Chemistry (P1/2) - Aluminium + Crude Oil + Ammonia
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Describe the Bayer process
Bauxite is set to react with sodium hydroxide in order to obtain sodium aluminate and get rid of impurities. Sodium aluminate is seeded with pure aluminium oxide and agitated with air to form a precipitate of aluminium oxide titrate. After 36 hours, the pure solid alumina is removed vacuum filtration then dehydrated in a rotary kiln at 1000 degrees Celsius. The sodium hydroxide is recycled
Describe the Hall-Heroult process
Pure aluminium oxide is dissolved in a mixture of molten cryoltie and aluminium floride or calcium fluoride in order to lower its melting point to around 1000 degrees Celsius. The mixture undergoes electrolysis in a tank lined with carbon (the cathode) with carbon blocks (the anode) bein gdipped into the molten electrolyte. At the cathode aluminium ions accept electrons and are converted to aluminium atoms. The molten aluminium which is 99.9% pure collects at the bottom of each cell and is siphoned off. At the anode, oxide ions lose electrons to form oxygen which often reacts with the anode to form carbon dioxide. Hence, the anode has to be replaced periodically and the carbon dioxide formed is led away through fume bonds. If at any point the concentration of aluminium oxide in the cell falls too much, dangerous fluorine gases evolve and the voltage required for the eleectrolysis rises. Which is why regular additions of alumina and calcium or aluminium fluoride are added to maintain constant electrolyte composition.
Describe the physical and chemical properties of aluminium which give way to its many uses (10)
Its soft, malleable and ductile nature make it capable of being drawn into wire and form thin sheets
It is an excellent thermal and electrical conductor
It is flexible and can be easily shaped by any industrial metalworking processes
It can be alloyed to increase its strength variably
It has a density similar to steel and can be used as a lighter substitute
It forms a thin compact coating of aluminium oxide which prevents further oxidation and mitigates effect of exposure to moisture, light, bacteria, odours and flavours
It is non-toxic and odourless
It has strong corrosion resistance
Polished aluminium is an excellent reflector of visible light and heat and can be used to make durable mirrors
It is 100% recycable and doing so only uses 5% of the energy used to produce aluminium from its ore
Describe the impacts the production of aluminium can have on the environment
Quarrying the bauxite ore can destroy landscape that could be repurposed for agriculture or left as an area of natural beauty
The quarries produce dust and fumes and the waste rocks from the bauxite ore may produce unsightly heaps
The waste products, such as red mud, from the purification of bauxite easily drain into soils, get into waterways and poison plants and animals due to the high concentrations of sodium hydroxide in the residue
The electrolysis of alumina uses vast amounts of electricity and creates dangerous greenhouse gases such as perfluorocarbons and carbon dioxides
The carbon cell linings of the electrolysis cells contain highly alkaline solutions, aluminium carbides and cyanides which are often just disposed in a landfill
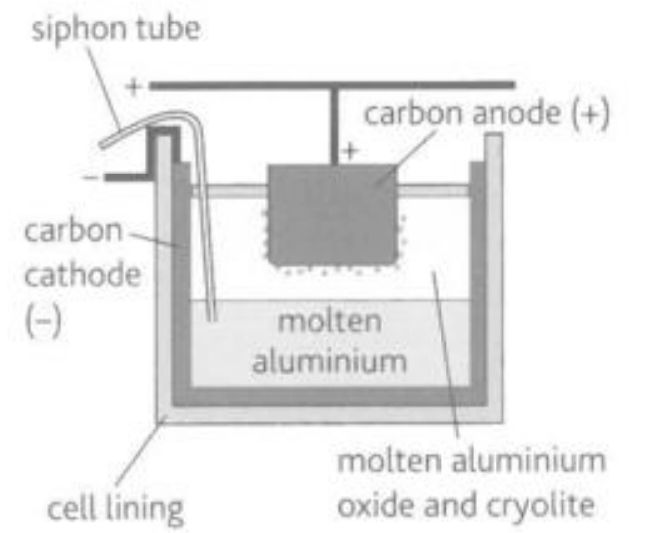
Describe this image
Electrolysis cell for the production of aluminum
Describe how crude oil is industrialized via distillation
The mixture of hydrocarbons is separated by distillation. Some natural gases are dissolved in the crude oil and removed by simple distillation. The remainder is transported to an oil refinery where it undergoes fractional distillation by being heated in a furnace to about 400 degrees Celsius. The vapour is fed into a fractionating tower which contains about forty trays containing bubble caps which allow thorough mixing of the vapour with descending liquid. The temperature gradient in the fractioning tower allows lighter hydrocarbons to be collected near the top and heavier hydrocarbons with higher boiling points to be collected near the base. After a component condenses on a particular tray it moves down to the tray below. When the ascending vapour reaches a tray containing liquid whose temperature is below the boiling point of the vapour, it starts to condense which heats the liquid in the tray and the more volatile components in the liquid evaporate.
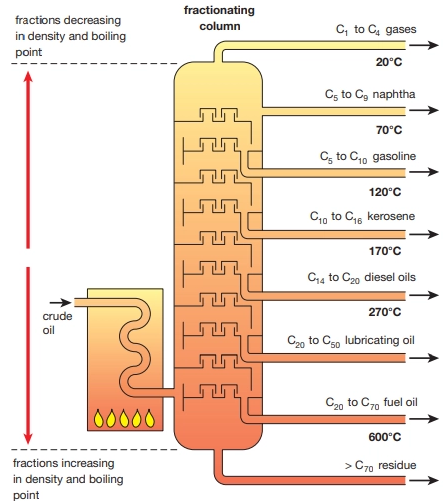
Describe this image
Fractional distillation of crude oil
State how oil refineries cater to the demand of lighter fractions of crude oil
Oil refineries convert the heavier fractions into lighter ones via cracking or reforming
Describe the process of reforming
It is the process in which alkanes are converted to cycloalkanes and cycloalkanes to arenes. When gasoline and naptha fractions are passed over a catalyst above 500 degrees Celsius, the straight-chain alkanes are converted to ring compounds, this is known as cyclisation. Platinium or molybdenum (VI) oxide catalysts catalyse the dehydrogenation while aluminium oxide catalyses any rearrangement of the carbon skeleton. Platinum and iridium atoms convert straight-chain alkanes to arenes while a catalyst containing platinum and rhenium atoms remove hydrogen from methylcyclohexane to form methylbenzene
List the uses of common crude oil fractions (8)
Refinery gas is used as gaseous fuel in heating appliances and can be liquified under pressure to produce LFG for domestic use.
Naptha is a raw material for making high octane gasoline, diluting bitumen, to produce alkenes and as a solvent in the manufacture of chemicals
Gasoline is used as a fuel for motor vehicles.
Kerosene is used as a fuel for jet engines and some rockets, fuel for domestic heating and can be cracked to produce gasoline
Diesel is used as fuel for diesel engines and can be cracked to produce kerosene and gasoline
Lubricating oil can be used to lubricate, in waxes, polishes, waxed paper, candles, solvent for ointment and fuel oil for large ships/factory boilers/large furnaces
Paraffin wax is used as a wax and serves as an insulator
Bitumen is used for road surfacing, roofing, waterproofing and sealing
State some of the impacts of the industrialization of crude oil (7)
Oil spills from oil wells or tankers can kill wildlife or severely harm them
The incomplete combustion of petroleum products results in toxic carbon monoxide being formed as well as carbon particles and unburnt hydrocarbons which can contribute to photochemical smog
Lead compounds from the addition of tetraethyl lead (IV) in gasoline can results in damage to the nervous system in children
Trace amounts of sulphur or nitrogen oxygen in fuels can react with oxygen and water in the air to form acid rain
Plastic made from petroleum products cause problems in terms of their disposal in the environment and their effect on wildlife
Some of the metals used as catalysts in the petroleum industry can escape into the air
Searching for oil often involves intense destruction to ecosystems
Describe the Haber Process
Nitrogen is obtained from fractionally distilling liquified air. Hydrogen is either made from natural gas reacting with steam in the presence of a nickel catalyst or by cracking ethane from the fractional distillation of crude oil. The carbon monoxide created from reacting natural gas with steam can poison the catalyst used in the Haber Process and is removed by reaction with more steam. A 1:3 gaseous mixture of pure nitrogen and pure hydrogen is compressed and passed into a converter which contains of promoter-supported iron-based catalyst at about 400-450 degrees Celsius at a pressure of 25-200 atmospheres. Under these conditions, up to 15% of the nitrogen and hydrogen are converted to ammonia which passes into an expansion chamber, cools condenses and is removed as a liquid. Any unreacted nitrogen and hydrogen are returned to the converter
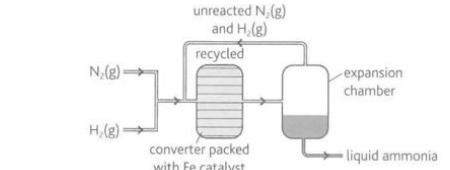
Describe this image
The Haber Process
Justify the conditions of the Haber Process
It is carried out at a high pressure so that the equilibrium shifts towards the right. It is also done at lower temperature because the reaction is exothermic and more heat/product can be consequently produced. Removing the ammonia by condensing also helps to improve yield as it can allow the equilibrium to reshift to the right in favour of fewer molecules
State common uses of ammonia
To manufacture nitric acid (for fertilizers, plastics, fabrics, dyes, sodium carbonate and pharmaceuticals
It is used in commercial and household cleaners
It is used in the food and beverage industry as a nitrogen source for yeast and to adjust the pH during fermentation
It is used as a refrigerant gas and a water purifier
State some of the effects of ammonia on the environment
Ammonia causes eutrophication.
Ammonia in the atmosphere can combine with nitrogen and sulfur oxides from vehicles to form particles which contribute to smog.
Ammonia can irritate the lungs and inhibit the uptake of oxygen by haemoglobin
It can also react with acids in the atmosphere to form ammonia salts which when inhaled over a period of time can cause bronchitis, asthma, coughing fits and farmer’s lung
An excess amount of ammonium ions in soil is converted by bacteria to nitrates, nitrates and hydrogen ions, the latter of which makes the soil more acidic
Ammonia gas can also settle on plant leaves and steams and cause damage because of its alkalinity
Detail the phenomenon of ammonia leading to eutrophication

eutrophication
the process by which excess quantities of fertilisers pollute rivers and lakes, causing an overgrowth of algae and bacteria leading to the death of aquatic organisms