Signal Transduction
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
44 Terms
Main target of regulation - cell/tissue?
Cell
What are the four main levels of regulation in organisms
Organism (neuro-endocrine)
Tissue/Organ
Cellular (unit of regulation)
Molecular (execution)
What system is the main mechanism for integration of regulation in organisms
The neuroendocrine system
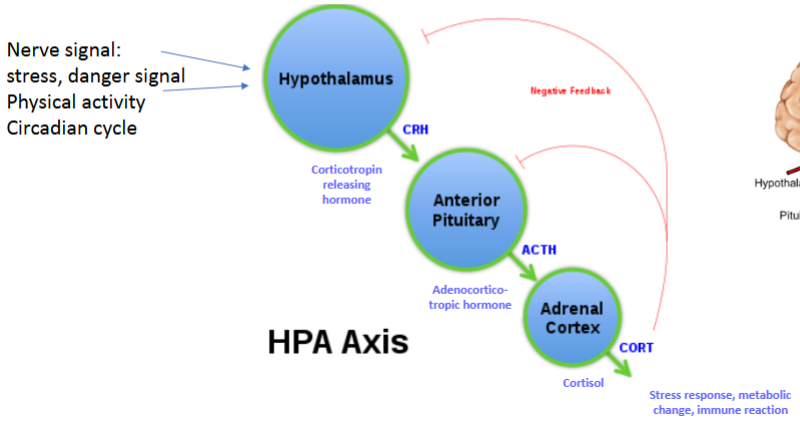
What is an example of a neuroendocrine axis?
The hypothalamic–pituitary–adrenal (HPA) axis.
List the key mediators of the HPA axis in order & name the structure from which they were released
Corticotropin releasing hormone (CRH) {hypothalamus} → Adrenocorticotropic hormone (ACTH) {anterior pituitary} → Cortisol {adrenal cortex}
Biological outcomes = Stress response, metabolic changes, immune reaction
What is always the ultimate target of regulation
Cell
Define signal transduction
The process linking a signal-activated receptor to the biological response
When is regulation initiated
When a signal (information) reaches the cell or is formed within the cell
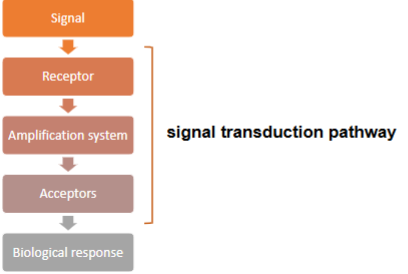
What is the flow of information in signal transduction from signal to biological response
What are the three broad origins of signals
From the environment (e.g. pain, temperature, light, smell).
From other cells in the organism (e.g. hormones, cytokines, metabolites).
From within the same cell (e.g. DNA damage, reactive oxygen species).
The two main types of signal transmission are short-range/local signals & long-distance signals. Name the 2 types of short-range/local signals
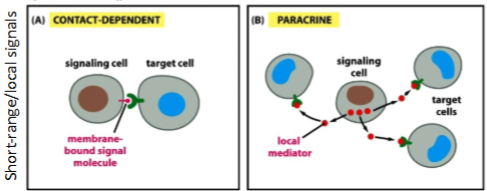
The two main types of signal transmission are short-range/local signals & long-distance signals. Name the 2 types of long-distance signals
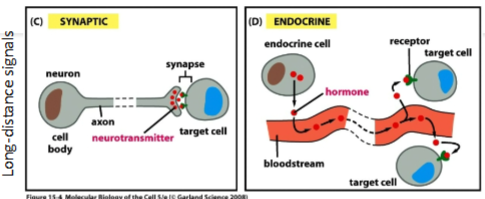
What are cytokines & what type of cell signalling are they involved in
Cytokines are small proteins used for communication with surrounding cells.
They fall under the paracrine category of cell signalling.
Give an example of cytokines being used in paracrine signalling (name the specific cytokine)
Attack of infected cells
T cell sees infection → releases IL-2 (cytokine) → activates nearby T cells → T cells produce more cytokines to tell other immune cells to act → infected cell gets destroyed
(release IL-2 if you are ill)
What is the definition of a receptor?
A specific recognitive element (protein)
Is it true that each signalling molecule has a unique receptor
Yes
what is a ligand
A ligand is any molecule (or sometimes ion) that binds specifically to a receptor or another target protein to cause a biological effect.
(don’t need - just for understanding)
What are the key features of receptor–ligand binding

What are the three main types of receptors (based on location)
Cell surface/transmembrane receptors
Nuclear receptors
Cytosolic receptors
Why is amplification necessary in cell signalling
Only a small number of the signalling molecules actually reach the cell receptor. Amplification is needed to make sure the cell is responding to the signal on a large enough scale
Name 3 mechanisms of amplification
Allosteric enzyme regulation
Interconversion cycles
Cascade
What does “allosteric” mean?
“Allosteric” means regulation occurs at a site other than the active site of the enzyme
(All other sites but its own)
What are the two natural states of an allosteric enzyme
Active form → active sites are functional and can bind substrate.
Inactive form → active sites are distorted/non-functional.
The enzyme naturally oscillates between the two.
How does an activator amplify a signal in allosteric regulation
By binding to a regulatory site, the activator stabilizes the active form, increasing the fraction of active enzyme molecules → a small amount of activator causes a large increase in enzyme activity (amplification).
How does an inhibitor contribute to allosteric amplification ?
By binding to the regulatory site, the inhibitor stabilizes the inactive form, shutting down enzyme activity. Even small amounts of inhibitor can strongly reduce product formation.
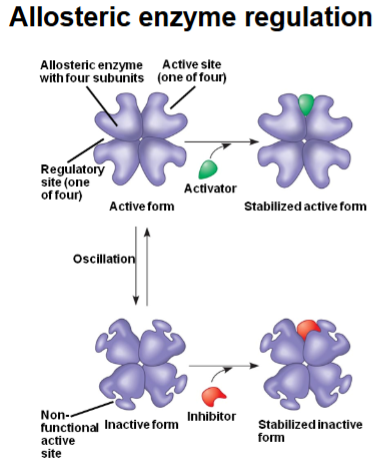
Why is allosteric regulation considered part of the “amplification system”?
Because a small change in regulator concentration (activator/inhibitor) can cause a big change in the enzyme’s catalytic output, amplifying the initial signal.
What is an interconversion cycle
A system where enzymes rapidly switch between active and inactive forms through reversible covalent modification
What is the most common interconversion cycle
Phosphorylation
Why is phosphorylation a good method of amplification?
Rapid and reversible: Adding/Removing a phosphate can quickly turn a protein on or off.
ATP is readily available: Phosphate groups come from ATP, which cells have in abundance.
Which enzymes control interconversion cycles
Protein kinase → adds a phosphate (ON switch).
Protein phosphatase → removes a phosphate (OFF switch)
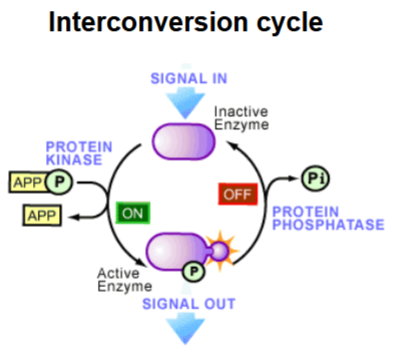
How does an interconversion cycle amplify signals
A small signal (like one kinase being activated) can convert an entire pool of enzymes to the active form → leading to a large increase in activity
Inactive Akt can interconvert to active Akt via two phosphorylation-dependent cycles. How do these 2 different mechanisms work
Phosphorylated Ser473 interacts with the linker between kinase and PH domains. (Serine 473 gets a phosphate added. This phosphate acts like a key that changes the shape of Akt so the PH domain moves a bit, letting the kinase domain work.)
Phosphorylated Ser477/Thr479 displace the PH domain. (Serine 477 and threonine 479 get phosphates. These phosphates push the PH domain out of the way, fully exposing the kinase domain so it can do its job.)
(Phosphates at certain spots on Akt either adjust or remove the “block” from the PH domain, turning Akt on.)
What’s another name for Akt
Protein kinase B
(AB)
What is a cascade?
A multi-step pathway where each activated enzyme activates many molecules of the next enzyme, producing an exponential amplification of the original signal.
How does amplification happen in cascades
One activated molecule → activates many downstream targets → each of those activates even more → the signal multiplies at each step.
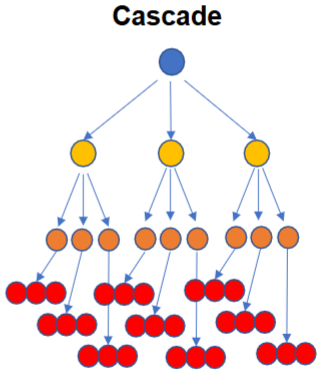
Give an example of an important enzyme cascade
The mitogen-activated protein kinase (MAPK) cascade
Explain the mitogen-activated protein kinase (MAPK) cascade & what it leads to
The MAPK cascade is a chain of proteins that pass a signal along inside a cell.
MAPKKK gets activated by the receptor → activates many MAPKKs → each MAPKK activates many MAPKs → each MAPK goes into the nucleus & takes effect
MAPK can activate cytosol proteins, trigger cytoskeleton proteins & switch on certain genes, causing metabolic changes, cytoskeleton remodelling & gene expression
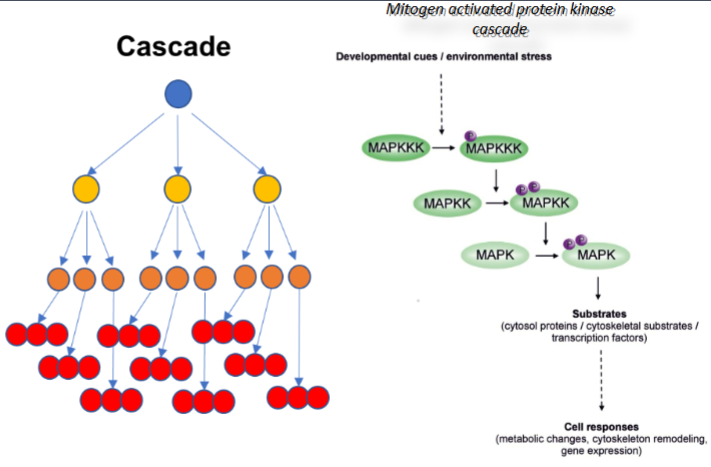
Compare the 3 mechanisms of amplification
Allosteric amplification: Amplification occurs within one enzyme due to regulator binding.
Interconversion cycle: Amplifies by flipping a pool of enzymes ON or OFF.
Cascade: Amplifies by multiplying the signal at each step of the pathway.
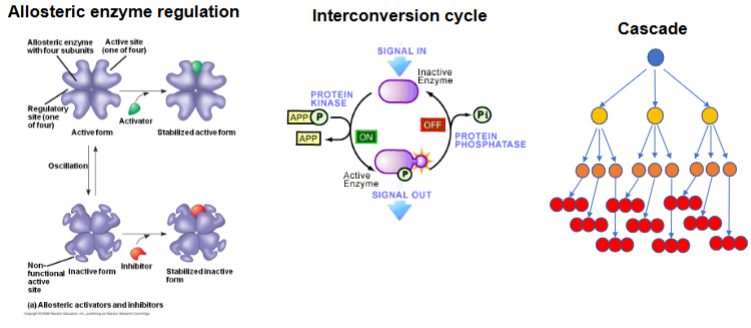
PKA activating glycogen phosphorylase is an example of which amplification system
Interconversion cycle
What do all signal transduction pathways ultimately control?
The function of proteins (enzymes, ion channels, transporters) that directly form biological responses.
What are the two broad categories of protein control?
Control of protein amount (gene expression, degradation, stabilization).
Control of existing proteins (with/without covalent modification).
What are the types of covalent modification control of proteins
Reversible and irreversible.
Signal transduction can influence gene expression by modifying histones. What are histones & what is their structure?
Histones are proteins that DNA wraps around to form chromatin.
2 copies of each of the 4 core histones assemble into an octamer that is shaped like a disk.
(Don’t need image - just to paint a picture)
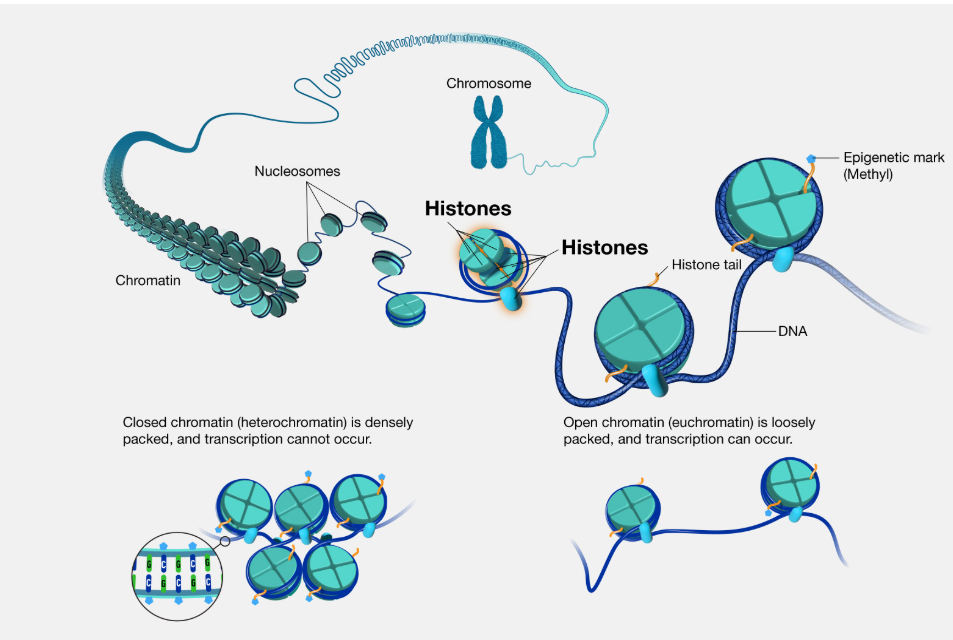
DNA is tightly wrapped around histones. How are they loosened to allow transcription factors to work
Histones normally have positive charges.
DNA has a negative charge.
This opposite charge interaction keeps DNA tightly wrapped around histones.
Histones are modified to reduce their positive charge, loosening the DNA around it