Reactions of Aldehydes & Ketones
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
*Basic conditions
Carbonyl —(1) Nuc, (2) H3O+—→
Nuc adds across C=O bond
*Acidic conditions
Carbonyl —(1) H3O+, (2) H—Nuc, (3) Base—→
Nuc adds across C=O bond
Ketone + H2O →
Ketone hydrate
Aldehyde + H2O
Aldehyde hydrate
Ketone + 2R—OH →
Acetal
Ketone + R—OH →
Hemiacetal
Aldehyde + ethylene glycol →
Cyclic acetal
2 Carbonyls —(1) ethylene glycol, (2) NaBH4, (3) H3O+—→
1 carbonyl protected, other reduced
Ketone —(1) H+, (2) Amine, (3) Anime, (4) H+, (5) deprotonation—→
Imine (C=N) or Schiff base
Imine —NaBH3CN or NaBH4—→
Adds two H’s, amine product
Aldehyde —H2CrO4—(Oxidation)→
Carboxylic acid
Ketone —Zn(Hg), HCl, H2O—→
Removal of O, replaced with H’s
Ketone —(1) hydrazine, (2) KOH, 𐤃—→
Removal of O, replaced with H’s
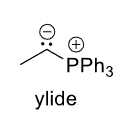
R—LG —(1) :PPh3, (2) Nuc—→
Ylide
Alkene product =>
Cleave C=C, ketone + ylid
Ylide =>
Replace Ph3 with Br, replace lone pair with H