gases
1/61
Earn XP
Description and Tags
ideal gas
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
law of combining volumes
gases combine at constant temperature and pressure, volumes are in ratio of simple whole numbers
avogadro’s law
v is directly proportional to n
equal volume of gas contain same number of molecules
V1/n1 = V2/n2
pressure and temperature is constant
more gas molecules = more space needed to move
molar volume
all gases have a certain volume that contains exactly 1 mole of particles
law of combining volumes formula
V1/n1 = V2/n2
combined gas law formula
P1V1T2 = P2V2T1
combined gas law
PV is directly proportional to T
number of gas moles is constant
formula of charles law
V1/T1 = V2/T2
charles law
V is directly proportional to T
when temperature increase - volume increase
when temperature decrease - volume decrease
moles of gas and pressure is constant
charles law explanation
temperature and volume are directly proportional
increasing temperature increases kinetic energy of gas molecules (directly proportional)
hence, speed of molecules increases causing an increase in collision frequency
To mantain the same pressure, the volume must increase to create more space for molecules to move
volume of gas increase
particles need to speed up to maintain pressure
travel far and colliding with same frequency
increase velocity
volume of all gasses
extrapolate to zero at the same temperature
-273.15 °C
P1V1 = k
initial measure of pressure and volume
P2V2 = k
final measure of pressure and volume after a change
Boyle’s law
inversely proportional
p is directly proportional to 1/v
pressure decreased - volume increase
pressure increased - volume decreased
number of moles and temperature is constant
Boyle’s law explanation
volume and pressure are inversely proportional
decreasing volume decreases the amount of space for gas molecules to move
hence increasing the collision frequency
therefore increasing the pressure
IMFs become stronger with more compacted gas particles - deviates from an ideal gas
formula for boyle’s law
P1V1 = P2V2
factors that affect gasses
pressure - kPa
volume - L
temperature - K
amount of moles - mol
Ideal gas law
inversely and directly proportional
all formulas derived from it
ideal gas
small molecules
kinetic energy proportional to absolute temperature
increased volume
negligable volume and IMFs due to large space between them
temperature ↑ and pressure ↓
molar mass
grams/moles
ideal gas law formula (moles given)
PV = nRT
P - pressure
V - volume
n - number of moles
R - Ideal Gas Constant
T - temperature
ideal gas law formula (mass given)
PV = mRT/M
P - pressure
V - volume
m - mass
M - molar mass
R - Ideal Gas Constant
T - temperature
ideal gas constant
8.314 kpa x L / K x mol
Density
ratio of chemical’s mass to volume it occupies
p = m/V
proportional to mass, moles, pressure, molar mass
sig digs for adding and subtracting
least number of decimals
sig digs for multiplying and dividing
count sig digs
solids
Definite shape
Definite volume
Particles vibrate around fixed axes
particles in contact, fixed
no thermal energy to overcome interactions
liquids
No definite shape (takes the shape of its container)
definite volume
Particles are free to move over each other but still attracted to each other
particles in contact, not fixed
enough energy to partially overcome intermolecular interactions
gases
No definite shape (takes the shape of its container)
No definite volume
Particles move in random motion (little/no attraction to each other)
particles not in contact/random
enough energy to completetly overcome intermolecular interactions
physical properties of gas
highly compressible
fill container
diffuse in any available space
affected by temperature
monoatomic
consists of one atom
diatomic
consists of two atoms
HOFINBrCl
KMT
smallest entities of a substance are
in continuous motion
colliding with each other/objects in their path
5 big assumptions
constant random motion
negligible volume
exerts no force on each other
collide elastically with each other/container walls
kinetic energy directly proportional to absolute temperature of gas
constant random motion
gas is composed of a large number of particles
higher energy than liquids or solids
negligible volume
distance between gas molecules is greater than the size of the molecules
easy to compress a gas - decrease distance
exerts no force on each other
Intermolecular interactions are weak - negligible
No attractions or repulsions
Treats all gases as a collection of particles that are identical in all respects except mass
Collide elastically
collision with each other and container walls are elastic
do not change the average kinetic energy of the molecules
Kinetic energy proportional to absolute temperature of gas
average kinetic energy depends on only the temperature
all gasous molecules have same average kinetic energy
pressure - ideal and real
ideal gas - low pressure
real gas - high pressure
temperature - ideal and real
ideal gas - high temperature
real gas - low temperature
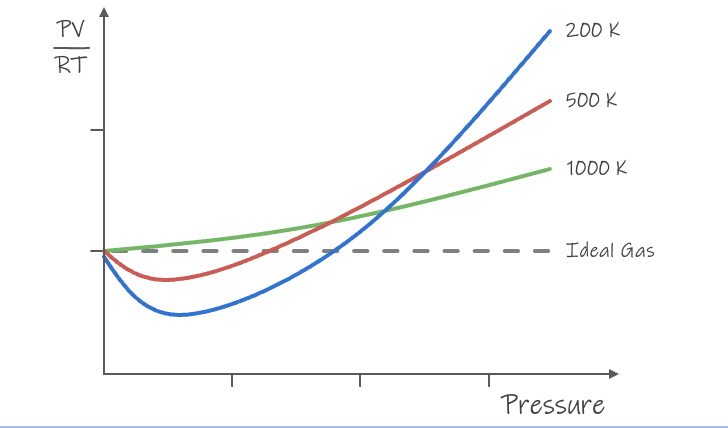
ideal gas graph
more pressure - deviates from an ideal gas
lower temperature - deviates from an ideal gas
ideal gas vs real gasses
temperature ↑ and pressure ↓
Small molecules behave more ideally than large gas molecules
strong IMFs = less like an ideal gas
ideal gas
no attractive/repulsive force between particles
no volume
collisions are elastic (no loss of kinetic energy)
real gas
small attractive forces between particles
small volume
collisions are not elastic (lose energy)
real and ideal gasses similarities
made of small particles that have mass
gases are mostly empty space
low density
converting temperature
measure of kinetic energy
higher temperature = greater kinetic energy
Fahrenheit
Celsius
Kelvin
STP
standard temperature and pressure
SATP
standard ambient temperature and pressure
celcius to kelvin
K = ºC + 273.15
pressure
concentration of force (per unit area)
p = f/a
atmospheric pressure
Earth’s surface experiencing a net pressure
atmospheric pressure decreases
volume increases
temperature decreases
converting units of pressure
atmospheres (atm)
millimetres of mercury (mmHg)
kilopascals (kPa)
ATM conversions
1 atm = 760 mmHg
1 atm = 101.325 kPa
mmHg conversions
760 mmHg = 1 atm
760 mmHg = 101.325 kPa
kPa conversions
101.325 kPa = 1 atm
101.325 kPa = 760 mmHg
gay-lussac’s law
P1/T1 = P2/T2
p is directly proportional to t
number of moles and volume is constant
gay-lussac’s law explanation
pressure and temperature are directly proportional
as temperature increases, the kinetic energy increases (directly proportional to temperature) (KMT)
hence, the collision frequency increases, resulting in an increase in pressure
limiting reactant
the reactant that is completely consumed
limits the amount of product formed
when it stops, the chemical reaction stops
excess reactant
substance left over after the reaction occured
graphing
title: y vs x
right side of table - y
left side of table - x
dependent/responding - y
independent/manipulated - x
upwards - y
horizontal - x