Physics Feud
1/286
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
287 Terms
Equation of Traveling wave of light
Ey(x,t) = E0 exp(ikx − iωt)
Wave Nature of Light
Electromagnetic wave with certain frequency
Explains interference, diffraction, refraction, and reflection
electric and magnetic fields are perpendicular to each other and the direction of propagation
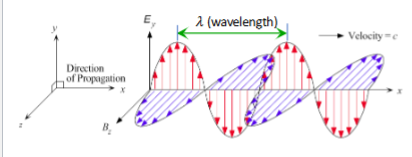
Intensity of light wave
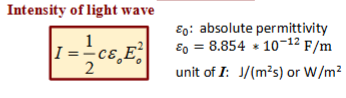
What does classical EM believe intensity only relies on (and is incorrect)?
Depends only on strength, not the frequency of light
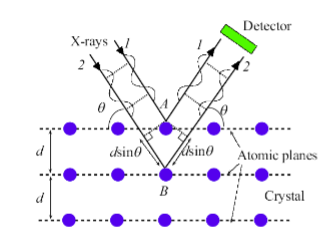
X-ray DIffraction
Uses constructive interference of waves being reflected by periodic atomic structure
Why are x-rays used for diffraction?
X-ray wavelength is comparable to the spacing between atomic planes
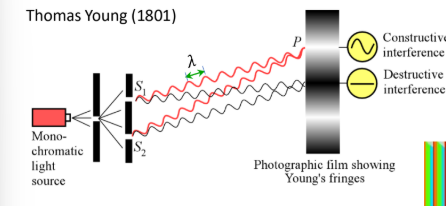
Young’s Double Slit Experiment
Demonstrates wave nature of light
experiment shows bright and dark fringes due to the constructive and destructive interference
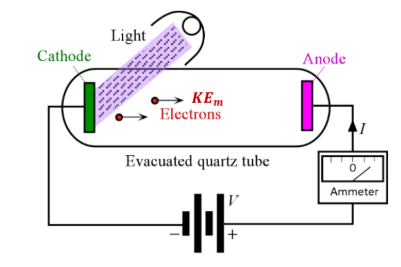
Photoelectric effect
electrons are emitted from a metal surface when light of a high enough frequency shines on it
Demonstrates particle nature of light
Work function
energy needed to free an electron for metal surface because PE of electron is lower than outside energy
varies per each element
Critical frequency
Frequency needed for the electrons to have enough energy to leave the metal surface
What happens when you apply a positive voltage to a photocathode
Electrons are accelerated towards the anode
Current increases until saturation
What happens when you apply a negative voltage to a photocathode
Reduces the current
Current becomes zero at the stopping voltage
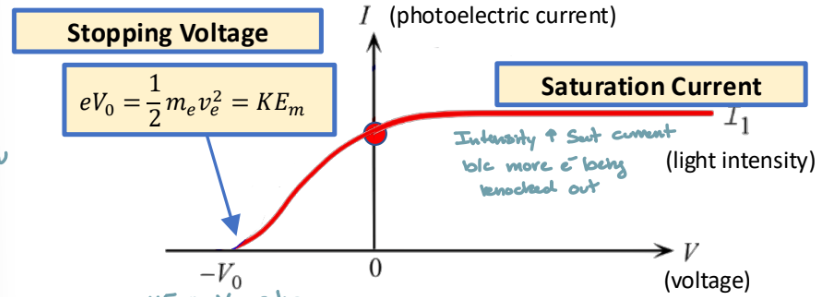
Stopping Voltage
minimum voltage to stop all electrons
increases with increasing frequency
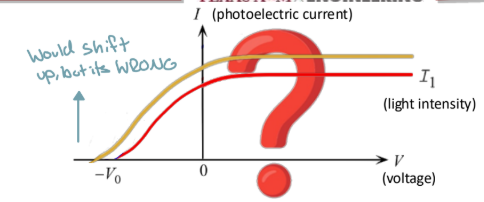
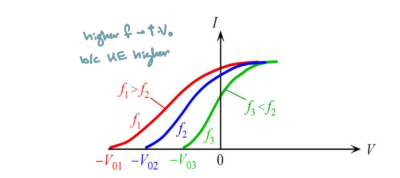
How would the current voltage plot of the photoelectric effect change according to classical theory?
The I-V curve would shift up, and the stopping voltage magnitude is greater
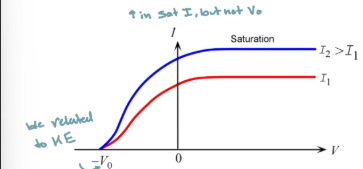
How does the I-V curve oft he photoelectric effect change when we change the intensify of light, but keep the frequency the same
saturation current increases with increases in intensity
Stopping voltage remains the same because the KE does not change
How does the I-V curve oft he photoelectric effect change when we change the frequency
Magnitude of Stopping voltage increases with increasing frequency
Max KE of electrons increases with increasing frequency
Main Quantum takeaways from photoelectric effect
Higher light intensity shall lead to higher electron kinetic energy, thus higher stopping voltage V0
Photoelectric current and stopping voltage shall not depend on light frequency f

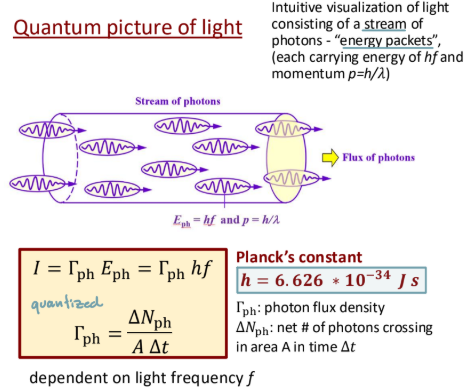
Quantum picture of light
Stream of energy packets that contain a frequency and momentum
Wave like behavior of light examples
Young’s double slit experiment
XRD
Particle like behavior of light examples
Photoelectric effect
Compton Scattering
Blackbody radiation
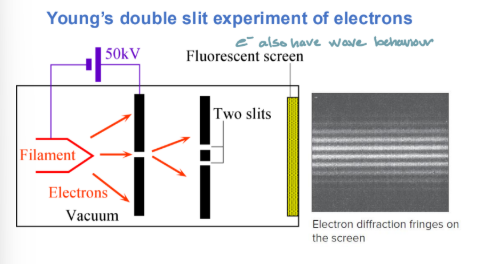
Young’s double slit experiment of electrons
electrons beam is fired through slits and produce a visible pattern striking a fluorescent screen
Shows wave behavior of electrons
De Broglie Relationship
relates the wavelength of electron to its momentums (branches wave and particle behavior)
Electron as a wave examples
Young’s double-slit experiment for electrons
Electron diffraction
Discrete emission and absorption spectra of hydrogen atom
Traveling wave general solution
u(x,t) = u0 exp(ikx − iωt)
Standing wave general solution (ends are fixed, has some reflection)
u(x,t) = 2u0exp(−iωt)cos(kx)
Probability of finding electron at a certain space and time
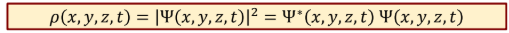
Total probability of finding electron in whole space at any time
1
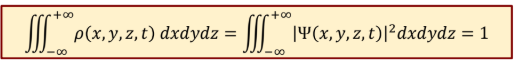
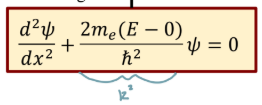
Time independent Schrodinger’s eq
Note that:
1. V and ψ are space-dependent function
2. E is energy of electron - a space-independent scalar value.
3. For a given V(x), we can solve Schrödinger equation and obtain ψ(x) and E, thus probability density
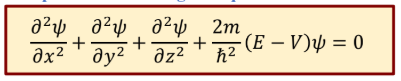
Time independent Schrodinger’s eq in 1D
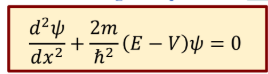
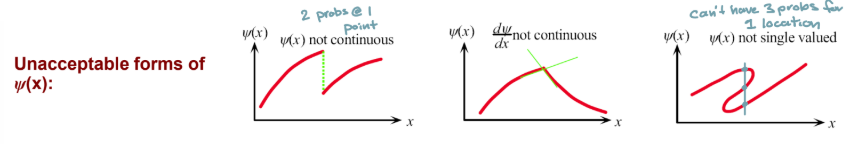
Constraints on ψ(x)
Must be continuous
Derivative of ψ(x) must be continuous
must be single valued
Expectation value in quantum mechanics
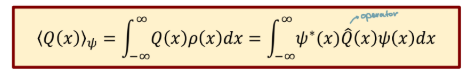
Infinite potential well
PE outside the well is infinite
PE of electron in well is 0, but always has a finite KE
Confined electron has quantized energy states that depend on well width and depth
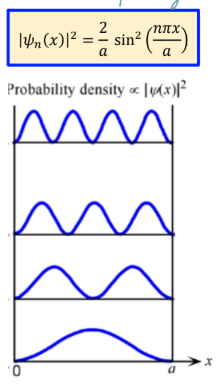
wavefunction has (n-1) nodes in the nth eigen-function (more oscillation, higher energy)
Probability at nodes is 0
Transition between 2 adjacent lever is also discretized (∆E = En+1 − En)
∆E approaches 0 as a goes to infinity (free electron)
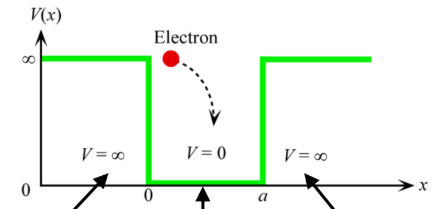
Boundary conditions for infinite potential well
ψ(x = 0) = 0
ψ(x = a) = 0
Steps to solve for energy in infinite potential well
Apply general solution: ψ(x) = Ae+jkx + Be−jkx
Apply boundary conditions (boundaries are 0)
Convert A(ejka -e-jka) = 0 to sin(ka) = 0
Apply ka = nπ
Plug (2meE)1/2a/ħ for k and solve E (E = (h²n²/8mea²))
Steps to solve for wavefunction in infinite potential well
Apply general solution: ψ(x) = Ae+jkx + Be−jkx
Apply boundary conditions (boundaries are 0)
Convert A(ejka -e-jka) = 0 to sin(ka) = 0
Use the normalization condition to set 1= integral of (2Asin(kx))² from 0 to a
Use identity integral of sin²(ax) = (x/2) - (sin(2ax)/4a)
Solve for A = 1/(2a)1/2
Eigenenergies in infinite potential well
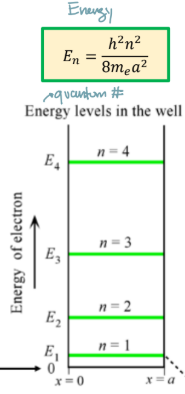
wave function in infinite potential well
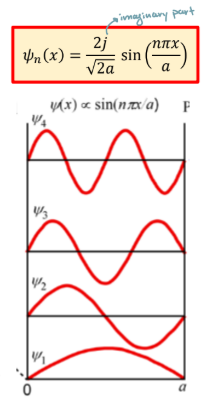
electron probability function of infinite potential well
Expectation value of PE in infinite potential well
0
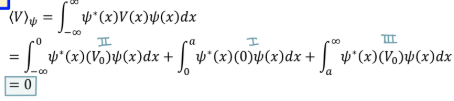
Expectation value of KE in infinite potential well
same as the eigenenergy
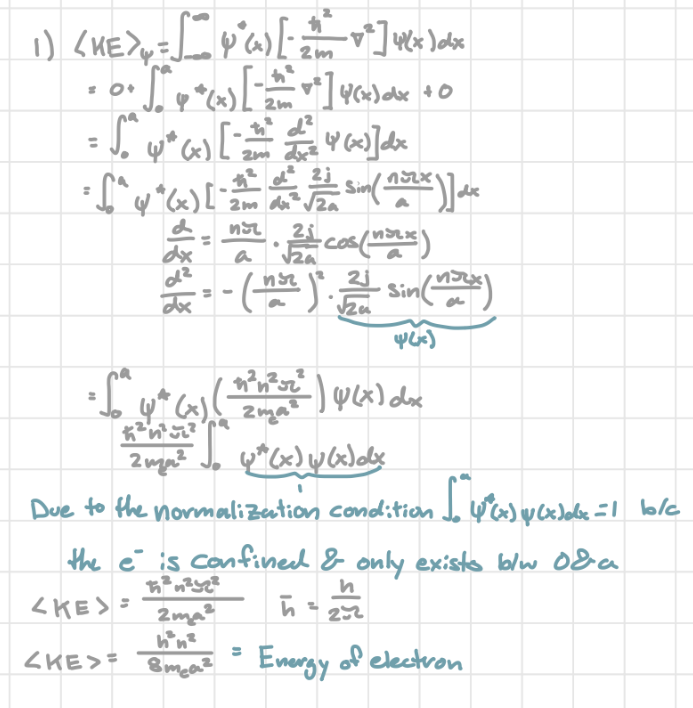
Electron in a finite well
Has a finite PE
electron has prob of penetrating in the barrier
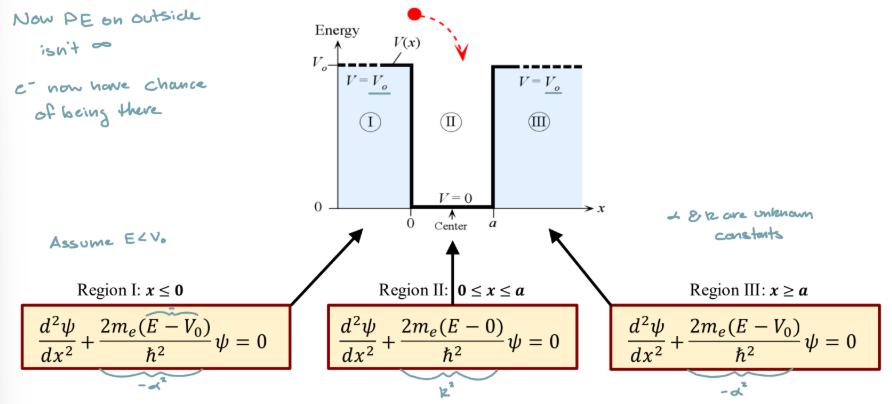
Equation for regions 1 and 3 in finite potential well

Equation for region 2 in finite potential well
Boundary conditions for finite potential well
function and derivatives are continuous at boundaries

Steps to solve finite potential well
Find general functions for Regions 1 and 3: ψI(x) = A1eαx and ψIII(x) = C2e−αx = C3e−α(x−a)
Reduce trial function of region 2 (ψII(x) = B1ejkx + B2e−jkx) into symmetric and antisymmetric wavefunction: ψIIas(x) = B1′ sink(x − a/2) and ψIIs(x) = B2′ cosk(x − a/2)
Apply boundary conditions and solve numerically to get: ψIIas(x) : α = −k cot(ka/2) and ψIIs(x) : α = +k cot(ka/2)
Intercept of α and k are the eigenenergies
Alpha for a symmetric wavefunction in finite potential well
+k cot(ka/2)
Alpha for an antisymmetric wavefunction in finite potential well
-k cot(ka/2)
How do the eigenenergies change if the finite quantum well width increases?
decreases: increasing a squeezes k in cot function which decreases where k intercepts alpha
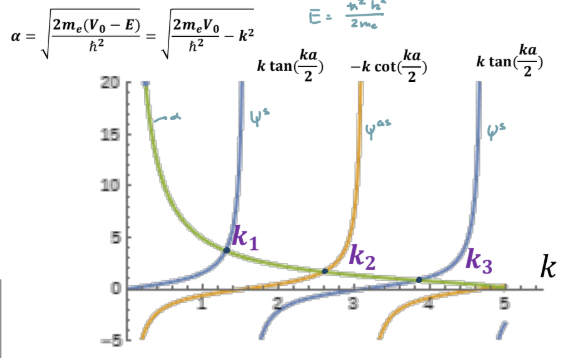
How do the eigenenergies change if the finite quantum well depth increases?
increases: increases outside potential which increases alphas, then increases k thus E
Finite potential well vs infinite potential well
Finite has a finite number of energy states, while infinite has infinite
Energy levels in finite potential well are lower because uncertainty is greater due to wall penetration, decreasing the energy
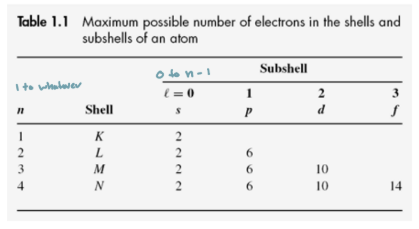
Principle Quantum Number (n)
Quantizes electron energy
Determines size/shell of orbital
n = 1,2,3,…
Orbital angular momentum quantum number
Quantizes the magnitude of orbital angular momentum
Determines orbital shape (s,p,d,f)
l = 0, 1, 2, ... (n − 1)
Magnetic Quantum number
Quantizes the orbital angular momentum component along a magnetic field Bz
Determines the orientation of orbital (px, py, pz)
ml = 0, ±1, ±2, ... , ±l
Spin magnetic Quantum number
Quantizes the spin angular momentum component along a magnetic field Bz
ms = ±1⁄2 (spin up vs spin down)
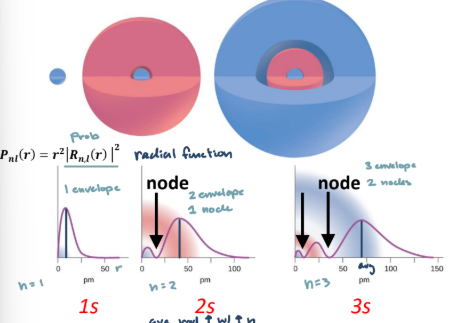
Hydrogenic atom model
Energy is quantized and determines by principle quantum number
Distance between energy levels decreases at higher and higher n
ψn,l,ml(r, θ, φ) = Rn,l(r)Yl,ml(θ, φ)
Radial function demonstrates nodes between subshells
Quantum tunneling
electron can leak through potential energy barrier
In region I, the incident and reflected waves interfere to give ψI(x)
In region II, the wave function tunnels through potential barrier and decays with x because E < Vo.
In region III, no reflected wave.
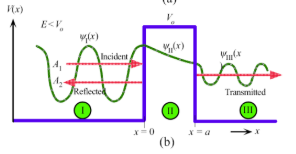
Schrodinger’s eq and general wavefunction for region 1 of quantum tunneling

Schrodinger’s eq and general wavefunction for region 2 of quantum tunneling
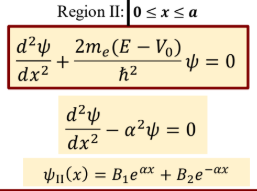
Schrodinger’s eq and general wavefunction for region 3 of quantum tunneling

Probability of tunneling
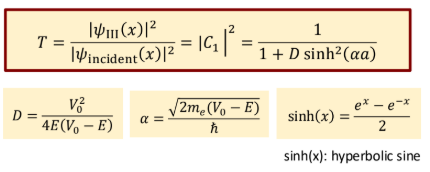
Probability of reflecting

Probability of tunneling for a wide or high barrier
exponentially decreases with wider or higher barriers
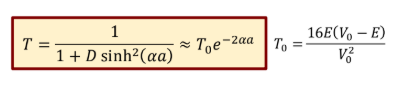
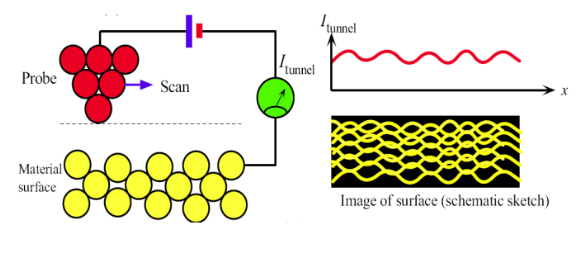
Scanning tunneling microscopy (STM)
Uses the tunneling effect to map atomic scale topology
Applies a voltage to lower PE barrier between tip and sample, creating a tunneling current
Pauli exclusion principle
In a given atom, no two electrons can have the same set of quantum numbers
max of 2 electrons allowed in an orbital, must pair their spins (± 1/2)
Boltzmann Distribution
Classical stats
can describe quantum particles when there are many more available states than particles (E-Ef » kt)
Prob at low energy levels can go to infinity
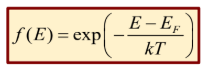
Deriving Boltzmann stats
Set forward and reverse rxn equal: P(E1)P(E2)= P(E3)P(E4)
Assume E2 = δE, E4 = 0: P(E1)P(δE) = P(E1 + δE)P(0)
Taylor series expansion that P(δE) ≈ P(0) + P′(0)δE & P(E1+ δE) ≈ P(E1) + P′(E1)δE
Plug expansion in and cancel out δE: P(E1)P′(0)
δE= P′(E1)P(0)δEUse P’(E)/P(E) = P’(0)/P(0) = constant, to get general solution: P(E) = Aexp(− βE)
P(E) = Aexp(-E/kT)
Fermi-Dirac Statistics
Quantum particles with half integer spin (electrons)
Obeys pauli exclusion principle
Prob at low energy levels goes to 1
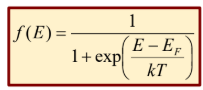
Fermi energy
electron occupancy probability is 50%
When can the fermi Dirac distribution be approximated by the Boltzmann distribution?
As the Energy increases ((E-EF»kT)
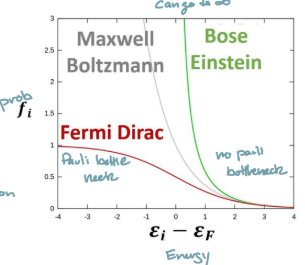
How does the Fermi Dirac distribution increase at higher temps?
Curves near fermi level become more gradual
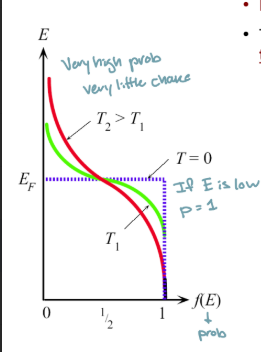
Bose-Einstein Statistics
Quantum stats for particles with integer spin (photons, phonons)
unlimited number of particles per state, at low energy levels, can reach infinity
Crystal
repeated, periodic, and infinite array of identical groups of atoms
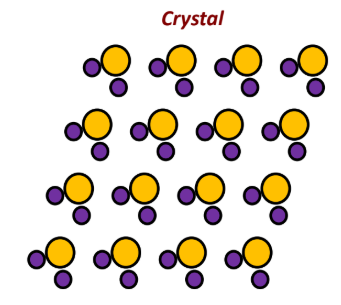
Basis
single repetitive group of atoms

Lattice
the sets of the periodic mathematical points which the basis is attached to
Translation Vectors
vectors a1, a2 (a3), such that the arrangement of atoms in the crystals looks the same when the entire lattice is translated
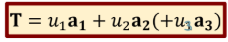
Primitive vectors
Translation vectors that result in the smallest volume of the parallelogram/parallelopiped
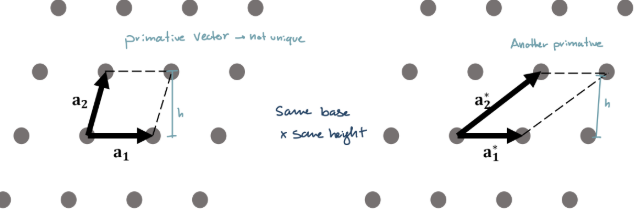
Primitive Cell
Parallelogram defined by the primitive vectors
All primitive have the same volume
Unit cell
contains all the essential information about the crystal and the entire crystal structures are repetitions of the unit cell.
A primitive cell isa unit cell, but a unit cell can be non-primitive
Wigner-Seitz Cell
a primitive cell and represents the highest level of symmetry
Wigner-Seitz Cell Construction
draw lines to connect a given lattice point to all nearby lattice points;
at the midpoint and normal to these lines, draw new lines or planes.
The smallest volume enclosed in this way is the Wigner-Seitz cell.
How many possible rotation operations are possible for a 2D lattice
5: 1-fold (360°), 2-fold (180°), 3-fold (120°), 4-fold (90°), 6-fold (60°)
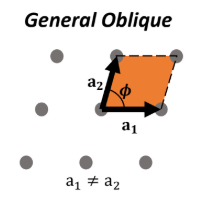
General Oblique lattice
a1 ≠ a2, no restriction on θ
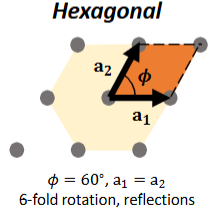
Hexagonal Lattice
θ = 60°, a1 = a2
6-fold rotation, reflections
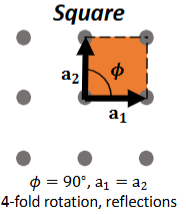
Square Lattice
θ = 90°, a1 = a2
4-fold rotation, reflections
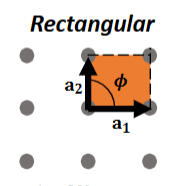
Rectangular Lattice
θ = 90°, a1 ≠ a2
2-fold rotation, reflections
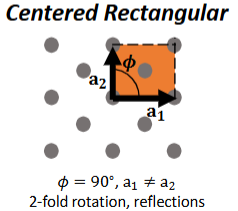
Centered Rectangular
θ = 90°, a1 ≠ a2
2-fold rotation, reflections
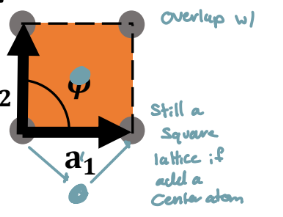
Why is the centered square not a special type?
a centered square lattice can just be reduced to a normal square lattice
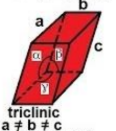
triclinic
a ≠ b ≠ c
α ≠ β ≠ γ ≠ 90
monoclinic
a = b ≠ c
α = γ = 90, β ≠ 90
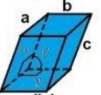
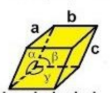
rhombohedral
a = b = c
α = β = γ ≠ 90
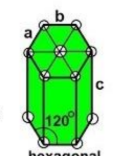
hexagonal
a = b ≠ c
α = β = 90, γ ≠ 120
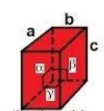
orthorhombic
a ≠ b ≠ c
α = β = γ = 90
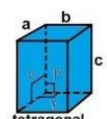
tetragonal
a = b ≠ c
α = β = γ = 90
Cubic
a = b = c
α = β = γ = 90
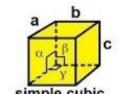
Primitive vectors and cell for simple cubic
(a,0,0), (0,a,0), (0,0,a)
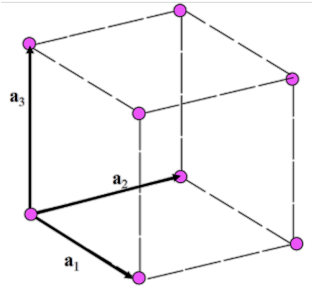
Number of atoms per cell and atom positions for simple cubic
1
(0,0,0)
Number of nearest neighbor of simple cubic
6
Primitive vectors and cell for bcc
(a/2,-a/2,a/2)
(a/2,a/2,-a/2)
(-a/2,a/2,a/2)
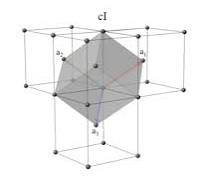
Number of atoms per cell and atom positions for bcc
2
(0,0,0)
(a/2,a/2,a/2)
Number of nearest neighbor of bcc
8