Transition Metals (Unfinished)
1/109
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
110 Terms
Why do the characteristics of transition metals arise?
Transition metals have a partially filled 3d energy sub-level in their atoms or ions
Why is zinc not considered a true transition metal?
When any transition metal loses electrons, they lose them for their 4s sub-level first
Zinc’s outer electron configurations is 3d10,4s2
Zinc only forms 1+ and 2+ ions meaning that it will never have a partially filled 3d sub-level which is a characteristic of true transition metals

What are the characteristic properties of transition metals?
Ability to form complexes
Form coloured ions
Exists in variable oxidation states
Good catalysts
What is meant by the term ligand?
Species that can donate one or more lone pairs of electrons to a transition metal or ion
What is meant by the term complex?
A complex is formed when transition metals or their ions form dative covalent bonds with ligands
What is meant by the term co-ordinate/dative covalent bonds in relation to transition metals?
Where the ligand provides both electrons in the covalent bond
What is meant by the term co-ordinate number?
The number of coordinate bonds formed to the central metal ion
What is meant by a monodentate ligand and give some examples
A ligand that forms one co-ordinate bond with the metal ion
H2O, Cl-, NH3, -CN
Which two uncharged monodentate ligands are similar in size?
Water and ammonia
Which monodentate ligand is bigger than water and ammonia?
Cl-
What is meant by the term bi-dentate?
Ligands that form two coordinate bonds since they donate two pairs of electrons (two donor atoms)
Name, write the formula and draw the two bidentate ligands that you should recall?
Ethane-1,2-diamine
NH2CH2CH2NH2
Ethanedioate ion
C2O42-
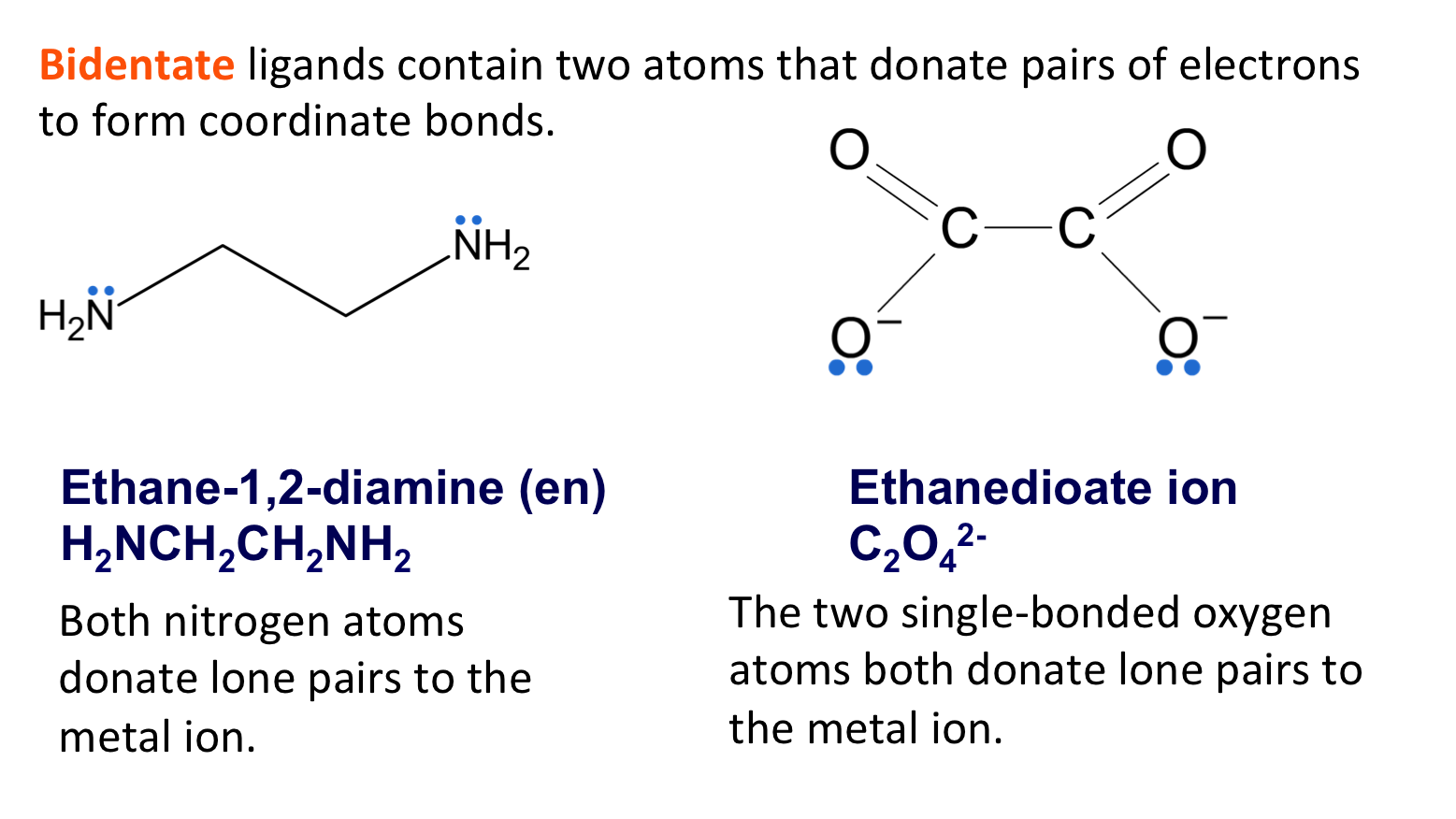
What is a multidentate ligand?
Ligands forming 3 or more dative covalent bonds since they donate at least 3 lone pairs of electrons
How many coordinate bonds does EDTA4- form?
6- it is a hexadentate ligand
How many coordinate bonds does porphin form?
4
What does EDTA stand for?
Ethane diamine tetra acetic acid
Recall the four most common shapes for transition metal complexes and their bond angles
Octahedral- 90o
Tetrahedral- 109.5o
Square planar- 90o
Linear- 180o
What are some ways we can form octohedral transition metal complexes?
6 monodentate ligands- water or ammonia, NOT Cl- as it is too large
3 bidentate ligands- ethane-1,2-diamine and the ethanedioate ion
1 hexadentate ligand- EDTA4-
How can we form tetrahedral transition metal complexes
Formed when a transition metal is bonded to 4 chloride ligands
Why do chloride ligands form tetrahedral complexes instead of octohedral?
Chloride ions are larger than other monodentate ligands
We can’t fit as many around the central metal atom- only enough space for four
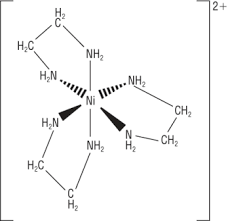
Draw the structure of [Cu(NH2CH2CH2NH2)3]2+
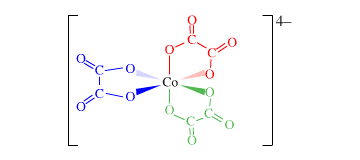
Draw the structure of [Cu[C2O4)3]4-
When do we normally form optical isomers of transition metal complexes?
Octahedral complexes with 3 bidentate ligands
What are optical isomers?
These exist as pairs of enantiomers
They are non-superimposable mirror images of each other
Why does cis-trans isomerism occur in transition metal complexes?
The ligands cannot rotate around the central metal ion
In which complex shapes can Cis-Trans isomerism occur and with what ligands?
Square planar with two of one type of ligand and two of another
Octahedral with 4 of one type of ligand and 2 of another
What is the difference between a cis and a trans isomer?
Cis- pair are next to each other at a 90o angle
Trans- the pair are on opposite sides to each other at a 180o angle
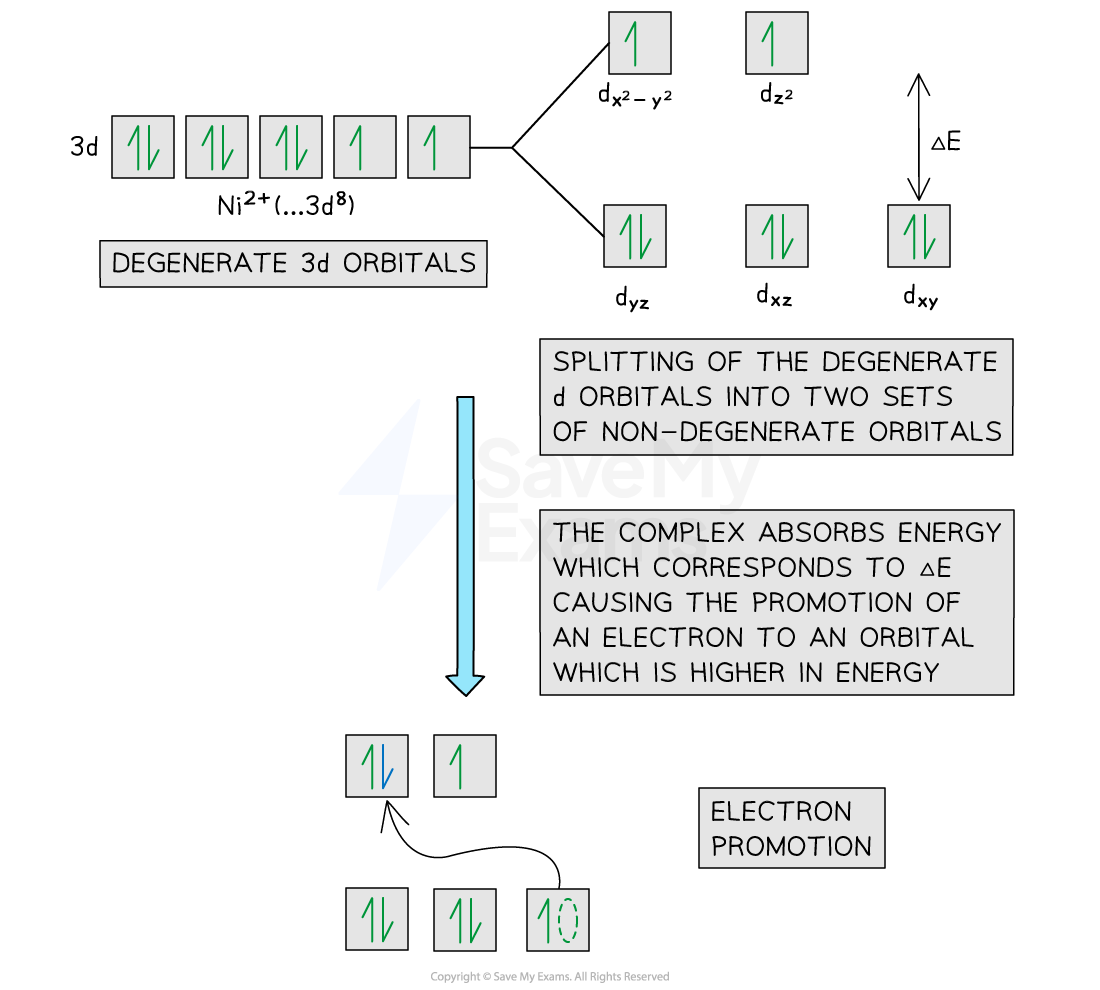
Explain with reference to d sub levels and electronic transitions why many transition metals appear coloured
When a transition metal is bonded to ligands, the d-orbitals split
When visible light is shone on the transition metal, the electrons are excited and an electron moves to a d-orbital higher in energy- this is called a transition
The light energy absorbed to excite the electron corresponds to certain parts of the visible electromagnetic spectrum
Visible light includes red, orange, yellow, green, blue, indigo and violet light with the red end being lower in energy and having longer wavelengths and the blue end being greater in energy and shorter wavelengths
If the change in energy is small, then lower energy wavelengths of light are absorbed (red, orange, yellow) and so shorter wavelength, high energy visible light is emitted (blue, indigo, violet) meaning that the compound will appear to be blue (for example
This is the inverse for transition energy changes that are larger
Why do some transition metals appear colourless?
Some like zinc have a full d sub level so transition cannot take place
Doesn’t absorb any visible light- it is all transmitted
What factors affect the colour of transition metals and how?
Type of ligand
Shape of the complex
Co-ordination number
Oxidation state of the metal
Metal
They affect the energy change that occurs when a transition happens. If any of these things change then the colour of the complex will change because the change in energy is different.
What equation give us the change in energy upon a transition?
ΔE = h x f (or v)
ΔE = The change in energy in Joules
h = Planks constant = 6.63 × 10-34 Js
f/v = Frequency (Hz/hertz or s-1)
What is the equation to give frequency?
f = c/λ
f = Frequency (Hz/hertz or s-1)
c = The speed of light 3.00 ×108 ms-1
λ = Wavelength in m
How can we link the two equations ΔE = h x f (or v) and f = c/λ to find the change in energy upon transition of an electron in a transition metal?
ΔE = (h x c) / λ
ΔE = The change in energy in Joules
h = Planks constant = 6.63 × 10-34 Js
c = The speed of light 3.00 ×108 ms-1
λ = Wavelength in m
We have just replaced frequency with the second equation
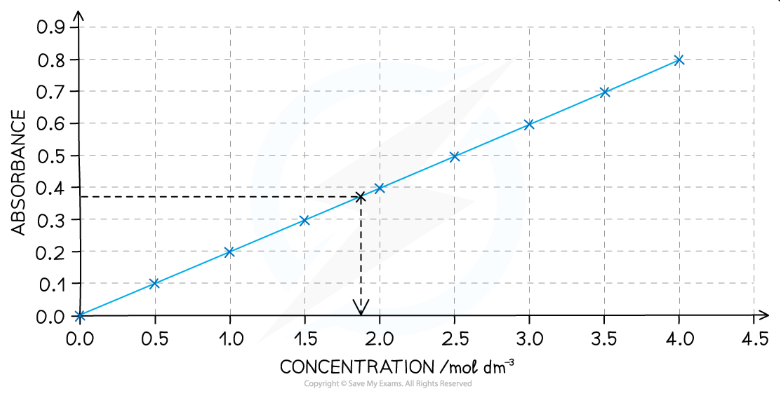
How do we make a calibration graph for a coloured transition metal solution?
Solutions of known concentrations are placed into the colorimeter and their absorption values are recorded
From these values, we can plot a graph of concentrations against absorption (a calibration graph)
What should a calibration graph look like and how would we use it to determine an unknown concentration of transition metal solution?
Graph of absorbance against concentration
Should be linear
If we can place a solution of unknown concentration is and measure the absorbance
We can then read across from absorbance and down to find the concentration
What happens in full ligand substitution reactions?
Ligands bonded to a central metal ion substituted for new ligand(s)
A complex with 6 monodentate ligands like water can be replaced by another 6 monodentate ligands or 1 hexadentate ligands for example
What happens in a ligand substitution reaction where chloride ligands are substitute monodentate ligands?
6 monodentate ligands can normally form a octahedral complex with a central metal ion
4 chloride ligands can be substituted for these monodentate ligands
Remember that chloride ligands are larger than ammonia and water so we can’t fit as many around the central metal ion
How is a haemoglobin molecule formed?
A central Fe2+ ion is bonded to the tetradentate ligand porphin
A globin molecule also form a coordinate bond to the central Fe2+ ion
This forms haemoglobin
How does haemoglobin transport different substances (oxygen, water, carbon monoxide) in the blood through ligand substitution reactions?
O2 form a dative covalent bond with Fe2+ to form oxyhaemoglobin
The bond is relatively weak so it breaks/dissociates at cells
At cells Fe2+ picks up water by forming a dative covalent bond with the oxygen and forms deoxyhaemoglobin
C≡O/carbon monoxide can form a dative covalent bond with Fe2+ but this covalent bond is very strong and does not dissociate from the new complex, carboxyhaemoglobin
What is entropy?
The measure of disorder in a system
This exists because everything in the universe tends towards disorder
Give the equation linking entropy, enthalpy change, temperature and free energy?
ΔG = ΔH - (T * ΔS)
ΔG: Change in Gibbs free energy- J or J/mol-1
ΔH: Enthalpy change- KJmol-1
T: Temperature (must be in Kelvin)
ΔS: Entropy change- JK-1mol
For a reaction to be feasible/spontaneous, what ΔG be?
Negative
When is the enthalpy change almost zero for ligand substitution reactions?
When you a breaking the same number of the same type of bonds
What is the chelate effect?
The greater stability of bidentate or multidentate ligand complexes compared to monodentate ligands for the same metal
What is en short for?
en is short for NH2CH2CH2NH2 or ethane-1,2-diamine we’re just using slang guys
Take the reaction:
[Co(NH3)6]3+ + 3en -> [Co(en)3]3+ + 6NH3
What happens to the entropy change of this reaction? What is the effect on Gibbs free energy and why does this mean the reaction is feasible?
6 Co-N bonds are broken and 6 Co-N bonds are formed so the enthalpy change is 0
This means that the reaction only happens due to the entropy change
When a reaction has fewer reactant particles producing more product particles, the entropy change increases
We know that ΔG must be negative for a reaction to be feasible/spontaneous
ΔG = ΔH - (T * ΔS) but since the enthalpy change is 0, ΔG = 0 - (T * ΔS)
Because our reaction has more product particles than reactant particles, our entropy change is positive meaning that 0 take away a positive number will give us a negative value for Gibbs free energy
Therefore the reaction is feasible
Take the reaction:
[Co(NH3)6]3+ + 3en -> [Co(en)3]3+ + 6NH3
Explain with reference to entropy why it’s difficult to reverse the above reaction?
If we attempt to reverse this reaction, the number of reactant particles in now greater than product particles- we are going from 7 particles to 4
This means that the entropy change is negative
ΔG = ΔH - (T * ΔS) but since the enthalpy change is 0, ΔG = 0 - (T * ΔS)
0 take away are negative value gives us a positive value for Gibbs free energy meaning the reaction is not feasible
In what scenarios does entropy change increase?
When going from a solid to a liquid to a gas
When we go from fewer reactant particles to more product particles
Recall the colours vanadium species 2+ to 5+ and an acronym to remember it
V5+ - Yellow
V4+ - Blue
V3+ - Green
V2+ - Purple
You’d Better Get Prepared
Will the reduction of Co2+ occur easier with water ammonia or ligands?
[Co(NH3)6]2+ + 2e- ⇌ Co + 6NH3 E
o= -0.43V[Co(H2O)6]2+ + 2e- ⇌ Co + 6H2O E
o= -0.28V
Water ligands
The electrode potential is more negative with ammonia ligands than it is with water
For the water ligands, it is more likely that the reaction moves forwards to form cobalt atoms/Co than Co2+
With ammonia ligands, it is more likely the reaction moves backwards to form the Co2+ ions rather than Co
What is the half equation for the oxidation of Fe2+ to Fe3+?
Fe²⁺ → Fe³⁺ + e⁻
What is the half equation for the oxidation of ethanedioate ions (C2O42-)?
C₂O₄²⁻ → 2CO₂ + 2e⁻
What is the half equation for the reduction of manganate ions/MnO4- to Mn2+ ions?
MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O
Give the full equation for the reaction of Fe2+ with MnO4- and the molar ratio of these two ions
MnO₄⁻ + 8H⁺ + 5Fe²⁺ → Mn²⁺ + 4H₂O + 5Fe³⁺
Fe2+ : MnO4- = 5:1
Give the full equation for the reaction of C2O42- with MnO4- and the molar ratio of these two ions
2MnO₄⁻ + 16H⁺ + 5C₂O₄²⁻ → 2Mn²⁺ + 8H₂O + 10CO₂
C2O42- : MnO42- = 5:2
What does aliquot mean?
A sub portion of a large sample
What is a heterogenous catalyst?
A catalyst that is in a different state to the reactants
How do heterogenous catalysts work?
The reactant particles diffuse to the surface of the catalyst
The reactants adsorb to the surface forming weak IMFs/attractive forces
This weakens the bonds in the reactant particles
This it takes less energy to break these bonds so the activation energy is lowered
Atoms then desorb from the surface
What is the point where reactants adsorb to the surface of a heterogenous catalyst?
The active site
What is catalyst poisoning?
impurities present in the reaction mixture can ‘poison’ the catalyst by blocking the active site
Why is catalyst poisoning bad for industrial reaction?
Reduces the efficiency of the catalyst
Increases cost of the catalyst- you may need to buy more to replaced the poisoned catalyst
Describe the structure of industrial catalysts and give two reasons for this structure
Structure- honeycomb support
Large surface area
Less precious metal needs to be used so reduced costs
Why did leaded petrol reduce the efficiency of cars?
The platinum and rhodium catalyst in a catalytic converter are poisoned by lead compounds
Give the equation for the Haber process and state the catalyst
N₂(g) + 3H₂(g) ⇌ 2NH₃(g)
Uses a catalyst of iron filings (essentially iron powder)
How can we produce the hydrogen needed for the Haber process and which is the method used most commonly?
Hydration of hydrocarbons with steam- this is used most frequently
Electrolysis of water
Equation for the hydration of methane with steam
CH₄(g) + H₂O(g) ⇌ CO(g) + 3H₂(g)
Why is using hydrogen produced from the hydration of methane with steam in the Haber process a problem?
Methane is usually contaminated with sulfur so the hydrogen produced will also be contaminated with sulfur
The iron filing catalyst used in the Haber process can be poisoned by sulfur
What is the contact process used for?
Producing sulfuric acid
Write the equation for the production of sulfur trioxide for sulfur dioxide in the contact process
SO2 (g) + ½ O2 (g) → SO3 (g)
Why does the contact process need a catalyst?
All the substances are in the gas phase
The particles are spread apart so there are fewer successful collisions than with a solid or a liquid
We could increase the rate by increasing temperature and pressure but these methods are expensive
What is the catalyst used in the contact process?
V2O5
How is the contact process catalysed? Use equations to show this
SO2 + V2O5 → SO3 + V2O4
½ O2 + V2O4 → V2O5
Overall equation: SO2 (g) + ½ O2 (g) → SO3 (g)
Keep in mind the two equations before are also full equations
What is a homogenous catalyst?
A catalyst in the same physical state as the reactants
What happens to a homogenous transition metal catalyst during a reaction?
The reaction often proceeds via an intermediate species
The transition metal temporarily changes oxidation state
Give the reaction between peroxodisuphate ions and iodine and identify a catalyst for this reaction?
S₂O₈²⁻(aq) + 2I⁻(aq) → 2SO₄²⁻(aq) + I₂(aq)
Fe2+ or Fe3+
Why is the reaction between S₂O₈²⁻ and 2I⁻ slow?
Both species are negatively charged
They will repel each other
Give the two overall equations that occur during the catalysis of this reaction
Step 1: Reduction of Peroxodisulphate by Iron(II): S₂O₈²⁻ + 2Fe²⁺ → 2SO₄²⁻ + 2Fe³⁺
Step 2: Oxidation of Iodide by Iron(III): 2I⁻ + 2Fe³⁺ → I₂ + 2Fe²⁺
If we cancel out species on either side then the overall equation is: S₂O₈²⁻(aq) + 2I⁻(aq) → 2SO₄²⁻(aq) + I₂(aq)
We can do either step first if we just change what products are added together first
What is auto-catalysis?
When one of the products of a reaction acts as a catalyst for the reaction?
In the reaction with MnO4- ions and C2O42- ions, why does it start off slowly?
Two negatively charged ions repel each other
In the reaction with MnO4- ions and C2O42- ions, what is the species that is an auto-catalyst?
Mn2+
What is the half equation to show the oxidation of Mn2+ to Mn3+
Mn2+ → Mn3+ + e-
Give equations to show how the autocatalysis of the reaction between MnO4- and C2O42- works
Step 1: 4Mn²⁺ + MnO₄⁻ + 8H⁺ → 5Mn³⁺ + 4H₂O (x2)
Mn3+ acts as an intermediate compound and is produced from the oxidation of Mn2+
Step 2: 2Mn³⁺ + C₂O₄²⁻ → 2Mn²⁺ + 2CO₂ (x5)
Overall: 2MnO₄⁻ + 16H⁺ + 5C₂O₄²⁻ → 2Mn²⁺ + 8H₂O + 10CO₂
Step 1 has to be multiplied by 2 and step 2 multiplied by 5 in order to get the same amount of Mn3+ on each side of the equation
This allows us to eliminate Mn3+ from the overall equations which is important as it never shows up in the overall equations
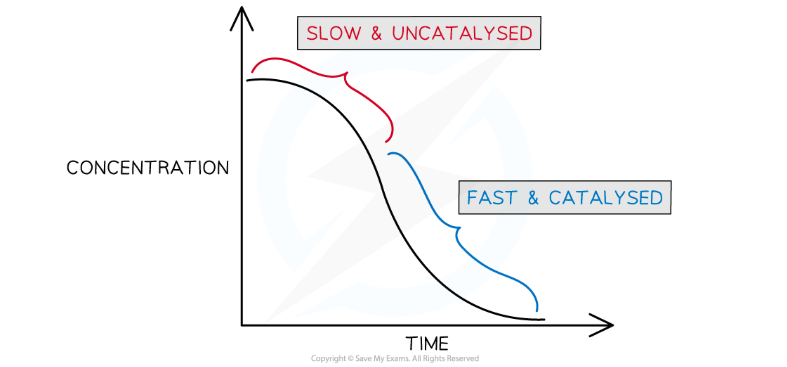
Describe and explain a concentration against time graph for an autocatalysis reaction (Use MnO4-’s reaction with C2O42- as an example)
Explain how this would be different if we added powdered Mn2+ at the start
At the start, the concentration of Mn2+ is building up but the negatively charged species are sill repelling each other
In the middle, the rate is the fastest as Mn2+ is acting as a catalyst
At the end, the concentration of reactants is low so the rate slows down as the collision frequency (and therefore the frequency of successful collisions, decreases
Adding powdered Mn2+
There would be no lag phase at the start because the Mn2+ can immediately begin catalysing the reaction
Give the general equation for the reaction of Cu2+ and Fe2+ in their metal aqua ion complex with OH- ions?
[M(H2O)6]2+ + 2OH- → M(H2O)4(OH-) + 2H2O
Give the general equation for the reaction of Fe3+ and Al3+ in their metal aqua ion complex with OH- ions?
[M(H₂O)₆]³⁺(aq) + 3OH⁻(aq) → [M(H₂O)₃(OH)₃](s) + 3H₂O(l)
What type of reaction is occurring when hydroxide ions are added to metal aqua ions complexes?
Hydrolysis
Give the colours of the following complexes:
[Fe(H₂O)₆]²⁺
[Cu(H₂O)₆]²⁺
[Fe(H₂O)₆]³⁺
[Al(H₂O)₆]³⁺
[Fe(H₂O)₆]²⁺ : Pale green solution
[Cu(H₂O)₆]²⁺ : Blue solution
[Fe(H₂O)₆]³⁺ : Yellow-brown solution
[Al(H₂O)₆]³⁺ : Colourless solution
What observations do we make when adding hydroxide ions to metal aqua complexes
Fe(OH)₂(H₂O)₄ : Dark green ppt and then turns brown on standing
Cu(OH)₂(H₂O)₄ : Blue ppt
Fe(OH)₃(H₂O)₃ : Brown ppt
Al(OH)₃(H₂O)₃ : White ppt
What observations do we make when adding hydroxide ions to metal aqua complexes in excess?
Fe(OH)₂(H₂O)₄ : No visible change
Cu(OH)₂(H₂O)₄ : No visible change
Fe(OH)₃(H₂O)₃ : No visible change
Al(OH)₃(H₂O)₃ : White precipitate dissolves
Why does Fe(OH)₂(H₂O)₄ turn brown on standing?
The iron 2+ ion is oxidised to iron 3+ which is brown
Why does Al(OH)₃(H₂O)₃ dissolve when excess OH- is added? Use equations to show how this occurs
Compounds of aluminium are amphoteric and so can react with OH- ions
Al(OH)₃(H₂O)₃ + 3OH- → [Al(OH)6]3- + 3H2O
The aluminium complex produced is charged so now it can form interactions with the water, causing it to dissolve
Why do neutral (not charged) complexes not dissolve in water?
They have no charge so they cannot form interactions with the water, therefore they do not dissolve- they are insoluble
Give the general equation for the reaction of 3+ metal aqua ion complexes when ammonia is added dropwise
[M(H₂O)₆]³⁺ + 3NH₃ → [M(H₂O)₃(OH)₃] + 3NH₄⁺
Give the general equation for [M(H2O)6]2+ reaction when ammonia is added dropwise?
[M(H₂O)₆]²⁺(aq) + 2NH₃(aq) → [M(H₂O)₄(OH)₂](s) + 2NH₄⁺(aq)
What type of reaction is occurring when ammonia is added dropwise to metal aqua ion complexes?
Hydrolysis
Give the observations made when ammonia is added to the following 4 compounds dropwise:
[Fe(H₂O)₆]²⁺
[Cu(H₂O)₆]²⁺
[Fe(H₂O)₆]³⁺
[Al(H₂O)₆]³⁺
[Fe(H₂O)₆]²⁺: Dark green precipitate
[Cu(H₂O)₆]²⁺: Blue precipitate
[Fe(H₂O)₆]³⁺: Brown precipitate
[Al(H₂O)₆]³⁺: White precipitate
Give the observations made when ammonia is added to the following 4 compounds in excess:
[Fe(H₂O)₆]²⁺
[Cu(H₂O)₆]²⁺
[Fe(H₂O)₆]³⁺
[Al(H₂O)₆]³⁺
[Fe(H₂O)₆]²⁺: No visible change
[Cu(H₂O)₆]²⁺: Deep blue solution
[Fe(H₂O)₆]³⁺: No visible change
[Al(H₂O)₆]³⁺: No visible change
Why does [Cu(H₂O)₆]²⁺ form a deep blue solution when ammonia is added in excess? Use equations in your explanation
[Cu(H₂O)₆]²⁺ + 4NH₃(aq) → [Cu(NH₃)₄(H₂O)₂]²⁺(aq) + 4H₂O(l) (This is an overall equation)
When ammonia is added dropwise, the copper aqua complex undergoes hydrolysis reactions with two of the water ligands at the axial positions
Ammonia can then also undergo ligand substitution with the rest of the water ligands
It breaks the O-H bonds at equatorial positions
What two equations occur when [Cu(H₂O)₆]²⁺ form a deep blue solution when ammonia is added in excess and what kind of reactions are occurring?
[Cu(H₂O)₆]²⁺(aq) + 2NH₃(aq) → [Cu(H₂O)₄(OH)₂](s) + 2NH₄⁺(aq) - Hydrolysis
[Cu(H₂O)₄(OH)₂](s) + 4NH₃(aq) → [Cu(NH₃)₄(H₂O)₂]²⁺(aq) + 2OH⁻(aq) + 2H₂O(l) - Ligand substitution
What is the name of the shape of the complex that is formed when [Cu(H₂O)₆]²⁺ reacts with excess ammonia?
Elongated octahedral