micro chapter 10
1/62
Earn XP
Description and Tags
metabolism
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
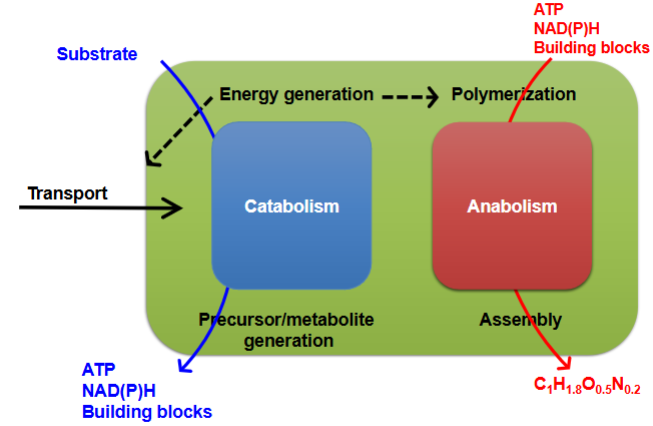
catabolism leads to the production of
key precursors: PEP, F6P, Acetyl-CoA from respiration, NAM/NAG (for peptidoglycan synthesis)
reducing power: electron carriers (NADH, FADH2)
energy: ATP
catabolism provides the ___ for anabolism
ATP
NAD(P)H
building blocks
redox reactions
explain the movement of electrons from donor to acceptor
standard reduction potential
describes the ability to gain electrons
negative E
donate electrons freely
no energy needed
positive E
accepts electrons freely
spontanteous, large negative delta G
chemotrophs
source of energy is by breaking chemical bonds, capture energy as ATP
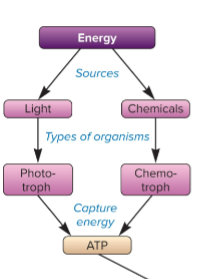
phototrophs
source of energy is from light, capture energy to ATP
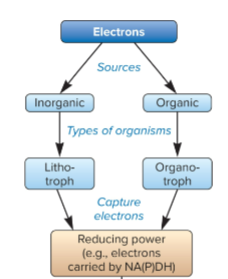
organotroph
source of electrons are from organic compounds, captured electrons carried NAD(P)H
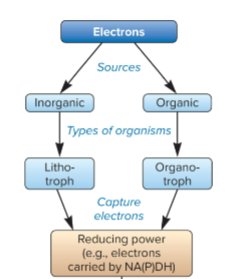
lithotrophs
source of electrons are from inorganic compounds, captured electrons carried NAD(P)H
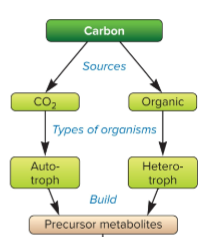
autotrophs
source of carbon is inorganic (from CO2), fixes CO2 into own source of carbon
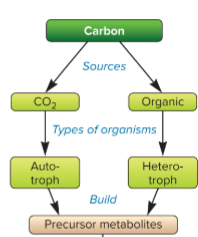
heterotrophs
carbon source is organic (glucose) and is consumed and made by another organism
photolithoautotroph
light energy source
co2 carbon source
inorganic electron source
photoorganoheterotroph
light energy source
organic carbon source
organic electron source
chemolithoautotroph
inorganic chemical energy source
co2 carbon source
inorganic electron source
important for nutrient cycling, biogeochemical cycels, and ecology
chemolithoheterotroph
inorganic chemical energy source
organic carbon source
inorganic electron source
chemoorganoheterotroph (most nonphotosynthetic organisms)
organic chemical energy source, often same as carbon source
organic carbon source
organic electron donor, often same as carbon source
glycolysis is actually 3 pathways
glycolysis (EMP pathway)
pentose phosphate pathway
ED pathways
glycolsis pathways are overall the conversion of
conversion of glucose to GAP by 3 different routes, eventually production of pyruvate
EMP pathway
glucose to 2 g3p (invests 2 ATP)
2 g3p oxidized to 2 pyruvate (payout 4 ATP, 2NADH)
(net 2 ATP)
substrate level phosphorylation
a direct metabolic process that produces ATP by transferring a phosphate group from a high-energy substrate molecule to ADP
done by kinases
EMP contributes to anabolic pathways
make amino acids, PLs, sugars (NAM, NAG)
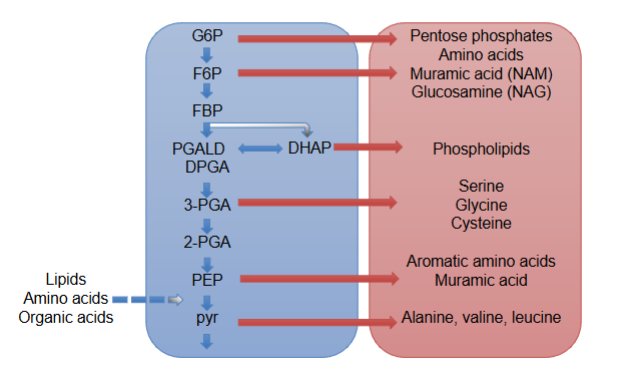
pentose phosphate pathway
makes NADPH, no ATP generated
oxidation-decarboxylation of G6P to Ru5P
Isomeration of ribulose to X5P and R5P (which makes nucleotides)
sugar rearranged, these sugars can make NAM and NAG, or sugars can funnel back into EMP to make pyruvate
source of precursors for some amino acids
some organisms rely solely on PPP
ED pathway
glucose → gluconate (making NADPH, using 1 ATP) → pyruvate and GAP
gap → pyruvate (making 2 ATP)
makes NADPH, NADH, net 1 ATP
only seen in prokaryotes
some organisms rely only on ED
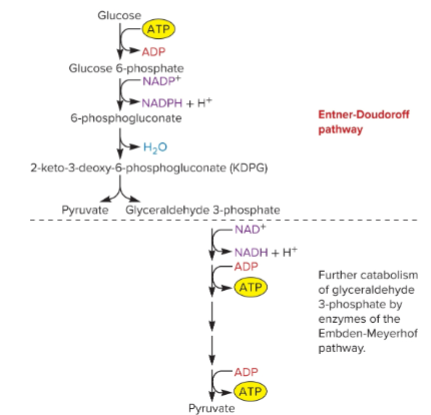
why is ED useful?
gives NADPH
use other sugars (aldonic acids),
allows for nutritional diversity, giving a competitive advantage
all these glycolysis pathways lead to the formation of..
pyruvate
reduced electron carriers
pyruvate oxidation
oxidatvely decarboxylation
pyr to acetyl coA, making NADH
TCA
acetyl coa input, generating 2 CO2, 3 NADH, 1 FADH2, 1 GTP/ATP
intermediates in TCA can make other amino acids
anaplerotic pathways
metabolic processes that replenish TCA intermediates when they get shuttled off, ensuring its continuous operation for energy production and biosynthesis
other carbs (breaking down lactose → galactose)
tagatose pathway, make gap
leloir pathway, uses UDP to exchange gal for glucose, proceeds to glycolysis to make gap
gap → lactic acid
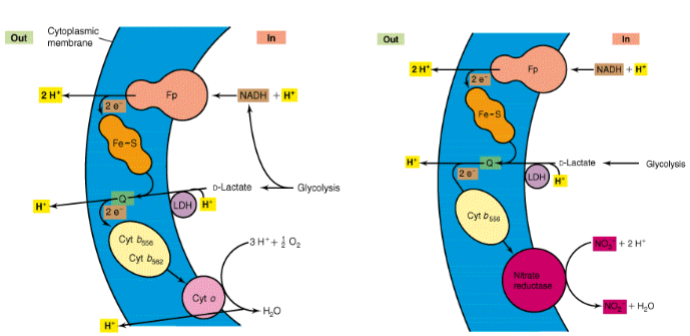
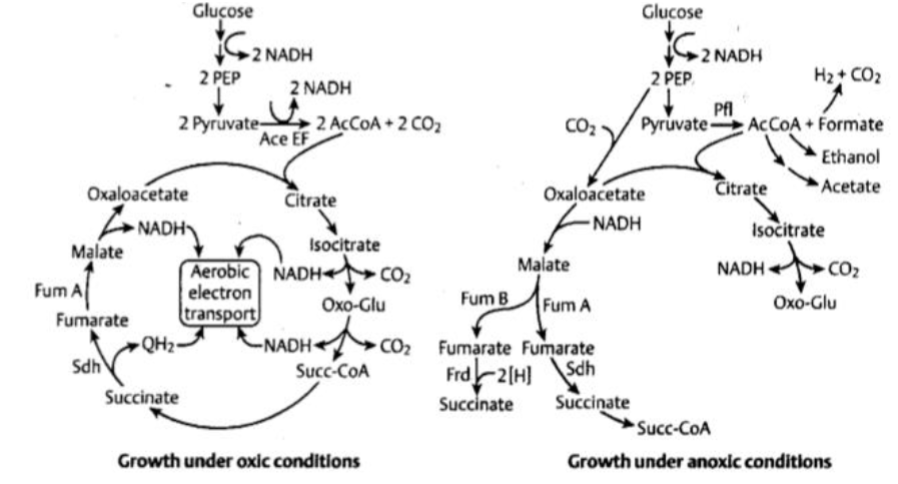
aerobic respiration/ ETC in bacteria
less complexes, shorter ETC, less energy made
protons pumped across plasma membrane instead of mitochondrial membrane, causing surrounding environment to change pH
bacteria can change its ETC complexes based on environmental conditions
ecoli uses two different cytochomr oxidases
2 pathways electrons can go during enviroment changes
high o2: use bo branch/complex
low o2: use bd branch, does not pump H+
terminal electron acceptor in aerobic respiration is usually
oxygen, highest reduction potential (E)
terminal electron acceptors in anaerobic conditions
nitrate, sulfate, other organic electron acceptors
smaller reduction potenital, not as good as O2, does not produce as much energy
organisms that only differ by aerobic or anaerobic respiration are still
chemoorganoheterotrophs
aerobic v anaerobic respiration in ecoli (a facultative anaerobe)
in aerobic: uses cyt O, pumps H+
in anaerobic: uses nitrate reductase instead of cyt O, needs NO3 present, no protons pumping, but less energy produced bc less proton motive force
nitrate reduction in paraoccus denitrificans
full denitrification (NO3- → N2
Com 1 → Coq → Nar (nitrate reductase) → Com 3 → cyt c → Nir (nitrite reductase) → Nor (nitric oxide reductase) → Nos (nitrous oxide reductase)
nitrate reduction chemical steps and enzymes
NO3 → NO2 → NO → N2O → N2
Nar → Nir → Nor → Nos
geobacter sulfurreducens
has a special conductive pilli to transfer e- to Fe 3+ → Fe2+
some species use ___ as a terminal electron acceptor, reducing it to be nontoxic
HAsO4 2-
anaerobic e- transfer pathways in shewanella oneidensis
gram -, rxns in periplasm
uses cym A: transfers e- to various e- acceptors
some e- acceptors are outside the cell: iron reductase and dmso (prevents toxic byproducts)
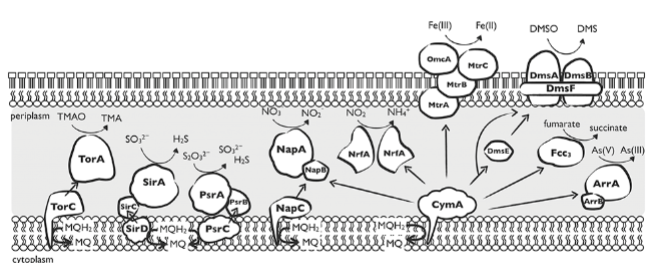
why do organisms like shewanella oneidensis have so many pathways?
enviroment is variable
hard to find particular resources, use what you can
full ___ makes more ATP than partial __ because
denitrification makes more ATP than partial denitrication (NO3→ NO2-)
because there is a bigger difference between reduction potenitals when reducing down to N2
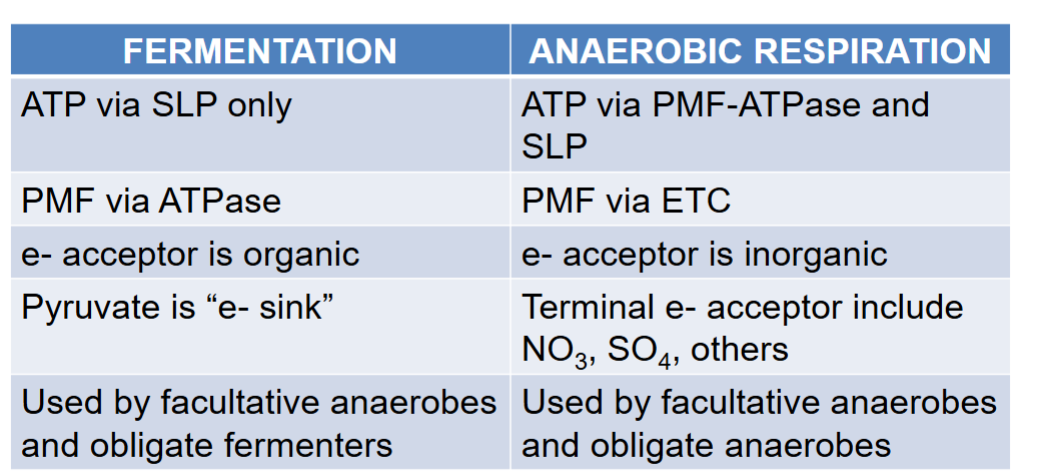
fermentations occur in bacteria that cannot
have an ETC
“fermenters”
3 types of facultative anaerobes depending on metabolism
1) ferment when anoxic (fermenters)
2) use anaerobic resp when anoxic
3) use anaerboic resp first, then switch to ferm when all TEAs run out
the main purpose of fermentation is to produce ___
produce NAD+ to be reused in glycolysis
glycolysis continues to make ATP (even tho not much)
SOMETIMES PRODUCE ATP VIA SUBSTRATE LEVEL P USING SLP
big problem for fermenters (abt oxidation?)
how to re-oxidize NADH without ETC?
if no NAD+ made, no glycolysis, no starvation
solution for fermentation problem
dump electrons on pyruvate (electron sink) additional pathways can reduce pyruvate to make more NAD+
main fermentation pathways
acid or alcohol product
atp can be made from SLP
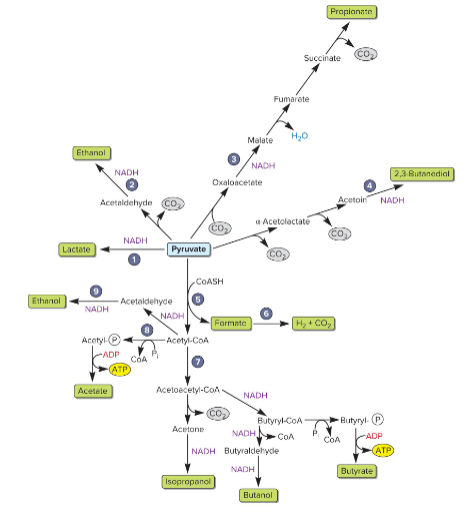
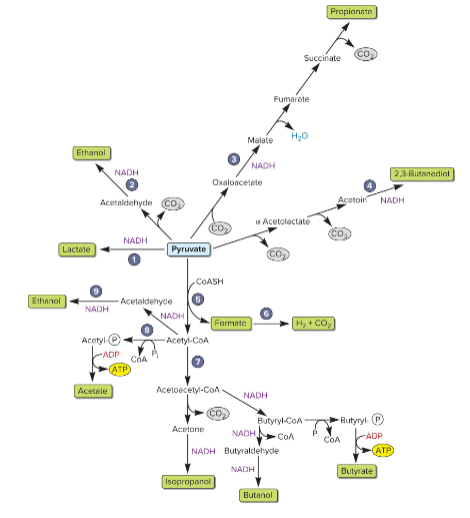
alcohol fermenters can produce
ethanol, isopropanol. butanol, 2,3-butanediol
prevents environment from becoming too acidic
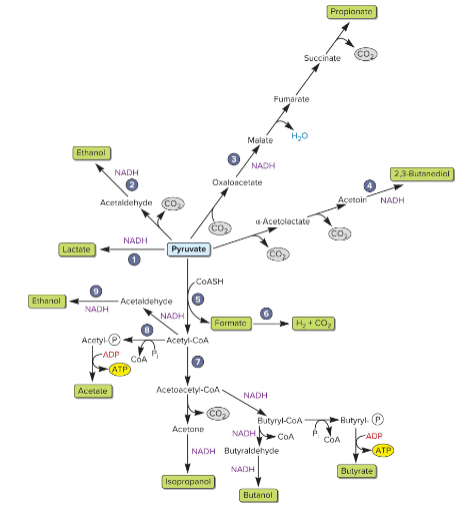
acid fermenters can produce
lowers environmental pH
makes lactacte, acetate (makes ATP), butyrate (makes ATP), formate (makes H2 and CO2), propionate (involves TCA intermediates)
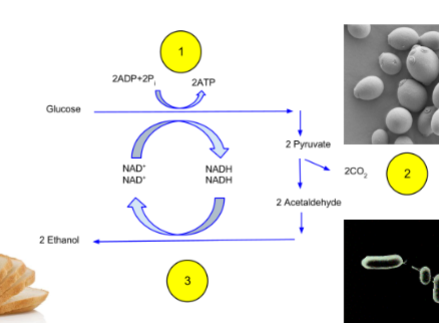
homoethanologenic
microbes that produce ethanol as primary fermentation product
Pathway: glucose → 2 pyruvate → 2 acetaldehyde + 2CO2 → 2 ethanol
acetaldehyde reducing to ethanol makes NAD+
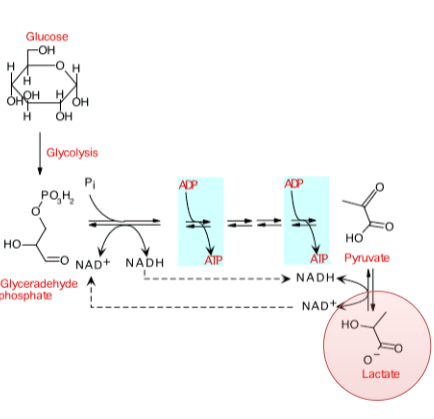
homofermentative lactic acid fermenters
only makes lactate, uses EMP
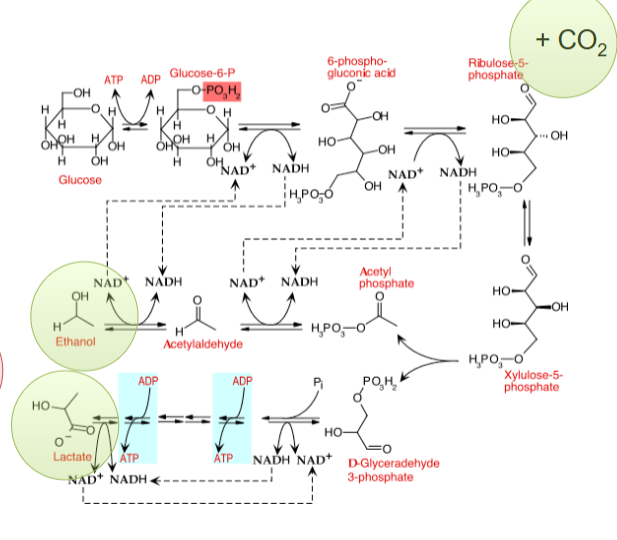
heterofermentative lactic acid fermenters
makes lactate, ethanol, CO2
uses PPP, EMP
mixed acid fermentation
pyruvate dehydrogenase is repressed (no ETC bc theres no TEA like O2 or NO3-)
pyruvate formate lyase (PFL) is active, making formate and acetyl coa
gas is produced if microbe makes formate hydrogen lyase
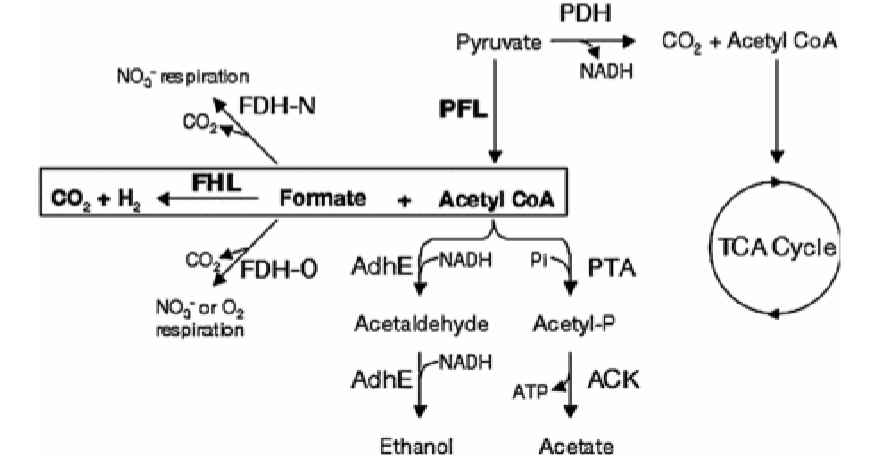
acetyl coA made during the PFL reaction from pyruvate can make
acetaldehyde (which goes on to make ethanol, NAD+)
acetate (makes ATP)
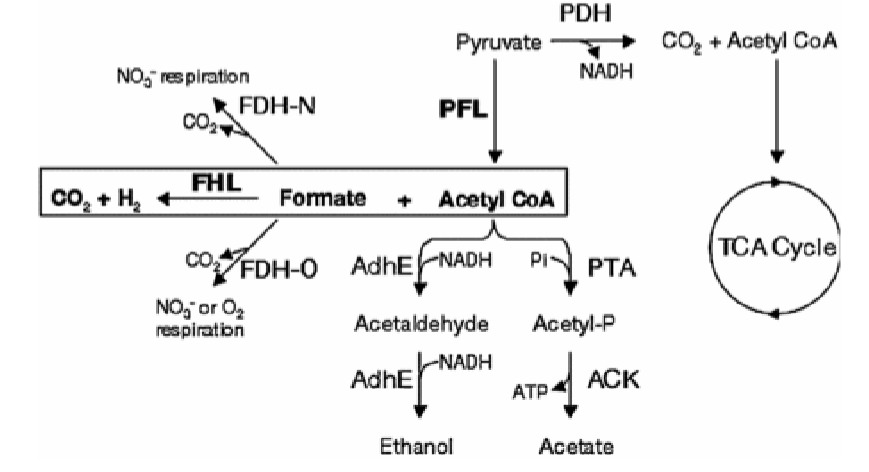
formate made during the PFL reaction from pyruvate can make
aka formic acid, break down to prevent too much acid build up
makes non acidic gases: CO2 and H2
mixed acid fermentation products are made
based on enviromental conditions
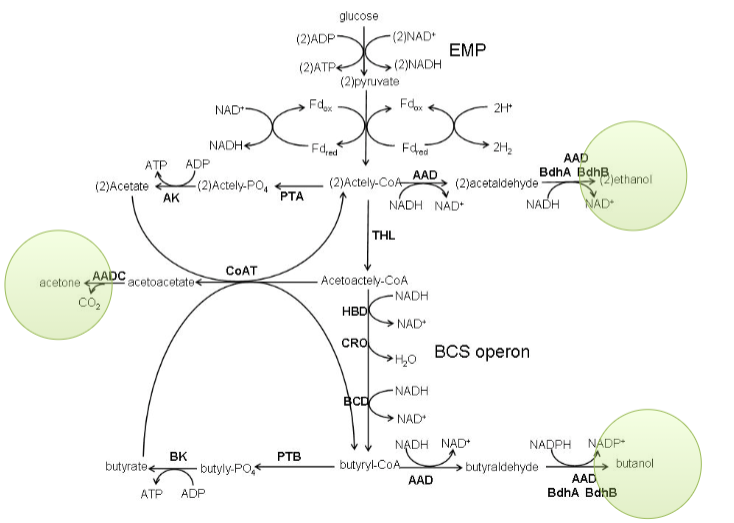
ABE fermentation
acetone-butanol-ethanol fermentations in clostridia (a strict anaerobe)
makes these products when stressed out during sporulation (low nutrients, low space)
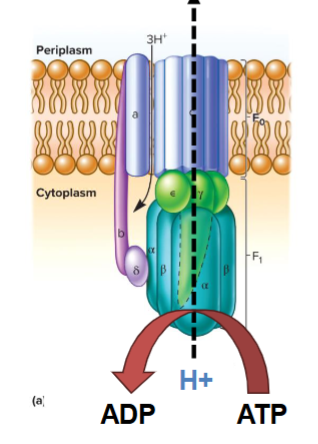
another problem fermenters must deal with
acidification: need to pump protons our to prevent internal acidifcation and make a proton motive force for transport
but have no etc
must run atp synthase backwards, pumps H+ out of cell
atp used to power ATPS can be from a fermentation reaction
horseshoe TCA cycle
makes NAD+, AcCoA, uses anaplueronic cycles
generates intermediates for anabolic processes
rank aerobic, anaerobic, and fermentation pathways based on amt of energy produced
most in aerobic, then anaerobic, then fermentation
fermenters grow slowly
fermentation vs anaerobic respiration
fermenterts reverse the ETC, e- acceptor is organic, pyruv is e sink