TAE020 - Sustainable Energy: Comprehensive
1/126
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
127 Terms
Energy Problem
High carbon emissions
Oil
Expanding global demand (1-2% increase per year)
Factors
Slow progress for fossil fuel alternatives
Changing limitations
Policy
Late response (yesterday was to act, act now)
Global Energy Use/Development
Consumption increased 2.5 times from 1970 to 2015 about 2% per year until 2013, and around 1.0 % from 2014-2016.
Most growth occurred up to 2000 in the developed countries and after 2000 in the developing countries.
Non-OECD (82% population) consumes energy at a rate of:
52 MBtu per capita
(compared to whopping 184 MBtu in OECD countries)
Energy per capita increased by __% in non-OECD countries, while it dropped by __% in the US and 8% in all other OECD countries
58, 15
Core indicators of human welfare/standards of living/education/income are reflected in the use of:
high-quality energy
Energy Use in the USA
The energy intensity of the economy improved by 27%, and per capita consumption dropped by 15%.
Electricity consumption increased significantly from 2000-2007
Petroleum consumption has decreased since 2006, and
reliance significantly (25-60%) on imports.
Agri-food sector currently consumes __ percent of the total energy demand globally,
30
Around _-_% of total final energy consumption is used directly in the agriculture sector, about _._% of total US primary energy consumption.
3-5, 1.9
Greenhouse Effect
Specific molecules transmit short-wavelength solar radiation but block irradiated long-wavelength
Equilibrium surface temperature increases
All energy establishing equilibrium is from the sun
Geothermal only accounts 0.1% of earth’s total heat
Equilibrium w/o atmosphere is __oC
-19
Solar power absorbed by the planet: Formula
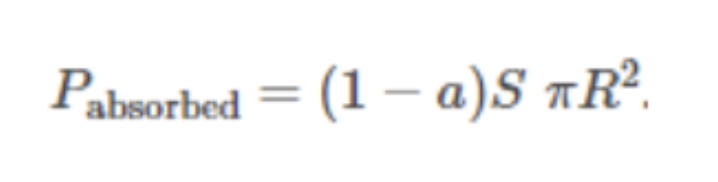
Solar power radiated from the planet: Formula
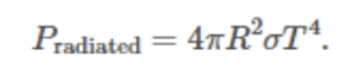
Global energy balance from the above two sources & sinks:
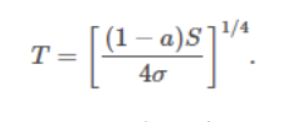
The earth's surface temperature will be higher (+__°C) than predicted by the simple model, which excludes the atmosphere (-__°C).
11, 19
The atmosphere traps heat, causing the greenhouse effect and increasing the earth's average temperature by more than __°C
30 (degrees)
Climate Change (Carbon Emissions)
Combustion of fossil fuels produces CO2
This CO2 adds to GHG → equilibrium raised
Evidence of Global Warming
Reduction in Arctic sea ice: The area has decreased by about 9 % in the past decade, and the thickness has decreased by 15 – 40 % over the past 30 years.
I.e. migration routes, geographic ranges
Increased sea levels
Predicted Temperature Changes for the Next Century
Global temperature increases of 2 to 8 C ° above the preindustrial level can be expected by 2100
Carbon Sequestration
Capturing and storing atmospheric carbon dioxide. It is one method of reducing the amount of carbon dioxide in the atmosphere with the goal of reducing global climate change.
Climate Change Scientific Consensus/Initiatives
Reduce energy consumption to mitigate enviro. effects
Major industrialized countries: binding 6-8% reduction in greenhouse gas emissions relative to 1990
IPCC
Paris Agreement/Accord
Kyoto Protocol
UNFCCC
100% Solution: Hypothetical Roadmap
Transition energy infrastructures to clean, renewable (Wind, Water, and Solar) (WWS) using existing technologies for 80% of all electricity, transportation, heating/cooling, industry, and agriculture/forestry/fishing by 2030 and 100% by 2050.
Energy
The ability/capacity to work
Work (W)
The product of force (F) and the distance (d) over which it acts
Force
Newton’s Law
F = ma
Work done against a gravitational field to lift an object to a height (h) is:
W = mgh
Power
The rate at which the work is done
Energy
The product of power (P) times time over utilization (t)
Kinetic Energy
Associated with the movement of an object
Two Types of Kinetic Energy
Translational motion (E = 1/2(m)(v)2)
Rotational motion (E = 1/2(l)(w)2)
Potential Energy
Most commonly associated with the energy of an object in a gravitational field given by:
E = mgh
Thermal Energy
Kinetic energy associated with the microscopic movement of molecules
E = 3/2(nRT)
A quantity of energy supplied Q supplied to material of mass and specific heat will increase the temperature by delta T.
Delta T = Q/mC
Chemical Energy
Energy associated with chemical bonds between atoms
Exothermic and endothermic reactions
Energy released in combustion reactions →
Nuclear Energy
Energy associated with bonds between neutrons and protons in the nucleus
Much greater than chemical energy
Energy release during an exothermic nuclear reaction → changes in total mass of system
Electrical Energy
Energy associated with flow of electreons in a conductor
Current (I) in conductor will experience voltage drop
Ohm’s law
V = IR
Energy of the electric and magnetic fields associated with electromagnetic waves (such as light).
Waves have a wavelength (λ) related to the frequency (f) and the velocity (c, speed of light).
Laws of Thermodynamics
0: Two systems both in thermodynamic equilibrium with a third system are in equilibrium
1: Energy is conserved
2: A closed system will move towards equilibrium
3: It is impossible to attain absolute zero temperature
Zeroth Law Thermodynamics
Implies that the thermodynamic state of system can be defined by a single parameter, the temperature
PV = nRT
Ideal Gas Law: linear relationship between temp and pressure
First Law Thermodynamics
When energy is applied through heat:
Internal energy of the gas increases if piston of tube is fixed
Energy is used to lift piston if piston moves
Second Law Thermodynamics
Heat naturally flows from hot to cold
Analogous to gravitational potential energy: object in a gravitational field only works if it moves from point of high potential to low
Total Energy in a system is calculated simply as:
= useful energy delivered + wasted energy
Energy efficiency (Ƞ) =
Useful energy delivered/energy input
Overall efficiency of a system =
product of all individual efficiencies
Efficiency
reducing the amount of energy input needed to meet our energy output needs
Conservation
reducing energy output needs or waste, e.g., through behavior/lifestyle changes
Types of Efficiency
Conversion efficiency
Functional/system efficiency
Heating your home
Specific Heat
Energy to raise a unit of mass by one degree
Heat Capacity
Ability of a substance to store heat
Solar Constant
Power density of the sun’s radiation at a distance from the Earth’s orbit
The solar constant is defined as the total power radiated by the Sun per unit _______ ____ of a _______ at Earth's orbit.
surface area, sphere
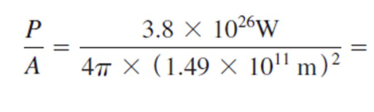
1367 W/m2
A blackbody radiation curve appoximates the solar radiation at ____K outside Earth’s atmosphere.
6000
The Earth’s atmosphere reflects and absorbs solar radiation. Certain _________ _______ in the atmosphere absorb at specific wavelengths.
molecular species
Total radiation on the Earth: _____ _________ times the _____-________ area of the Earth's disk.
Solar constant, cross-sectional
About ____ of radiation is absorbed or reflected by the Earth’s atmosphere.
half
Average (horizontal) insolation is the ________ ___________ arriving at the Earth’s surface divided by the _____ ____ of the earth.
Horizontal insolation, surface area
Distribution of Insolation
Solar insolation depends on:
Time of day
Day of year
Latitude
Overcast Day: Insolation/Solar Intensity
10-25% of that on a clear day, represents a 75-90% drop
Distribution of Insolation
Azimuth
Altitude angle
Zenith angle
Angle of incidence
Azimuth
Angle between the projection of sun rays and line due north/south
Altitude Angle
Angle between the horizontal and the line to the sun
Zenith Angle
Angle between the sun ray and the vertical directly overhead
Angle of Incidence
Angle between a sun ray and the perpendicular line to a surface at the point of contact
Electromagnetic Spectrum: Trends
Shorter wavelengths: higher energy intensity
Longer wavelengths: lower energy intensity
99.9% of solar energy is between UV and Infrared wavelengths
Electromagnetic Spectrum: Composition
UV 2%
Visible 47%
Infrared 51%
Electromagnetic Spectrum/PAR
Photosynthetically active radiation (400-700nm)
Photometric Sensor
Measures light as seen by the human eye in lux or lumens or foot-candles.
A good relative indication of the intensity and uniformity of lighting for human perspective.
Radiometric Sensor (Pyranometer)
Measurement of electromagnetic radiation (or radiant energy) emitted by the source, often defined in terms of power, W/m2.
Provide a relative indication of solar intensity for the whole spectrum.
PAR Sensor
Units of measurement: μmol m^-2 s^-1.
Compare PAR values at various points in the plant canopy and check PAR uniformity and intensity
Heat Transfer
Conduction, convection, radiation
Conduction
Heat transfer by conduction through a piece of material:
Temperature difference across the material (Th - Tc)
Cross sectional are of the material (A)
Thickness of the material (l)
The thermal conductivity of the material (k)
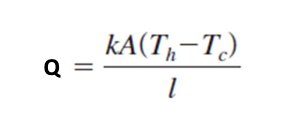
Thermal Conductivity
Refers to the ability of a given material to conduct/transfer heat.

Convection
Transport of thermal energy by fluid (gases or liquids) movement.
Free convection
Air or water moves away from the heated body as the warm air or water rises and is replaced by cooler air or water.
Forced convection
Air or water is forcibly moved across the body surface (such as in wind or wind-generated water currents) and efficiently removes heat from the body
Radiation
All objects radiate energy according to Stefan Boltzmann
Perfect Black Body
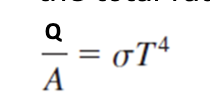
Not Perfect Black Body
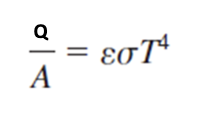
Most common materials have emissivities of around eta = 0._
0.9
Emissivity
Measures a material's efficiency in emitting thermal radiation compared to a perfect blackbody.
Earth Emissivity
Most natural Earth surfaces is a unitless quantity and ranges between approximately 0.6 and 1.0
Emissivity: Less than 0.85
restricted to deserts and semi-arid areas
High emissivities above 0.95 in thermal infrared wavelength
Vegetation, water, ice
Solar Energy Conversion
Conversion to electricity (photovoltaic effect);
Conversion to usable heat (for example, via thermal collectors);
Conversion to matter / fuel (for example, production of biomass through photosynthesis)
Solar Electric Generation
Use of photoelectric device
Conversion of solar radiation into heat → run a heat engine to drive a generator
Suitable temperature for operating a heat engine:
90 to 105ºC
Three most notable solar devices
Parabolic troughs
Dishes
Central receivers (solar towers)
PV cells are composed of ____________ material.
Semiconductor
Function of a semiconductor
Exposed to light; it absorb the light's energy and transfer it to negatively charged particles in the material called electrons. Extra energy allows the electrons to flow through the material as an electrical current.
PV Efficiency is regarded simply as:
the amount of electrical power coming out of the cell compared to the energy from the light shining on it.
______ efficiency is lower than individual cell efficiency.
Module
Solar cell efficiencies vary from _% for amorphous silicon-based solar cells to __._% with multiple junction production cells, and __._% with multiple dies assembled into hybrid packages.
6, 44.0, 44.4
Solar cell energy conversion efficiencies for commercially available Si PV are around __-__%.
14-22
_______ is, by far, the most common semiconductor material used in solar cells, representing approximately 95% of the modules sold
silicon
About 95% of solar panels on the market today use either _______________ silicon or _______________ silicon
monocrystalline, polycrystalline
Monocrystalline silicon
Comprise of one crystal structure
Efficiency higher → electrons move freely
Expensive
Polycrystalline silicon
Made up of many different crystals
Less expensive manufacturing
Thin Film Solar Cells
deposited layers of PV material on supporting material → glass, plastic, metal
amorphous silicon, cadmium telluride, and copper indium gallium selenide.
light, flexibility
Efficiencies 7-18%
Curved surfaces: greenhouses
Photo-selective Solar Cells: Opaque

Non-selective Solar Cells
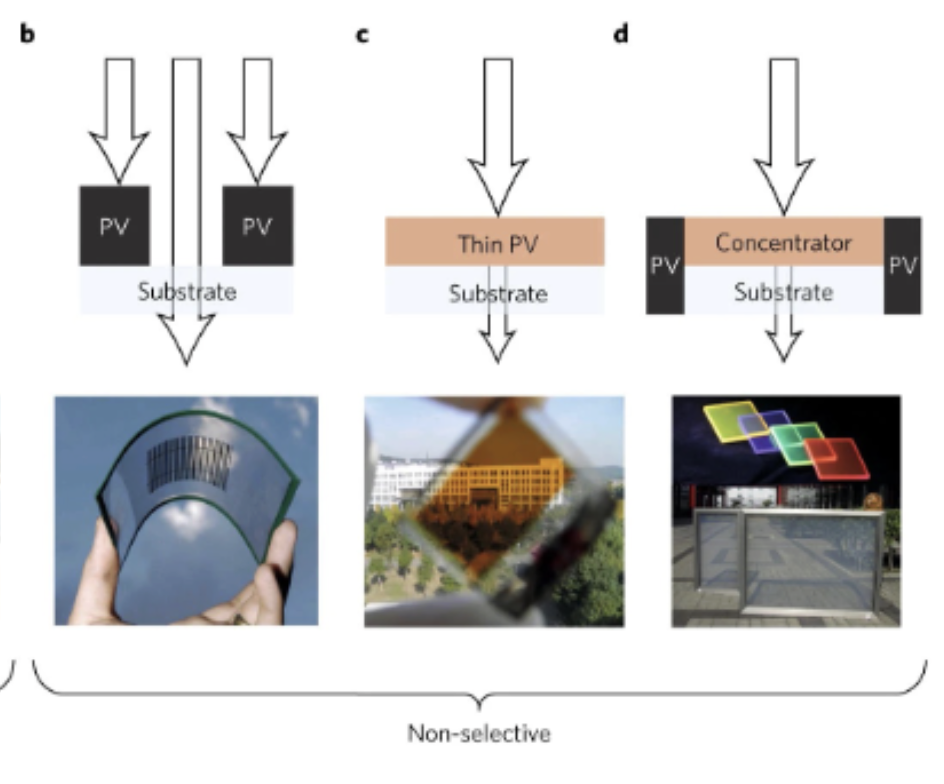
UV/NIR-selective Solar Cells
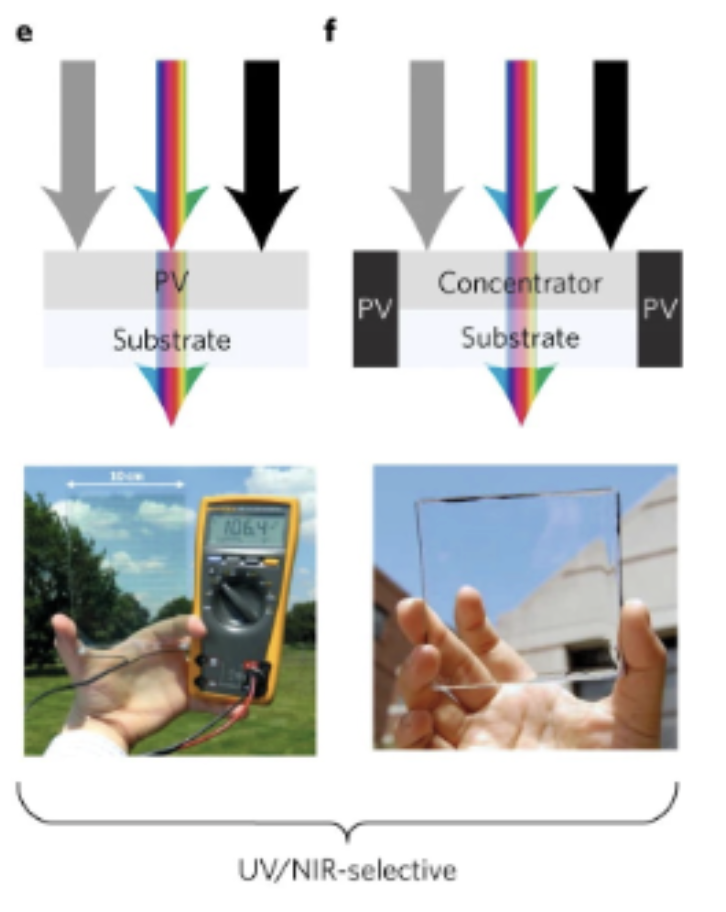
Factors of PV Performance
Transparent glazing (top surface)
Encapsulant: thin sheets of ethyl vinyl acetate holding top surface, solar cell, rear surface
Rear layer: substrate
Frame: typically aluminum
Electrical connection