Chapter 13: ethers & epoxides ; thiols & sulfides
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
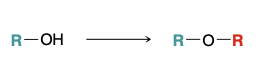
Willamson ether synthesis
1) NaH
2) RX
Willamson ether synthesis mech
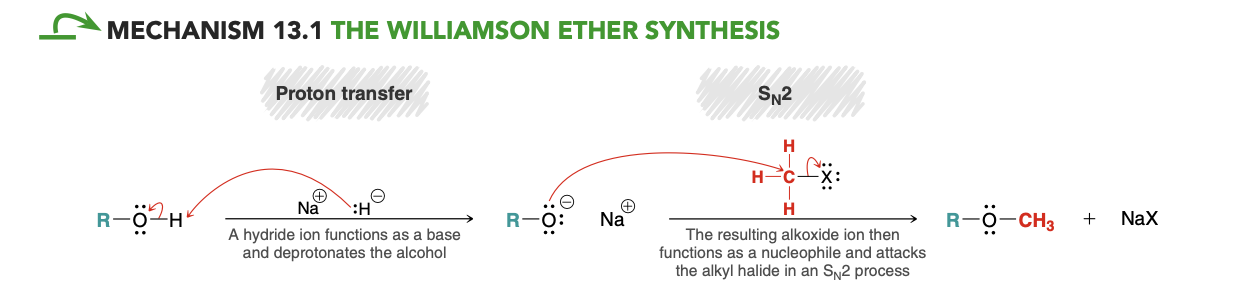
.
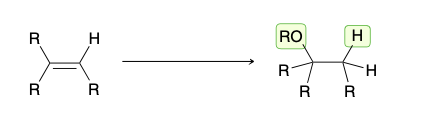
alkoxymercuration-demercuration
1) Hg(Oac)2, ROH
2)NaBH4
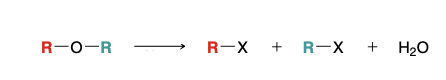
Acidic cleavage of an ether
excess Hx, heat
acidic cleavage of an ether mech
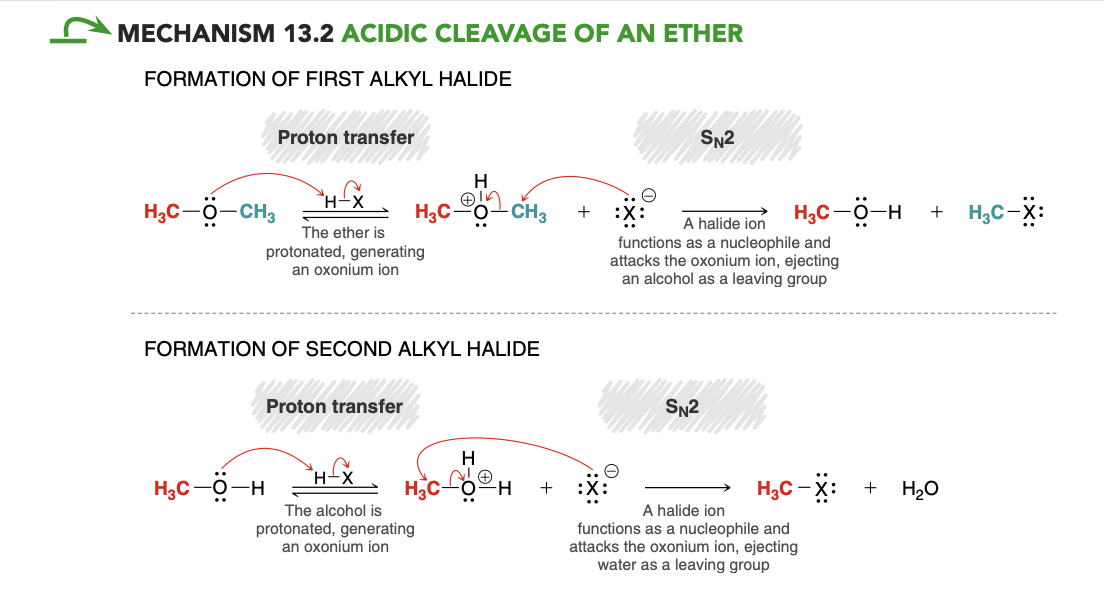
.
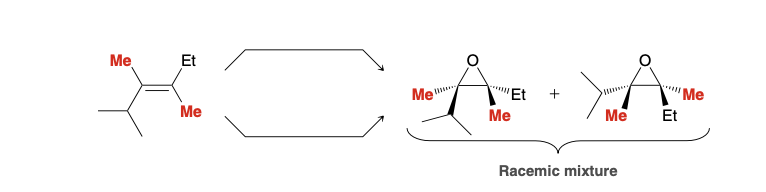
preparation of epoxides
MCPBA
or 1) Br2, H2O 2) NaOH
preparation of epoxides mechanism
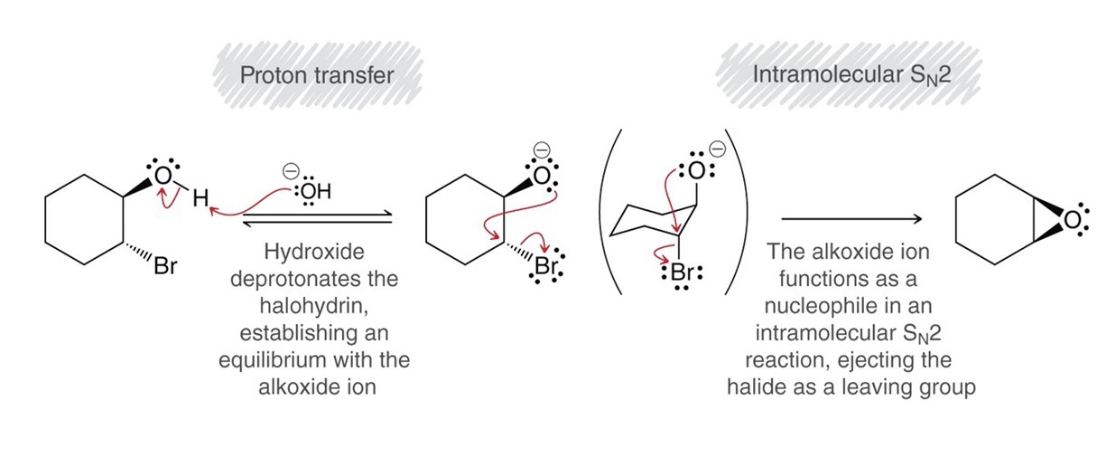
.
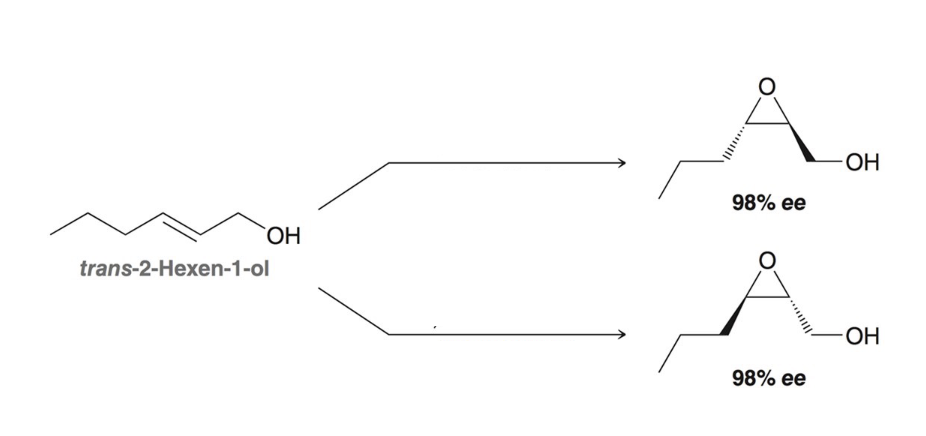
Enantioselective epoxidation
.

Sharpless asymmetric epoxidation. +DET forms epoxide ____, -DET forms epoxide ____
Above, below
Epoxide ring opening with a strong nucleophile mech
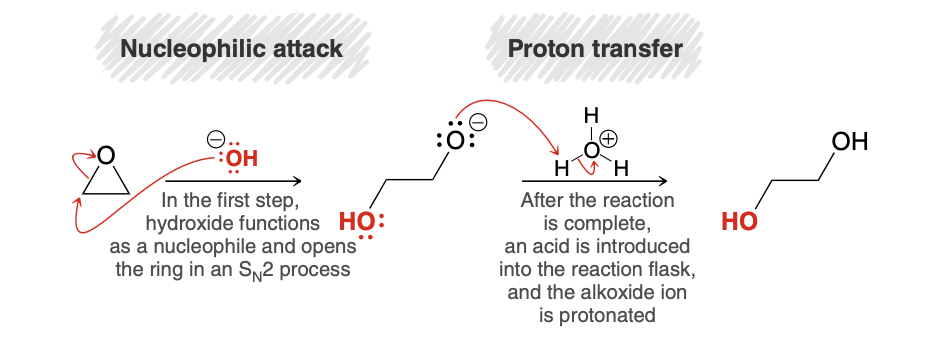
.
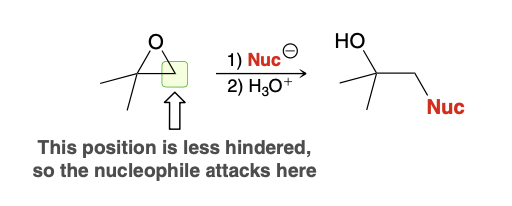
Unsymmetrical epoxide opening. Where does nuc attack?
Nucleophile attacks at least sub
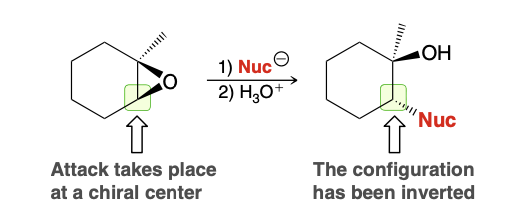
Symmetrical epoxide ring opening. what is observed?
Inversion of configuration is observed
Epoxide ring opening: Adding OR group
1) NaOR
2) H3O
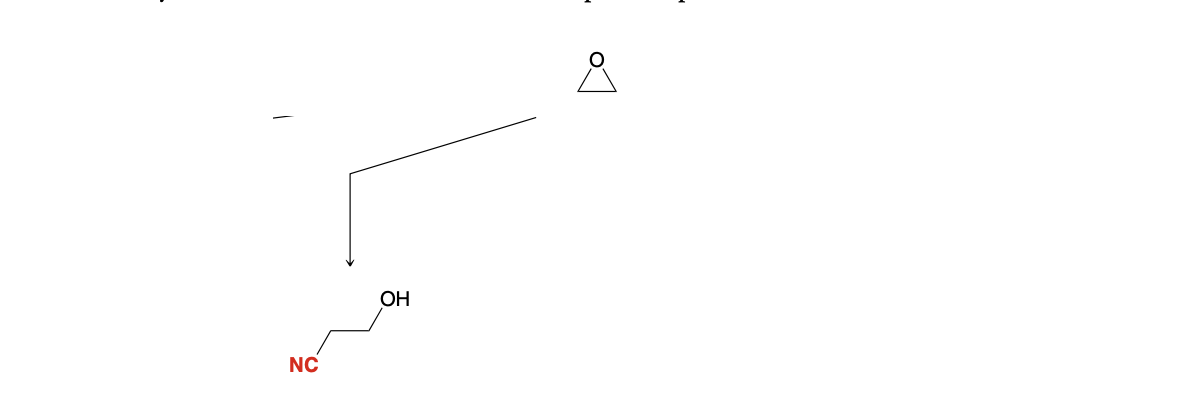
Epoxide ring opening: Adding CN group
1) NaCN
2) H3O
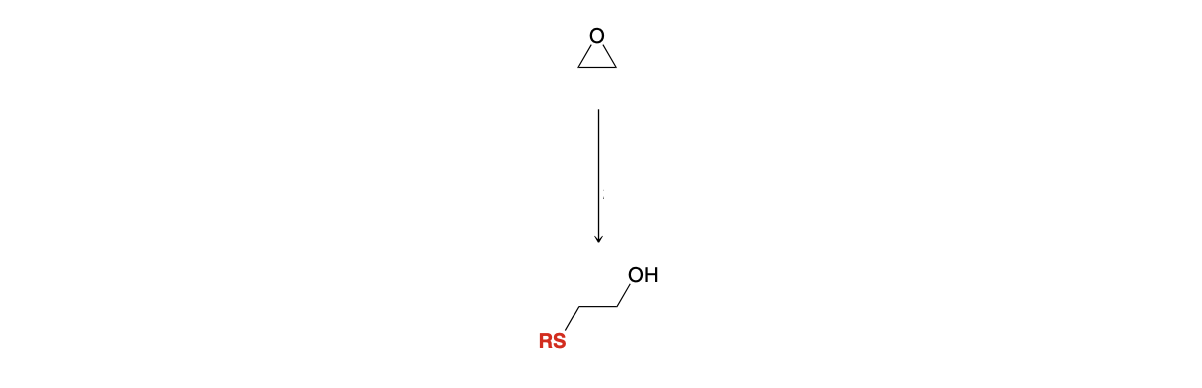
Epoxide ring opening: Adding SR
1) NaSR
2)H3O
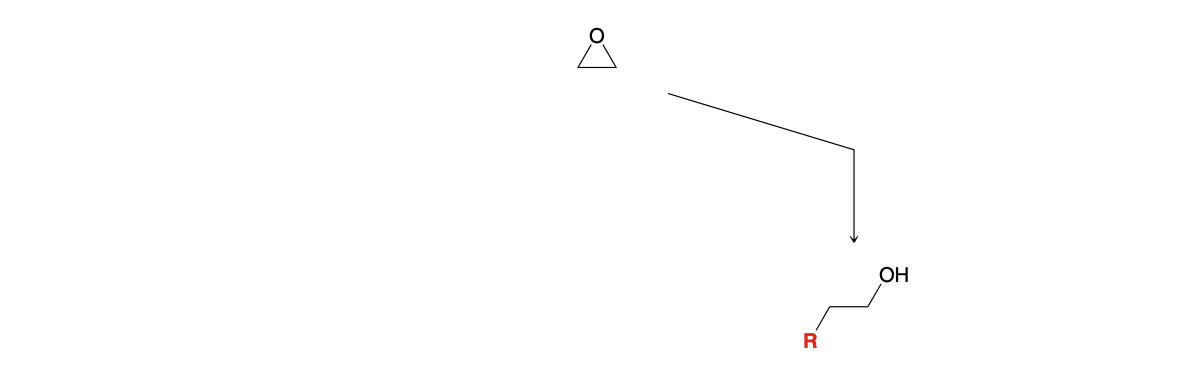
Epoxide ring opening: Using Grignard
1)RMgBR
2)H3O
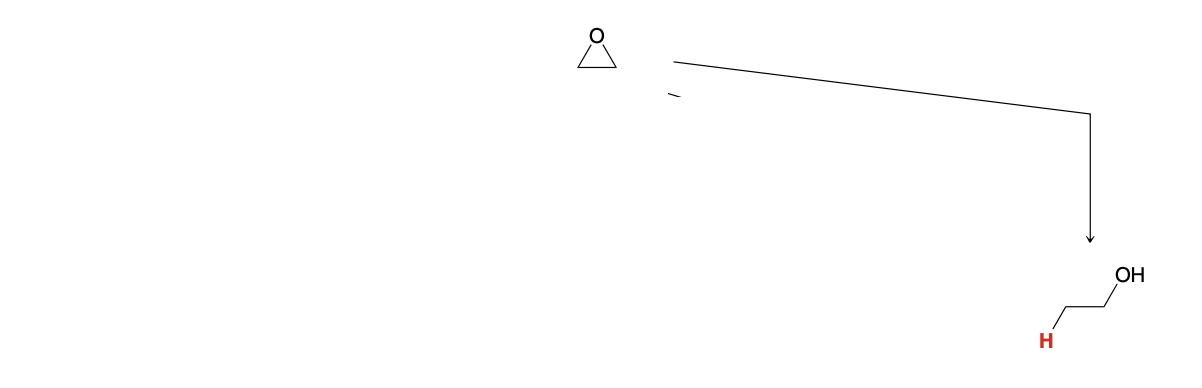
Epoxide ring opening: Adding H
1)LiAlH4
2)H3O
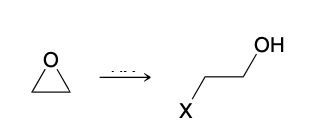
Acid Catalyzed Ring Opening
HX
Acid Catalyzed ring opening of epoxide mech
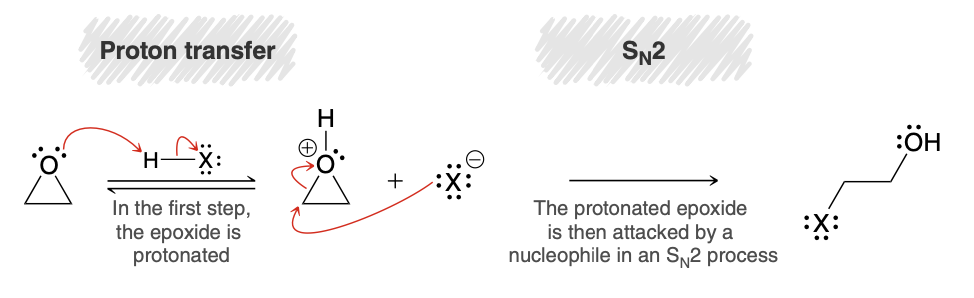
.
Ring opening of epoxides: using water mech
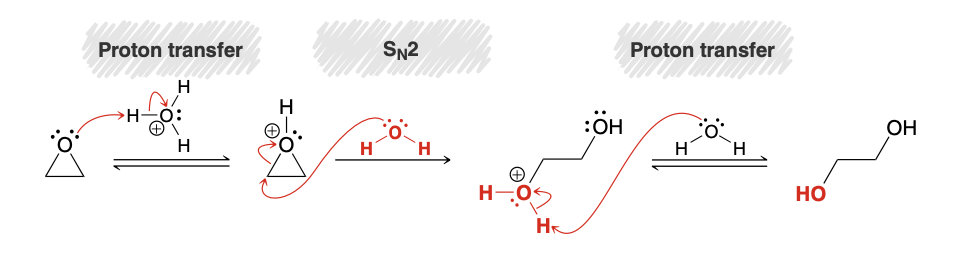
.
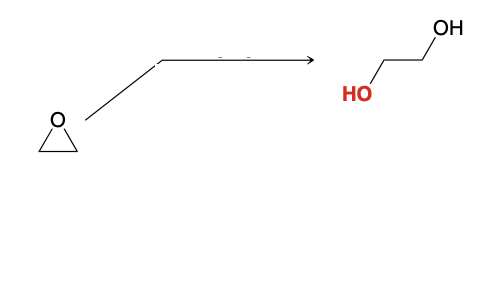
Ring opening of epoxides using water
1) [H2SO4]
2) H2O
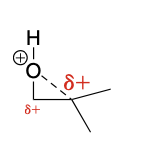
Ring Opening degree of substitution: 1° vs 2°. Where does the nuc attack?
Less hindered primary position
Ring Opening degree of substitution: 1° vs 3°. Where does the nuc attack?
tertiary position, because of the electronic effect.
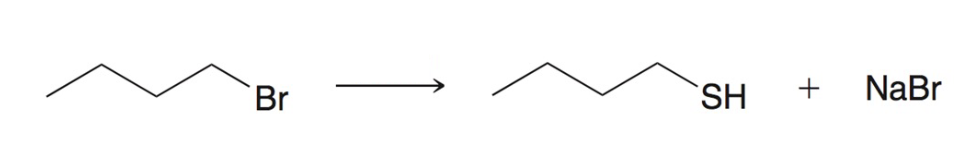
Preparing thiols using SN2
NaSH
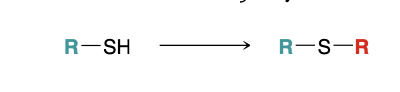
Preparing Sulfides
1)NaOH
2)RX
preparing sulfides from thiols mech
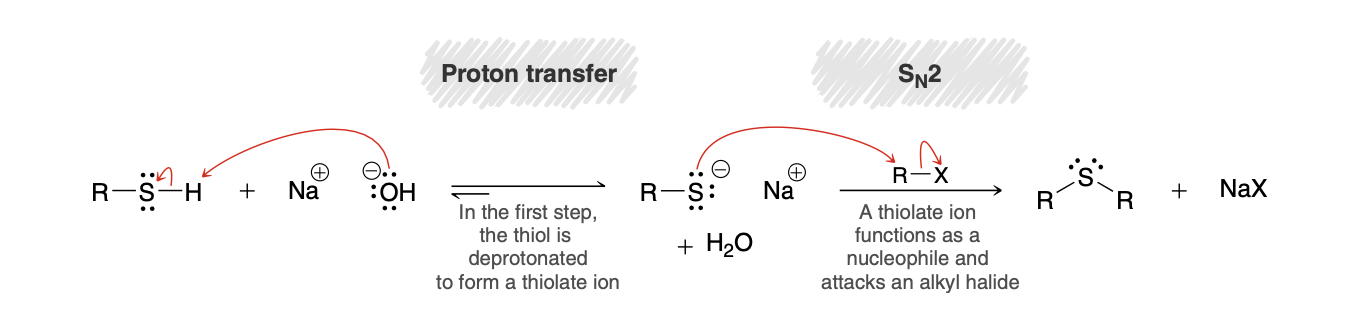
.
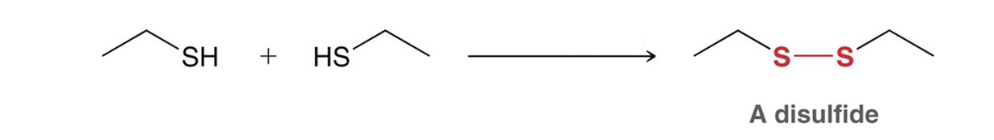
oxidation of thiols to disulfides
NaOH, Br2
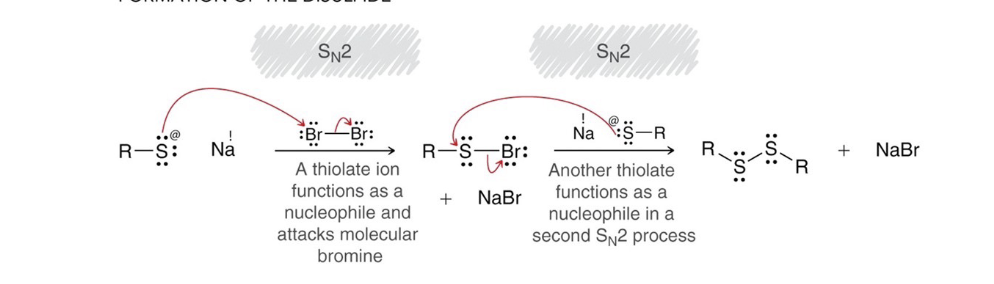
formation of disulfides mech
.
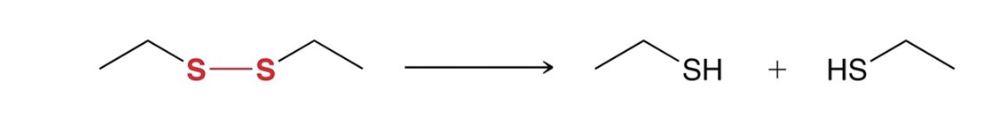
reduction of disulfides to thiols
HCl, Zn
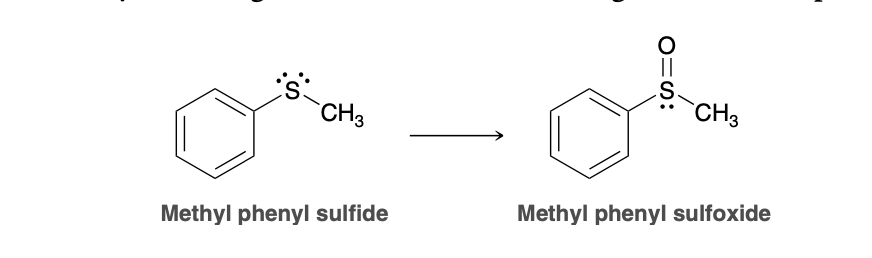
Sulfide to sulfoxide
NaIO4
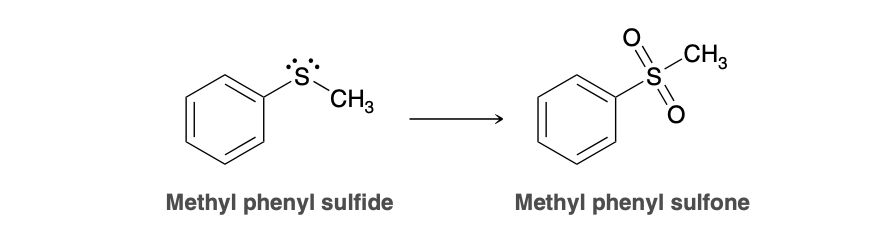
sulfide to sulfone
2 H2O2