sleep physiology pt 3 a ha ha haha
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
What is a hormone
A hormone is a chemical messenger secreted by an
An endocrine cell that travels through the circulation to
act on distant target tissues expressing specific
receptors.
what do circadian-controlled hormones do?
– Rhythmic across the 24-hour day
– Persist under a constant routine
– Sleep modulates amplitude but does not generate the
rhythm
What do sleep-dependent hormones do?
– Released primarily during sleep or specific sleep stages
– Strongly reduced by sleep loss or fragmentation
– Poorly predicted by clock time alone
why melatonin and cortisol are considered primarily circadian-regulated hormones
Melatonin: Why it is circadian
Controlled directly by the SCN → pineal gland pathway
Secretion rises at night based on internal clock timing, not because you are asleep
Its role is to signal biological night
Cortisol: why it is circadian
Controlled by the SCN via the HPA axis
Shows a strong daily pattern:
Low at night
Peaks in early morning (before waking)
How does light differentially affect melatonin and cortisol secretion?
cortisol:
Light has minimal direct effect on timing
Cortisol rhythm is:
More stable
Less acutely suppressed by light compared to melatonin
melatonin:
Strongly light-sensitive
Light exposure at night:
Suppresses melatonin secretion
Can delay or shift circadian phase
How does sleep loss differentially affect melatonin and cortisol’s secretion
Melatonin
Still occurs even if you don’t sleep
Timing remains largely intact
Sleep mainly affects how much is released (amplitude), not whether it happens
not sleep dependent
Cortisol
Persists during sleep deprivation
Still rises to a morning peak even without sleep
Even shifting sleep to daytime:
Does not shift the cortisol peak appropriately
Confirms it is clock-driven, not sleep-driven
Explain how growth hormone and prolactin secretion depend on sleep onset
GH:
GH shows a large, rapid surge shortly after sleep begins
This surge is:
Specifically tied to sleep onset
Strongest during early-night slow-wave sleep (SWS)
The timing is not based on clock time → it happens whenever sleep starts
Prolactin:
PRL levels:
Increase soon after sleep begins
Stay elevated across the sleep period
Like GH, this rise is:
Linked to sleep onset
Not dependent on time of day
Explain how growth hormone and prolactin secretion depend on sleep architecture,
Growth Hormone
GH release coincides with peaks in SWS/SWA
Enhancing SWS → increases GH secretion
Reducing SWS → reduces GH secretion
Prolactin
PRL is less tightly linked to SWS than GH
Instead:
It shows a sustained elevation across the entire sleep period
Not just during deep sleep
Describe how aging alters hormonal profiles GH and cortisol
Reduced Sleep-Dependent Growth Hormone (GH)
Older adults show:
Much smaller GH peak after sleep onset
Reduced total GH secretion over 24 hours
The large early-night GH pulse seen in young adults is blunted or absent
Cortisol
Older adults show:
Higher cortisol levels in the evening and early night
A higher nadir (baseline) instead of the normal low nighttime levels
Sometimes a slightly earlier circadian phase (earlier peak timing)
Describe how testosterone exhibits mixed sleep-dependent and circadian regulation
Sleep-dependent component (dominant)
Testosterone rises after sleep onset
Peaks during the last part of the sleep episode
This happens regardless of clock time
Even though sleep drives secretion:
The timing of the peak is shaped by circadian rhythms
Typically aligns with early morning
So:
Circadian system = sets timing window
Sleep = enables actual secretion
How do estrogen and progesterone influence sleep despite being cycle regulated
Estrogen and progesterone are driven by the menstrual cycle, not by sleep or circadian timing.
Estrogen effects: Stable estrogen levels are
generally associated with better sleep, while rapid
declines before menses or during peri- and post-
menopause often disrupts sleep.
• Progesterone effects: Progesterone has more direct
sleep-promoting effects via GABAergic mechanisms,
making it the more strongly sleep-facilitating ovarian
hormone
what are some of the consequences of insufficient or mistimed sleep
Disrupted sleep and hormonal signaling
contribute to age-related changes in
metabolism, stress regulation, recovery, and
overall health.
Explain why sleep is an active cardiovascular state, rather than a period of cardiovascular
shutdown,
During sleep, cardiovascular regulation is reorganized rather than shut down, with characteristic changes in blood pressure (BP), heart rate (HR), and autonomic balance that differ from wakefulness
How does nightly repetition of sleep across decades influence long-term cardiovascular health
Because sleep recurs every night for decades, even modest sleep-related changes in autonomic and vascular control can accumulate into meaningful protection or stress for the cardiovascular system.
Describe how autonomic nervous system balance regulates heart rate and blood pressure
• HR is regulated by the balance of parasympathetic and
sympathetic nervous system activity
• BP is primarily regulated by sympathetic control of
cardiac output and vascular tone
What are the distinct roles of sympathetic and parasympathetic activity during sleep
During sleep, parasympathetic activity predominates in NREM sleep to lower heart rate and blood pressure and promote cardiovascular recovery (rest and recover).
Sympathetic activity is suppressed during deep sleep but reemerges during REM sleep and arousals to produce variability and enable rapid cardiovascular responses (activation and responsiveness)
Why does the suppression of sympathetic activity during NREM sleep lower cardiovascular
load, leading to reduced heart rate, vascular resistance, and blood pressure
With sympathetic suppression:
Heart pumps less frequently and with less force bc PNS is dominating
Blood vessels offer less resistance (vasodilation)
Since both HR and vascular resistance occur, this lowers BP (nocturnal dipping)
Overall:
↓ oxygen demand
↓ mechanical stress on vessels
↓ strain on the heart
what is nocturnal dipping and the morning surge
Nocturnal dipping is the normal decline in BP during
sleep, typically about a 10–20% reduction from daytime
levels
• The morning surge is the physiological rise in BP that
occurs in the first few hours after waking
explain how nocturnal blood pressure dipping is shaped by sleep state and circadian timing
How sleep state shapes it
During NREM sleep (especially slow-wave sleep):
↓ Sympathetic activity
↑ Parasympathetic activity
This leads to:
↓ Heart rate
↓ Vascular resistance
↓ Blood pressure
The deepest drops in BP:
Occur during slow-wave sleep
Align with periods of lowest sympathetic tone
So dipping is actively generated by sleep physiology, not just inactivity
explain how the morning surge is shaped by sleep state and circadian timing
How circadian timing shapes it
The circadian system prepares the body for wakefulness by:
↑ Sympathetic activity
↑ Hormones like cortisol
Upon waking:
The sympathetic system activates strongly
BP rises quickly
-The morning blood pressure surge occurs during the transition from sleep to wakefulness rather than a specific sleep stage, as circadian-driven increases in sympathetic activity combined with arousal from sleep rapidly elevate heart rate, vascular resistance, and blood pressure.
explain the linkage of sleep stage to sympathetic nerve activity and blood
pressure, including differences between NREM and REM sleep
NREM sleep (especially deep sleep / SWS)
↓ Sympathetic activity
↓ Blood pressure
BP is low and stable
Strongest cardiovascular recovery phase
REM sleep
↑ Sympathetic activity (more variable)
BP becomes more unstable
Transient spikes in BP
Cardiovascular system is more active, not resting
Explain why cardiovascular events cluster at specific times of day
Cardiovascular events cluster at specific times of day because circadian rhythms produce predictable increases in sympathetic activity, blood pressure, and heart rate—especially during the early morning surge—creating periods of heightened cardiovascular stress and vulnerability.
Evaluate how circadian disruption and misalignment, such as daylight saving time transitions
and rapid time zone travel, acutely alter cardiovascular regulation and risk.
The spring shift is followed by a sharp increase in heart attacks,
while the fall shift is associated with a reduction, indicating that
even a one-hour change in sleep and timing can acutely alter
cardiovascular risk
Why disruption increases risk (core mechanism)
Loss of coordinated timing between:
Autonomic activity
BP rhythms
Hormone release
This leads to:
Poor nighttime recovery
Inappropriate daytime or nighttime activation
Greater cardiovascular strain
Assess sleep duration as a modifiable regulator of blood pressure, using sleep extension
studies to distinguish causal effects from correlational associations
Sleep duration is a modifiable regulator of blood pressure because randomized sleep extension studies show that increasing sleep in habitual short sleepers lowers 24-hour systolic and diastolic blood pressure, demonstrating a causal effect (not correlational tho ) between short sleep and hypertension.
Why it’s hard to prove it’s correlational:
The studies cannot prove causation because short sleepers may also differ in stress, diet, activity, caffeine use, or health status. So correlation alone cannot tell us whether short sleep causes higher BP.
what is sleep arousal
Sleep arousal is a brief transition toward wakefulness marked by increased brain activity and
autonomic activation, often without full awakening
Explain how sleep fragmentation and arousals act as acute cardiovascular stressors and
how repeated sympathetic surges contribute to cardiovascular risk in conditions such as
restless legs syndrome and obstructive sleep apnea
Auditory arousals caused rapid increases in mean and diastolic blood pressure If arousals happen frequently
These repeated sympathetic surges lead to:
Sustained ↑ blood pressure
Increased vascular strain
Conditions like restless leg syndrome or sleep apnea:
Show repeated systolic and diastolic BP spikes
Restless leg is linked to higher rise in hypertension
Sleep apnea is linked to an increased risk of hypertension, arrhythmias, and sudden cardiac death
Explain what wearable sleep trackers measure and how they infer sleep.
• Wearables estimate sleep using movement (accelerometry)
and cardiovascular signals such as heart rate and heart rate
variability (HRV) measured by photoplethysmography
• These signals are used to infer sleep timing and stages, but
wearables do not measure brain activity, eye movements, or
muscle tone
• As a result, they are most reliable for sleep duration and timing
and less accurate for arousals and sleep architecture
Explain why sleep is a vulnerable state for breathing
Loss of wake-related neural drive
During wake, breathing is supported by voluntary
control, upright posture, cortical input, and behavioral
backup.
• Sleep removes these stabilizing influences, allowing
latent respiratory instability and disease to emerge.
Reliance on automatic brainstem control
In sleep:
Breathing is controlled only by:
Brainstem (medulla + pons)
This system:
Maintains breathing using CO₂, O₂, and pH signals
It is the only control system during sleep
Differentiate between voluntary and automatic control of breathing
Voluntary
Controlled by higher brain centers, primarily the cerebral cortex
• Allows conscious modification of breathing for speech, exercise, or breath holding
• Temporarily overrides brainstem respiratory signals
• Limited by rising CO₂, which restores involuntary breathing
Automatic control of breathing:
Controlled by the brainstem centers in the medulla and pons
• Generates the automatic respiratory rhythm
• Driven by blood gas and pH regulation through chemical and reflex inputs
• Maintains O₂ delivery and CO₂ removal
• Sole control during sleep
identify the roles of cortical input versus medullary and pontine respiratory center for breathing
Cortical input:
Function: Conscious control of breathing
Talking, singing, breath-holding, exercise adjustments
Can temporarily override automatic breathing
The medulla oblongata generates the basic respiratory rhythm
– The dorsal respiratory group (DRG) produces rhythmic inspiratory bursts that activate the diaphragm and external intercostals during quiet breathing
– The ventral respiratory group (VRG) is recruited during increased demand to drive actively or forcefully expiration
• The pontine respiratory group (PRG) is a set of
neurons in the pons that modulate the medullary
breathing rhythm
Apneustic activity promotes and prolongs inspiration
Pneumotaxic activity terminates inspiration
• Their balance shapes the timing and smoothness of
breathing
Describe how breathing patterns and respiratory muscle activity change across wake, NREM
sleep, and REM sleep
Wake
• Faster and more irregular breathing due to cortical and
behavioral input
NREM sleep
• Breathing slows and becomes more regular
REM sleep
• Breathing speeds up and becomes irregular
How do we interpret breathing states through EMGs?
Regular diaphragm rhythm → NREM
Irregular diaphragm + low airway tone → REM
High, variable activity everywhere → Wake
Sudden spikes → Arousal
Explain the role of serotonin in stabilizing breathing during sleep
• Serotonin stabilizes brainstem respiratory control
during sleep without generating the breathing rhythm
• Serotonergic neurons are in the raphe nuclei of the
brainstem and are part of the ARAS
• These neurons project to medullary respiratory
centers, including the DRG and VRG, to support
reliable breathing during sleep
explain serotonin’s effects on inspiratory neurons, chemoreflex sensitivity, and responses to hypoxia and hypercapnia.
• Serotonin increases the firing rate of inspiratory neurons in the DRG + strengthens diaphragm activation
Serotonin affects chemoreflex sensitivity
Enhances the responsiveness of these systems
Makes the brainstem more sensitive to changes in blood gases
This leads to faster and stronger ventilatory responses to disturbances
how does serotonin affect the response to hypoxia
• ↓ Arterial O₂ (hypoxia) → activation of peripheral
chemoreceptors in carotid and aortic bodies →
excitatory input to medullary respiratory centers
(DRG/VRG) → 5-HT amplifies inspiratory neuron
firing → increased rate and depth of breathing →
restoration of arterial O₂
How does serotonin affect the response to hypercapnia
↑ CO₂ (hypercapnia) → ↓ CSF pH (acidosis) →
activation of serotonergic neurons in the medullary
raphe → increased 5-HT release → stimulation of
medullary respiratory centers (especially DRG) →
increased rate and depth of breathing → CO₂
removal and restoration of blood pH
How does orexin provide tonic excitatory drive?
• Orexin increases breathing rate and strength by
exciting brainstem respiratory centers
• These effects occur even when CO₂ and O₂ levels
are normal
• This indicates orexin provides tonic excitatory drive
independent of chemoreflexes
how does orexin help with maintenance of upper airway stability
• Orexin increases the activity of upper airway motor circuits
• This helps maintain airway openness during wake
• Loss of orexin signaling during sleep reduces airway tone, increasing the risk of hypoventilation and
obstruction
how does orexin support state-dependent respiratory control
Orexin stabilizes brainstem respiratory control during
wake without generating the breathing rhythm
• Orexin neurons in the lateral hypothalamus (LH) are a
core component of the sleep-wake switch and arousal
network
• These neurons project widely, including to the respiratory
centers in the medulla and pons, supporting stable
breathing during wakefulness
Explain how do opioids disrupt respiratory control during sleep, including the μ-opioid receptor
effects on brainstem drive, REM sleep, and respiratory vulnerability.
Brainstem drive
μ-opioid receptors are expressed in the brainstem respiratory
control circuits
• Activation of μ-opioid receptors suppresses automatic
breathing (reduced breathing rate and depth)
Rem:
• Opioid use reduces both the percentage of REM sleep and the number of REM episodes across the night
• As a result, REM sleep becomes less frequent and less stable, altering normal sleep-state transitions
Respiratory vulnerability
opioid-mediated inhibition of the brainstem
respiratory centers
• Automatic breathing is less controlled
what is apnea
Apnea is a temporary cessation of breathing lasting
≥10 seconds during sleep
• It reflects either loss of respiratory drive (central
apnea) or airway collapse (obstructive apnea)
Describe how developmental vulnerability of brainstem serotonin systems contributes to
SIDS
Impaired serotonergic function may reduce autoresuscitation and arousal, increasing risk
during sleep
Differentiate obstructive sleep apnea and central sleep apnea based on the underlying
physiological mechanisms, not just symptoms or definitions
OSA
Obstructive sleep apnea arises from sleep-related reductions in upper airway muscle tone and structural airway vulnerability that cause inspiratory collapse despite preserved respiratory drive
CSA
Central sleep apnea results from instability or suppression of brainstem respiratory control, often due to altered chemoreflex sensitivity and delayed CO₂ feedback, leading to absent inspiratory effort (no drive to breathe)
What is obstructive sleep apnea
OSA occurs when the upper airway repeatedly collapses or becomes blocked during sleep, despite normal neural signals to breathe
Describe how anatomical factors contribute to airway collapsibility in obstructive sleep
apnea
Anatomical factors increase airway collapsibility in obstructive sleep apnea by reducing airway size, increasing surrounding pressure, and weakening structural support.
A smaller upper airway provides less space for airflow, so even minor reductions in muscle tone during sleep can lead to complete obstruction.
Increased soft tissue volume, such as enlarged tonsils, tongue, or soft palate, further narrows the airway and adds internal pressure that promotes collapse.
Craniofacial structure also plays a key role, as features like a recessed jaw or shortened lower face reduce the skeletal support that normally helps keep the airway open.
In addition, a larger neck circumference, often associated with excess fat deposition around the airway, applies external pressure that further compresses and narrows the airway.
Together, these anatomical factors create a structurally narrow and unstable airway that becomes highly susceptible to collapse during sleep when neuromuscular tone is reduced, leading to obstructive events.
What is central sleep apnea
Breathing pauses occur due to reduced or absent neural drive from the brainstem respiratory centers, not airway obstruction
Explain how instability in brainstem ventilatory control leads to central sleep apnea,
including reduced versus excessive chemosensitivity and periodic breathing patterns
Central sleep apnea arises from instability in brainstem ventilatory control, particularly in how the system regulates breathing in response to changes in carbon dioxide (CO₂). Under normal conditions, medullary respiratory centers adjust ventilation to maintain stable CO₂ levels, increasing breathing when CO₂ rises and decreasing it when CO₂ falls. In central sleep apnea, this feedback system becomes unstable in two key ways.
In cases of reduced chemosensitivity, the brainstem has a blunted response to rising CO₂, resulting in insufficient stimulation of inspiratory neurons and a failure to generate adequate respiratory drive, which can lead to periods of absent breathing.
In contrast, excessive chemosensitivity produces an overactive response to small increases in CO₂, causing hyperventilation that lowers CO₂ below the threshold required to sustain breathing.
When CO₂ falls too low, respiratory drive is temporarily suppressed, resulting in apnea. As CO₂ rises again, the system overcorrects once more, creating a repeating cycle of hyperventilation followed by apnea known as periodic breathing, commonly seen as Cheyne–Stokes respiration. These oscillations are often exacerbated by delayed feedback in the system, such as prolonged circulation time in heart failure, which causes mismatches between blood gas levels and the brainstem response. During sleep, the absence of wake-related neural input further increases reliance on this unstable automatic control system, making these fluctuations more pronounced and increasing the likelihood of central apneas.
Compare major treatment strategies by identifying their primary physiological targets, such
as airway splinting, ventilatory support, neuromuscular activation, or anatomical
modification.
Airway Splinting
– Continuous positive airway pressure (CPAP) delivers
continuous positive pressure that splints the airway open
– Oral appliances advance the mandible to enlarge the upper
airway
– Oral pressure therapy (OPT) pulls the tongue and soft
palate forward using negative oral pressure
Ventilatory Support
– Bilevel positive airway pressure (BIPAP) provides
inspiratory pressure support and reduces work of breathing
– Adaptive servo ventilation (ASV) adjusts pressure support
breath by breath to stabilize ventilation
Neuromuscular activation
- Hypoglossal nerve stimulation activates tongue muscles
to maintain airway patency
Anatomical Modification
These interventions permanently alter airway structure
to reduce obstruction.
– Surgery (uvuloplasty) removes or repositions obstructing
tissue or modifies airway anatomy
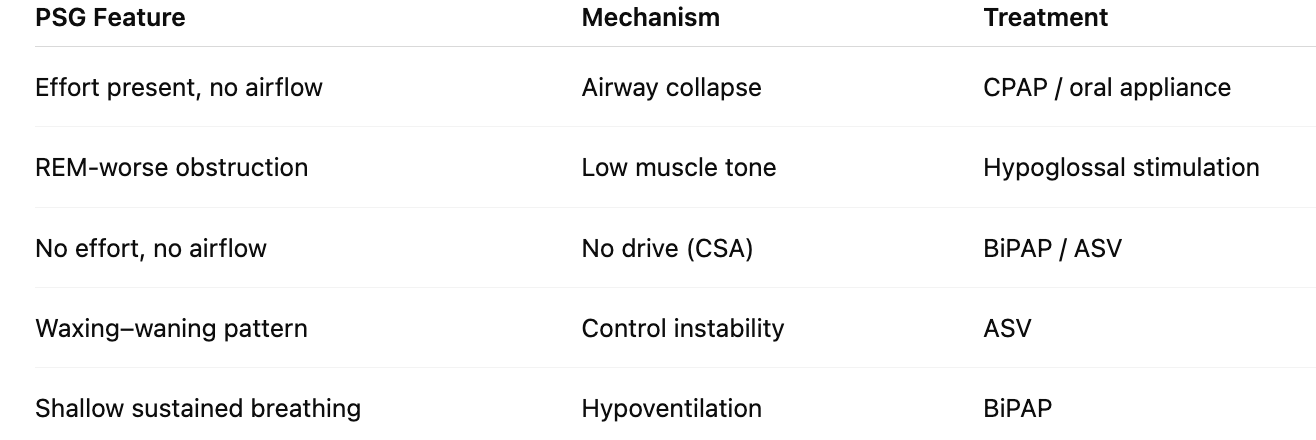
Predict appropriate treatment approaches for different patient phenotypes based on PSG
features and underlying pathophysiology rather than disease labels alone
Explain how thermoregulation is controlled by the hypothalamus
Thermoregulation by the hypothalamus
• The preoptic area of the hypothalamus (POA) is the
primary integrator of thermal information
– Receives input from peripheral & central thermoreceptors
– Generates coordinated autonomic and behavioral responses
Peripheral effectors regulate production through
• Cutaneous vasodilation
– Warm-sensitive POA neurons
(primarily GABAergic) increase
firing as local temperature rises →
GABAergic inhibition reduces
sympathetic vasoconstrictor tone →
increased skin blood flow promotes
heat loss at the body surface
• Sweating
– Warm-sensitive POA neurons
activate downstream sympathetic
pathways to eccrine sweat glands
→ evaporative cooling dissipates
excess heat
peripheral effectors regulate heat loss through
• Cutaneous vasoconstriction
– Cold-sensitive POA neurons increase
activity as local temperature falls →
increased sympathetic vasoconstrictor
tone → reduced skin blood flow limits
heat loss
• Shivering thermogenesis (ST)
– Cold-sensitive POA pathways increase
somatic motor output → rhythmic
skeletal muscle contractions generate
heat
• Non-shivering thermogenesis (NST)
– Cold-sensitive POA pathways increase
sympathetic drive to brown adipose
tissue → increased metabolic heat
production
define the thermoneutral zone
The thermoneutral zone (TNZ) is the range of ambient temperatures where:
The body maintains core temperature
With minimal metabolic effort
predict how moving above or below the thermoneutral zone alters metabolic cost and sleep architecture

Describe how the circadian regulation of body temperature and melatonin release promotes
sleep onset.
Circadian regulation promotes sleep onset through the coordinated timing of a
decline in core body temperature
and an increase in melatonin release.
Melatonin
increases sleep propensity but also
facilitates peripheral vasodilation, particularly in distal regions such as the hands and feet, which enhances heat loss from the body. This redistribution of heat from the core to the periphery accelerates the decline in core body temperature. The combined effects of reduced core temperature, increased heat loss, and rising melatonin levels create a physiological state that lowers metabolic activity and promotes the transition from wakefulness to sleep.
Explain how distal skin vasodilation and the distal–proximal temperature gradient facilitate
sleep initiation.
Distal skin vasodilation increases blood flow to the hands and feet, promoting heat transfer from the core to the periphery and raising distal skin temperature, which creates a positive distal–proximal temperature gradient that reflects enhanced heat loss; this redistribution of heat lowers core body temperature and facilitates sleep initiation by promoting a physiological state conducive to sleep
Compare thermoregulatory control during NREM and REM sleep, including which responses
are preserved, reduced, or suspended.
• NREM sleep
– Body shifts toward heat loss at sleep onset
– Thermoregulation remains active but less sensitive
– System prioritizes thermal stability and low energy use
– Lower metabolism and cooler core temperature support
restoration
• REM sleep
– Active thermoregulation is suspended (not occuring)
– Body temperature becomes less actively regulated
Predict how ambient temperature and brain temperature differently affect NREM and REM
sleep.
Ambient
• Below the TNZ, NREM predominates while REM is suppressed
• Within the TNZ, both NREM and REM sleep are maximized
• Above the TNZ, both NREM and REM decline, with REM most affected
Brain temp
• Brain temperature is highest during wake due to sustained neuronal firing and high metabolic demand
• During NREM sleep, the brain temperature falls as neuronal activity and metabolism decrease
• During REM sleep, brain temperature rises again due to intense cortical activation and increased cerebral blood flow, despite muscle atonia
what are night sweats
Night sweats are episodes of excessive sweating
during sleep that soak clothing or bedding
• They occur even when the room is not hot, and the
individual is not overdressed
• Night sweats reflect abnormal activation of
thermoregulatory or autonomic systems during sleep,
not simple overheating
Apply thermoregulatory principles to explain real world phenomena such as night sweats,
exercise timing, and the effects of cooling sleep environments.
Thermoregulatory principles explain that sleep is promoted by a decline in core body temperature and increased peripheral heat loss, so excessive or dysregulated heat loss can cause night sweats and disrupt sleep, elevated core temperature from late exercise can delay sleep onset, and appropriately cool sleep environments enhance heat dissipation and facilitate sleep initiation, whereas extreme temperatures can impair sleep by increasing thermoregulatory demands.
