OZ 4: Ozone- here today gone tomorrow
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Steady state
ozone is being made as fast as it is used up
Equation for rate of reaction
Rate of reaction= change in property / time taken
What are 5 changes in property?
Measuring volume of gases
Measuring mass change
pH measurement
Colorimetry
Chemical Analysis
What are two pieces of equipment that can measure the volume of gas produced?
Measuring cylinder
Gas syringe
What do you use to measure mass change?
electronic scale
What do you use to measure pH?
pH probe
universal indicator
A colorimeter measures
the change in colour of a reaction
A colorimeter detects
how much light is absorbed by the solution
A higher absorbance of a colorimeter means
more of the coloured species is present
A lower absorbance of a colorimeter means
the coloured species is being used up
How can you use chemical analysis?
take a sample of reaction mixture at regular intervals
stop reaction in sample
analysis
quench
stop reaction
Example of chemical analysis
titration
What are the 5 factors that affect rate of reaction?
Concentration
Temperature
Intensity of Radiation
Surface Area
Catalyst
collision theory
The idea that reactions occur when particles collide with enough kinetic energy.
Activation enthalpy
minimum kinetic energy required by a pair of colliding particles before a reaction will occur
Reactions occur when particles
collide with activation energy
Why don’t all collisions lead to a reaction?
Particles must collide with enough energy to overcome the activation energy barrier
What does the peak of an enthalpy profile represent?
transition state
transition state
point where old bonds stretch and new bonds start to form
How do catalysts affect a reaction?
They provide an alternative pathway with lower activation energy, increasing the rate.
The distribution of kinetic energy at a given temperature is given by the
Maxwell- Boltzmann distribution
At a higher temperature, more particles
have minimum energy to react
If temperature is raised, M-B graph
shifts to the right and the peak is lower
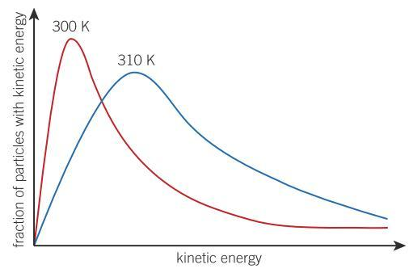
Reactions go faster at higher temperatures since
a larger proportion of colliding molecules have minimum activation enthalpy needed to react
peak of a M-B diagram represents
most probable energy
area under M-B curve
all particles
activation energy in M-B curve is on the
right
What effect does adding a catalyst have to a M-B curve?
curve remains the same
Ea shifts to the left so more particles can react
How does concentration affect the rate of reaction?
Higher concentration increases the frequency of collisions.
Why does increasing temperature speed up a reaction?
Particles move faster and collide more often with greater energy.
How does radiation intensity affect reaction rate?
More radiation provides energy to break bonds faster.
Why do smaller particles react faster than larger ones?
They have a larger surface area, leading to more collisions.
How do catalysts increase the rate of reaction?
They lower the activation energy, allowing more collisions to be successful.