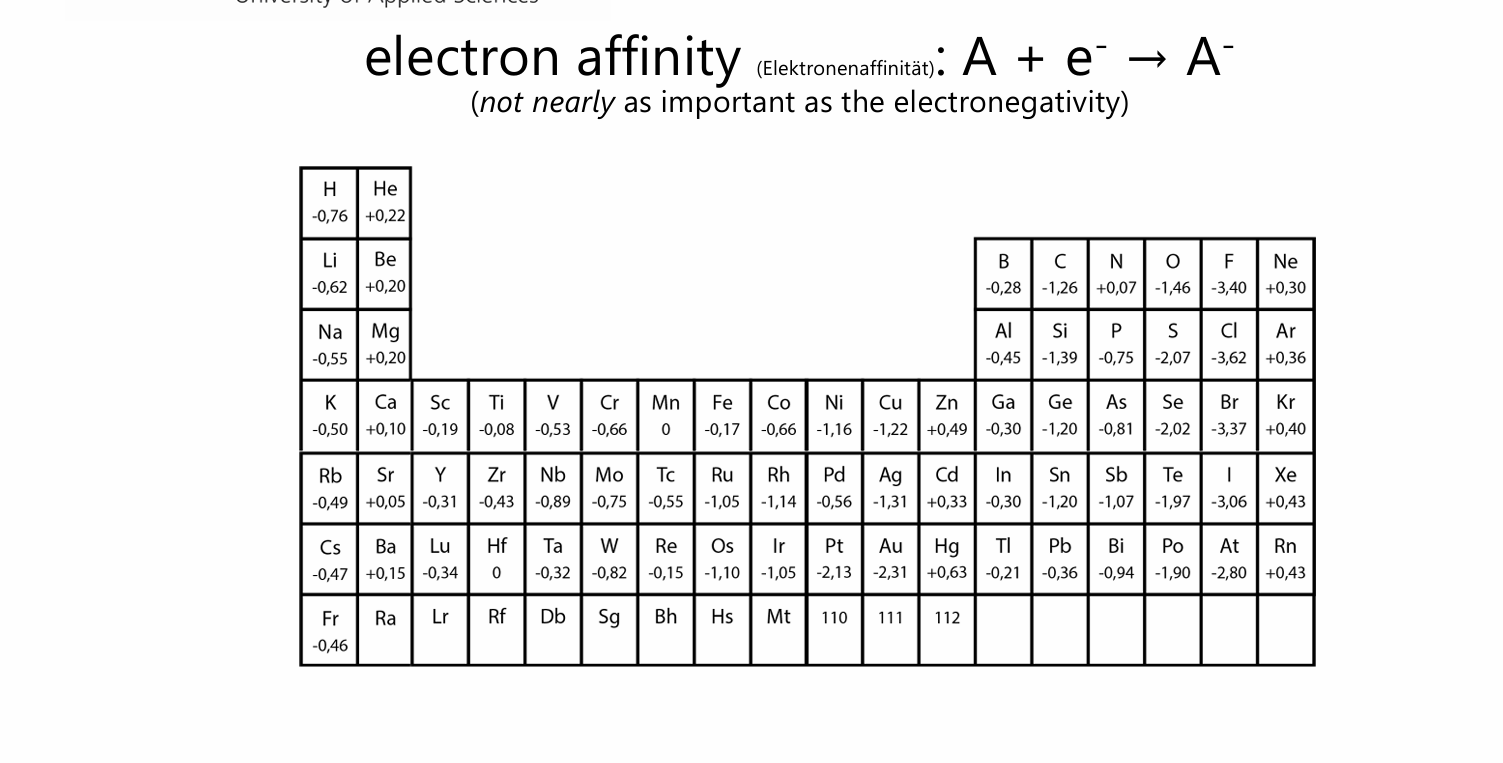
what group has the lowest electron affinity
what is a radical?
A radical in chemistry is an atom, molecule, or ion that has at least one unpaired valence electron, making it highly reactive
draw as a dot
two lone electrons diradikal
what is a O=O bond?
explosive
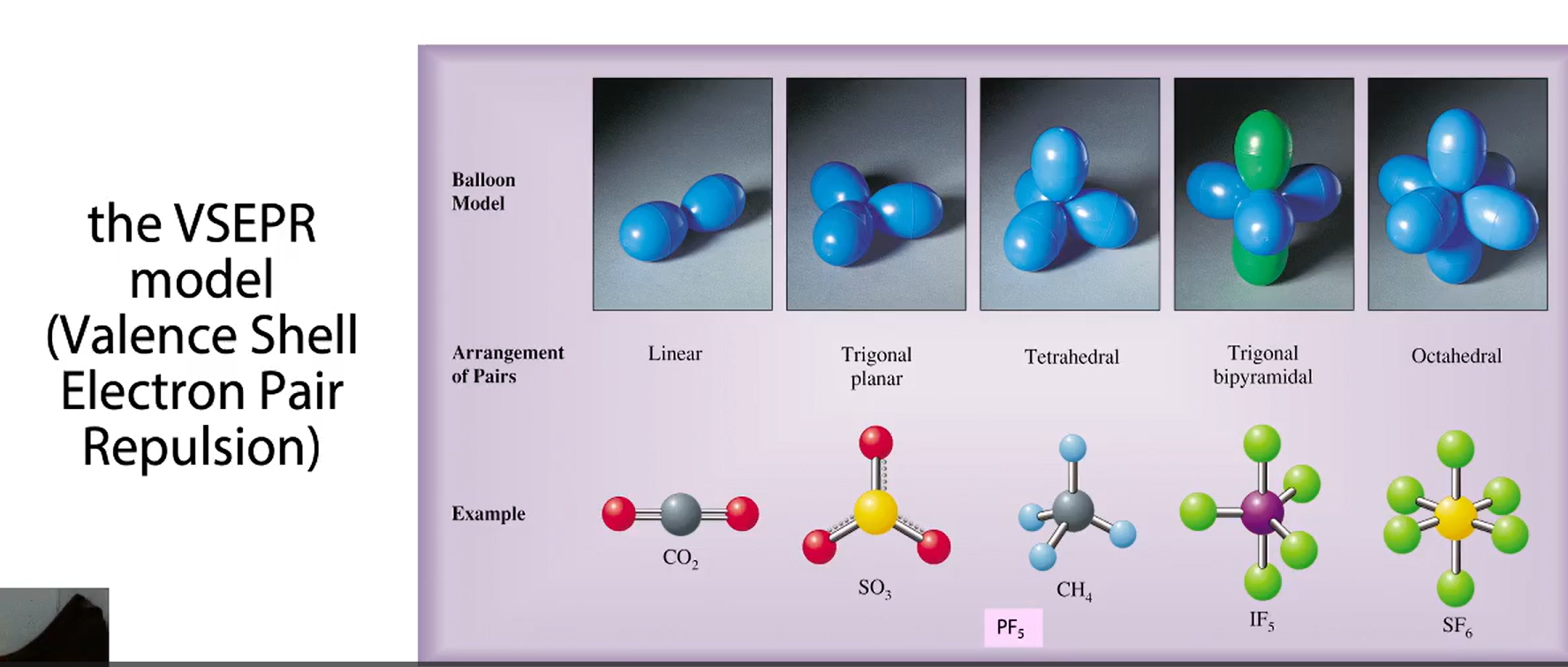
the VESPER model
2 binding partners, 3 binding partners, 4 binding partners + the degrees
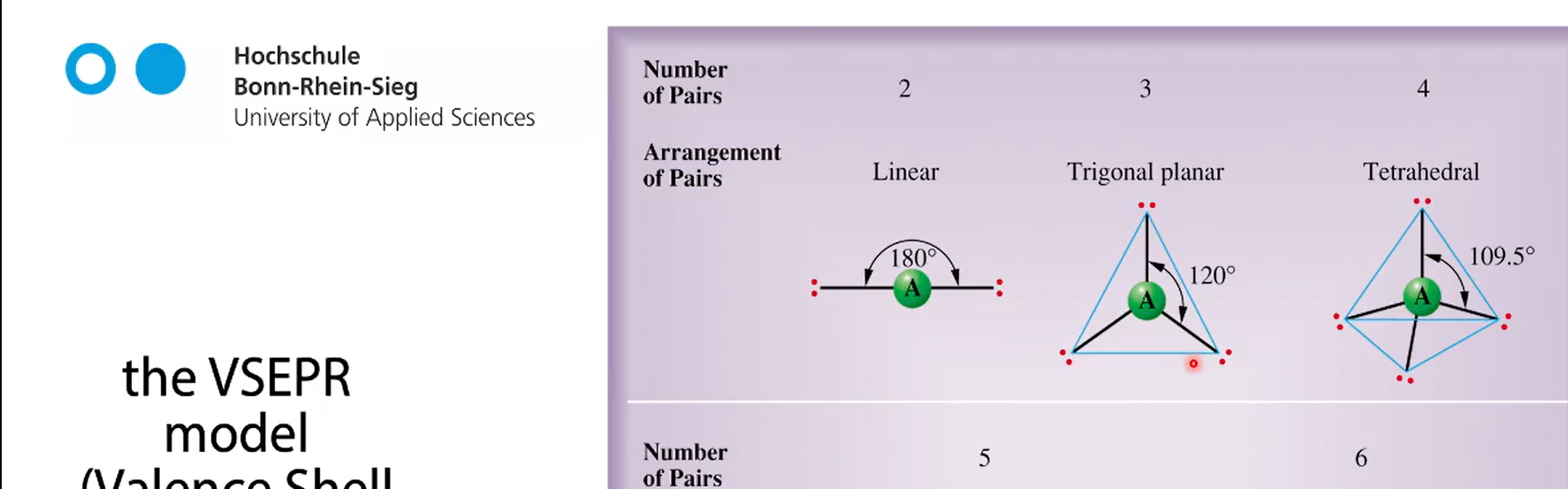
5 binding partners + degree
6 binding partners + degree
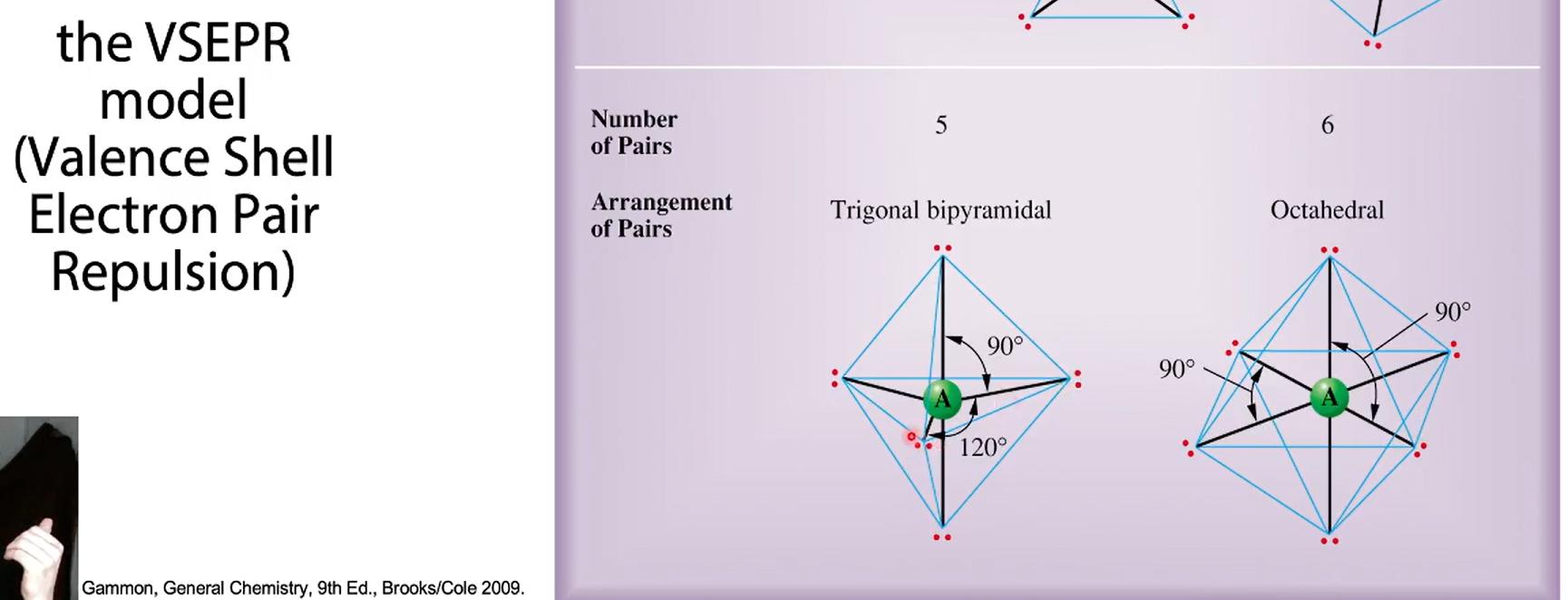
name in example for binding partners 2-6
sulfur trioxide
for n=3,4 with lone pairs
what geometrical from would sulfur dioxide take? what about ammonia? what about H2O?
angular
trigonal pyramidal
angular

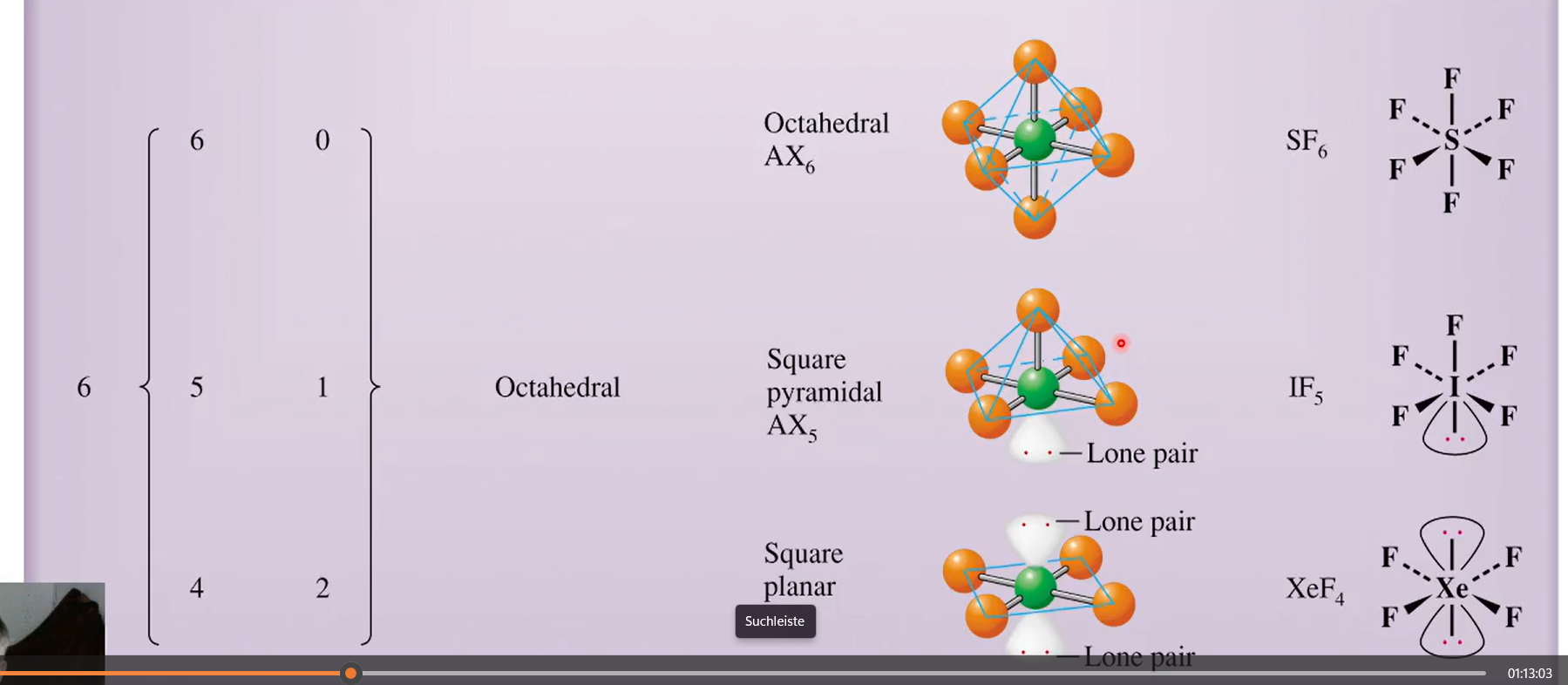
for n=6 with lone pairs
what about SF6 vs IF5. what about XeF4?
octahedral
square pyramidal
square pyramidal
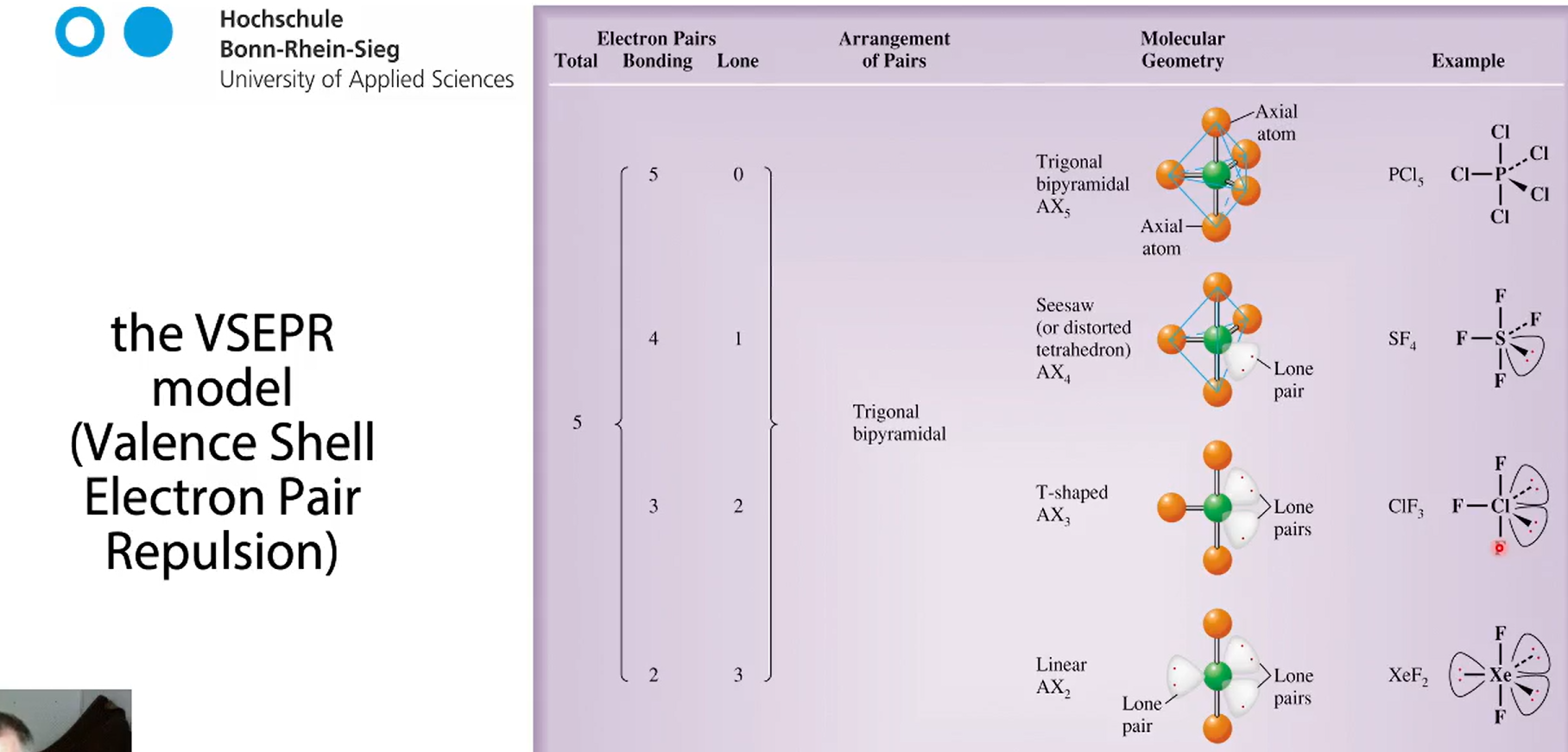
n=5 with lone pairs PCl, ClF3, SF4
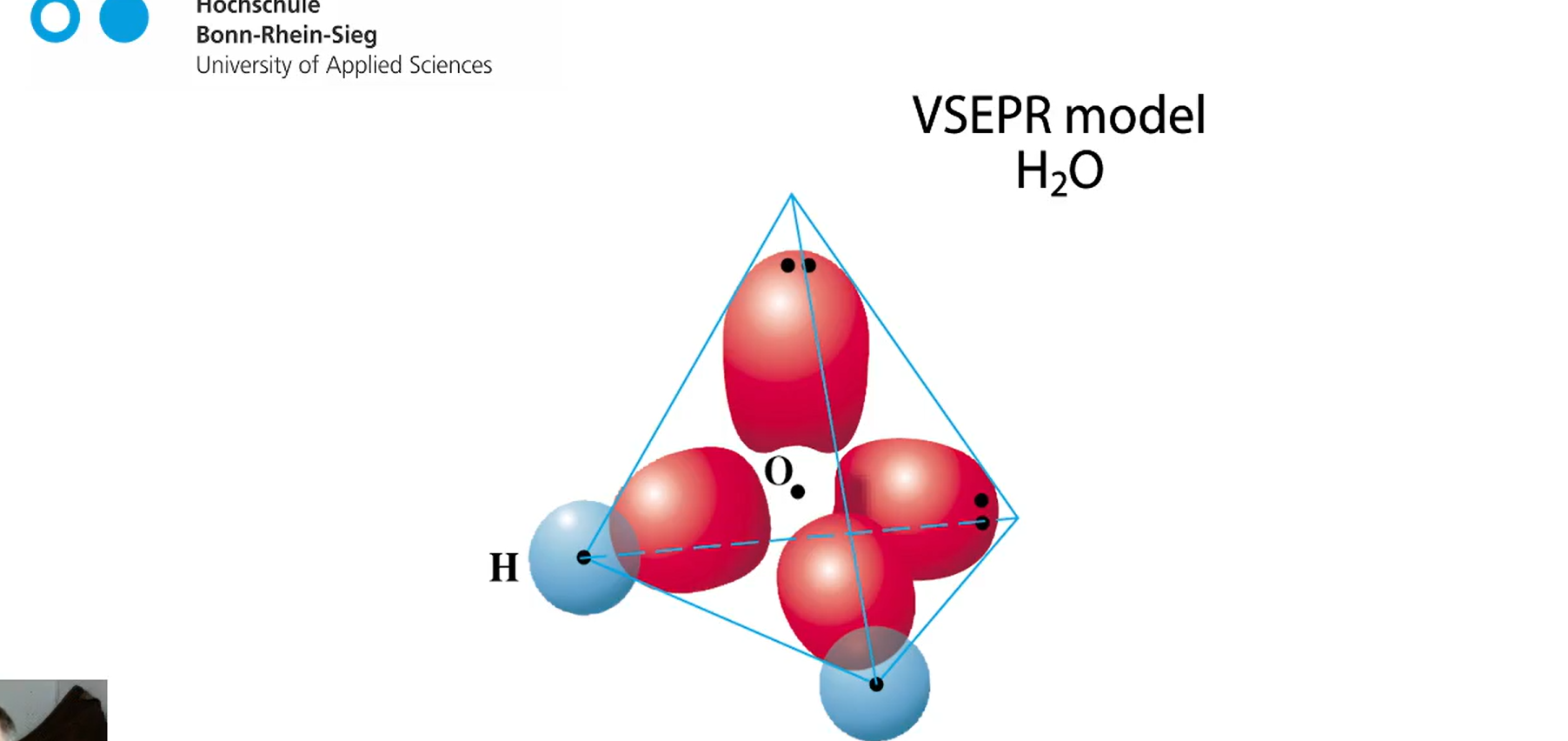
VESPER model of H2O
its angular of trigonal planar
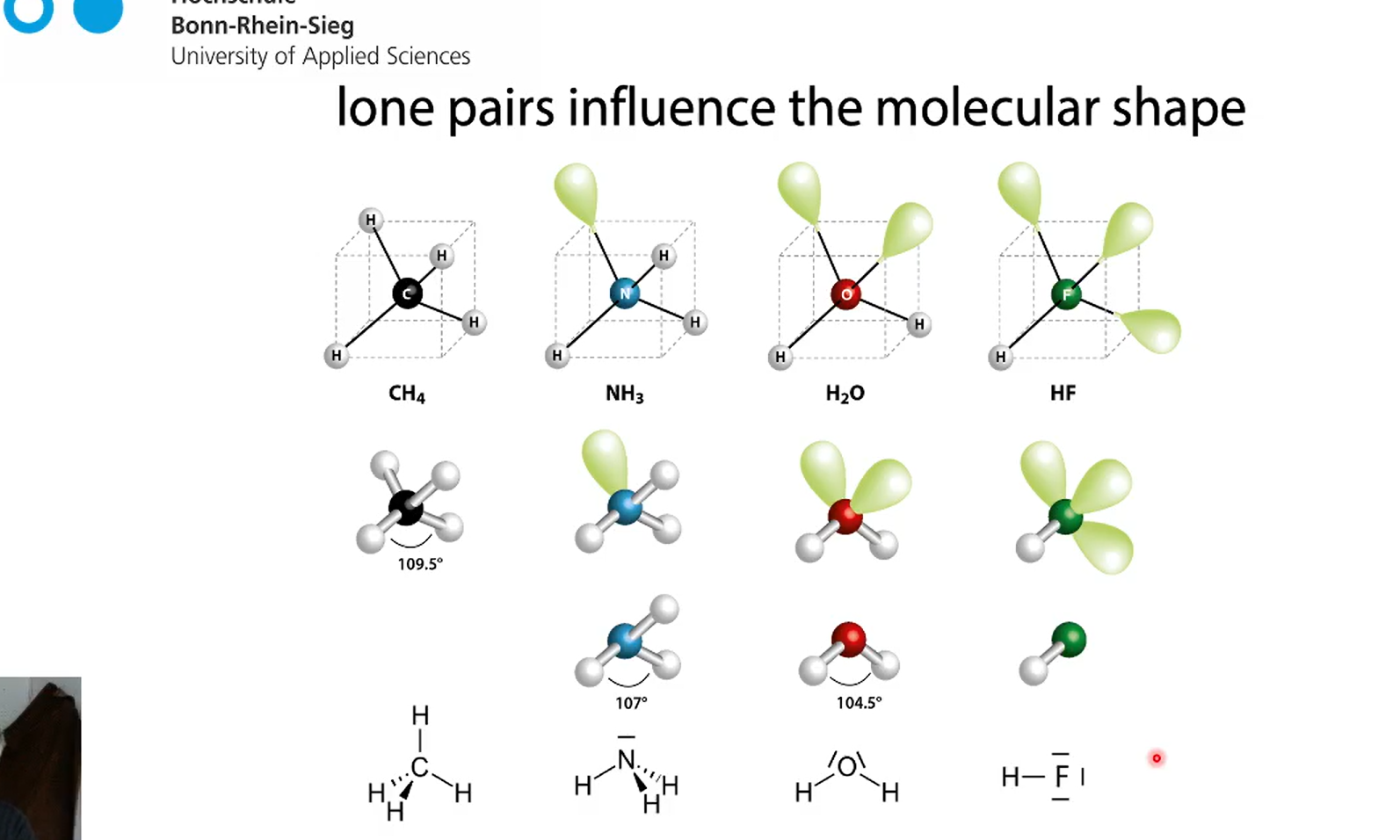
CH4, NH3, H2O and HF
we start at tetrahedal
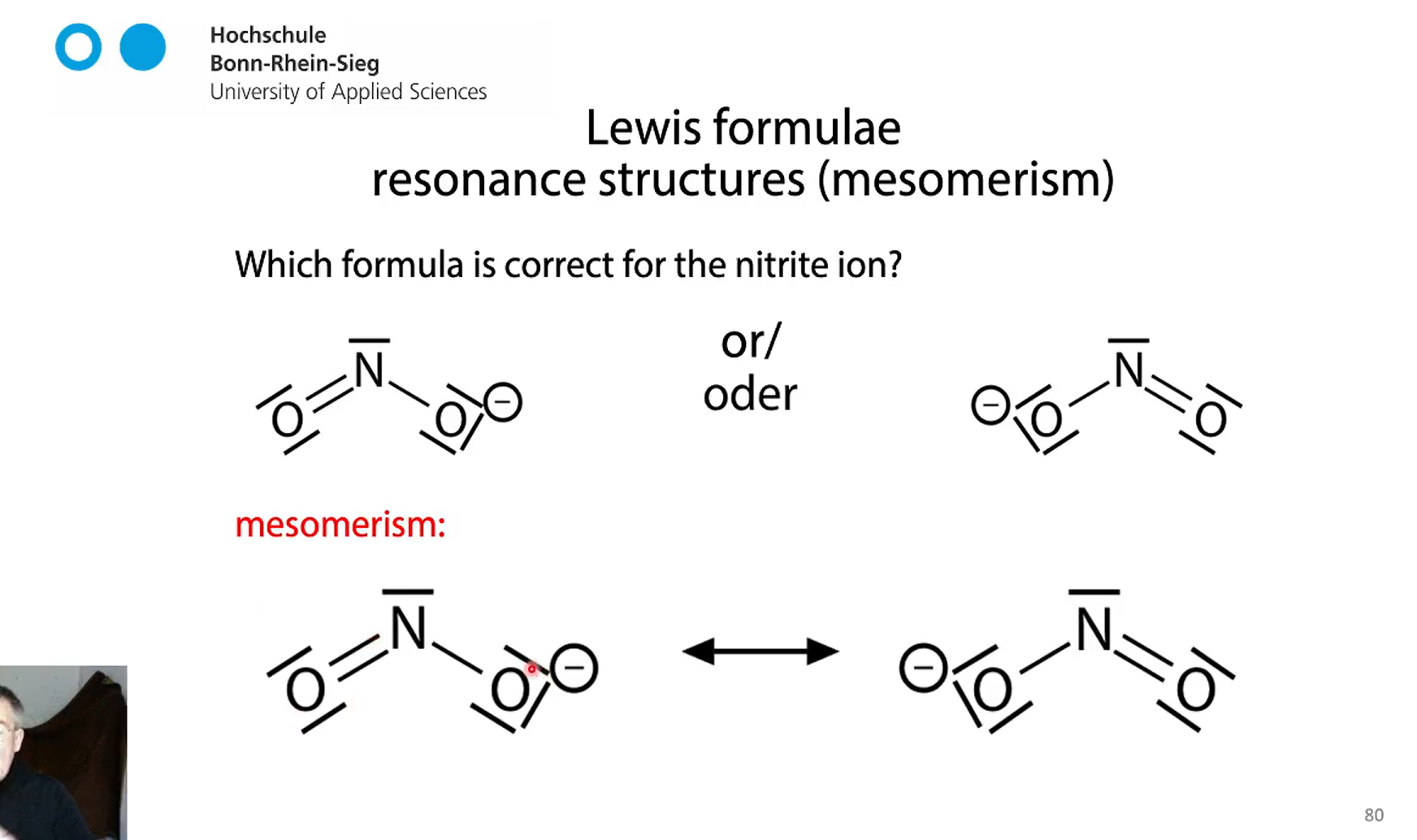
lewis formula nitrite ion

mesomerism
negative charge is equally distributed between two oxygen atoms. NOT a dynamic process the actual nitrite formula is somewhere in the middle but we have no lewis fromula for that
both descriptions are right
which rings are chemically unfavorable?
3 or 4 membered rings

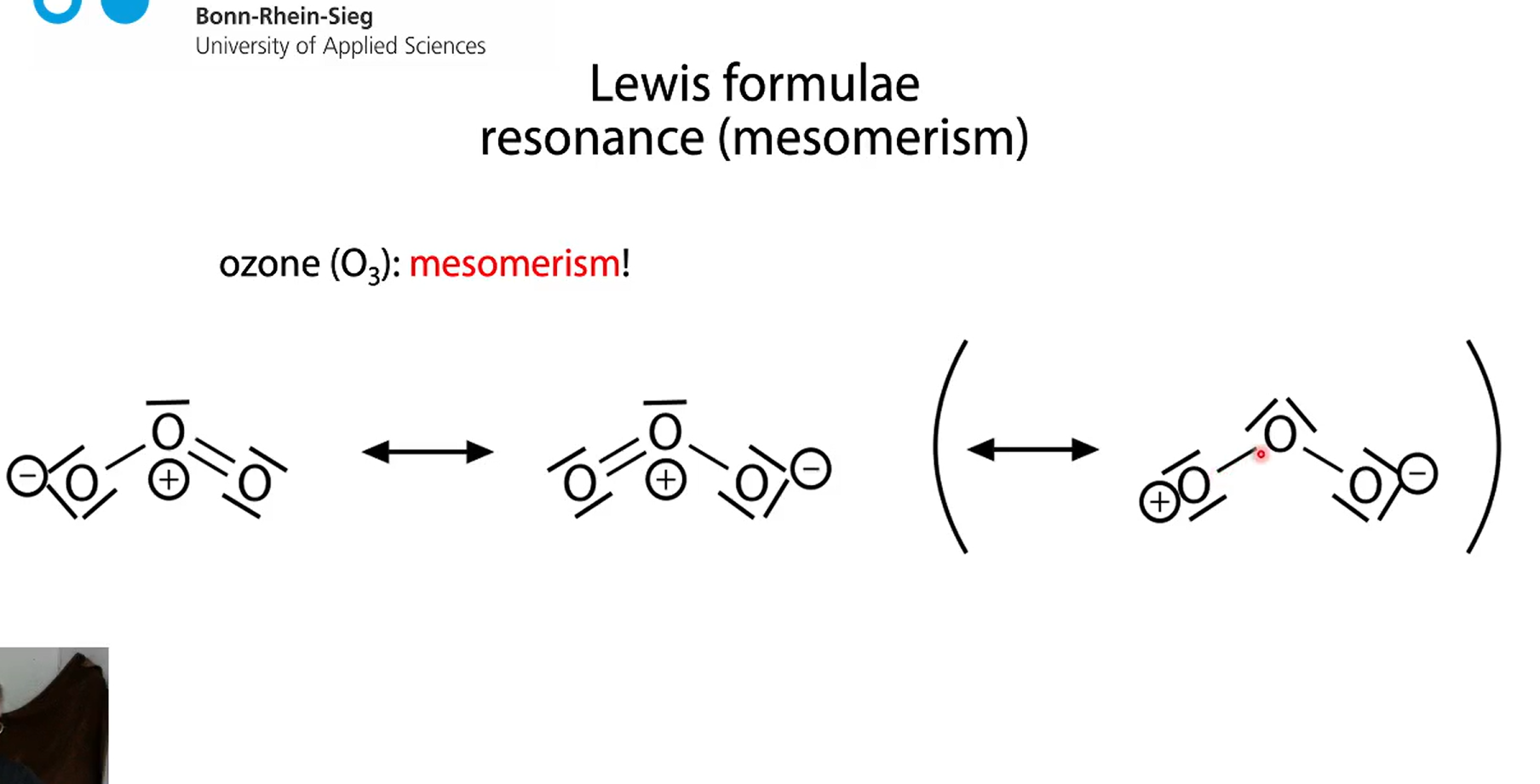
ozone O3
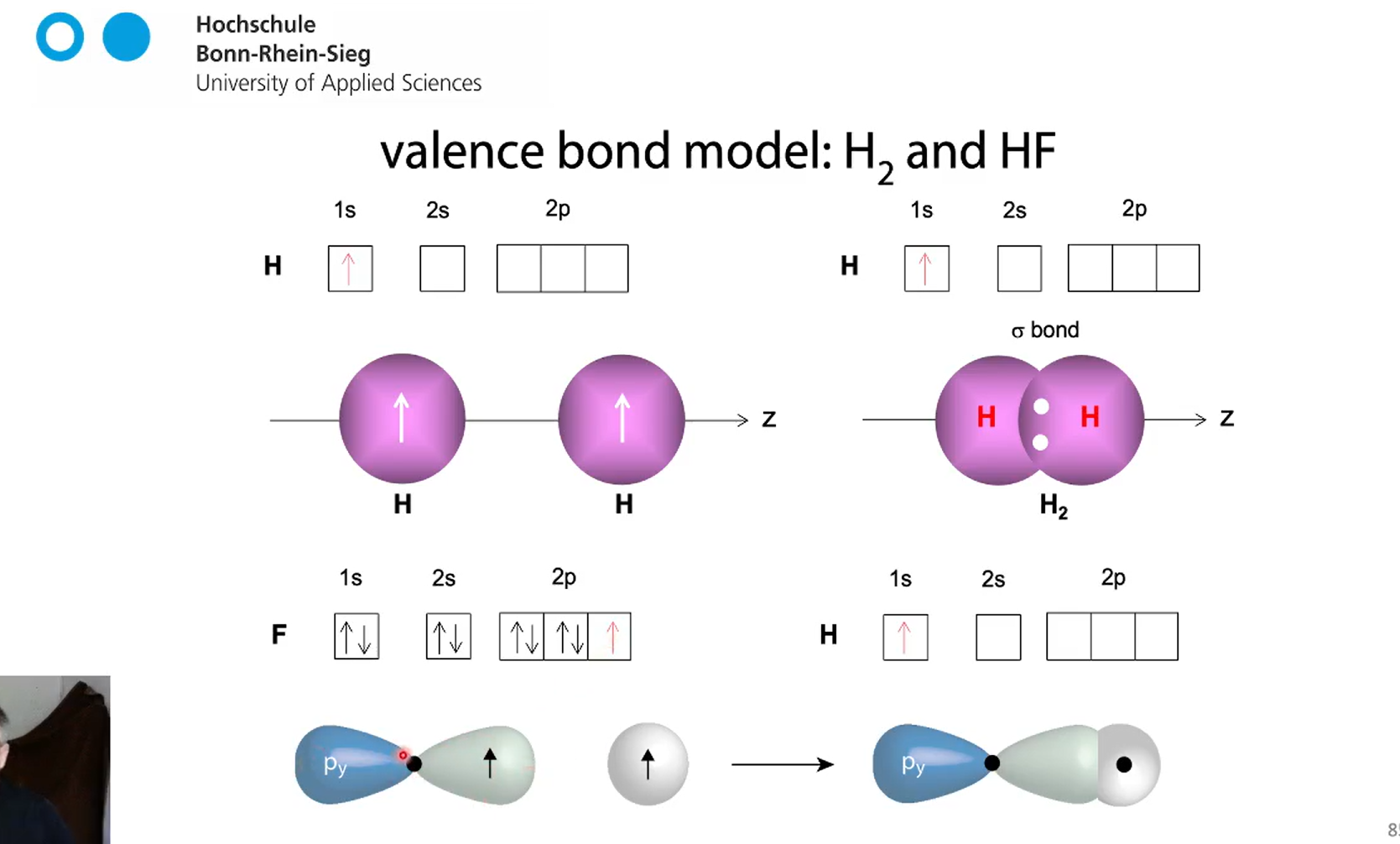
what are sigma bonds and when do they from?
A sigma (σ) bond is the strongest type of covalent bond, formed by the head-on overlap of atomic orbitals along the internuclear axis, with electron density concentrated between the nuclei.
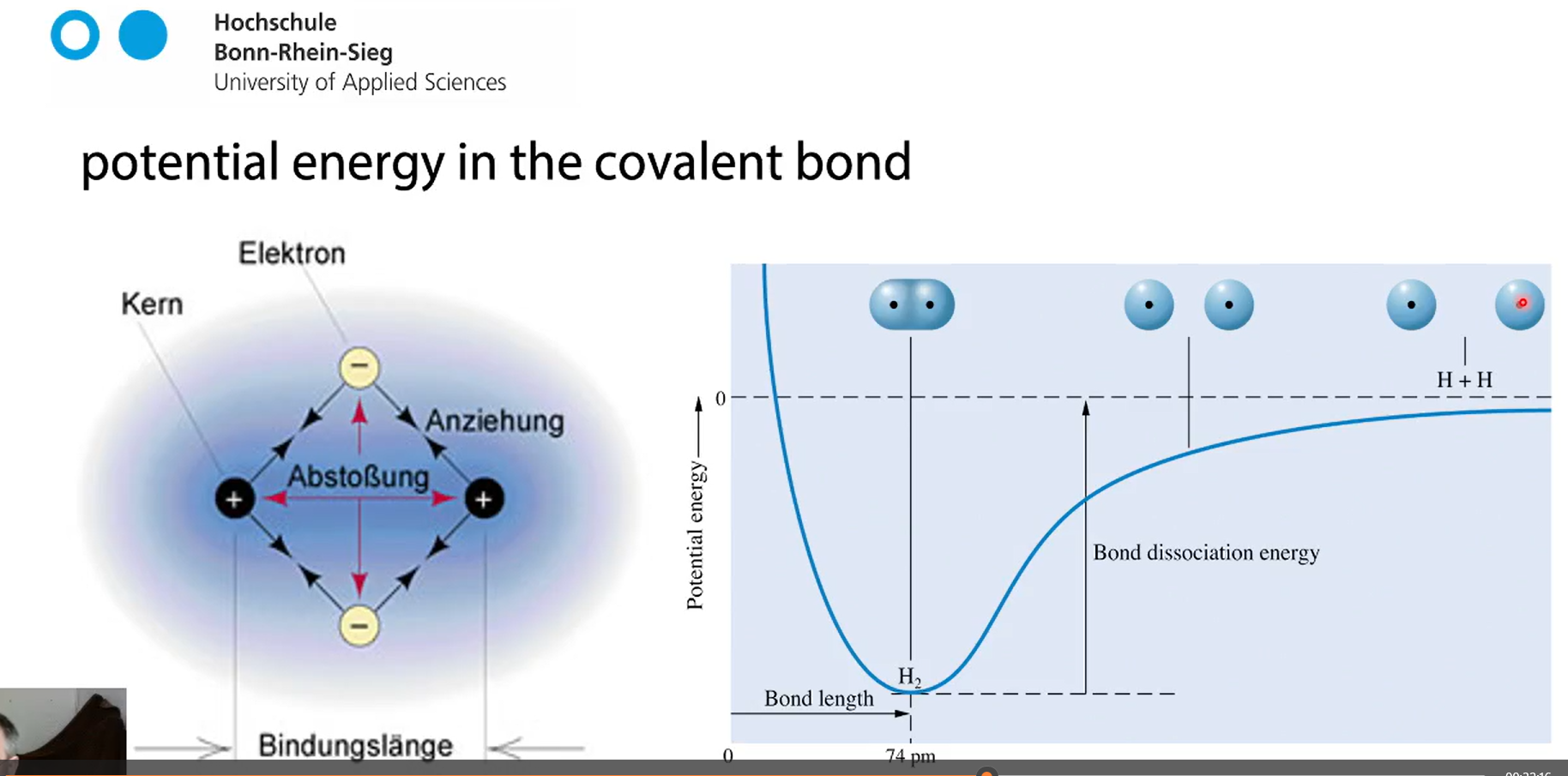
bond lenght
ideal distance between the atoms
the closer they are the lower the energy,
if you push them closer together the repulsion of the nuclei kicks in which raises the energy
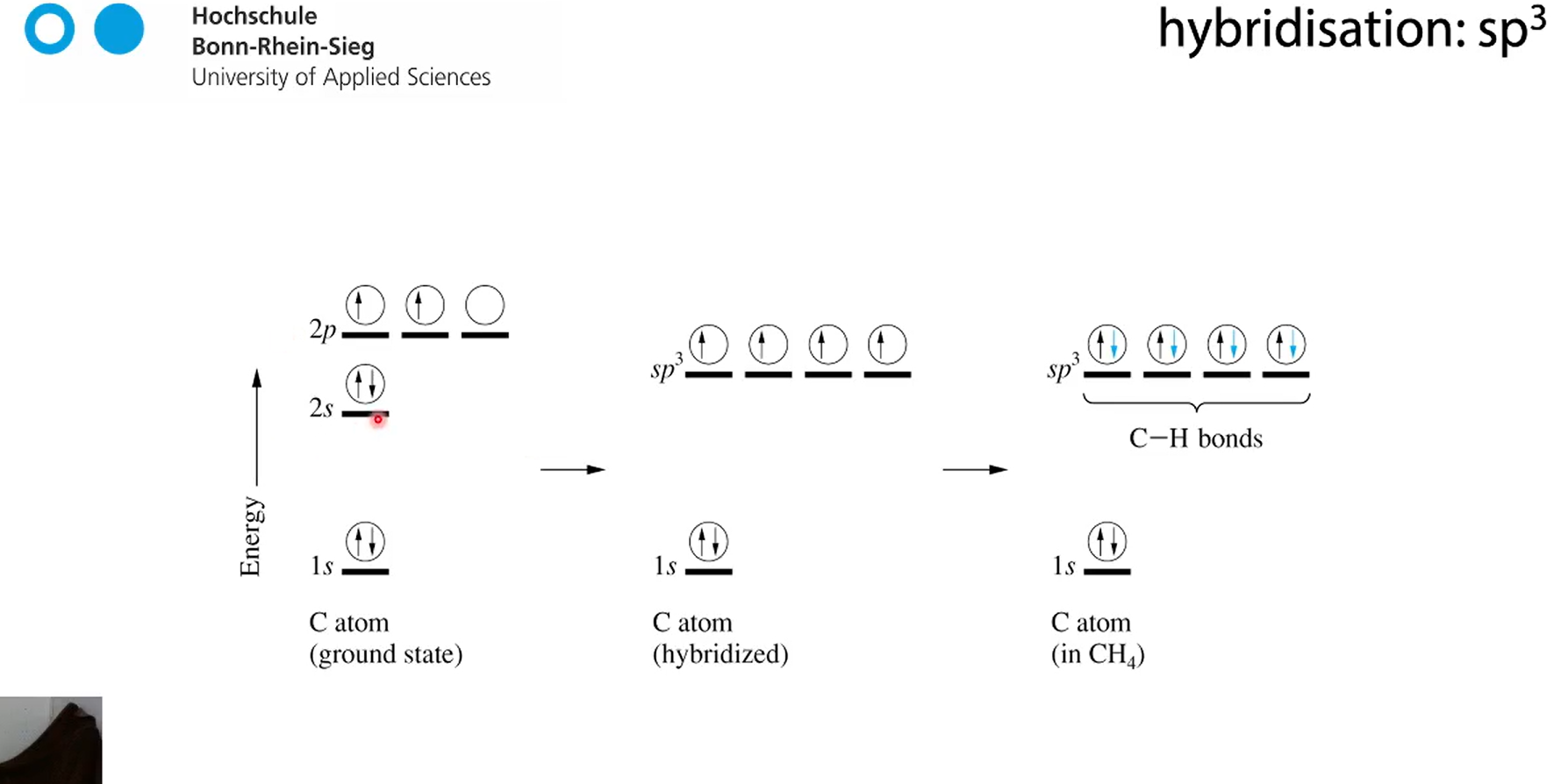
hybridisation
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals

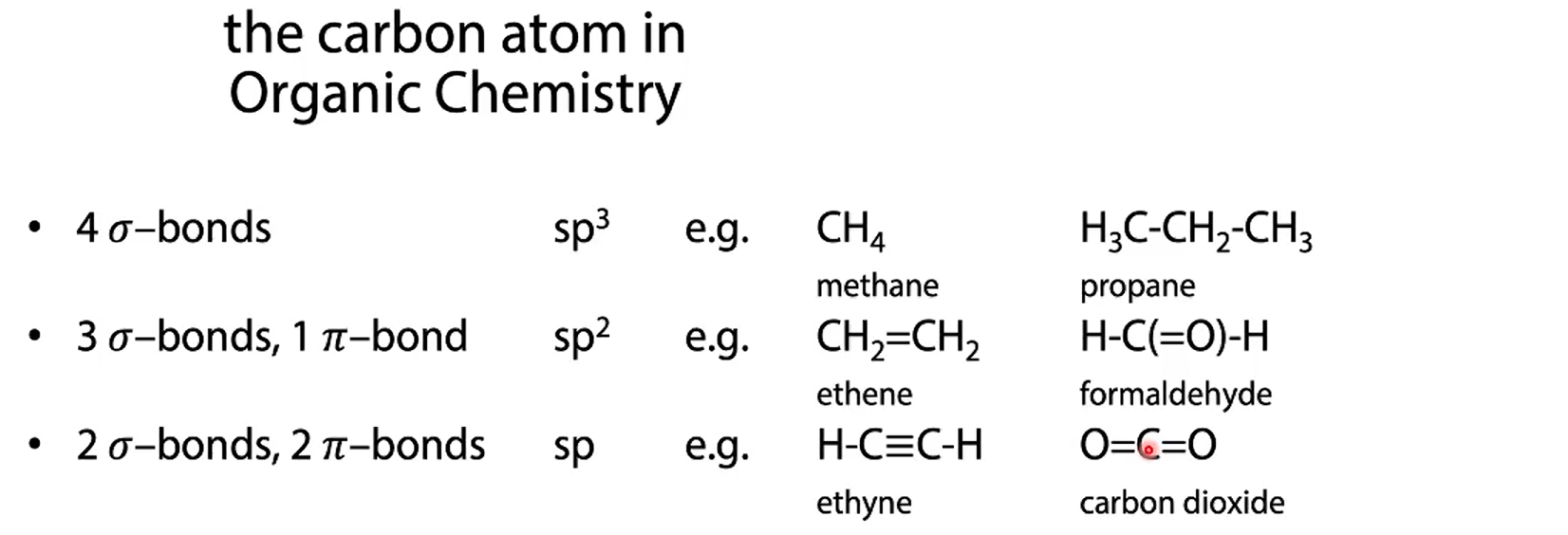
how do we make carbon have 4 bindings with hybridisation?
so now C can form 4 bonds with hydrogen because binding pairs are disrubted now sp3 orbitals
sp3 orbitals are now degenerate
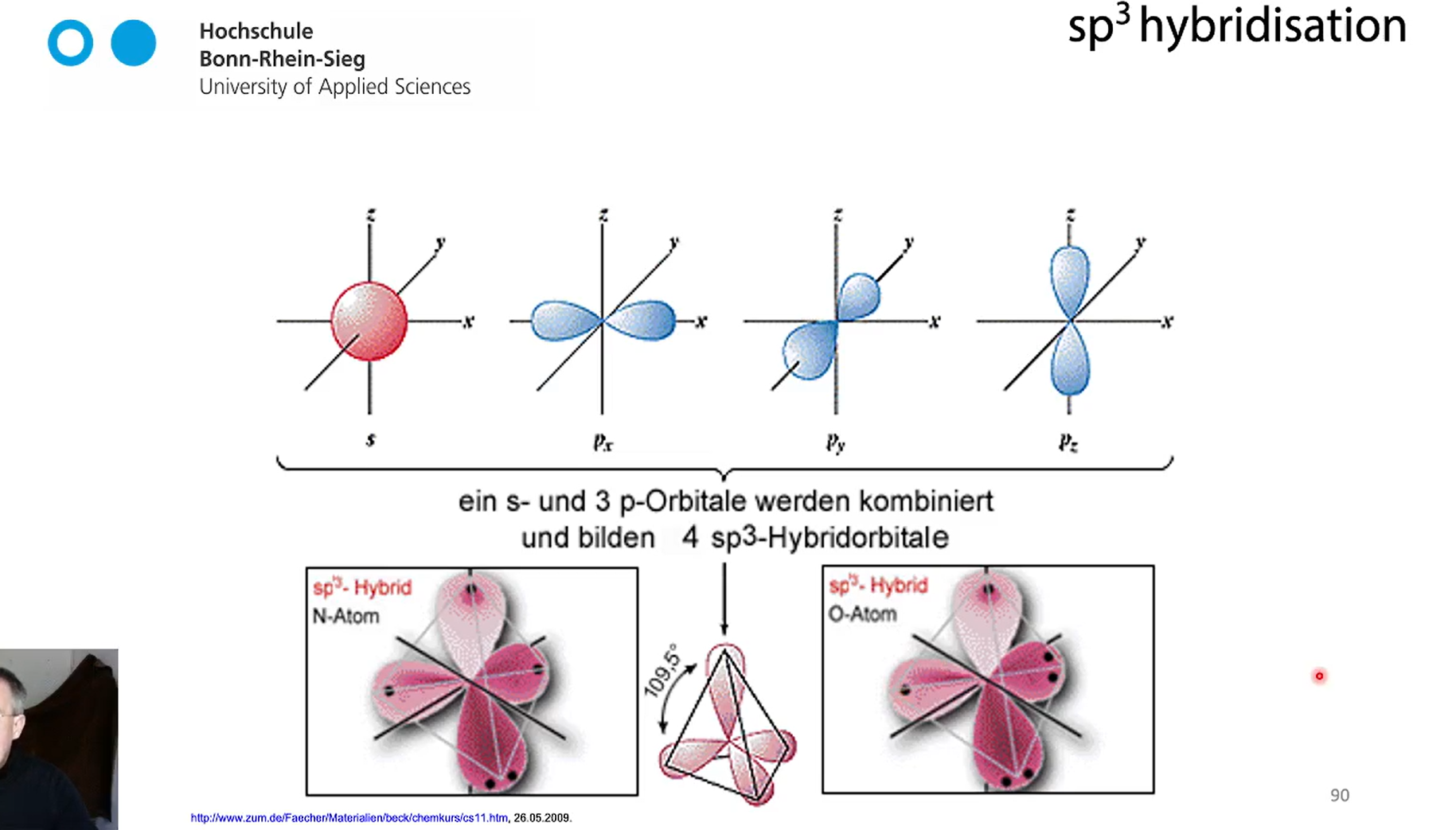
hybridisation of N atom and O atom
shape of a single sp3 orbital vs 4 sp3 orbitals
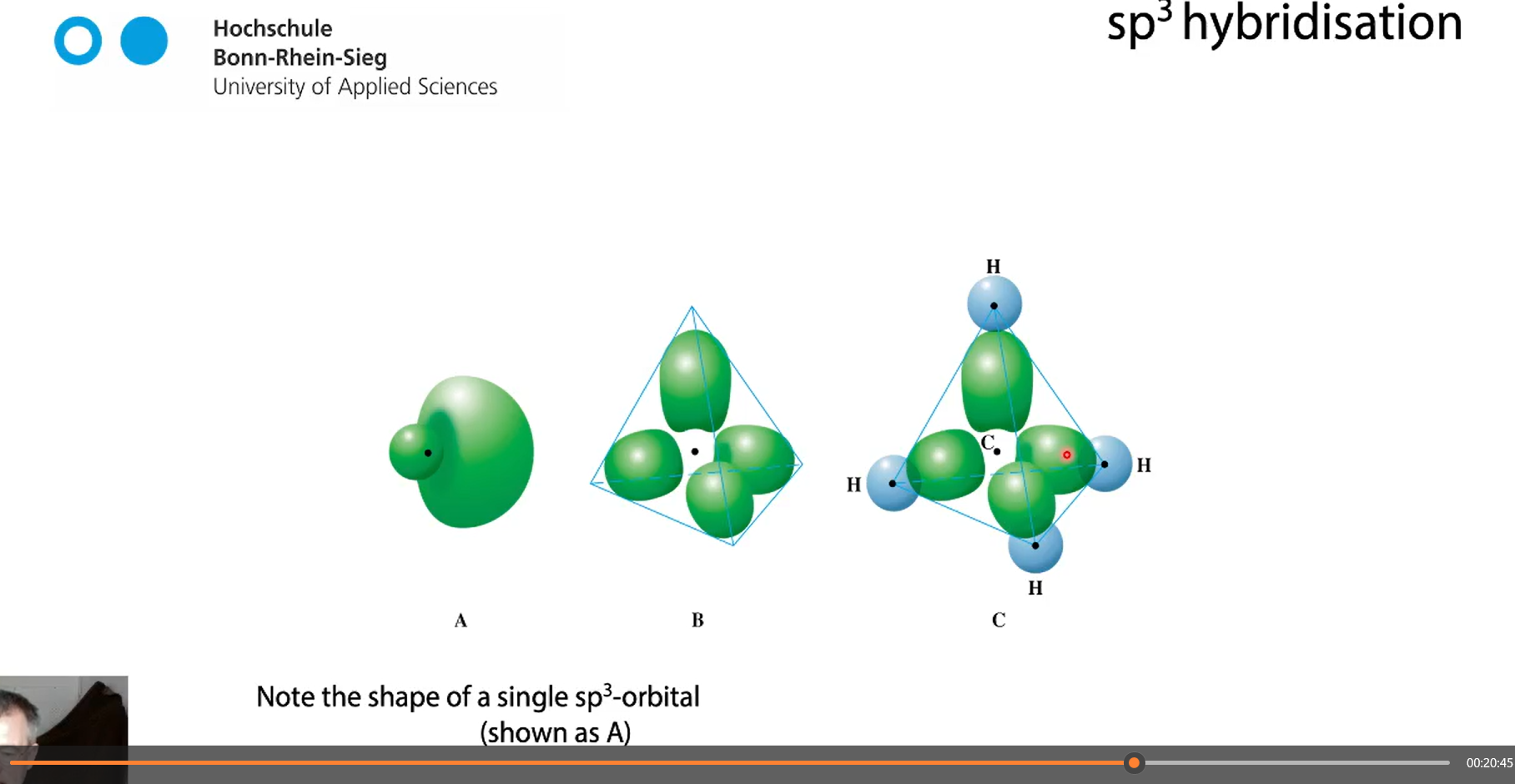
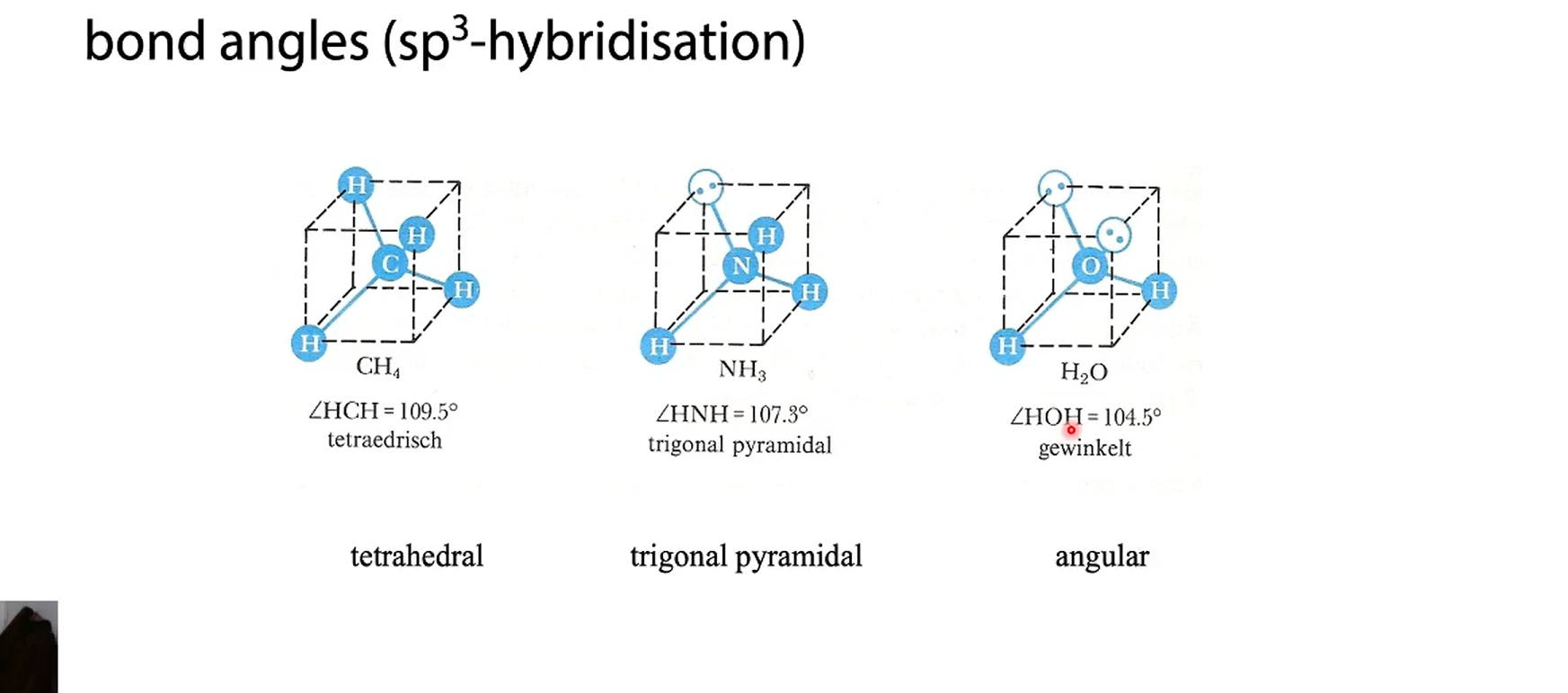
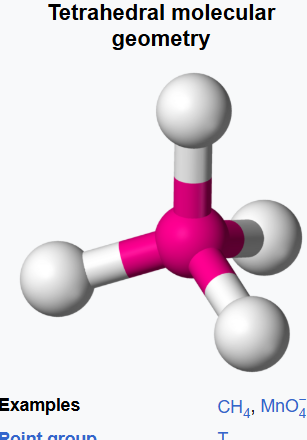
tetrahedrally surrounded
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron.
homolytic cleavage
The electron pair separate evenly to each part, and as a result both products contain a single electron.

all C-C and C-H bonds are ____
sigma bonds
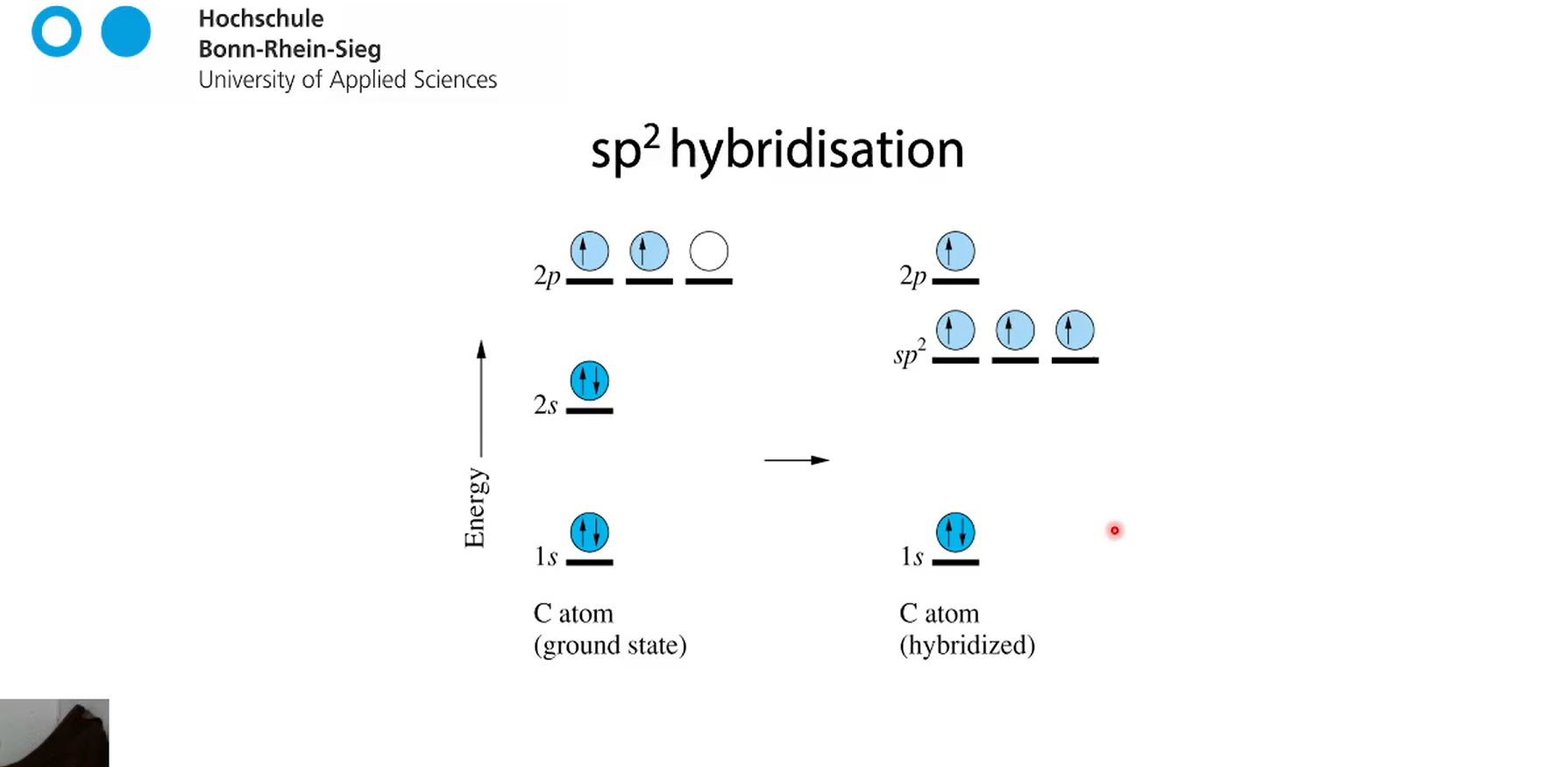
sp2 hybridsation (C-atom in ground state)
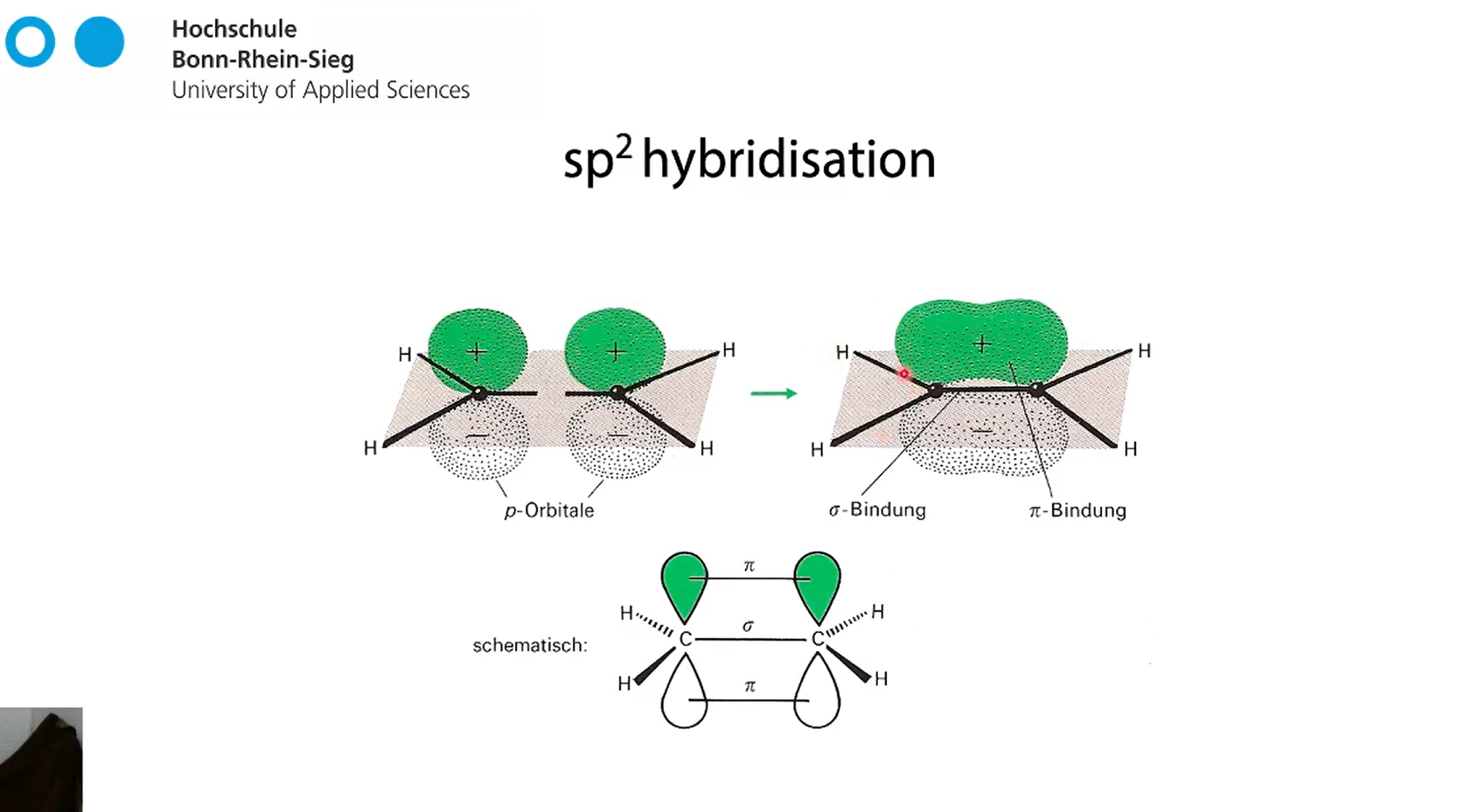
sp2 hybridisation of two carbon atoms. what bonds are happening?
3 sigma bonds between C-C and H-C for the hybridised orbitals
but since the free p orbitals are parallel they can also overlap forming pi-bonds NON hybridised
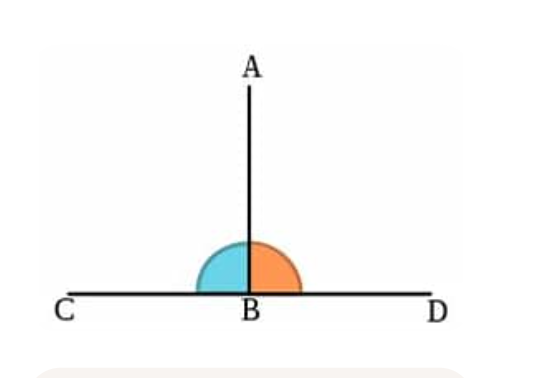
perpendicular
at a 90 degree angle
diamond vs graphite
diamond: tetrahedically surrounded C-C 154 pm
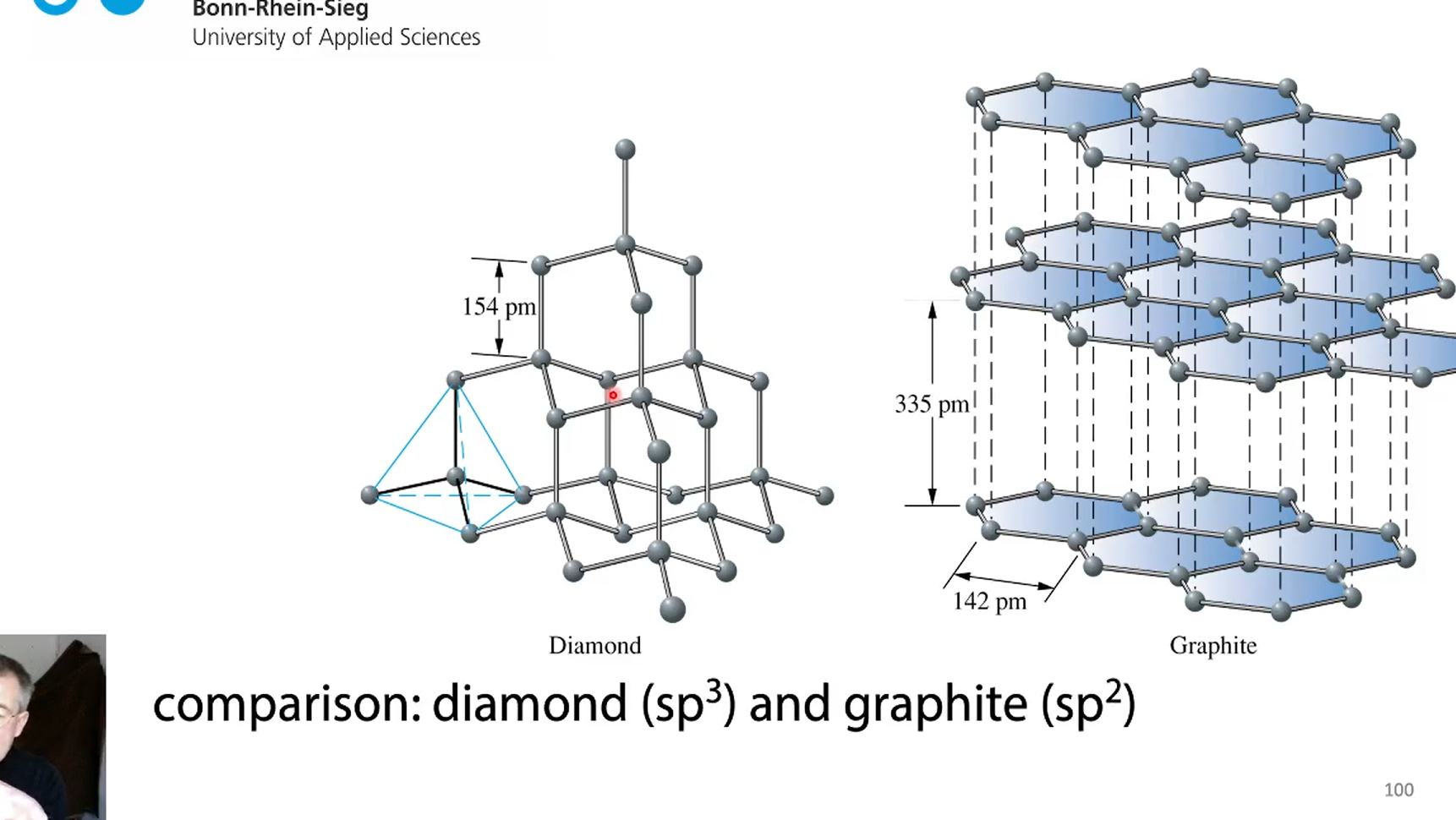
how long are C-C bonds?C=C and C=-C bonds
single bond 154 pm
douple bond 135 pm
triple bonds 122 pm
sp hybridisation
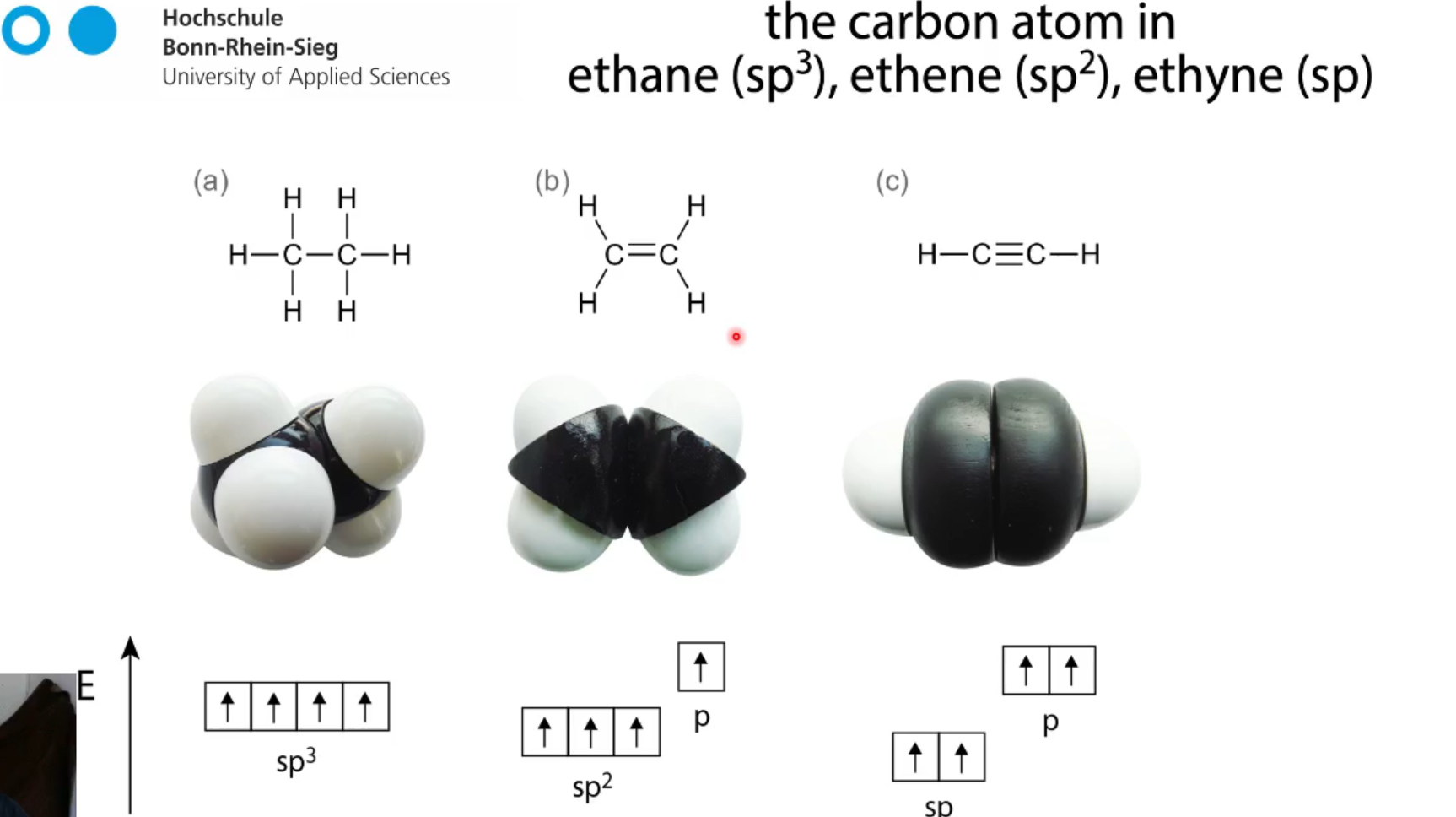
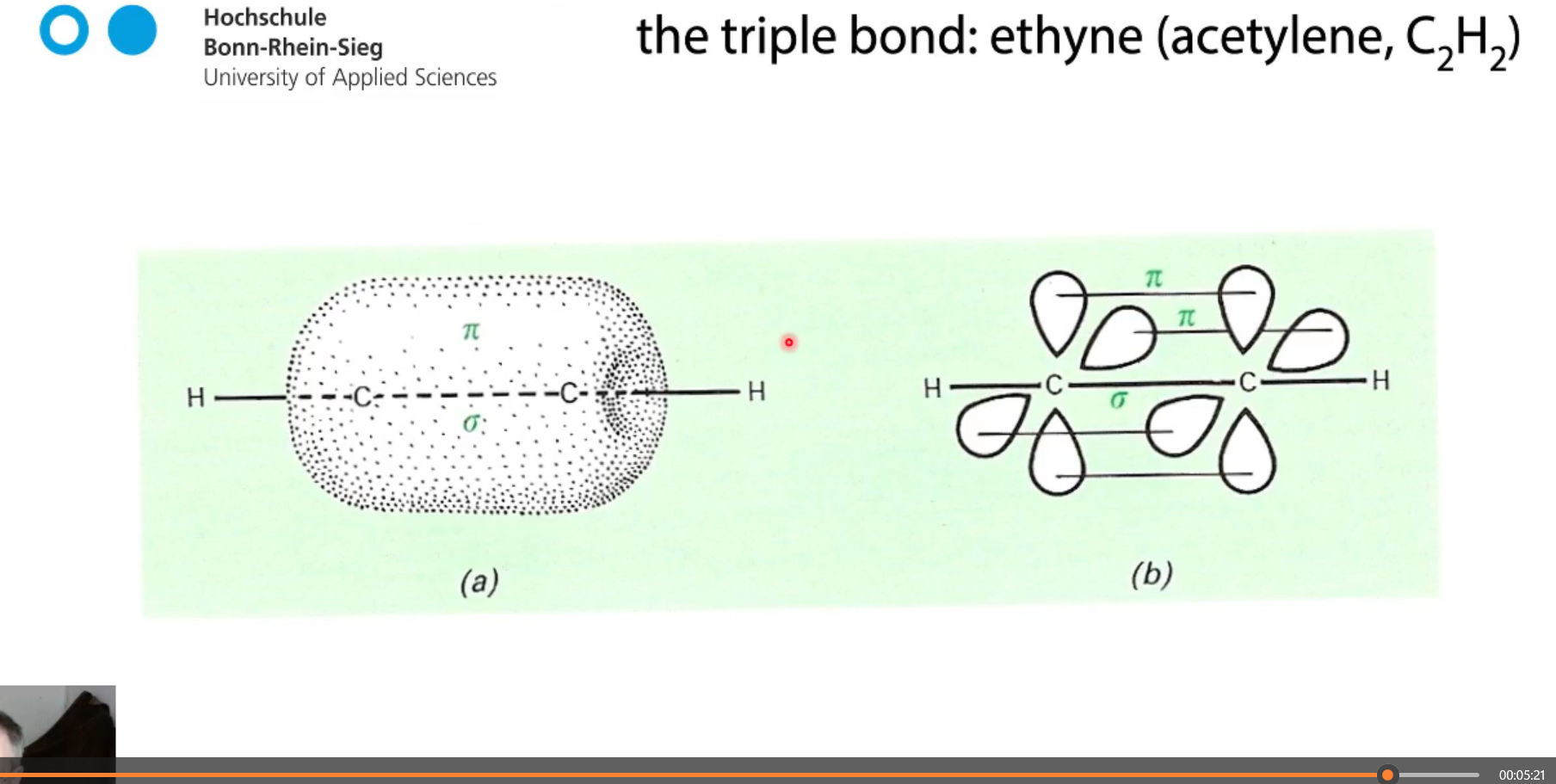
ethyne C2H2 how many bonds?
1 sigma bond and two pi bonds
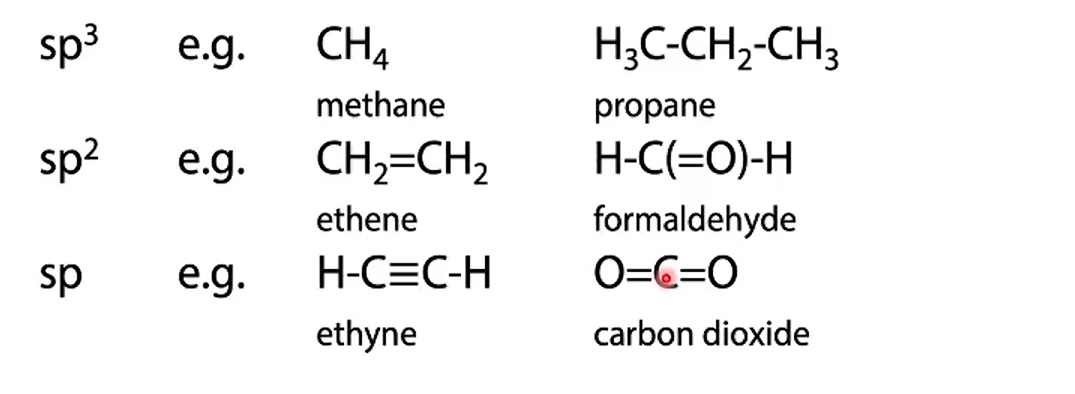
name the bonds