substitution reactions
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
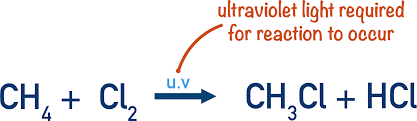
halogenoalkanes by free-radical substitution
alkanes react with chlorine or bromine in the presence of UV light
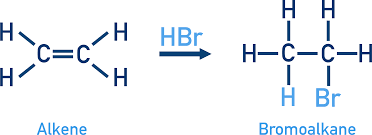
halogenoalkanes from electrophillic addition
alkenes react with halogens or hydrogen halides
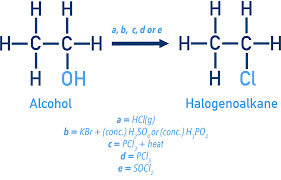
halogenoalkanes by alcohol substitution
alcohols react with hydrogen halides, PCl3 under heat and PCl5 under room temperature
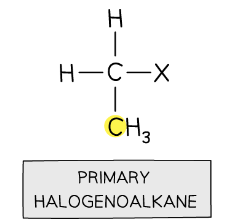
primary halogenoalkanes
halogen is bonded to carbon atom with only one alkyl group
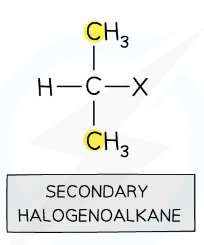
secondary halogenoalkanes
halogen is bonded to carbon atom with 2 alkyl groups
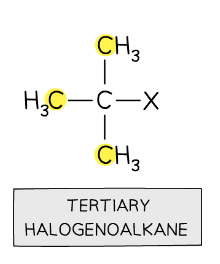
tertiary halogenalkanes
halogen is bonded to carbon atom with 3 alkyl groups
general formula
CnH2n+1X
reactivity
decreases down the group…more reactive than alkanes due to polar nature between carbon and halogen atom
nucleophilic substitution
reaction in which a nucleophile attacks a carbon atom with a partial positive charge resulting in the replacement with an atom carrying a partial negative charge by the nucleophile
substitution reactions with aqueous alkali
halogen atom in the halogenoalkane is replaced by an OH- to form an alcohol
hydrolysis of a halogenoalkane
using reflux apparatus including a condenser and anti-bumping granules…halogenoalkane is broken down by water to form an alcohol, H+ and a halogen anion
fastest nucleophilic substitution
take place with iodalkanes
slowest nucleophilic substitution
take place with fluoroalkanes
substitution with cyanide ions in ethanol
use an ethanolic solution of potassium cyanide and heat under reflux with halogenoalkane…cyanide ion adds an extra carbon to carbon chain…forms halogen anion and newly formed chain
substitution with ammonia in ethanol
heated under pressure with excess ammonia dissolved in water…forms an amine and hydrogen halide