Required practical 7 - measuring the rate of reaction
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
Outline of the method for the Iodine clock:
In a smaller beaker have H2O2
In a larger beaker have H2SO4 + KI + Na2S2O3 + starch
Mix the smaller beaker to the larger beaker, and start the stock watch immediately, stop the timer when the colour change turns blue-black
Repeat the experiment by varying the concentration of KI and water - but everything else remains the same.
Then in a separate table have the volume of KI and the rate (1/time)
Then determine the rate order.
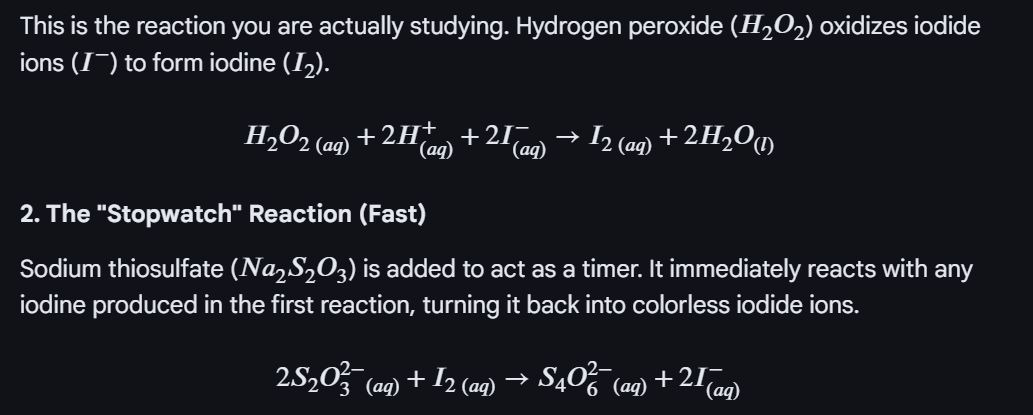
Practical considerations for Iodine clock:
Use wet equipment: Any remaining water will dillute the concentration of the reactants and effect the rate of reaction
Temperature changes: any changes in the temperature effects the rate of the reaction, so make sure to repeat all the experiments under constant temperature using water bath
Colour change is subjective and reaction time of humans can effect the reading when they stop the stockwatch.
Outline the method: measuring rate using volume of gas
Pour 40 cm cubed of HCl into a conical flask
Clamp a syringe and the plunger is pushed all the way in
Get a magnesium strip and place in the conical flask, and quickly place a bun and start stock watch
As the gas is produced this will increase the pressure and push the plunger giving a reading on the volume of gas.
Every 30 seconds record the volume of gas produced and record
Repeat the experiements under same temperature, and pressure and surface area of magnesium ribbon but DIFFERENT concentration of hydrochloric acid.
Plot a graph of volume of hydrogen produced on y-axis against time in seconds on x-axis for both concentrations of hydrochloric acid
Draw a tangent at t = 0s and find gradient to find intial rate and compare these values to find the reaction order.

Other ways of measuring the rate of reaction:
Measuring the mass lost
Measuring pH
Quenching: When a reaction mixture is sampled at different times, but cooling/ diluting or neutralising the reaction. Then titrating the solution to find the concentrations of reagent present
colorimeter where the absorbance is proportional to the concentration of Iodine.