Topic 14 Redox II
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
What is meant by standard electrode potential?
the voltage/potential difference of a cell when a half-cell is connected to a standard hydrogen electrode measured under standard conditions
Conditions of standard electrode potential:
298K temperature
100kPa pressure of gases if gases are involved
1.0 moldm-3 concentration of ions
Features of a standard hydrogen electrode
more complex and less reliable as it is difficult to maintain the pressure of gas at 100kPa (as a gas is involved)

Why is a reference electrode necessary?
contains a standard electrode potential of 0V under standard conditions
used to measure the standard electrode potential of another half cell
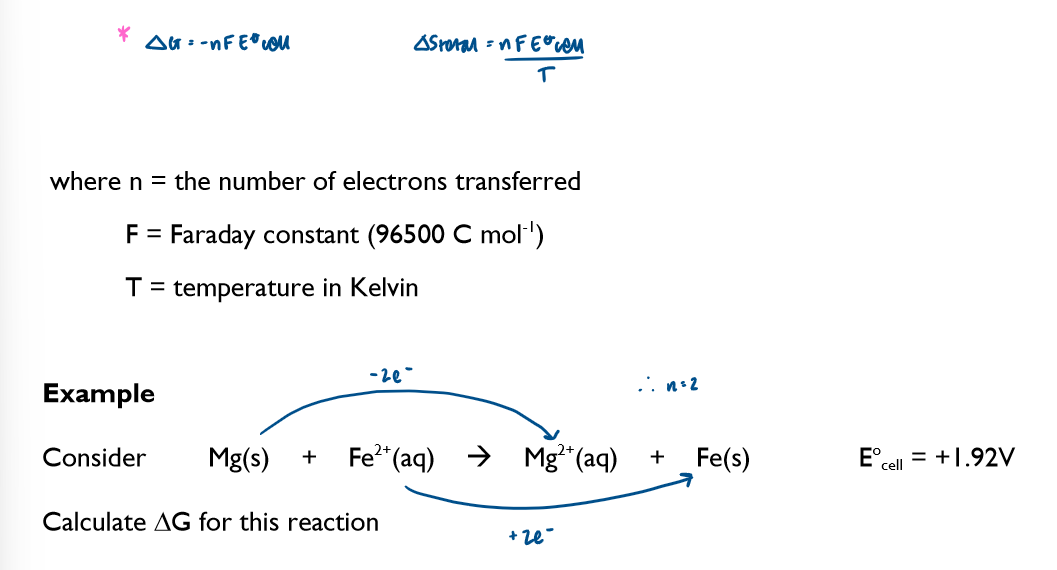
Calculate the emf by combining two standard electrode potentials
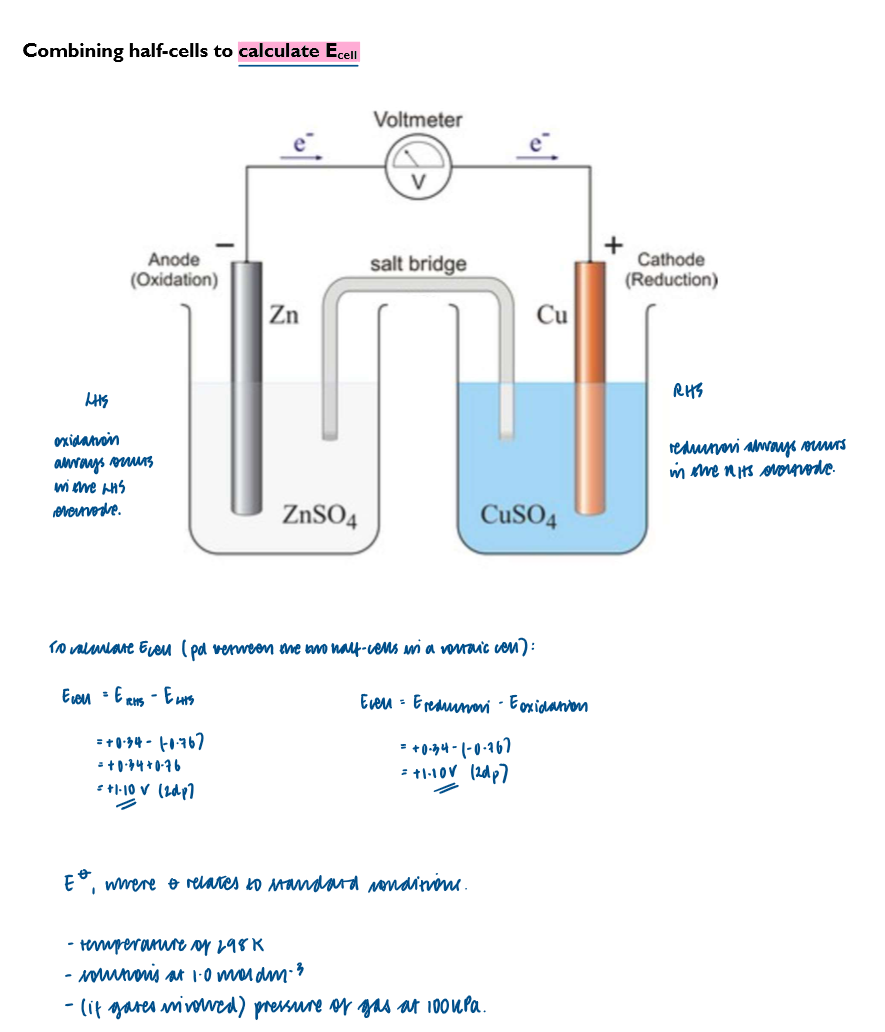
Write cell diagrams using conventional representation of half-cells

Importance of conditions when measuring the electrode potential, E
temperature of 298K
If using a gas, 100kPa
Solutions of 1.0moldm^-3
Predict the thermodynamic feasibility of a reaction using standard electrode potentials
In order for a reaction to be thermodynamically feasible, the Ecell value must be positive. This does not mean that the reaction will happen, as kinetic factors, such as a high Ea, could cause the rate of reaction to be effectively 0.

Why can’t HCl be used to acidify potassium permanganate?

What is the electrode potential of a cell directly proportional to?
the total entropy change for a reaction
lnK for a reaction
(therefore a positive E cell will have an overall positive entropy change - spontaneous)
What are the limitations of predictions made using standard electrode potentials
kinetic inhibition
departure from standard conditions
While a reaction may be thermodynamically feasible it may occur at such a slow rate that it doesn’t actually occur.
Limitations are non-standard conditions or high activation energy.
What can standard electrode potentials be listed as?
an electrochemical series
How does disproportionation relate to standard electrode potentials?
Disproportionation definition => an element in a single species is simultaneously oxidised and reduced

What are different methods used to measure standard electrode potentials of?
metals or non-metals in contact with their ions in aqueous solution
ions of the same element with different oxidation numbers
What is the energy released on the reaction of a fuel with oxygen utilised in?
a fuel cell to generate a voltage
Methanol and other hydrogen-rich fuels
Electrode reactions that occur in a hydrogen-oxygen fuel cell:

Carry out both structured and non-structured titration calculations including Fe2+/MnO4-:

Methods used in redox titrations (MnO4- ):
redox titration uses the same techniques as an acid-base titration but is applied to the reactants in a redox reaction
MnO4- ion acts an as oxidising agent:
acts as an oxidising agent in acidic solution
The mole ratio is 5 mol Fe2+: 1 mol MnO4-
potassium manganate (VII) acts as its own indicators during titration with aqueous iron (II) ions
dilute H2SO4 must also be added to the conical flask to supply the H+ ions
purple MnO4- (aq) ions are reduced to colourless Mn2+ (aq) ions
pale green Fe2+ (aq) ions are oxidised to pale yellow Fe3+ (aq) ions
the end-point of the reaction occurs when a hint of pink is observed in the flask showing a slight (one drop) excess of MnO4- (aq) when all Fe2+ (aq) has reacted
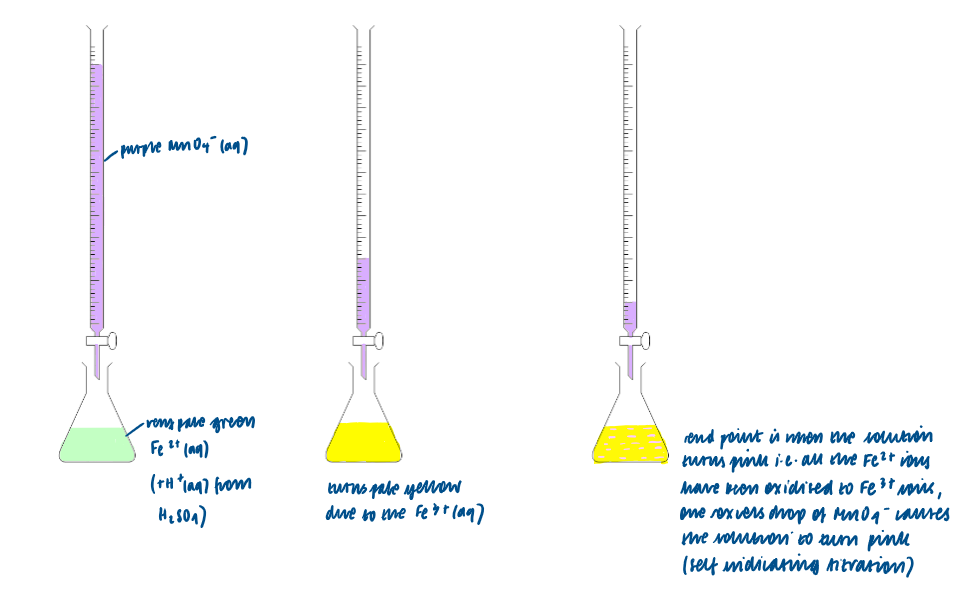
Half equations and overall equation of MnO4-/Fe2+:

Carry out both structured and non-structured titration calculations including I2/S2O32-:

Methods used in redox titrations (S2O32-):
Sodium thiosulphate can act as a reducing agent:
mole ratio is therefore I mole of I2: 2 mole S2O32-
none of the ions resent in the iodine / thiosulphate reaction are coloured
starch, which forms a deep blue complex with iodine, can be used as an indicator to show a clear end point for the reaction
a small quantity of starch solution is added just before the end point when the iodine solution is a pale straw colour (i.e. when most of the iodine has reacted)
the end point is when the intense blue colour of the starch-iodine complex finally disappears
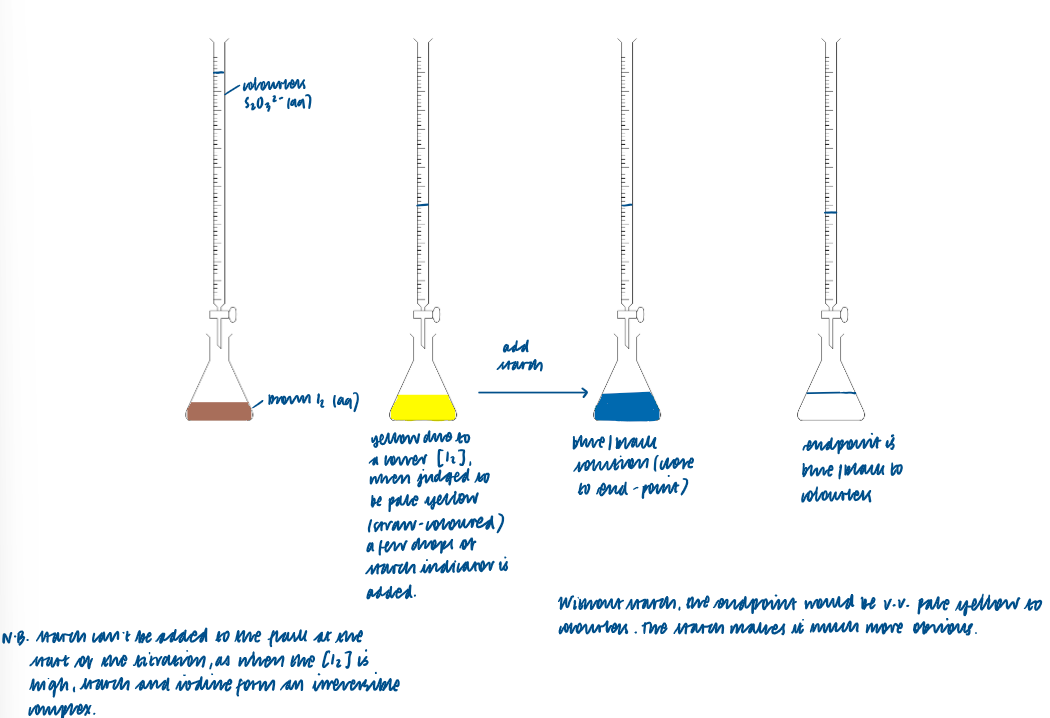
Half equations and overall equation of I2/S2O32-


Using redox titrations to work out the oxidation state of an element - Example 1:


Using redox titrations to work out the oxidation state of an element - Example 2:


Using redox titrations to work out the oxidation state of an element - Example 3:


Using redox titrations to work out the oxidation state of an element - Example 4:


Using redox titrations to work out the oxidation state of an element - Example 5:

What must a half-cell contain?
Both the oxidised and reduced species from a half-equation
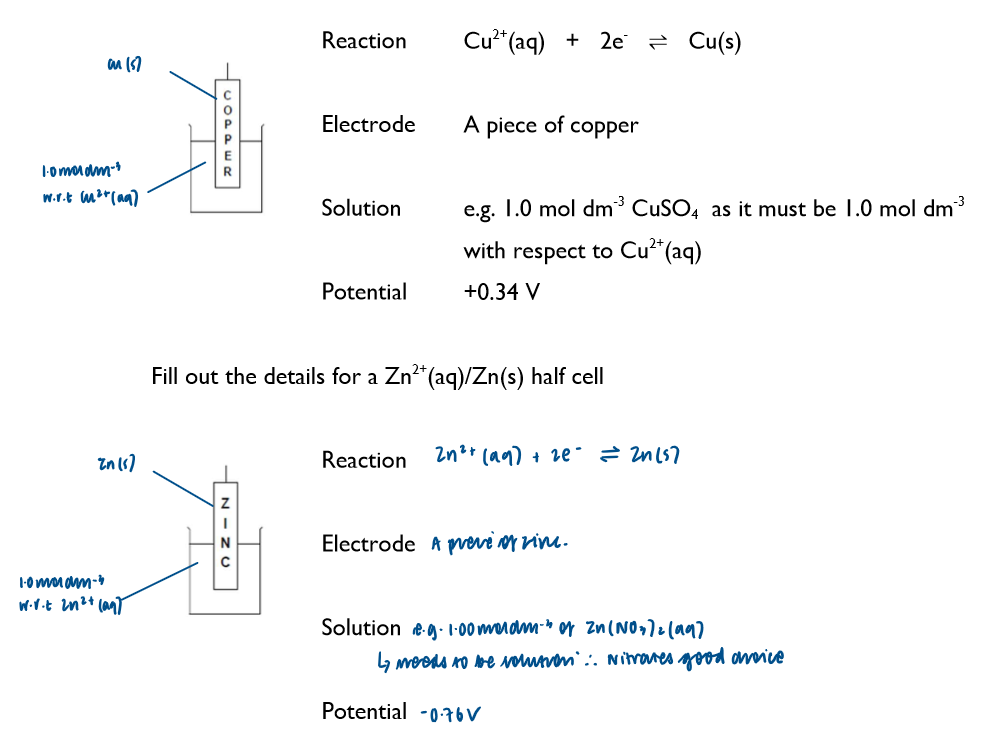
Half-cell with metal in contact with a solution that contains metal ions:
What is an electrochemical cell?
2 half cells joined together
How do you set up an electrochemical cell?
use sandpaper or emery paper to rub off any metal oxides formed on surface of metal during oxidation of the metal which acts as an insulator
place each metal in a solution of ions of the same metal for example using a zinc electrode placed in solution of zinc nitrate
place a salt-bridge (filter paper soaked in saturated potassium nitrate) across both beakers - has to be submerged in solution
allows ions to move and carry charge to complete circuit (electrons can’t move across this)
Why use a high resistance voltmeter in an electrochemical cell?
prevents current from flowing
if replaced by lightbulb, the current will flow, and the bulb will light
the light bulb will go out eventually when one of the reagents is used up
as voltmeter measures the voltage between two half cells (Ecell)
Why is platinum used as an electrode when 2 aqueous ions are involved?
metallic so conduct electricity but are also inert so won’t interfere with reaction
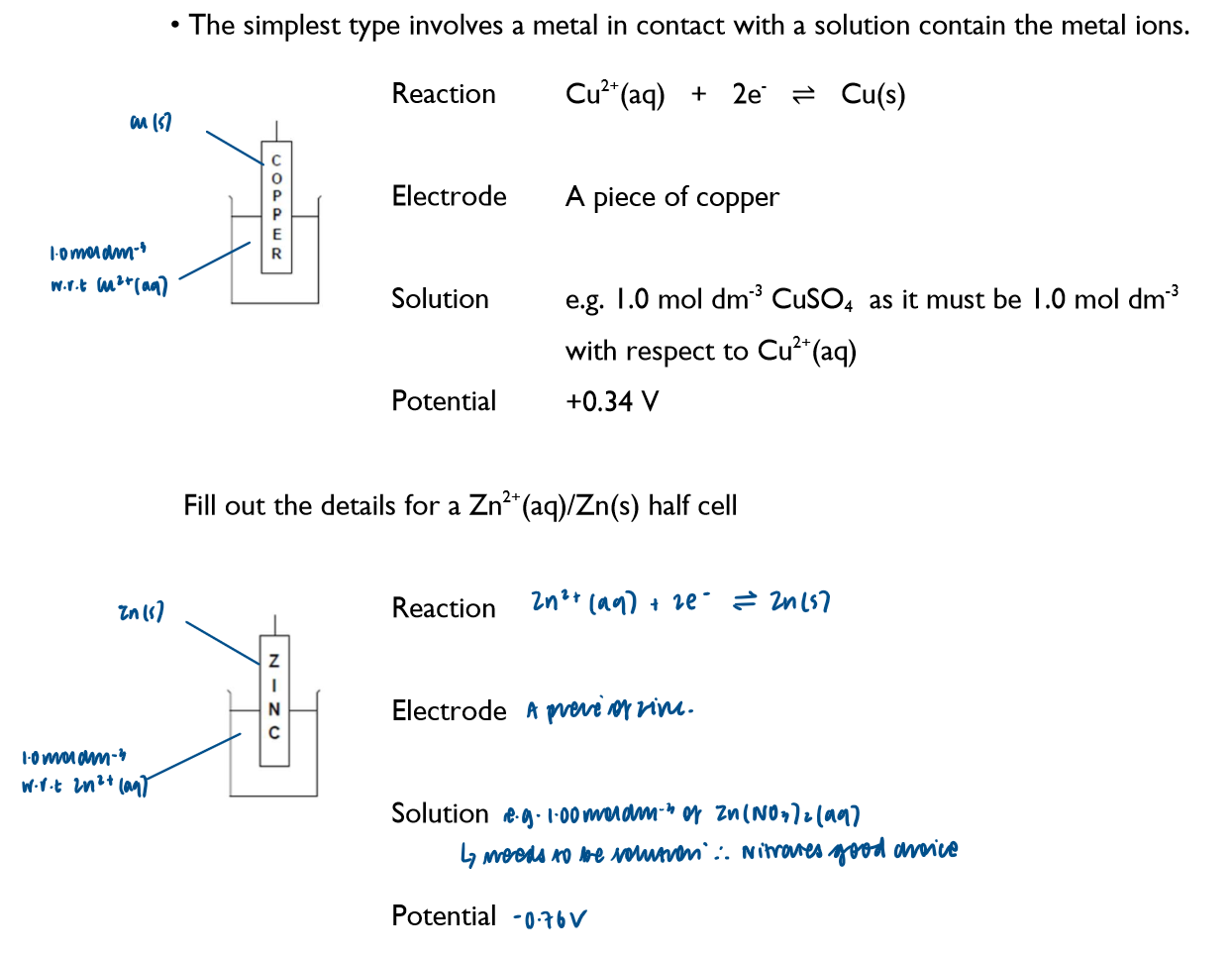
Explain what happens at a Zn2+/Zn half cell:
although it looks as though nothing happens in a half-cell, some of the metal atoms get oxidised forming ions, leaving the electrons on the piece of metal
the reversible arrow shows that the reaction happens in both directions
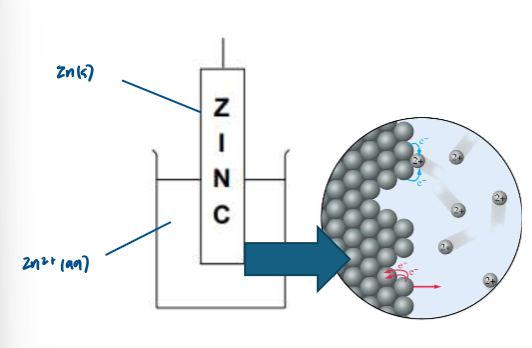
Half equation of previous reaction:
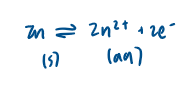
Explain what happens at a Cu2+/Cu half-cell:
as Cu is less readily oxidised compared to Zn, there’d be less of a build-up of electrons on the piece of Cu (s) metal
Half equation of previous reaction:
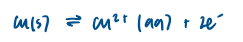
Details relating to a Zinc/Copper voltaic cell
when the two half-cells are connected they form a voltaic cell
a voltaic cell is an example of an electrochemical cell
a wire allows electrons to flow from one half-cell to the other
a salt bridge completes the circuit by allowing the flow of ions
What happens at the Zn electrode?
the Zn electrode is the negative electrode, as there is a greater build-up of electrons on the Zn electrode compared to the Cu electrode
Why is the fact that the voltmeter is high resistance important?
the voltmeter is a high resistance voltmeter which prevents much current from flowing
this enables us to measure the pd (or the cell emf) when two-half cells are combined
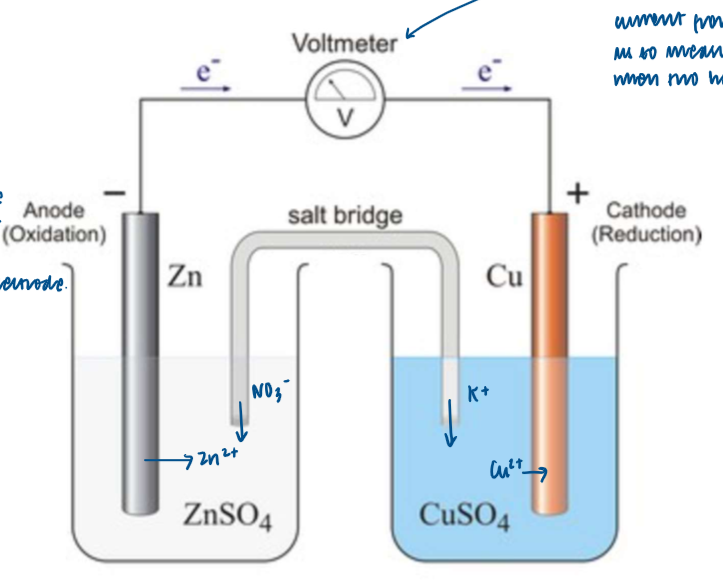
Explanation of this voltaic cell
no reaction/ very little reaction takes place while the high-resistance voltmeter is in the circuit
if it’s replaced by a bulb, or if a wire joins the two electrode together, then electrons will flow freely from left to right
once this happens, the size of the Zn electrode will get smaller due to Zn→ Zn2+ + 2e- (oxidation) and the size of the Cu electrode will get bigger due to the Cu2+ + 2e- → Cu (reduction).
the salt bridge completes the circuit
it is usually made from a strip of filter paper soaked in saturate aqueous KNO3
in the LH half-cell, zinc metal is oxidised to Zn2+ ions
the Zn2+ ions enter the solution, two NO3- ions from the salt bridge enter the solution to maintain neutrality
in the RH half-cell, Cu2+ ions are reduced to Cu metal
the [Cu2+] in the solution therefore decreases, and to maintain neutrality for each Cu2+ ion that is reduced, two K+ ions enter the solution from the salt bridge
Write down the key features of a half cell where the metal is in contact with the solution containing its metal ions (using Cu and Zn as an example)
Write down the key features of a half-cell which involves a gas, using H2 as an example

Write down the key features of a half-cell which involves a gas, using Cl2 as an example
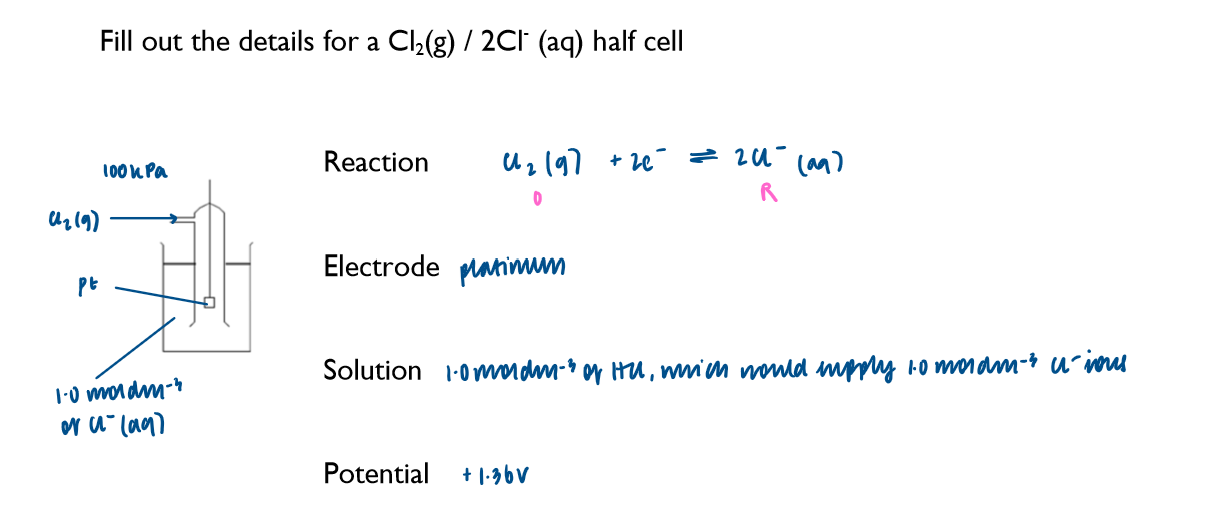
Write down the key features of a half-cell involving both the aqueous ions of the same element, using Fe2+ and Fe3+ as an example
(when you add two equal volumes of 2 solutions, it halves the concentration of each ions)

Write down the key features of a half-cell involving both the aqueous ions of the same element, using V2+ and V3+ as an example
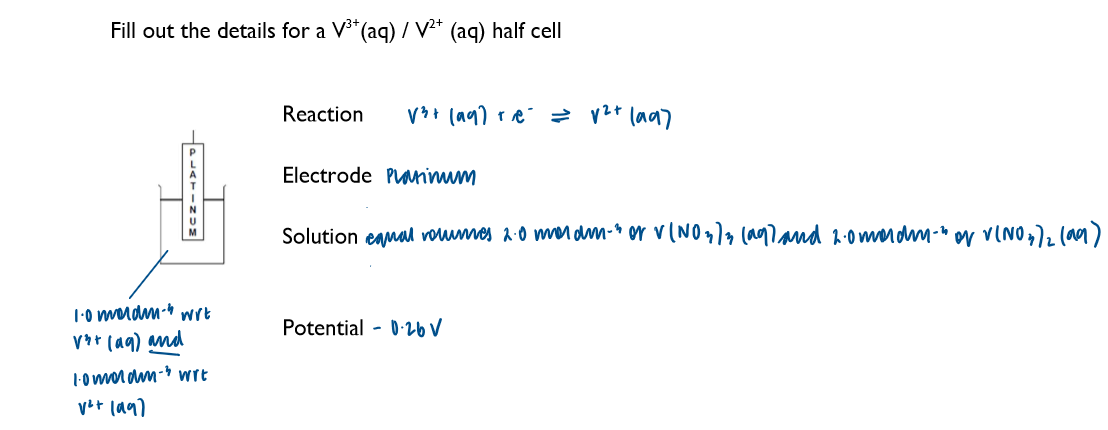
Write down the key features of a half-cell when it contains an oxidising agent (using MnO4- as an example)
For oxidising agents, it is important that the solution is acidic

Write down the key features of a half-cell when it contains an oxidising agent (using Cr2O72- as an example)
For oxidising agents, it is important that the solution is acidic

Define standard electrode potential
the potential difference of a cell when a half-cell is connected to the standard electrode under standard conditions
What is the value of the electrode potential affected by?
temperature
pressure of gases
concentration of the solution
By convention, what side does the standard hydrogen electrode go on?
the left hand side
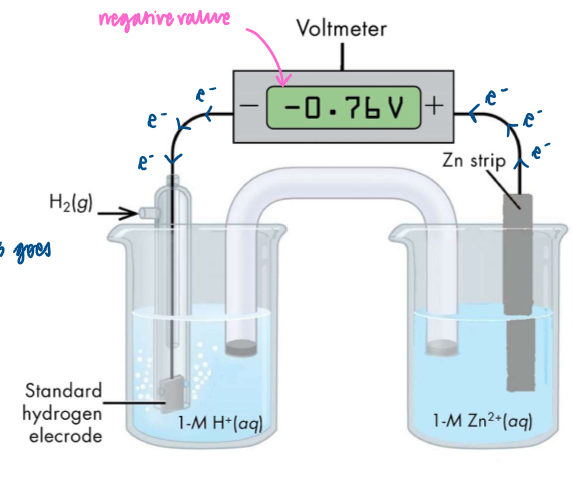
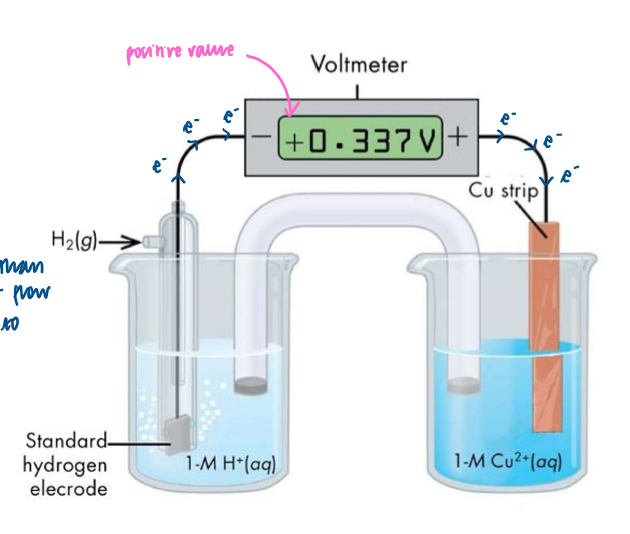
How to measure the potential difference between the standard hydrogen electrode and the Zn2+ (aq)/ Zn(s) half cell
set up voltmeter as shown
Zn is a better reducing agent than H2, so Zn is oxidised and electrons flow around the circuit from right to left
Ecell = ERHS - ELHS = -0.76V
How to measure the potential difference between the standard hydrogen electrode and the Cu2+ (aq)/ Cu(s) half cell
set up voltmeter as shown
H2 is a better reducing agent than Cu, so H2 is oxidised and the electrons flow around the circuit from left to right
Cu2+ (aq) + 2e- → Cu (s)
Ecell = ERHS - ELHS = +0.34V
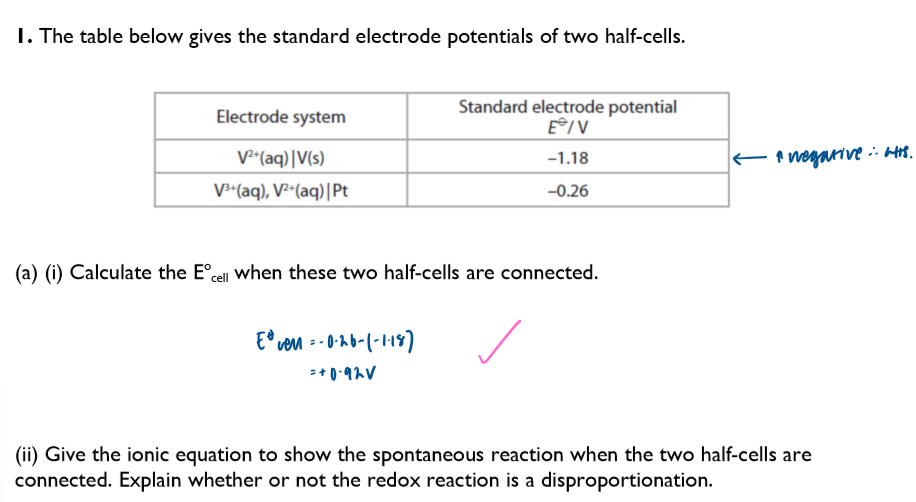
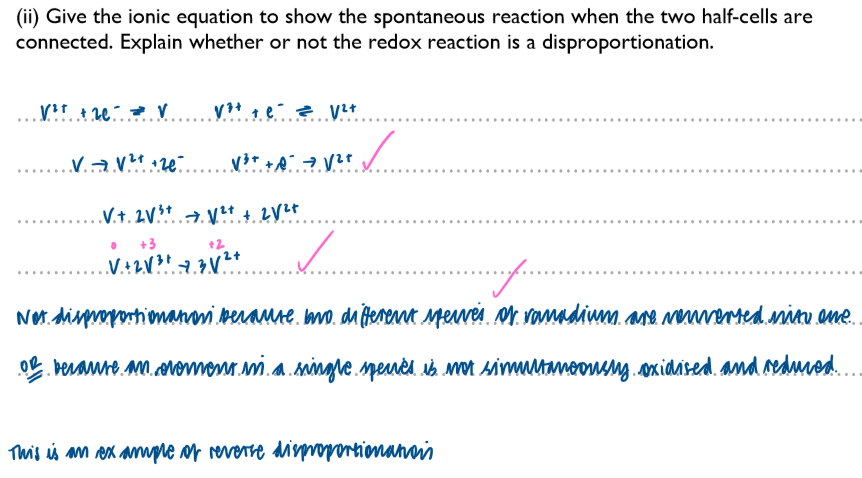
A question regarding disproportionation and Ecell:
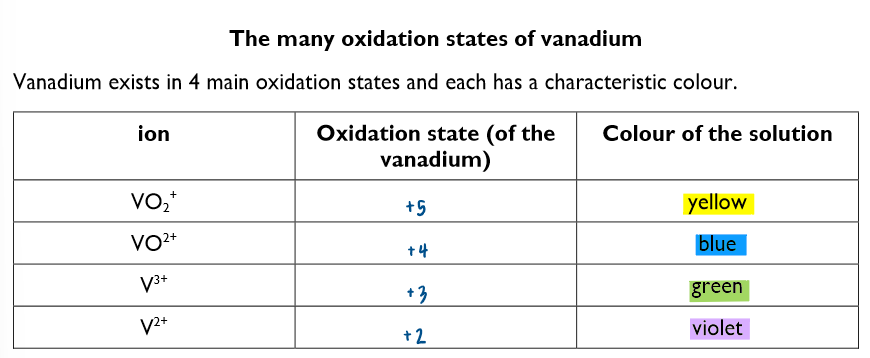
Name the oxidation states and their associated colours of Vanadium:
Show the reaction that occurs when iodide ions are used to reduce vanadium +5

Show the reaction that occurs when tin is used to reduce vanadium +5

Show the reaction that occurs when zinc is used to reduce vanadium +5

What must be true if a reaction is thermodynamically feasible?
the total entropy must be positive
gibbs free energy must be negative
Ecell must be positive
What is true about the relationship between the Ecell of a redox reaction and the total entropy change and lnK for a reaction?
they are directly proportional
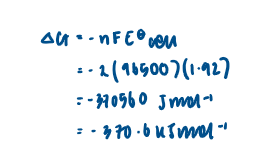
What must you think about if non-standard conditions are used?
le Chatelier’s principle - equilibrium
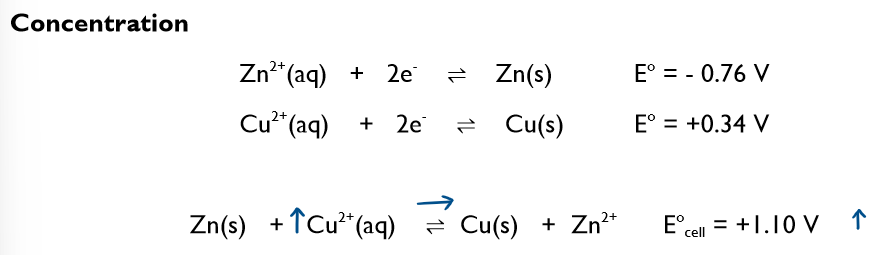
What happens if you increase the concentration of Cu2+?
This would cause the equilibrium to shift to the RHS so Ecell for the forward reaction would become more positive.
Any change that would result in the position of equilibrium shifting to the RHS will result in the value of the Ecell becoming more positive.
What is the effect of changing the temperature on the value of Ecell?
The majority of voltaic cells are exothermic in the spontaneous direction. You would have to be told the value of delta H or whether the reaction was exothermic to justify changing the temperature.
Assuming the reaction is exothermic, an increase in temperature would cause the position of equilibrium to shift in the backward direction, so the value of Ecell, for a spontaneous reaction, would be less positive.
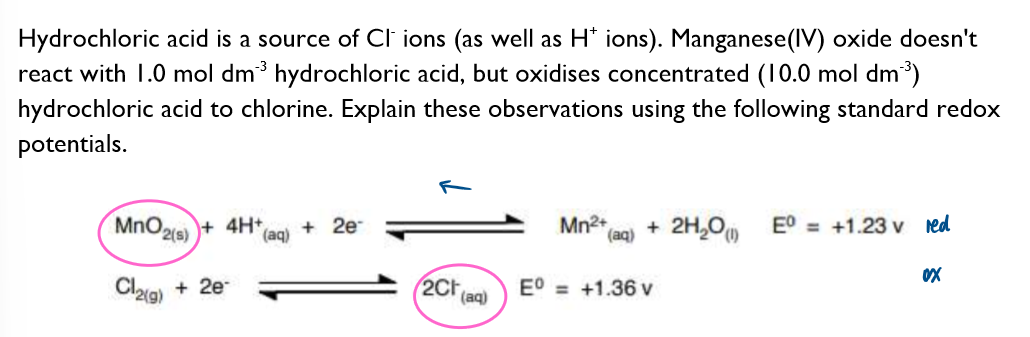
An increase in [Cl-] would cause the position of equilibrium to shift to the RHS and so the value of Ecell will become more positive / less negative. If the value of Ecell>0V, then the reaction becomes thermodynamically feasible, and that’s why MnO2 can oxidise conc HCl to Cl2 gas.
![<p>An increase in [Cl<sup>-</sup>] would cause the position of equilibrium to shift to the RHS and so the value of E<sub>cell</sub> will become more positive / less negative. If the value of E<sub>cell</sub>>0V, then the reaction becomes thermodynamically feasible, and that’s why MnO<sub>2 </sub>can oxidise conc HCl to Cl<sub>2 </sub>gas. </p>](https://assets.knowt.com/user-attachments/7b95f30c-60c9-468e-850e-0e81ff791bbf.png)
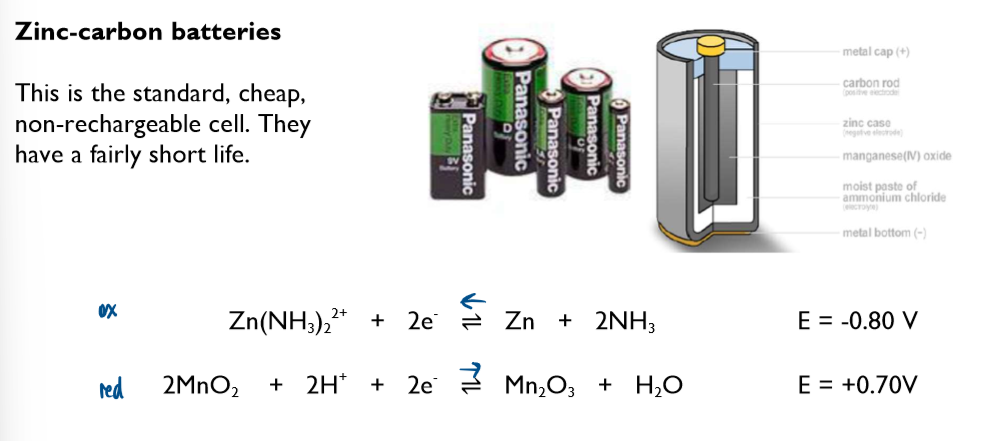
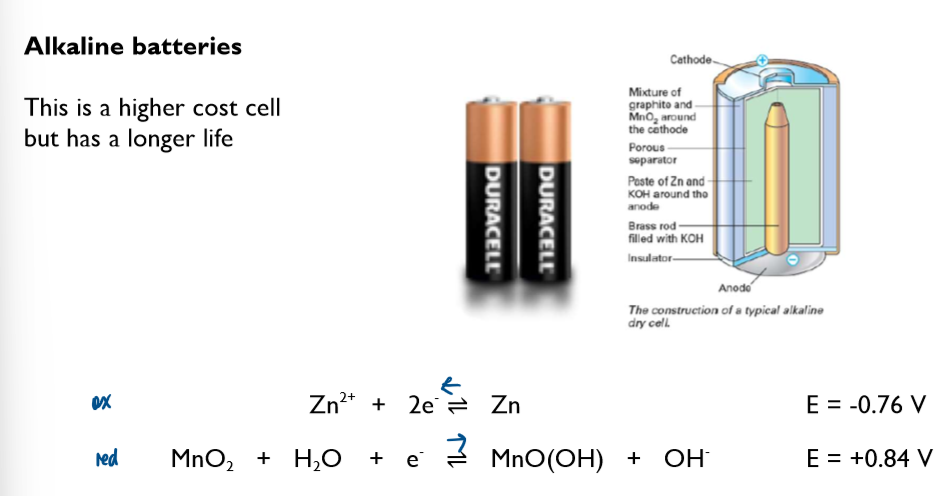
Commercial cells (batteries)
electrochemical cells can be used as a commerical source of electrical energy. Important of cell include non-rechargeable, rechargeable and fuel cells.
the great advantage of this is that it is a source of portable electricity.
note that a battery is more than once cell joined together (e.g. a car battery is made from six cells joined together) - what the general public call a battery, chemists call cells.
Non-rechargeable cells
in these cells, the chemicals are used up over time and the Ecell drops. Once one or more of the chemicals have been completely used up, the cell is flat and the Ecell is 0 volts
note the lack of the standard symbol sign. Conditions in these cells are not standard.
these cells cannot be recharged and have to be disposed of after their single use
Ecell of batteries is sometimes referred to as emf (electromotive force)
you are not required to learn any of these half-equations, just to be able to use them
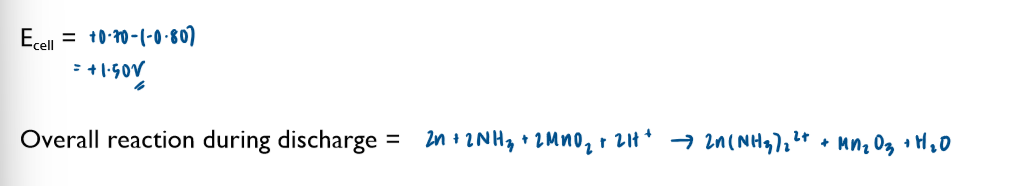
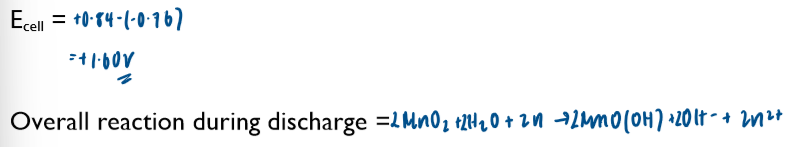

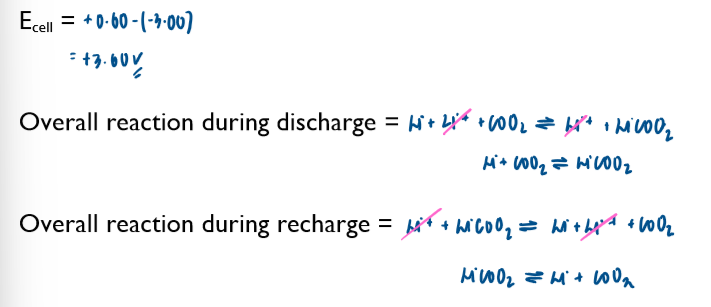

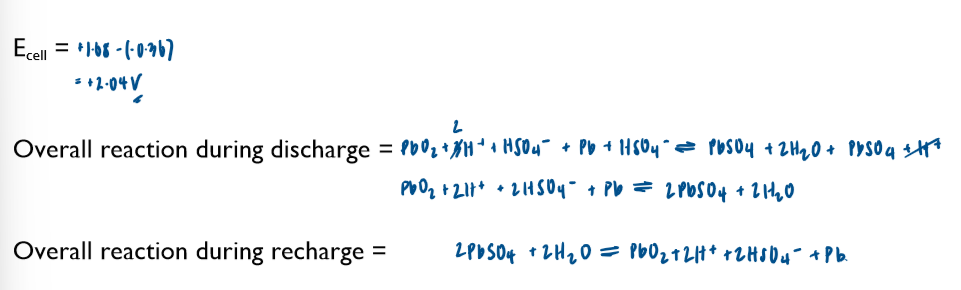

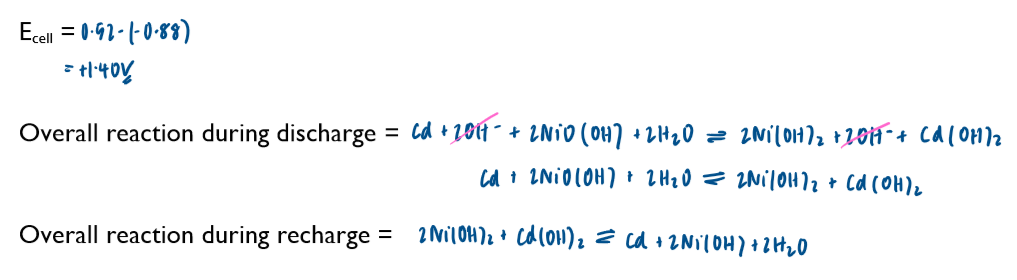
What are fuel cells?
continuous supply of the chemicals into the cell and so neither run out of chemicals nor need re-charging
use energy from the reaction of a fuel with oxygen to create a voltage
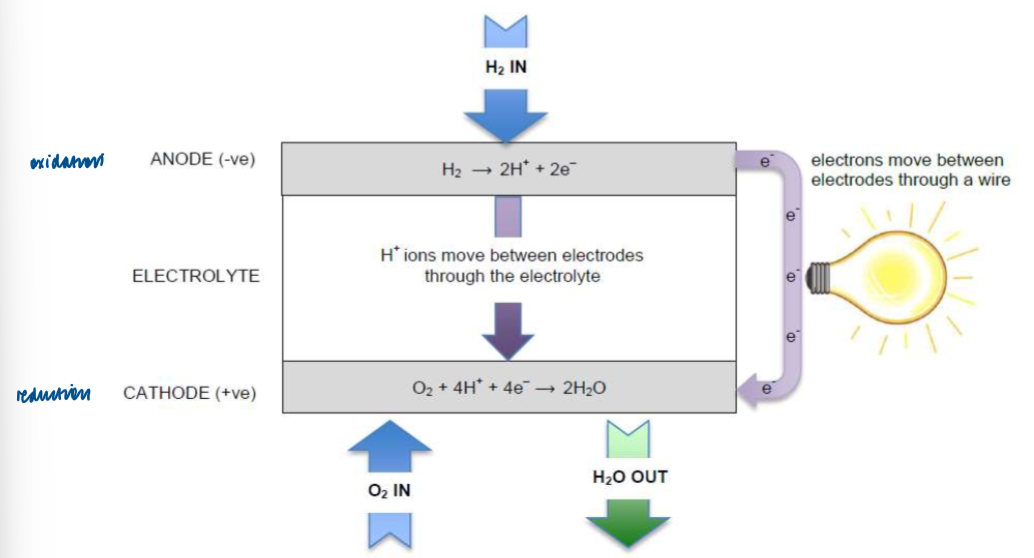
most common fuel cell is a hydrogen-oxygen fuel cell
Hydrogen-oxygen fuel cell in acidic conditions:
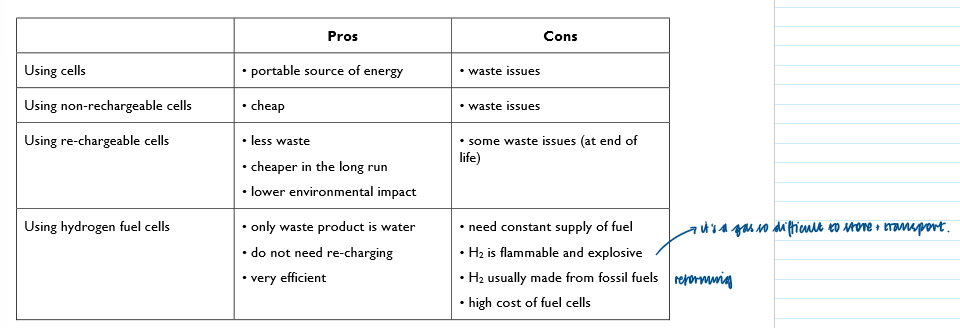
Pros and cons of using cells:
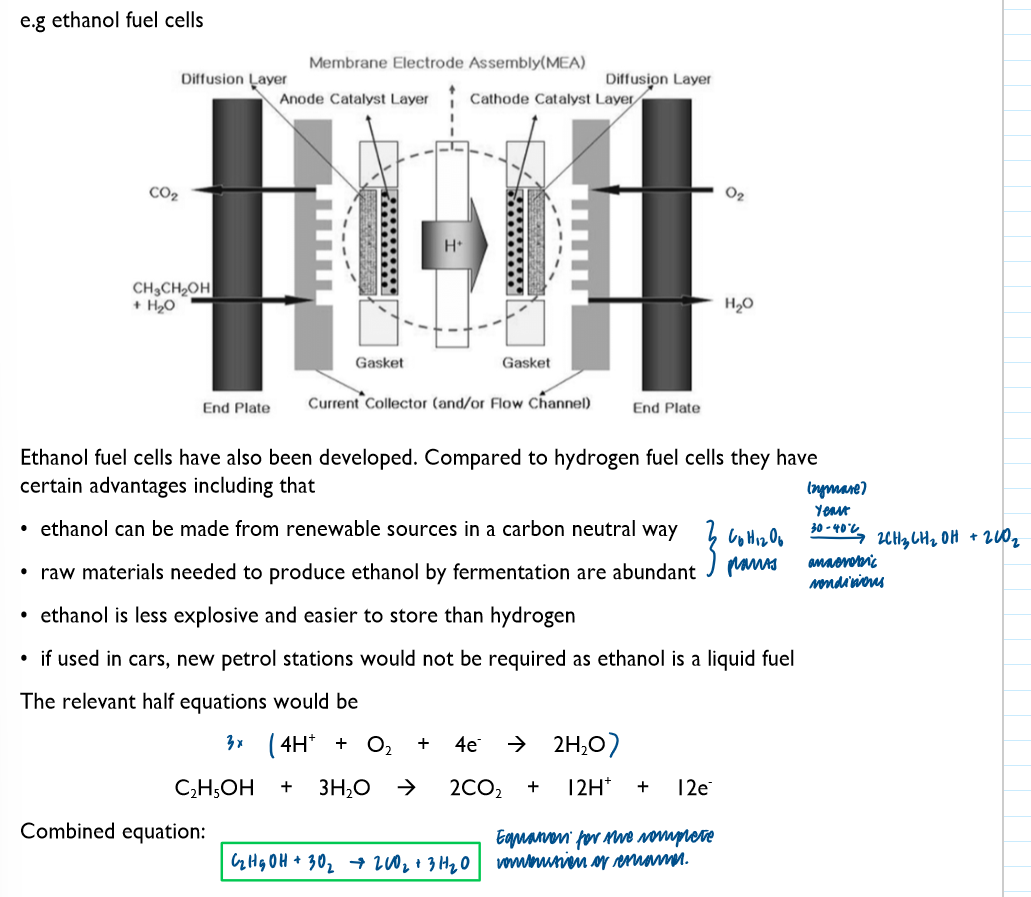
Ethanol fuel cells:
Methanol fuel cells:
