Part 5
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Suspensions (mixtures)
Mixtures where something solid will settle out of solution over time, ie in muddy water, mud will eventually settle to the bottom
Colloids
Particles that are too large to dissolve but small enough not to settle out of the solution, instead they stay suspended, ie proteins
Solution
A homogenous mixture of two or more substances in which a solute is uniformly dissolved in a solvent
Partial Pressure
the pressure an individual gas in a mixture would exert if it occupied the entire volume alone at the same temperature

Strong vs Weak acids
Strong acids dissociate 100 percent in solution (ie HCl), weak acid are acids that do not dissociate 100 percent in solution (ie lactic acid)
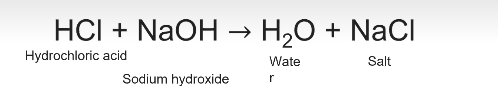
Neutralization reaction
Acid plus base producing water and a salt