Perodic Table Trends
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
Periods
Rows of the table, elements in a period show patterns that repeat
Groups/Families
Columns of the table, elements in the same group have similar properties
Non-metal properties
Dull, non conductive, brittle, gases or liquids.
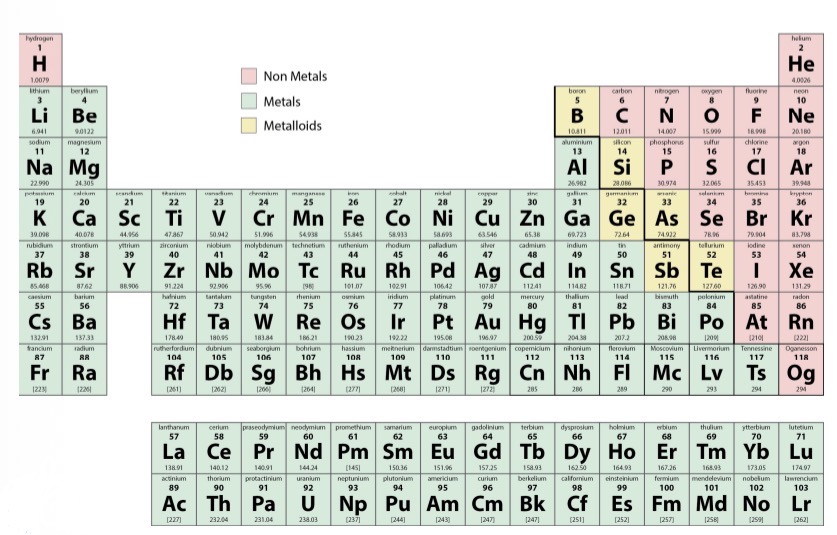
Metal Properties
Conductive, shiny, malleable, ductiles
Metalloid Properties
between metal & non metal
Main Group
Group 1A, 2A (Very reactive) and Groups 3A-8A (More inert). They are labeled with an A.
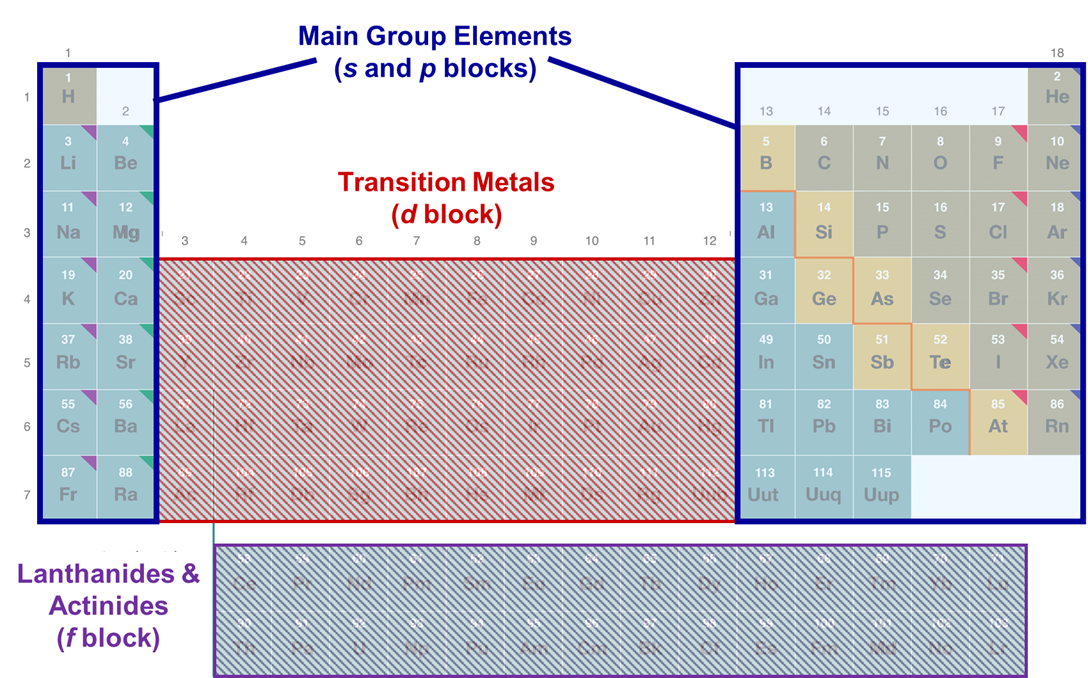
Transition Metals
In between the main groups. They are labeled with a B.
Atomic Radius
½ distance of a bond between to atoms
Factors that effect atomic radius (3)
Effective nuclear charge increases (dependent on # of protons)
Number of electrons
Orbital Size
Atomic Radius Trend on Table
Increases more on the LEFT and increases even more moving DOWN.
Ion Electron Configuration
“A-group” ions adopting the nearest noble-gas configuration. Many ions are isoelectronic (=equal number of electrons)
ex: [Ne], F-, O2-, N3-, Na+, Mg 2+, etc.
Ionic Radii
Size of Ions
Ionic size depends on: (3)
# of protons
# of electrons
orbital in which electrons reside (Cations are smaller than parent atoms, anions are larger than parent items)
Ionic Radii Trend on table
Increases more on the LEFT and increases even more moving DOWN..
Ionization Energy
amount of energy required to remove an electron from the ground state of a gaseous atom or ion
Ionization requires the input of energy to remove the electron. The lower the IE, the ___ the electron is to be removed.
easier
1st ionization
Removing the 1st electron from a neutral atom
2nd ionization
Start w/ +1 cation and remove an electron
Ionization Energy Table Trend
IE increases as it moves UP a group and RIGHT. IE decreases as we go down because less tightly held electrons whereas increases as we move across because smaller atoms = tightly held.
Ionization Energy Trend Exceptions
Group 6A due to half-filled shells
Electron Affinity
Increases Left → Right. Metals have low EA whereas nonmetals have high EA.
Metal Atoms tend to…
give up electrons to form positive ions; They have low IE, cations, and low EA
Nonmetal Atoms tend to…
gain electrons to form negative ions; they have high IE, anions, and high EA