Organic Reaction Tables
1/10
Earn XP
Description and Tags
Identify the reagants and reaction conditions needed for the reaction as well as what kind of reaction it is
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
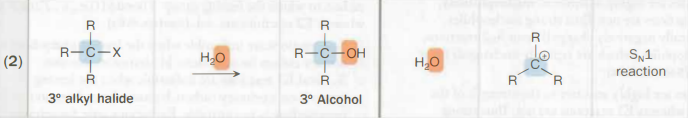
10 alkyl halide → 10 alcohol
Na—OH
any base with an OH-
The OH is the nucleophile
SN2
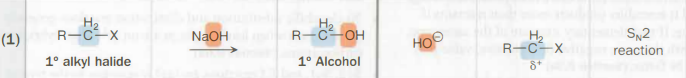
30 alkyl halide → 30 alcohol
H2O
The oxygen is the nucleophile
SN1
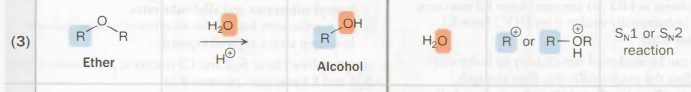
ether → alcohol
H2O
acidic conditions, H+
SN1 or SN2
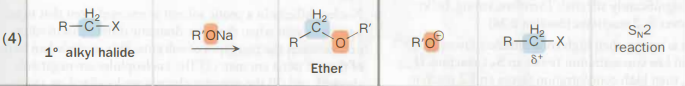
10 alkyl halide → ether
R’ONa
An aloxide salt binded to a soluble group 1a element
SN2
Low steric hindrance, therefore SN2
Also called Williamson ether synthesis
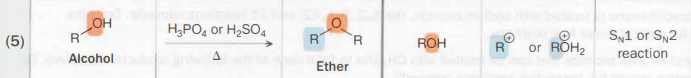
Alcohol → ether
H3PO4 or H2SO4
heat is needed as well
SN1 or SN2
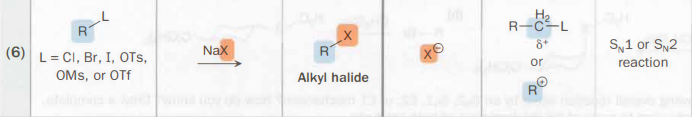
R—L (alkyl bonded to good leaving group) → alkyl halide
Na—X
Sodium salt with a good nucleophile
SN1 or SN2
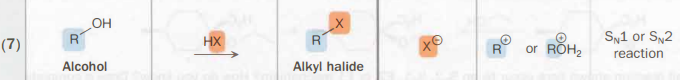
Alcohol → Alkyl halide
H—X
A halogenous acid
SN1 or SN2
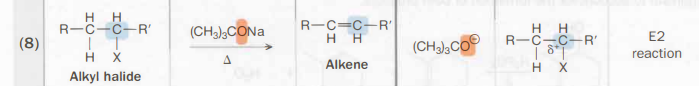
Alkyl halide → Alkene
(CH3)3CONa
Also called NaOtBu, it is a strong base that is non-nucleophilic
Heat also needed
E2 reaction
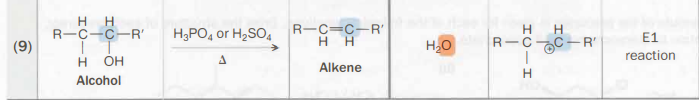
Alcohol → alkene
H3PO4 or H2SO4
heat also needed
E1 reaction
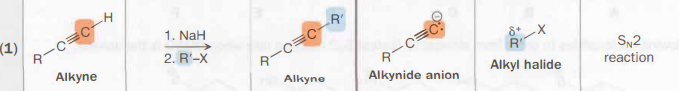
alkyne → haloalkyne
NaH (strong base)
R’—X, any alkyl halide
SN2
carbon skeleton-altering
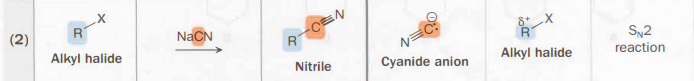
Alkyl halide → nitrile
NaCN
SN2
carbon skeleton-altering