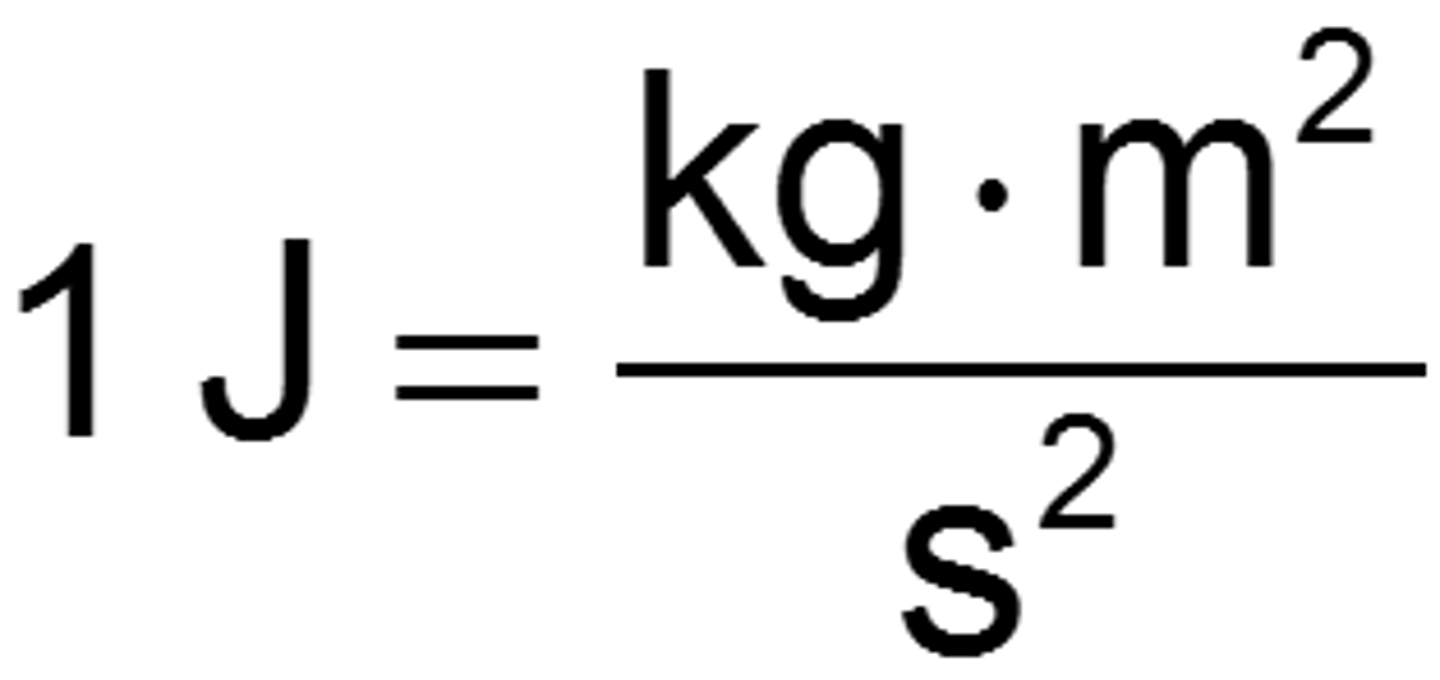MYE 24.25 L4 Vocabulary Diagram | Quizlet
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|

59 Terms
TERM
Low Precision, Low accuracy
DEFINITION
not repeating, not in the same place
TERM
High Precision, low accuracy
DEFINITION
Has repeated the darts in the same place, but is not hitting the bullseye
TERM
High accuracy, low precision
DEFINITION
All are close to the bullseye, but not all in the same place
TERM
High accuracy and high precision
DEFINITION
Darts are clustered in the same place, and are clustered in the bullseye
Accuracy
How close to a correct answer a measurement is to a true or known value
Calibration
How close a measuring tools markings are to a known, and correct, value
Precision (tool)
The smallest value the tool (ruler, scale, etc.) measures
Estimated digit (digital tools)
The smallest value measured by the tool (digit place farthest to the right on the display)
Tare (digital tools)
Reset the digital device to zero before massing, measuring, etc.
Mass
A measure of the amount of matter in an object, which can be converted into energy through a variety of means: nuclear or gravitational potential.
1st law of thermodynamics
Energy cannot be created or destroyed, it is constant in context of the universe.
Radiation
The direct transfer of energy by electromagnetic waves, does not need matter to travel through.
Work
The physics definition: Force * displacement. Measured in joules (J).
Surroundings
Anything that is not part of the (energy) system being studied at a particular time
Internal Energy
The sum of all the energy (both potential and kinetic energy) in a system
Chemical energy
A potential energy stored in the bonds of atoms and molecules. Batteries, biomass, petroleum, natural gas, and coal are examples of chemical energy. Chemical energy is converted to thermal energy when people burn wood in a fireplace or burn gasoline in a car's engine.
Mechanical energy
A potential energy stored in objects by tension. Compressed springs and stretched rubber bands are examples of stored mechanical energy.
Gravitational energy
A poteintial energy stored in an object's height. The higher and heavier the object, the more gravitational energy is stored. When a person rides a bicycle down a steep hill and picks up speed, the gravitational energy is converting to motion energy. Hydropower is another example of gravitational energy, where gravity forces water down through a hydroelectric turbine to produce electricity.
Radiant energy
A kinetic electromagnetic energy that travels in transverse waves. Radiant energy includes visible light, x-rays, gamma rays, and radio waves. Light is one type of radiant energy. Sunshine is radiant energy, which provides the fuel and warmth that make life on earth possible.
Motion energy
The kinetic energy associated in the movement of objects. The faster they move, the more energy is present. It takes energy to get an object moving, and energy is released when an object slows down. Wind is an example of motion energy. A dramatic example of motion energy is a car crash—a car comes to a total stop and releases all of its motion energy at once in an uncontrolled instant.
Electrical energy
A kinetic energy delivered by tiny charged particles called electrons, typically moving through a wire. Lightning is an example of electrical energy in nature
Pie Chart
Use pie charts to compare parts of a whole. They depict a single point in time rather than changes over time. A pie chart can display percent composition, such as the composition of air. The area of each sector is proportional to a percentage.
Line graph
Line graphs show continuous data over periods of time. Before setting up a line graph, determine the dependent and independent variables. The independent variable may be a scalar (numeric) or ordinal (order) quantity. The independent variable always goes on the x-axis. If an experiment requires taking data points every 5 seconds for a minute, or every day for a month, it is appropriate to use a line graph.
Area Graph
Similar to a line graph, an area graph is used to track changes over time for one or more groups. An area chart works well for data that both changes over time and indicates where the sum and the parts are important.
Atom
the smallest particle of an element that retains the properties of the element
(chemical) bonds
the attractive force that holds atoms or ions together
carbon
a nonmetal element, not a good conductor

copper
a metal element that is a good conductor of electricity

electrons
negatively charged subatomic particles
element
A pure substance made of only one kind of atom
incandescent (light bulb)
-A source of electric light that works by incandescence
-An electric current passes through a thin filament, heating it until it produces light
-The enclosing glass bulb prevents the oxygen in the air from reaching the hot filament, which otherwise would be destroyed rapidly by oxidation

molecules
Groups of two or more nonmetallic atoms held together by covalent bonds
LED light bulbs consume up to ___% less energy than a standard incandescent lightbulb
The Light bulb saves the most money and is the most efficient, where most (90%) of the electrical energy goes into producing light

Precision (data)
The degree to which repeated measurements show the same result.
Estimated digit (analog tools)
usually 1/10 of the smallest measurement of the tool, unless directed differently by your instructor
Thermodynamics
The study of Energy, Heat, and Work
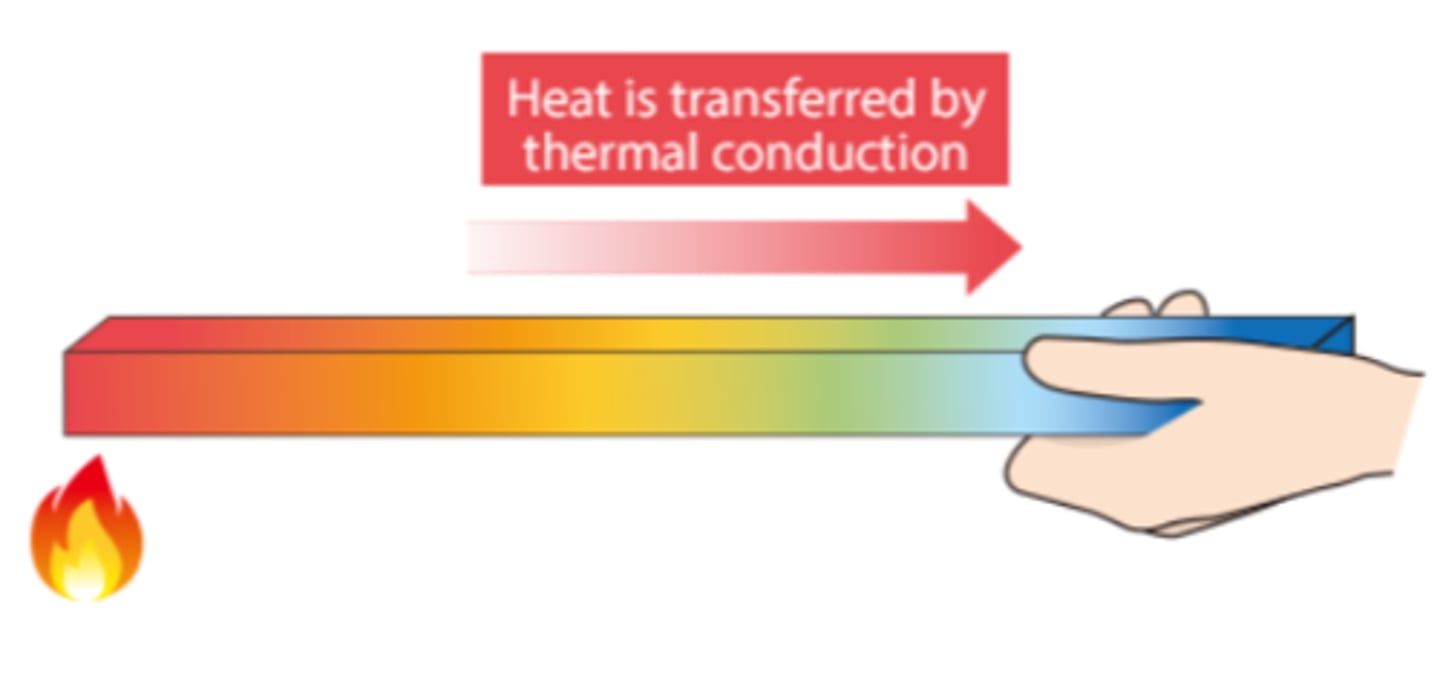
Conduction
Form of heat transfer where thermal energy is directly transferred between molecules through molecular collisions or direct contact.
System
The area being studied regarding energy. It can be as big or as small as needed. The observer defines the system.
Nuclear Energy
A potential energy stored in the nucleus of an atom—the energy that holds the nucleus together. Large amounts of energy can be released when the nuclei are combined or split apart.
Thermal energy
Also known as heat, is the kinetic energy that comes from the movement of atoms and molecules in a substance. Heat increases when these particles move faster. Geothermal energy is the thermal energy in the earth.
Sound
A kinetic energy that is the movement of energy through substances in longitudinal (compression/rarefaction) waves. Sound is produced when a force causes an object or substance to vibrate. The energy is transferred through the substance in a wave. Typically, the energy in sound is smaller than in other forms of energy.
Energy
The ability to do work or produce heat, measured in joules (J)
Heat
The transfer of energy through thermal means: convection, conduction, and convention. Measured in joules (J)
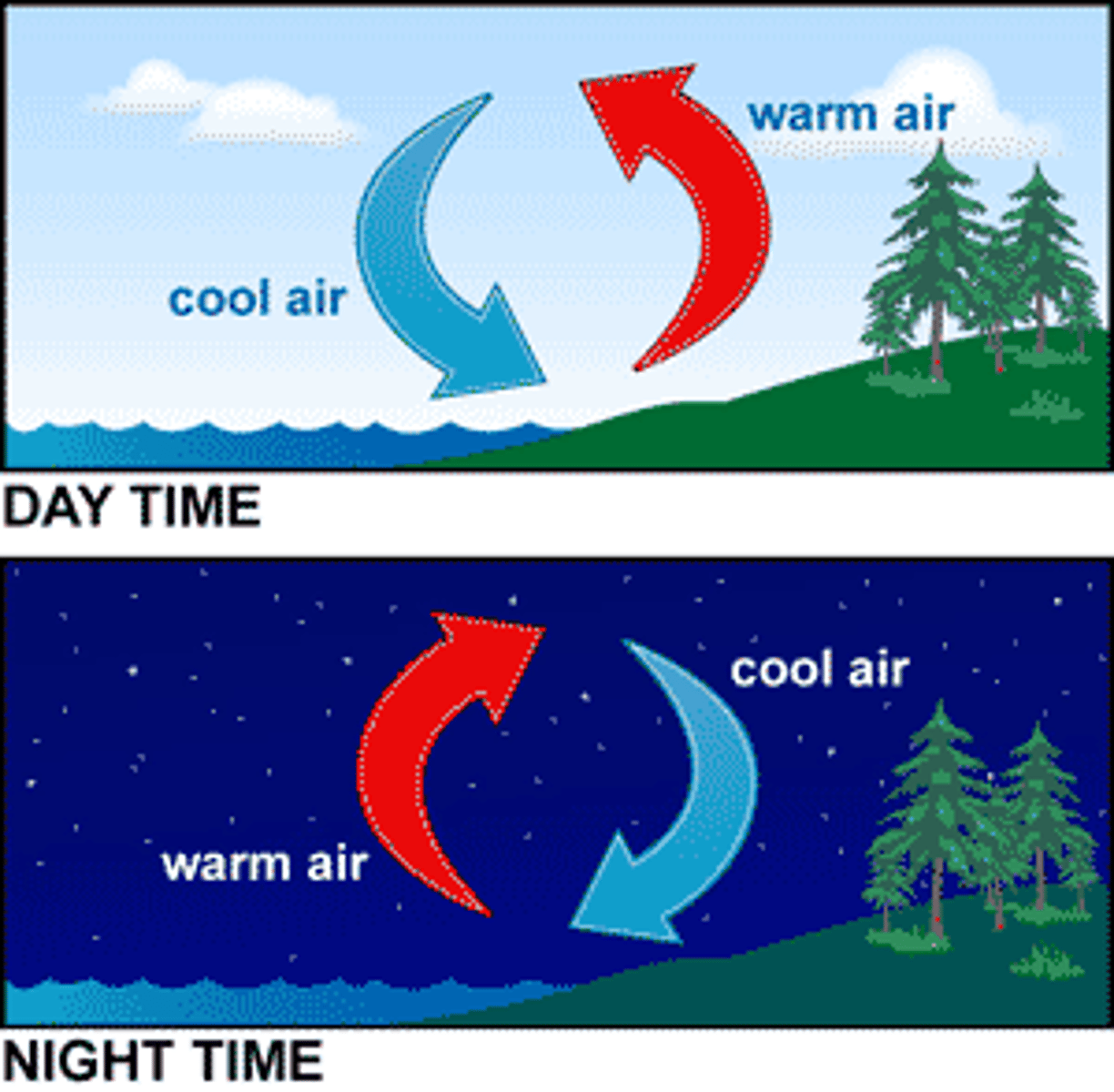
Convection
The transfer of thermal energy by the movement of a fluid. Warm fluids rise, and cold fluids sink.
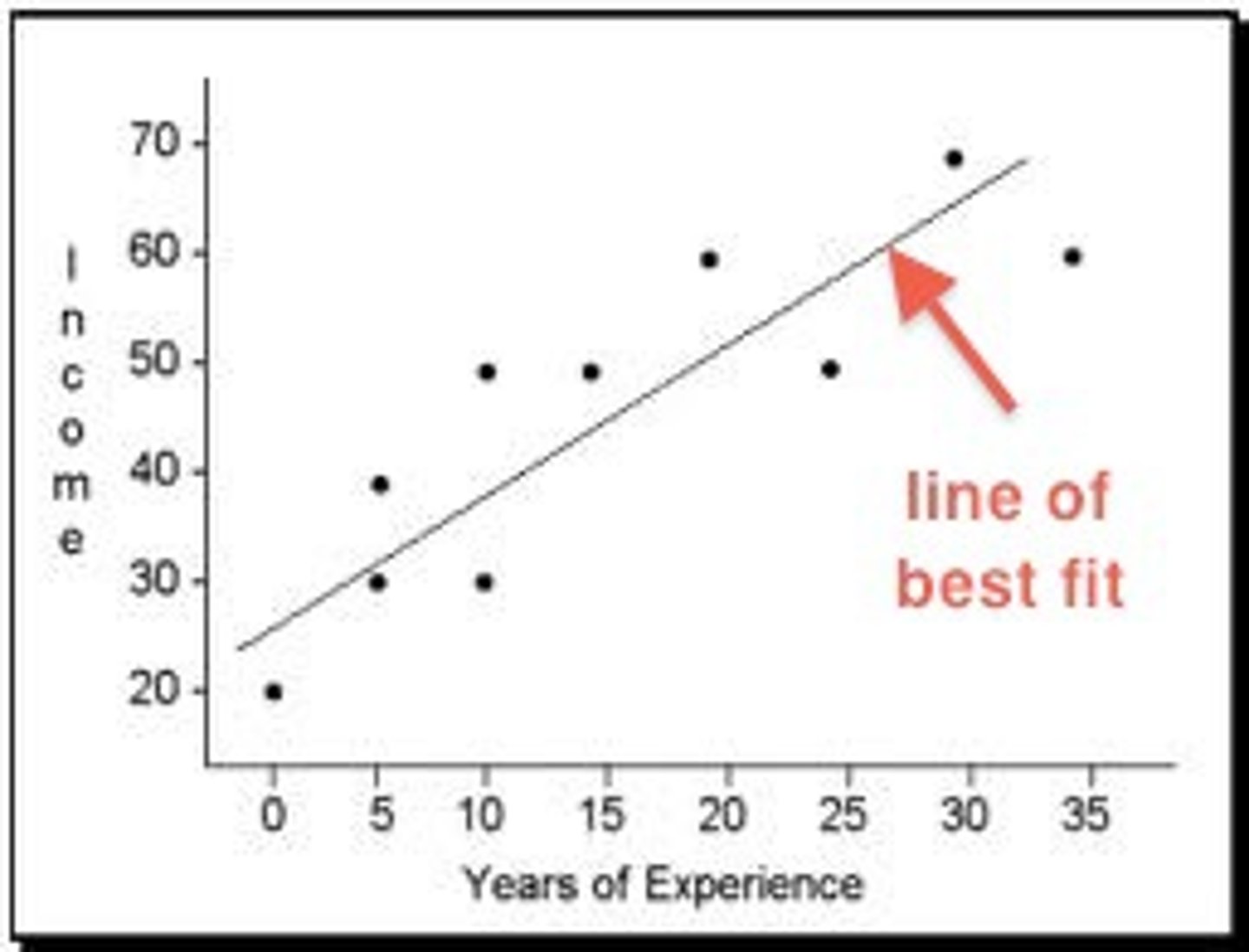
X-Y Scatter Plot
A scatter plot is often used to show relationships between independent and dependent variables. Instead of connecting data points with a line, a best-fit line can be used to find a trend in data. Scatter plots are frequently used for creating a standard curve in chemistry, as is shown in the graph below. An equation for the trend line can be determined
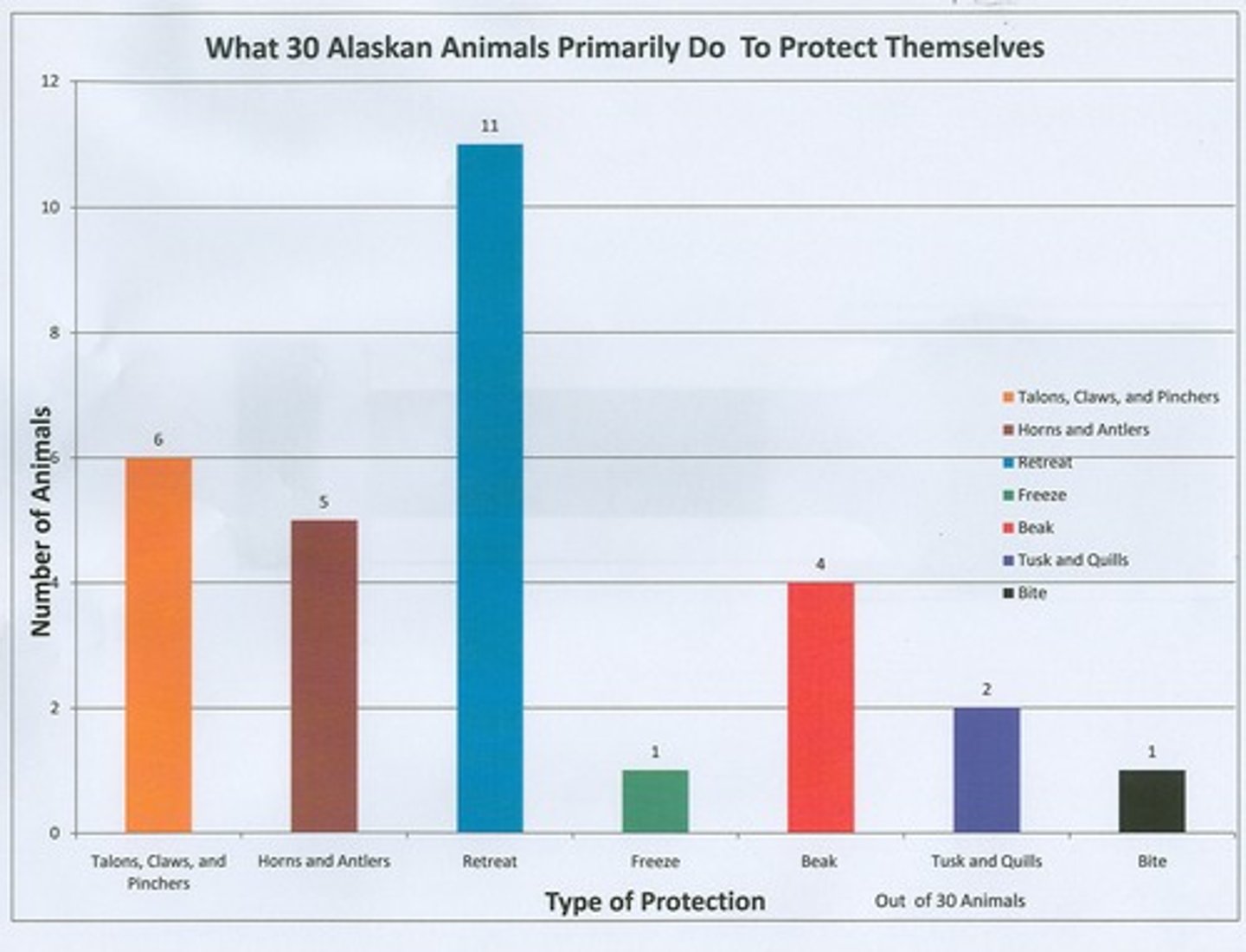
Bar Graph
Bar graphs are used to compare different groups or to track a change over a period of time. Bar graphs compare data that do not continuously change over time, and they are helpful when you need to compare information collected by counting. A bar graph should be used if you are not looking for trends over time and when the items are not part of a whole.
Transform
to change in form or appearance
Transfer
to move from one place to another
Electrical resistance
The property of a material that resists the flow of charged particles through it; measured in ohms
Open system
A system in which exchanges of matter or energy occur across system boundaries
Closed system
A system in which matter and energy exchanges do not cross system boundaries into the surroundings. And the objects and energies in the surroundings does not affect the system.
Fluorescent light bulb
A ligh bulb that contains a gas that gives off ultraviolet radiation when electricity flows through it; about 30% of electrical energy is changed into light.

Volt
A volt is a measurement of how many joules of energy a electric charge can carry
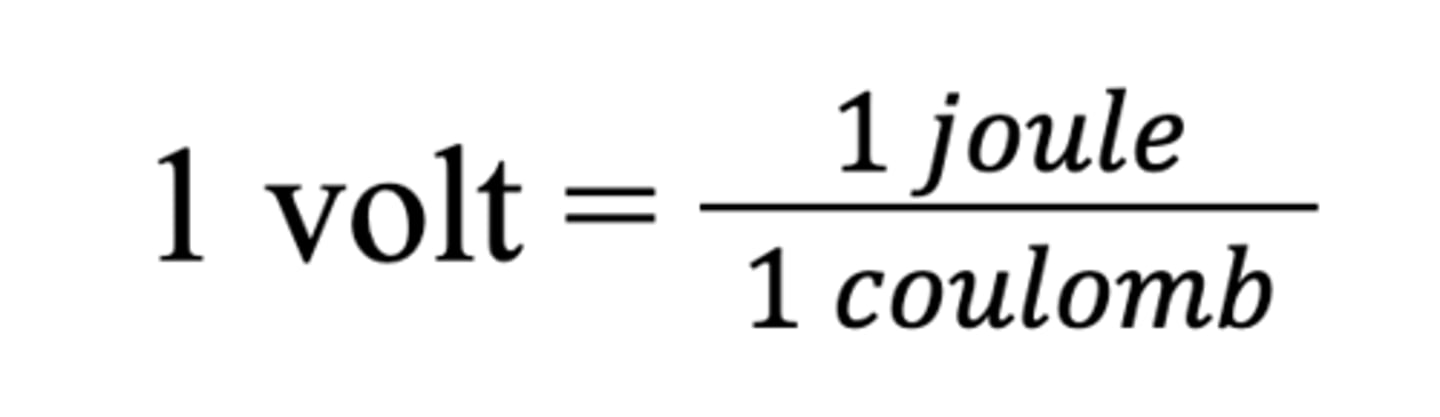
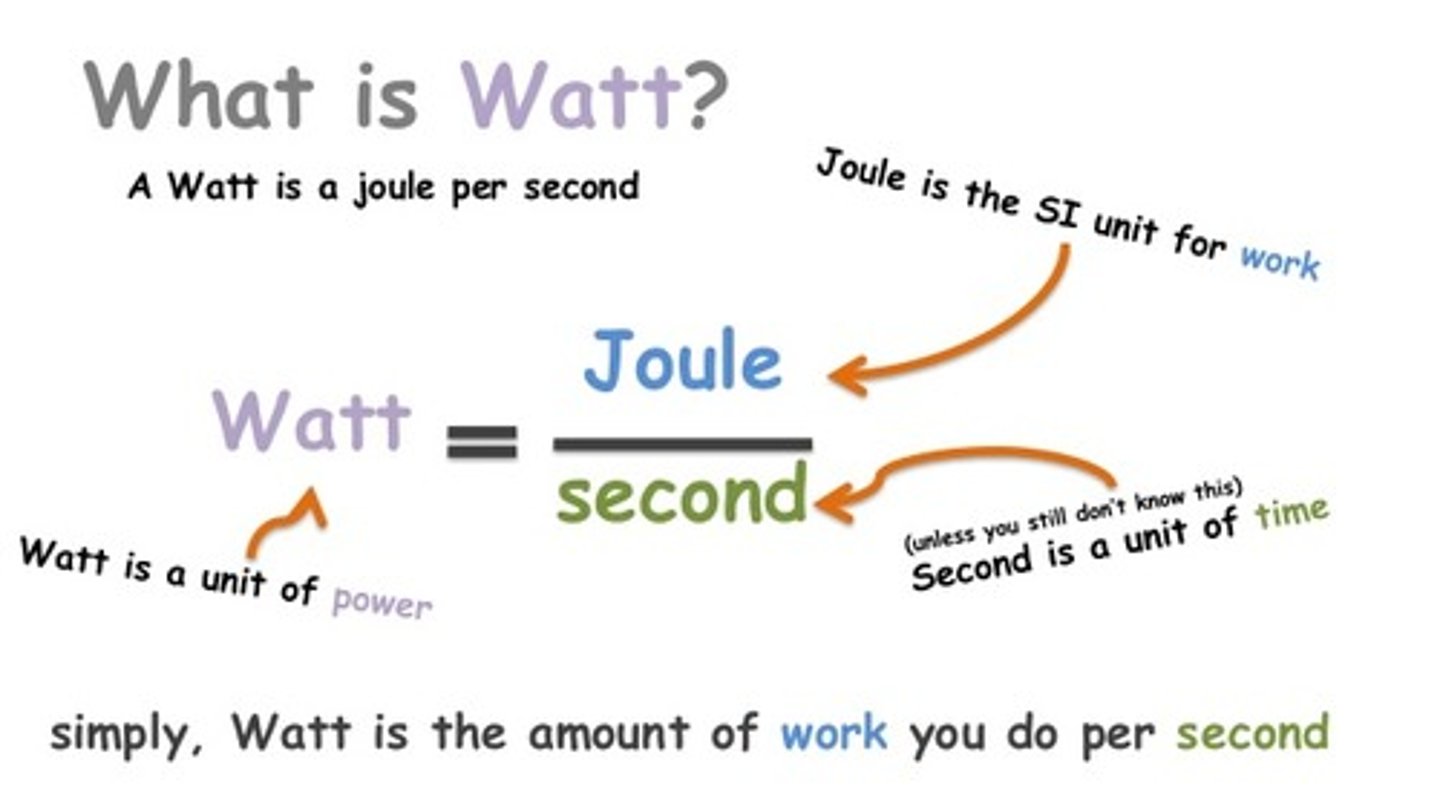
Watt
the SI unit of power, equal to one joule per second
Ampere
a unit of electric current equal to a flow of one coulomb per second.
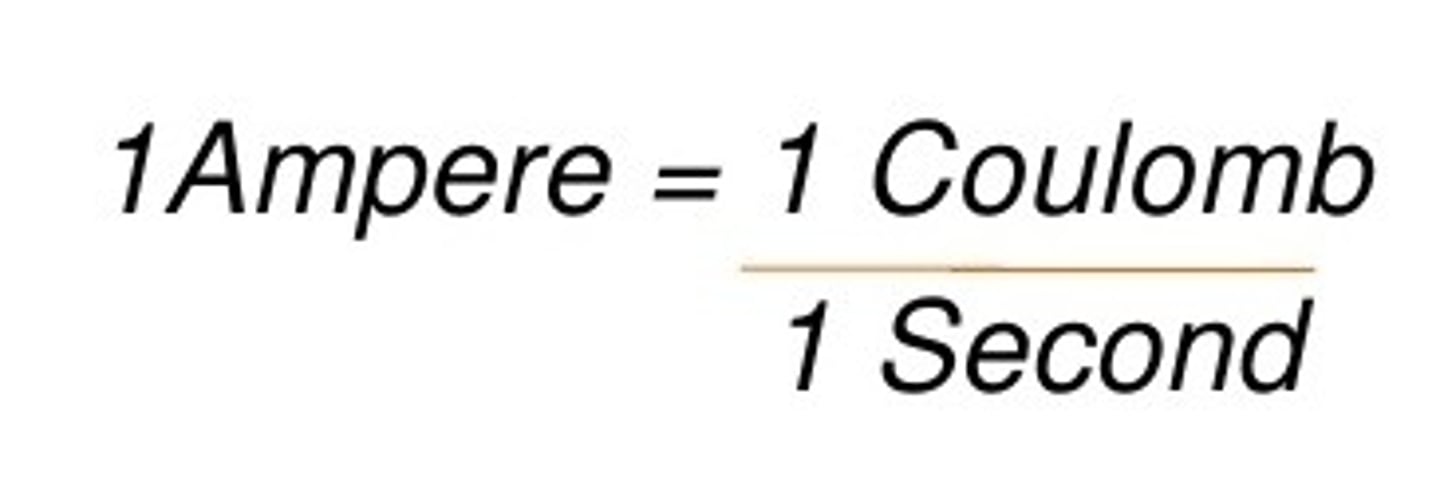
Coulomb
SI unit of electric charge
Current
A flow of electric charge through an electrical system, measured in amperes
Power
how much energy is used each second
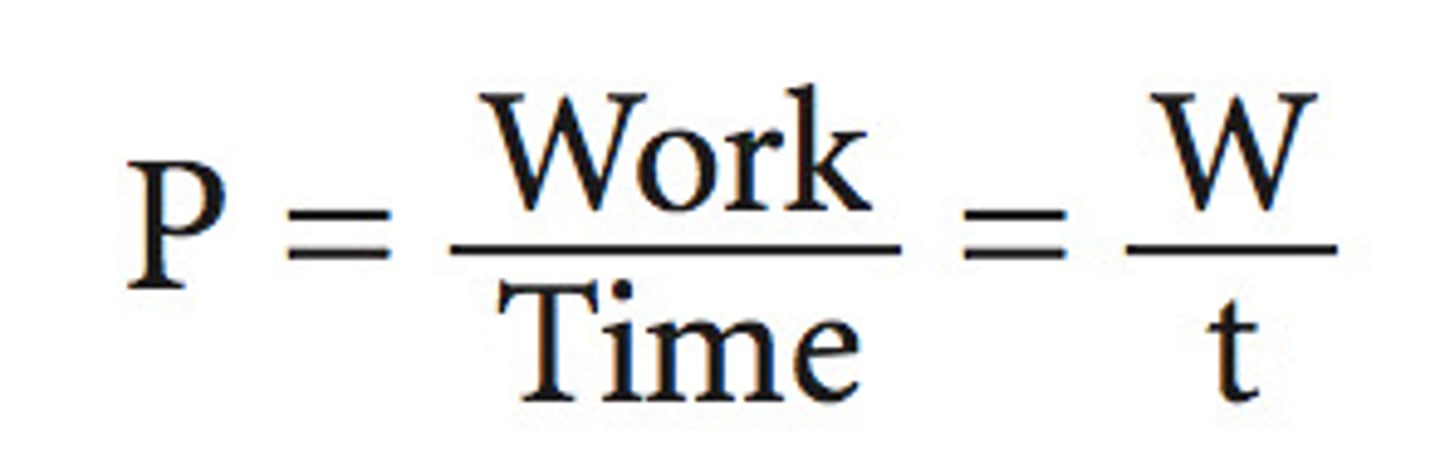
Joule
SI unit of energy