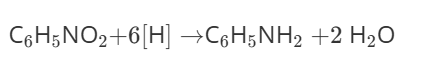Yr2 Organic chem-Amines CH28
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
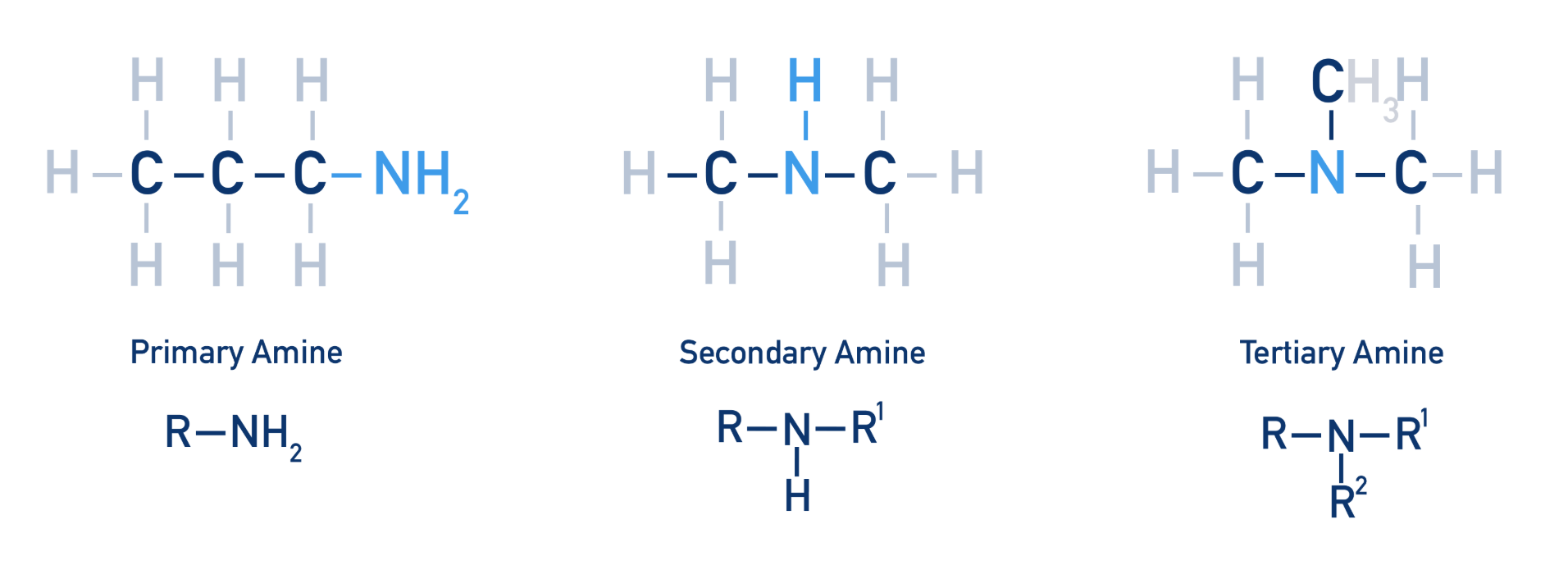
What are the primary, secondary and tertiary forms of Amines?
What is the general formula for amines, their naming, properties: shape, boiling point, solubility and reactivity:
RNH2
Amines are named using the suffix name of -amine
Properties:
The shape of ammonia is Trigonal pyramidal, with 3 bonding pairs and 1 lone pair
Amines with the RNH2, have difference in electronegativities, where Nitrogen is more electrongeative, this causes hydrogen bonding between molecules, so they have a high Boiling point
Shorter chain amines are more soluble in water and alcohols, most amines are soluble in less polar solvents
phenylamine is not very soluble in water, because benzene ring is very stable and cannot form hydrogen bonds
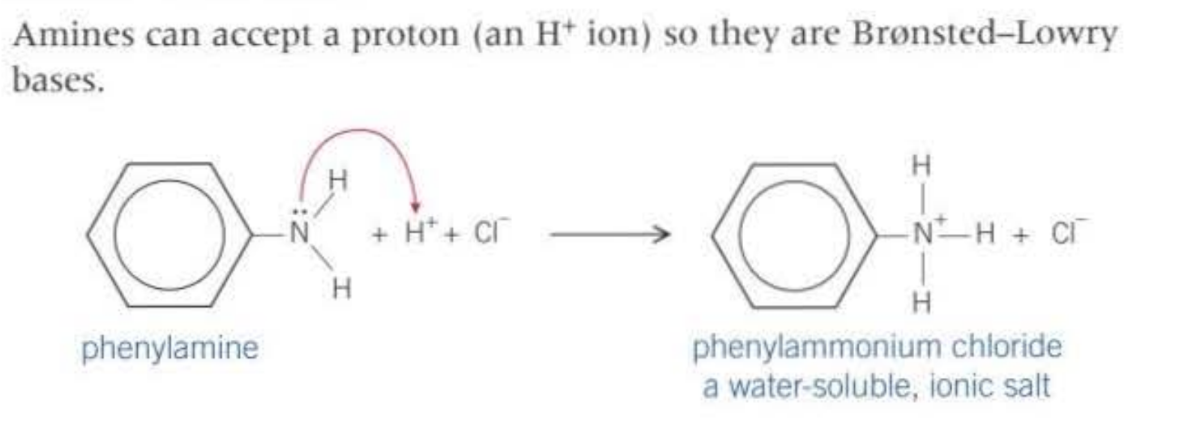
Reactivity of amines: There is a lone pair on the Nitrogen, so attraction with H+, additionally, the positive charge on the carbon is electron deficient - allowing nucleophiles to attract the nitrogen
The properties of amines as bases:
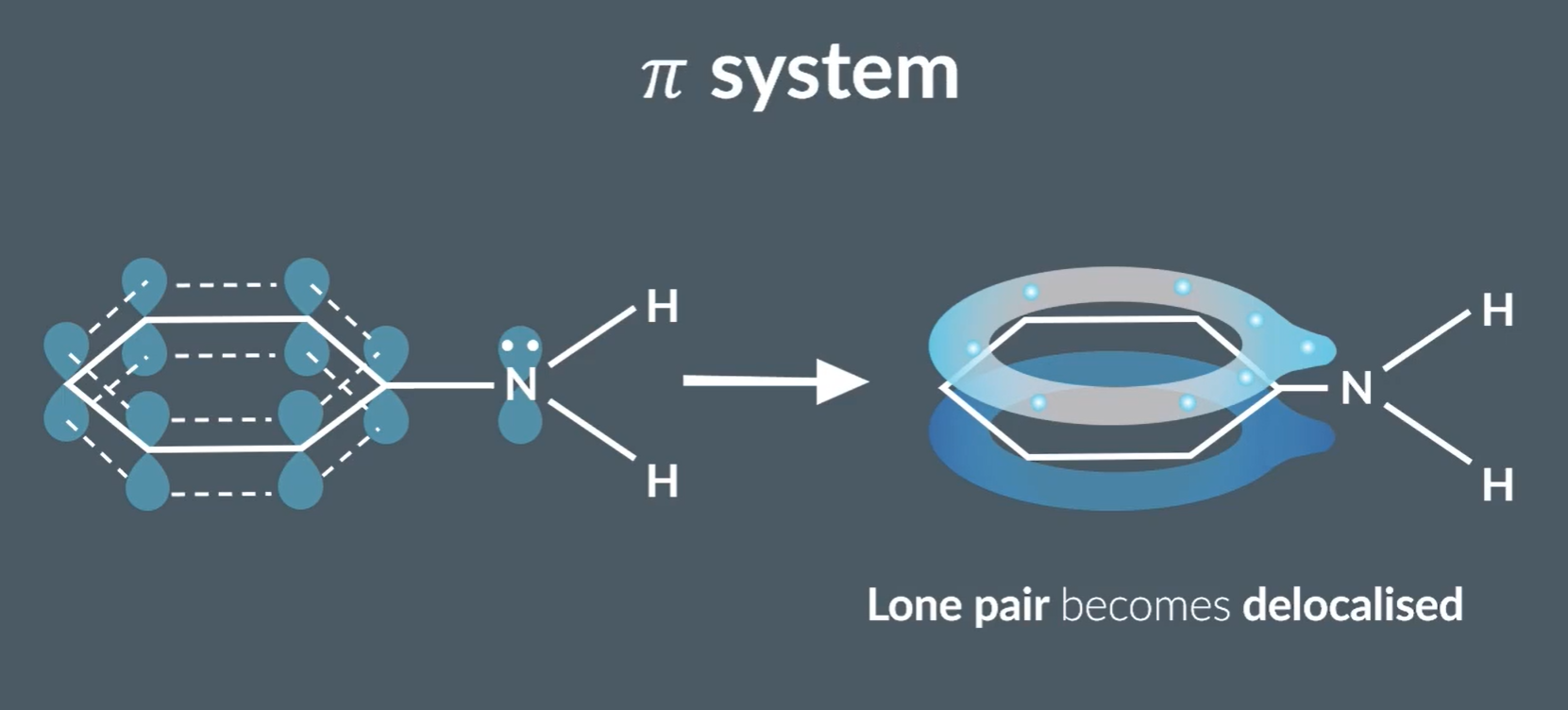
Lone pair becomes delocalised, and participates in the benzenes’ delocalised electron ring, no nitrogens’ lone pair is less available to the nitrogen, acting as a weaker base (proton acceptor)
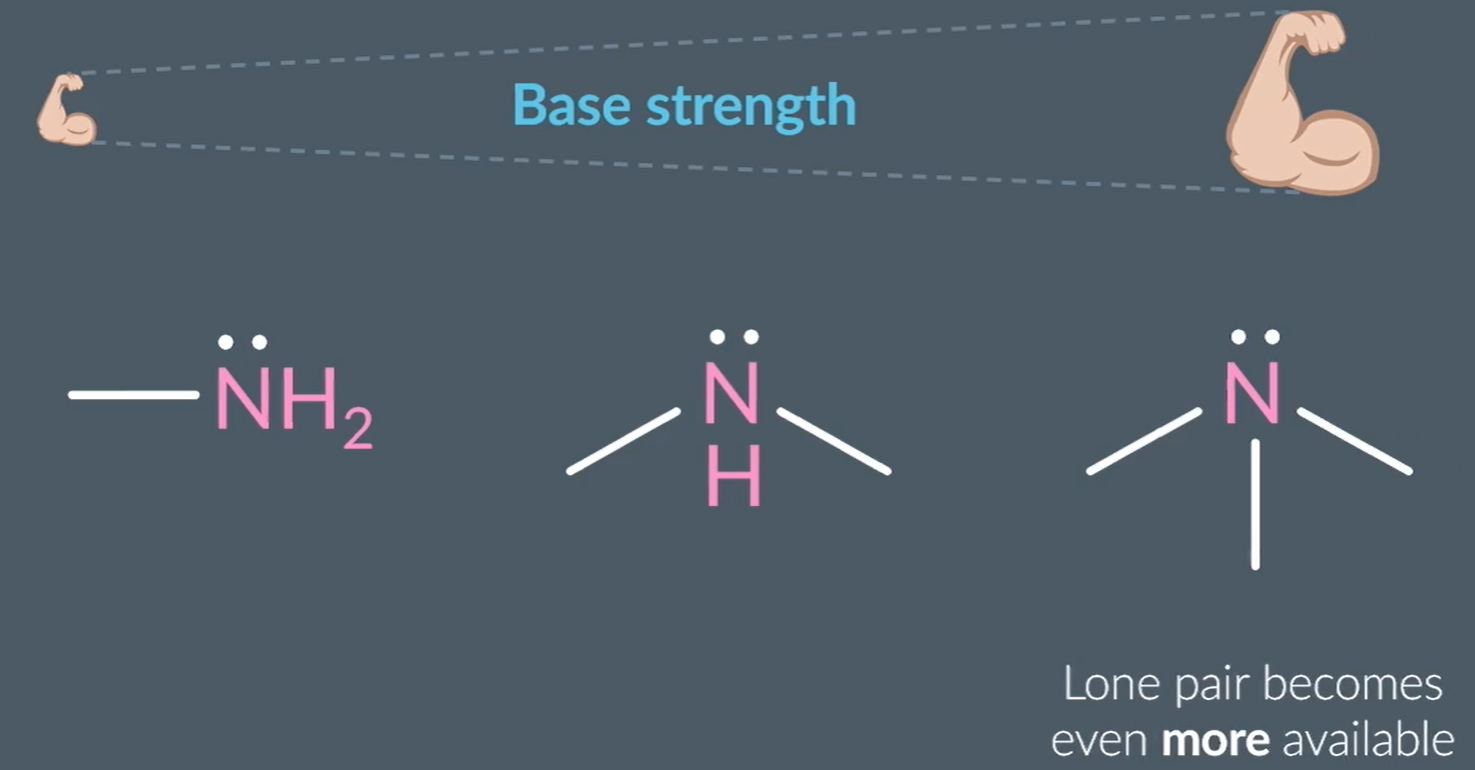
Based on the positive inductive effect, where the alkyl groups donate electron density towards nitrogen, increasing basic strength.
So, overall…
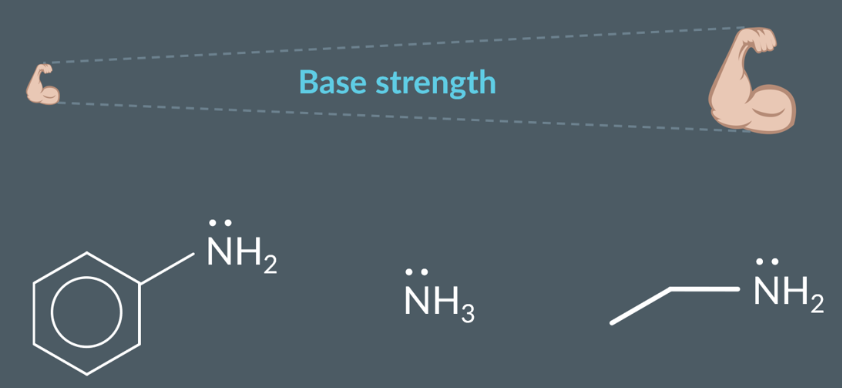
Key point: Tertiary alkylamines are not stronger bases than secondary alkyl amines, as tertiary alkylamines are not soluble in water.
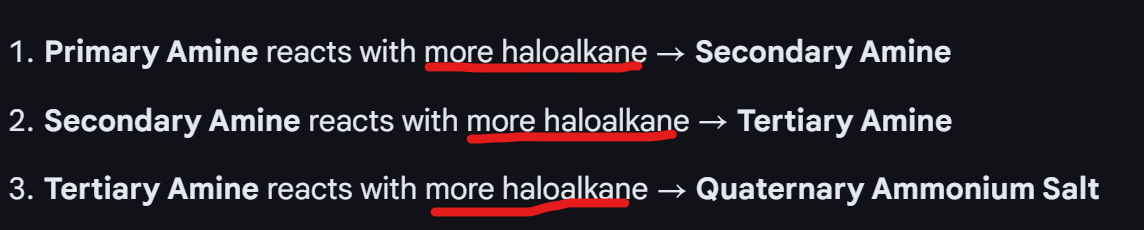
What is the process of making a quaternary ammonia salt?
Nucleophillic substitution: With halogenoalkane and ammonia
Ammonia, primary amine, secondary amine, tertiary amine all have a lone pair of electrons - so can act as nucleophiles.
Conditions: High pressure, heat, excess ammonia and dissolve ammonia in ethanol.
So, the final product from the series of nucleophillic substitution is quaternary ammonium ion.
So, when the quaternary ammonium ion, is with another anion it is called a quaternary ammonium salt.
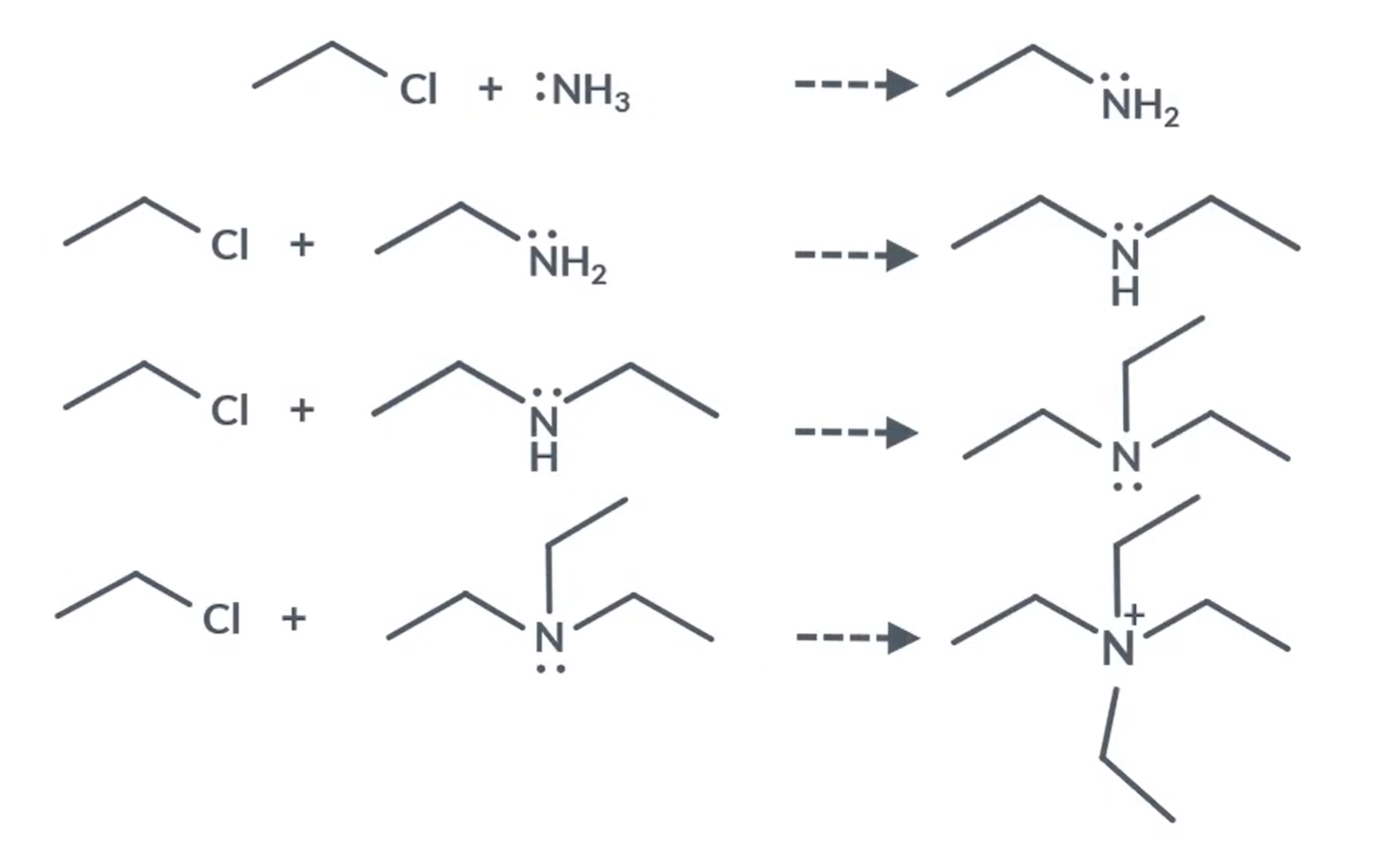
For example:
To form primary amine as major product, use an excess of ammonia.
What can a quaternary ammonium salt act as?
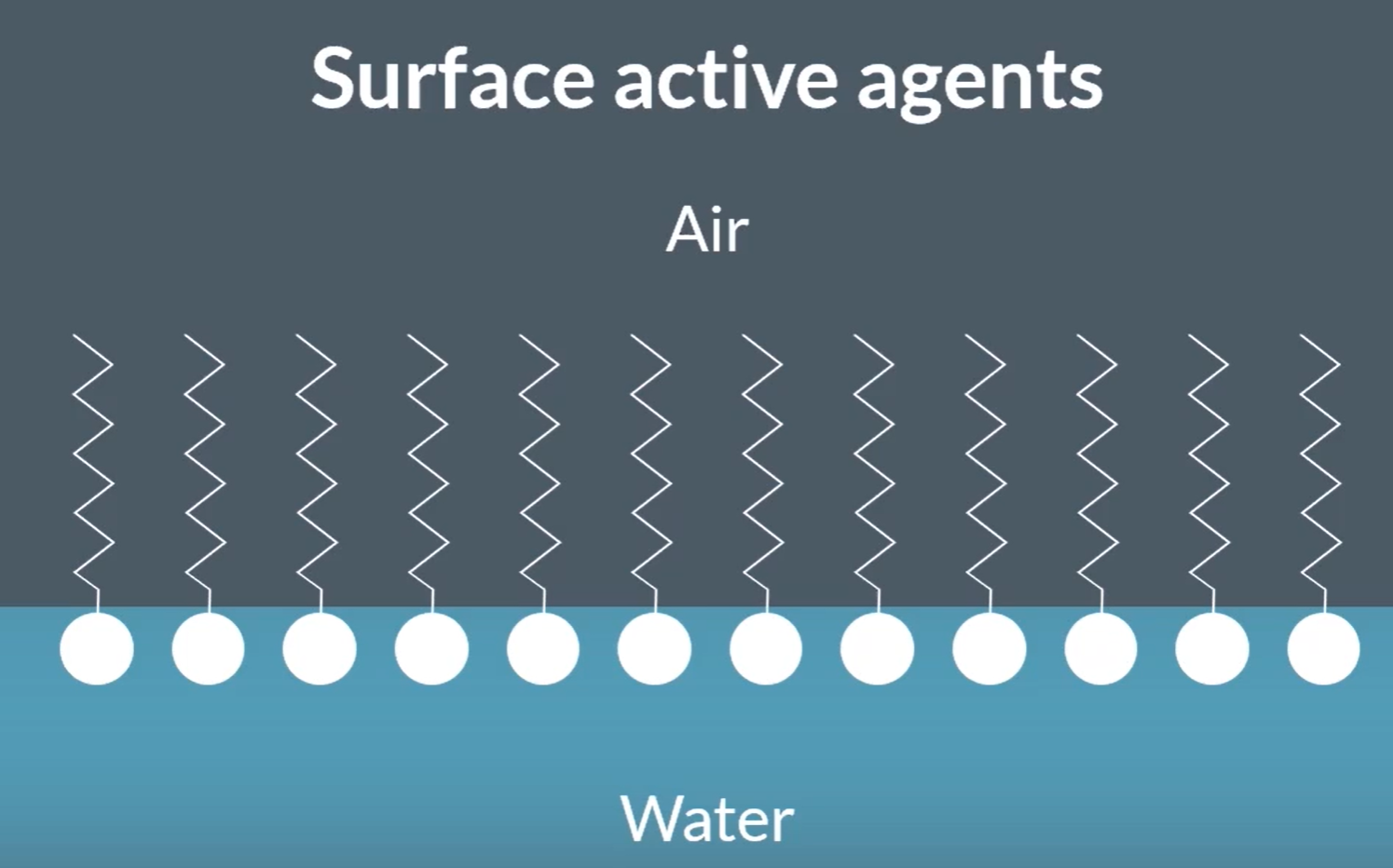
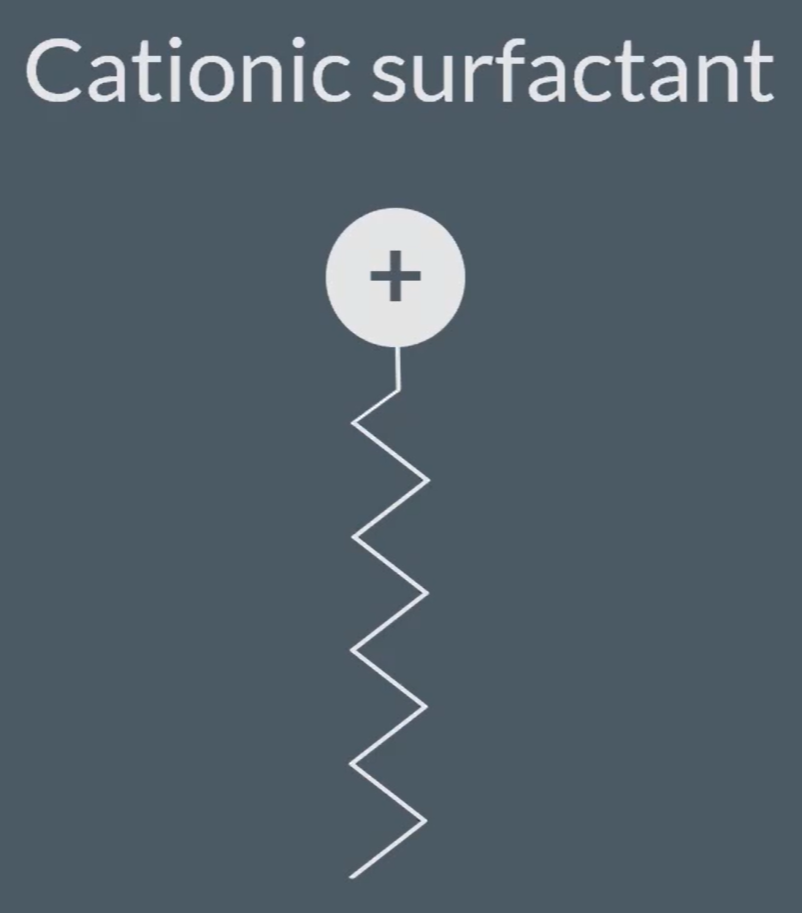
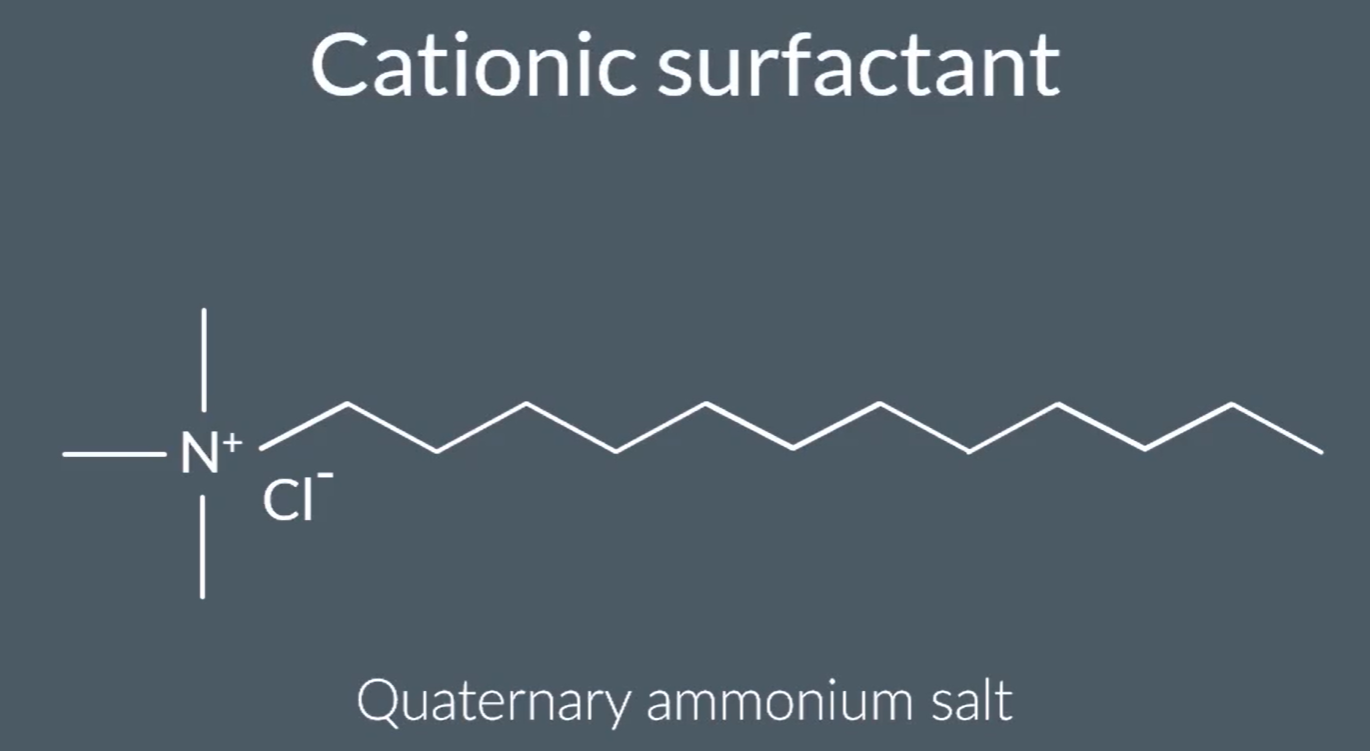
So, Quaternary ammonium salt can be used as a ‘Cationic surfactant’=> both are required, found in soap and shampoo.
How to prepare a primary amine from a nitrule?

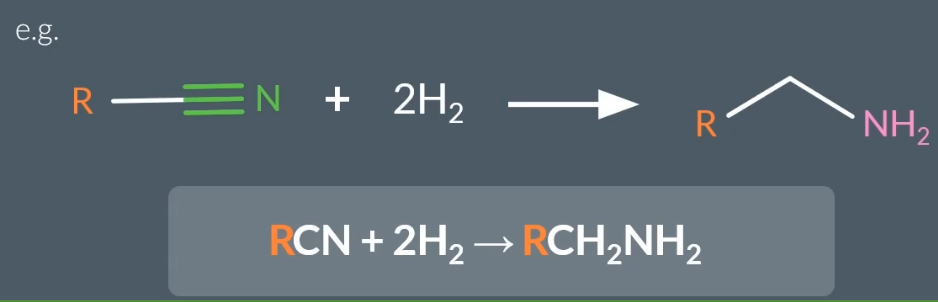
Which method is better in preparing an amine via Nucleophillic substitution or reduction?
By reduction
Atom economy is 100%
No further reaction occuring from reduction, but from nucleophillic substitution further reactions are possible
How can you form phenylamine?
Aromatic amines are used to manufacture colorful dyes.
Step 1: Concentrated nitric and sulfuric acid, forms nitrobenzene
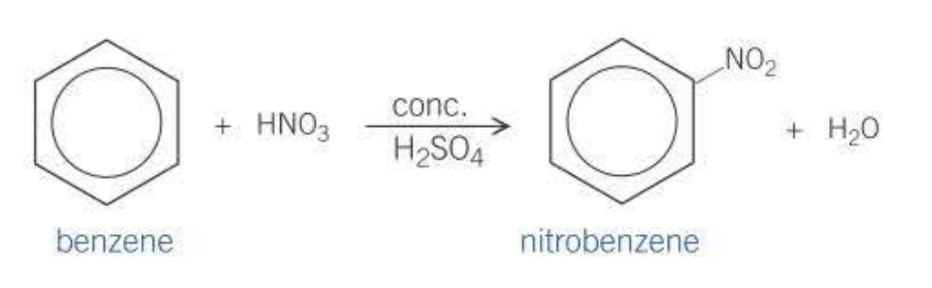
Step 2: Nitrobenzene is reduced to phenylamine by concentrated HCl or Tin which act as reducing agents