How fast 5.1.1
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
what are the units for rate
mol dm⁻³s⁻¹
What is the rate expression?
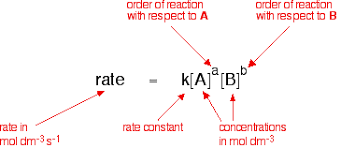
what is the unit for Concentration
mol dm⁻³
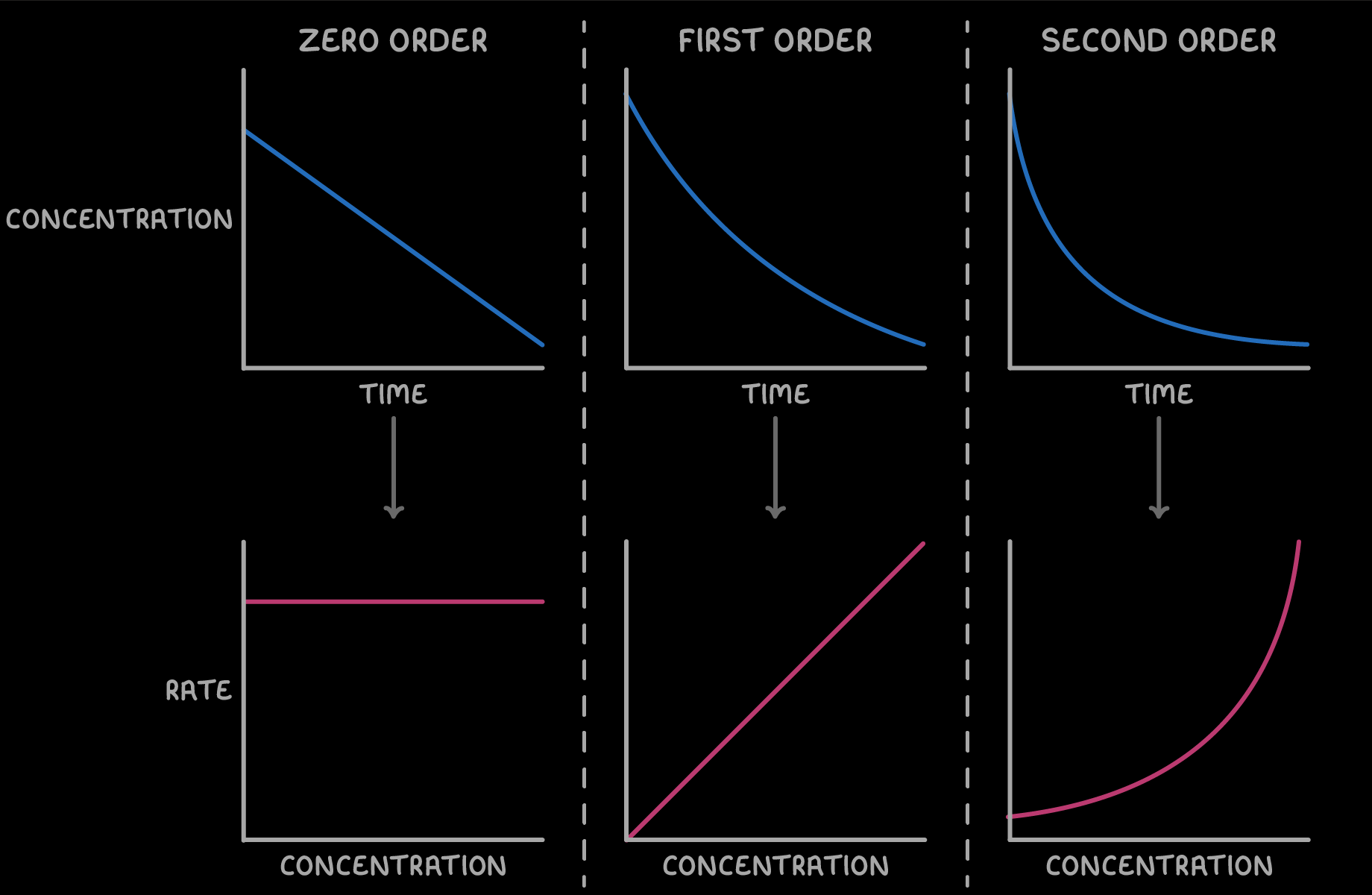
what are the two types of graphs
Concentration ( mol dm⁻3) / Time (s) graph
Rate ( mol dm⁻3 s⁻1) / Concentration ( mol dm⁻3) graph
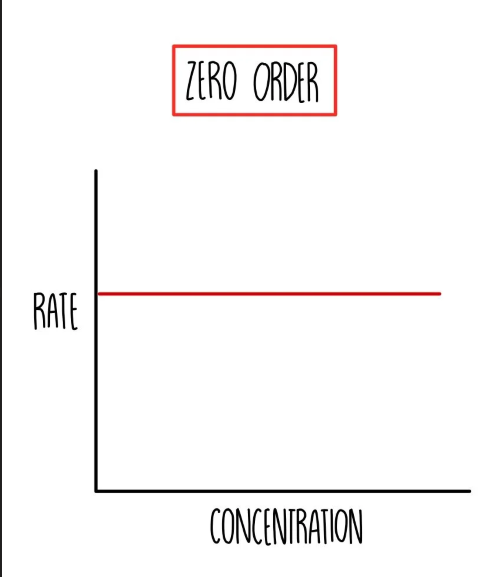
Zero order Graph —> Rate / Conc
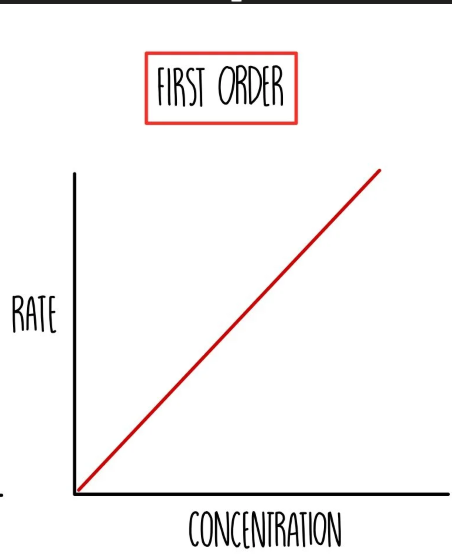
First order graph —> Rate / conc
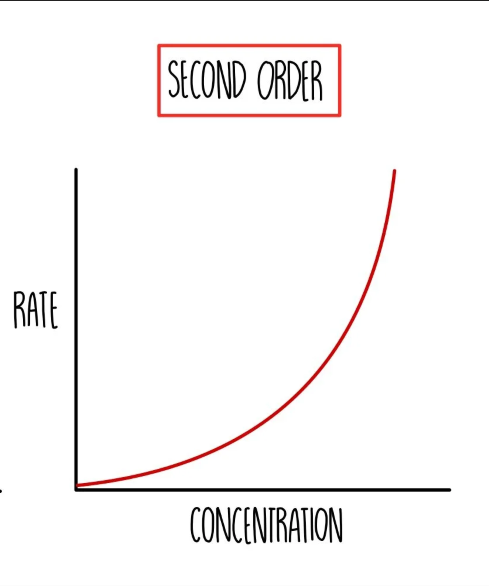
2nd Order graph —> Rate/ conc
what does Zero order mean
changing concentration has no effect on the rate
rate stays the same
what does first order mean
Rate ∝ [ x ]¹
what does 2nd Order mean
Rate ∝ [ x ]²
Should you include Zero order in rate equation?
No ( ignore it )
temperature increasing effect on K
K increases
what does a larger K result in
Faster rate
how to work out overall order
add the orders together
[ x ] = ¹
what are units of 0 order overall
mol dm⁻³ s⁻¹
what are the units of 1st order overall
s⁻1
what are the units for 2nd order overall
s⁻¹mol⁻¹dm³
what are the units for 3rd order overall
s⁻¹mol⁻²dm⁶
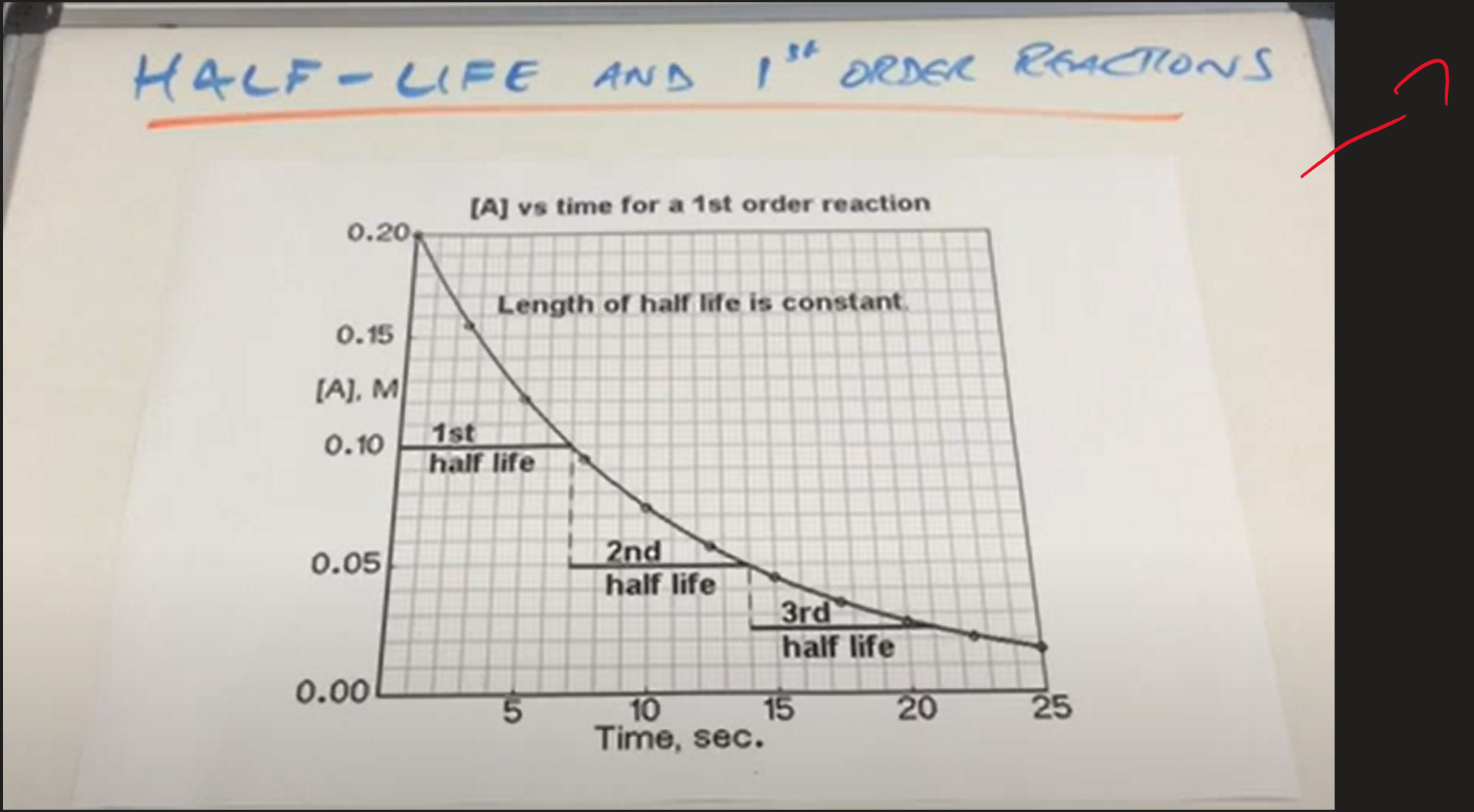
what does half life mean?
time taken for the concentration to half
what is the rule for the half life of a 1st order reaction
constant half life
examples of constant half lifes
Radioactive decay
Pharmacokinetics of drugs
Zero order concentration/ time graph

First order concentration/ time graph
First order half life graph ( Image)
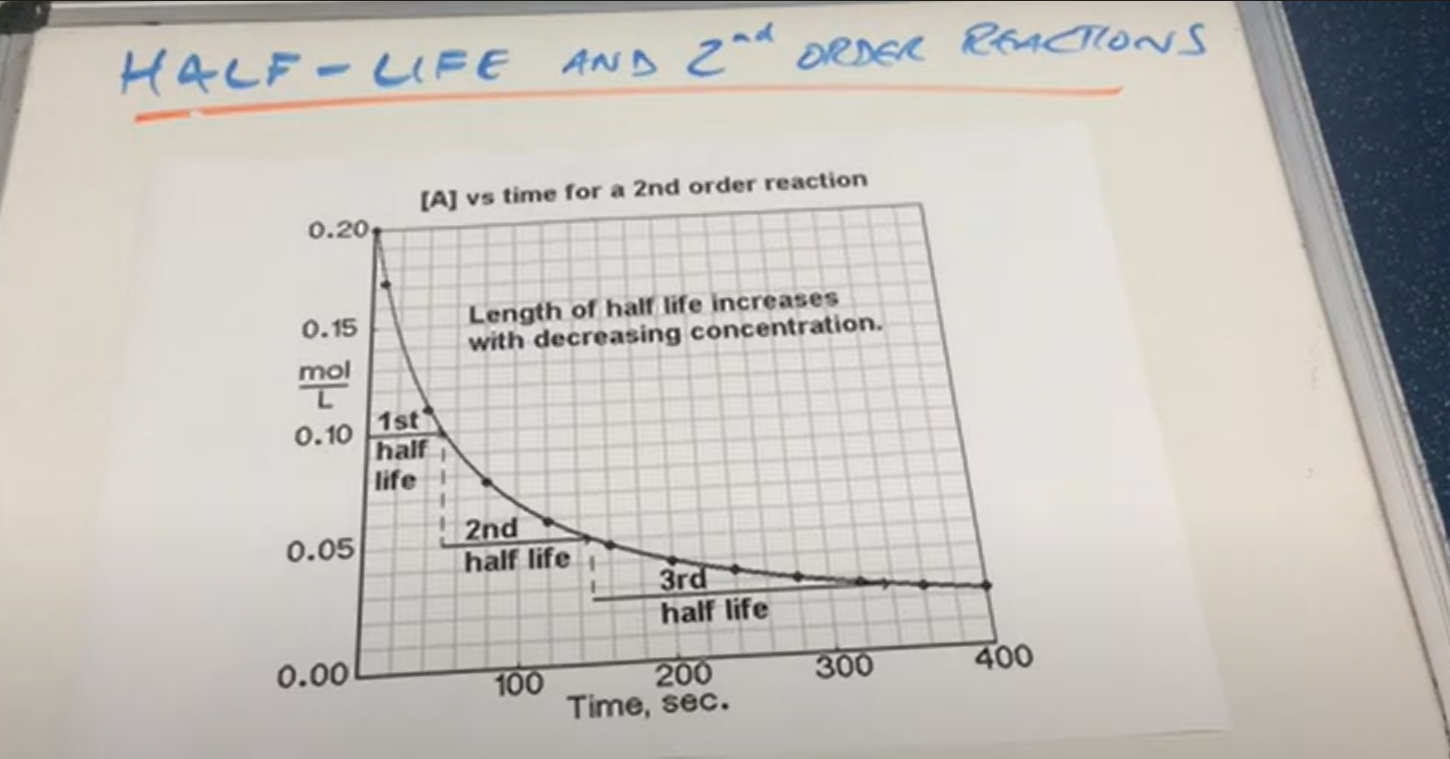
Rule for 2nd order half life
As concentration decreases, half life increases
2nd Order half life graph ( Image)
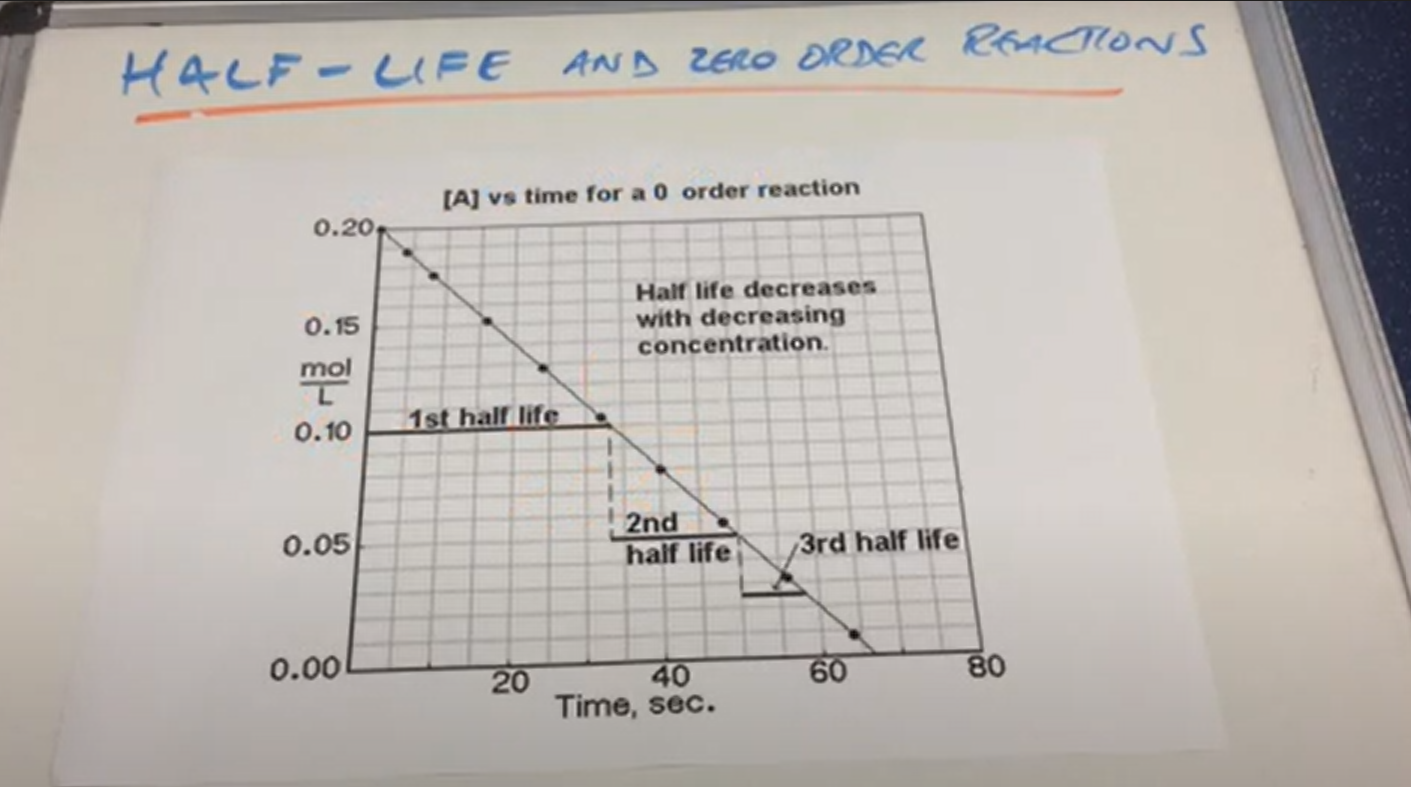
Rule for Zero order half life
As concentration decreases , half life decreases
Zero order half life ( image)
Initial rates questions which experiments to use?
other reactants concentration are constant / the same
what are the Arrhenius equations
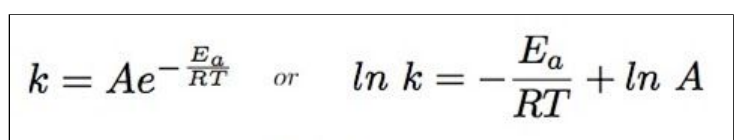
what does K represent in the Arrhenius equation
Rate constant
what does Ea represent in the Arrhenius equation
Activation energy ( J mol⁻¹ )
how to convert from KJ to J
x1000
how to convert from J to KJ
/1000
what does T represent in the Arrhenius equation
Temperature (K)
what does R represent in the Arrhenius equation
8.31 ( constant)
what does A represent in the Arrhenius equation
exponential factor
same units as Rate constant
what is the effect of increasing Ea on the rate constant
As Ea increases, K gets smaller
what is the effect of decreasing Ea on the rate constant
K increases
what is the effect temperature increasing on rate constant
K increases
why does Rate increase as temperature increases
kinetic energy if particles increases —> Faster moving particles —> many more particles have energies greater then the Activation energy —> more frequent and successful collisions.
which Arrhenius equation to use when working out any of the powers ( Ea / R/T )
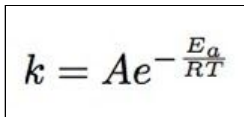
which Arrhenius equation to use when working out A
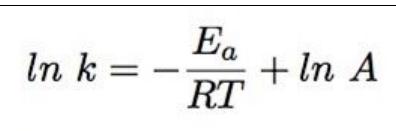
Explain qualitatively why increasing temperature has a greater effect then increasing concentration
reactions occur when particles have energy greater then the activation energy
increasing temperature causes many more particles to have energy greater then the Ea
increasing concentration increases number of particles with the energy
Arrhenius plots ( Image)
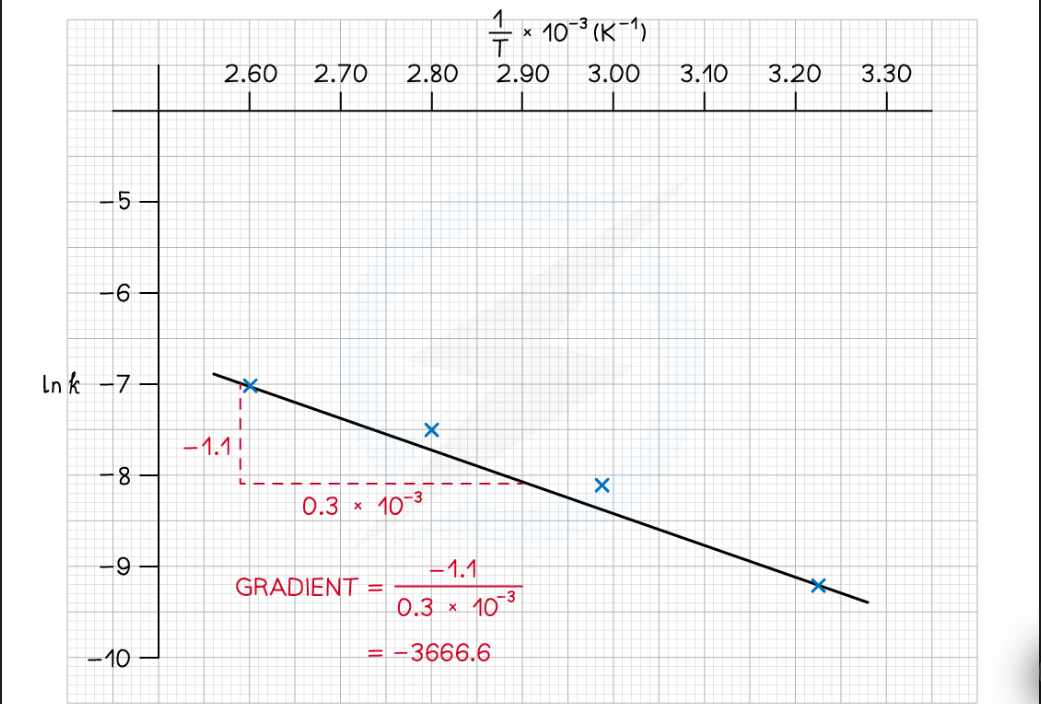
what are the Y and X axis for Arrhenius plot and units
Y= In K
X= 1/T ( K⁻¹)
Arrhenius equation to work out Plots
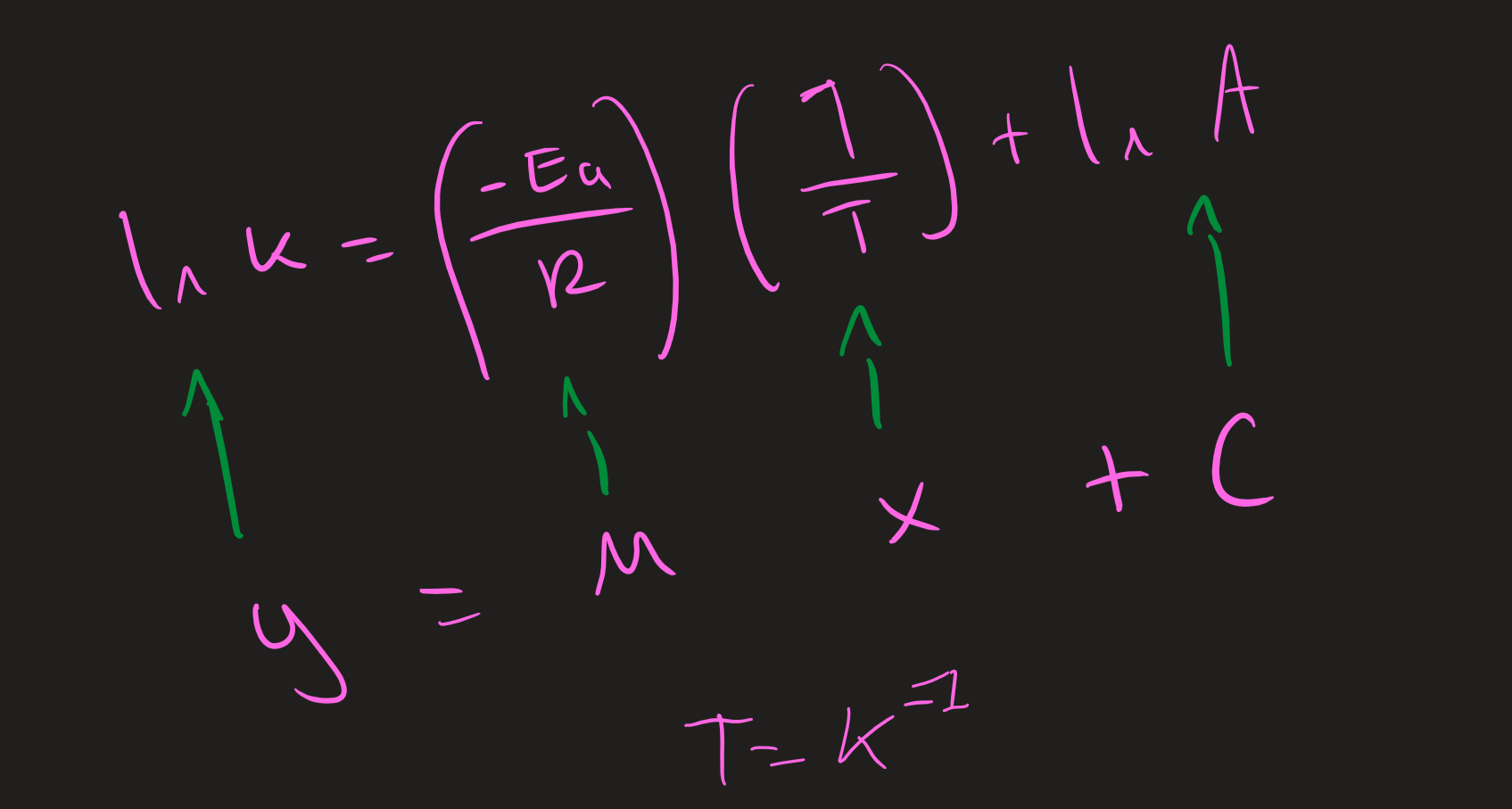
method for working Ea in kjmol⁻¹ ( Arrhenius plots)
1) plot the graph X = 1/T ( K⁻¹) Y = In K
2) work out the gradient ( dy/dx)
3) Gradient x ( -8.31) = Ea ( J mol⁻¹)
/1000 = Kj mol⁻¹
How to convert from J mol⁻¹ to KJ mol⁻¹
/1000
How to convert from KJ mol⁻¹ to J mol⁻¹
X 1000
How to work out A ( Arrhenius equation)
Y intercept value ( point Line of best fit crosses Y axis) = In K
( -EA / RT) - ( In K) = In A ( x)
e×
how to work out A in Arrhenius plots ( Image)
To work out T = 1/T
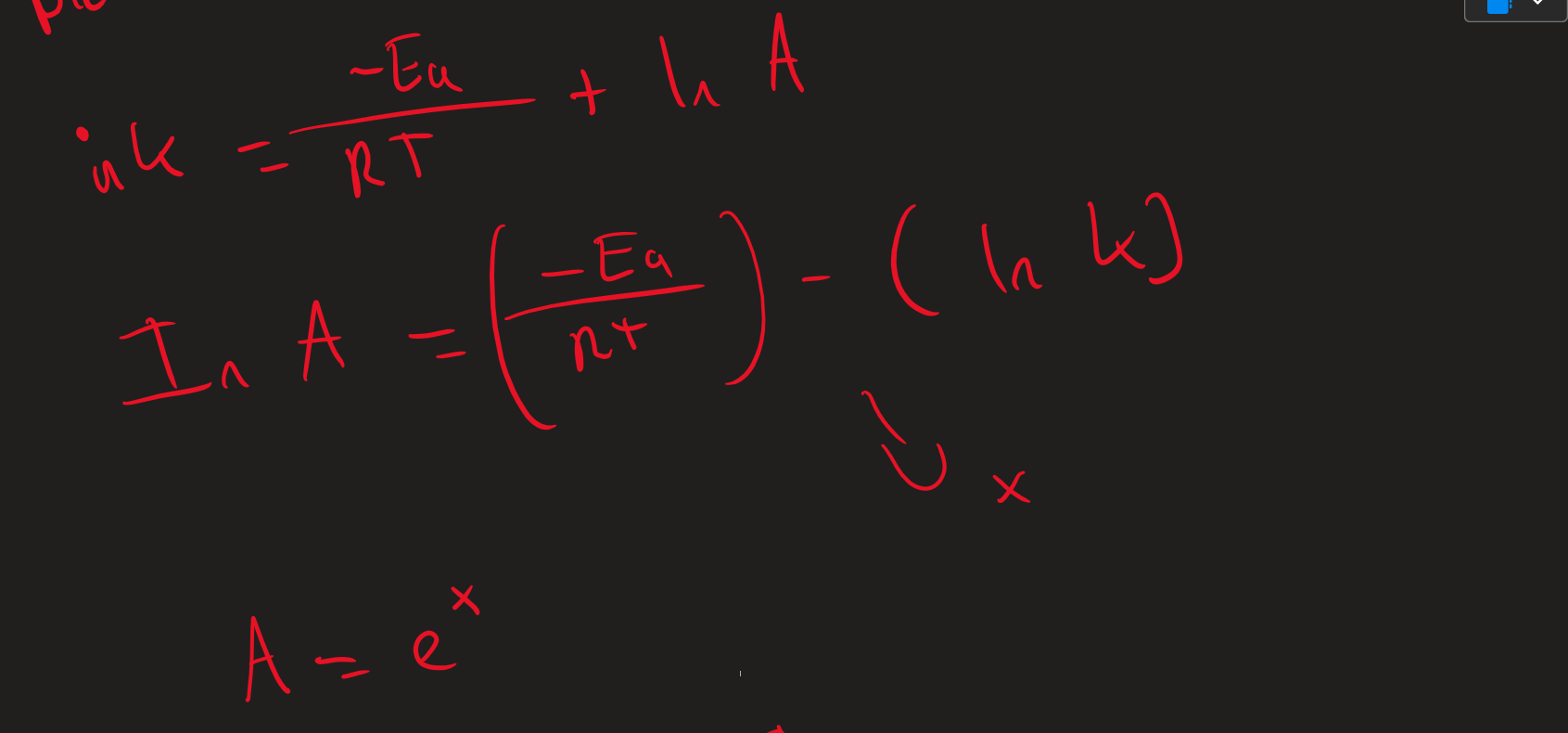
what is the rate determining step
the slowest step in a reaction
what is the rate equation and orders for this reaction
A + 2B → X + Y
Rate = K [ A] [ B]²
First order → A
Second order → B
In R.D.S rate equation what does the Order show
the molar ratio of a reactant ( how many of the reactant there is )
Working out R.D.S method ?
The R.D.S and Step 2 equation = the overall equation
only the products and reactants from R.D.S and Step 2 can cancel out
Product from step 2 and overall equation has to be the same
to work out the R.D.S products = combine the reactants !!!!
overall equation has more then 1 mole of reactant → both equations reactant ( both contain 1 mole of reactant) add up to make moles of reactant
If step 2 and R.D.S cancel to make overall equation = Correct
maximum of 2 reactants in step 2
what is the catalyst in a R.D.S equation
catalyst is a Reactant that gets reformed as a product after getting consumed in the previous equation
what is the intermediate in a R.D.S
A reactant that gets formed in the first step then consumed in the second step
Why is excess other reactant used in ( calculate order of reaction experiments)
keep the reactant constant
2 ways to measure volume of gas
Gas syringe method
measure mass loss of reactant
suggest apparatus that allows volume of gas to be collected ( with scales of working
1000cm³/ 1 dm³ measuring cylinder
why does absorbance decrease in reactions with colorimeter
limiting reactant gets used up
the products are colourless
method to working out how to obtain concentrations from experiments
mix 10cm³ of each( named) reactant
dilute each solution with water
total volume is control variable
how to tell if ions are acting as catalyst
catalyst → ion is regenerated
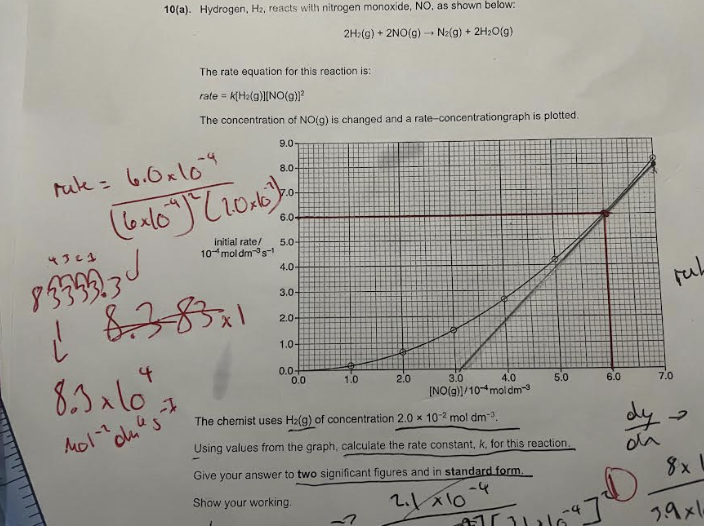
standard form / incorrect past paper question ( IMAGE)
R.D.S is the overall equation apart of the steps in mechanism?
no overall equation isn’t a step
reasons why it is unlikely for R.D.S reactions to occur in one step
collisions are unlikely with more then 2 species
rate equation doesn’t match overall equation
how to monitor concentration of reactant in conc/time graph
use colorimeter to measure reduction of colour of ( named) reactant
how to work out K in half-life for first order equation
In(2) / time
Reasons why conditions are different in industry
High temp → High energy usage
Low pressure → Slow rate of reaction