10.1 - Depicting Molecules and Ions With Lewis Dot Structure
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
define resonance structure
when can write lewis structure in more than 1 way. BUT OVERALL CHARGE MUST REMAIN THE SAME
how to calculate fractional bond orders
#of bonds/ # connections

equation to calculate formal charge
# of valence electrons - # of lone atoms - # of bonds
share the rules for choosing the most valuable resonance form
smaller charges (+ or -) are preferred
Avoid like charges on adjacent atom
A more negative formal charge should reside on most electronegative atom
what are the electron-deficient atoms (WHEN BONDED COVALENTLY) and what # is the necessary electrons
Be (only 4 electrons)
B (only 6 electrons)
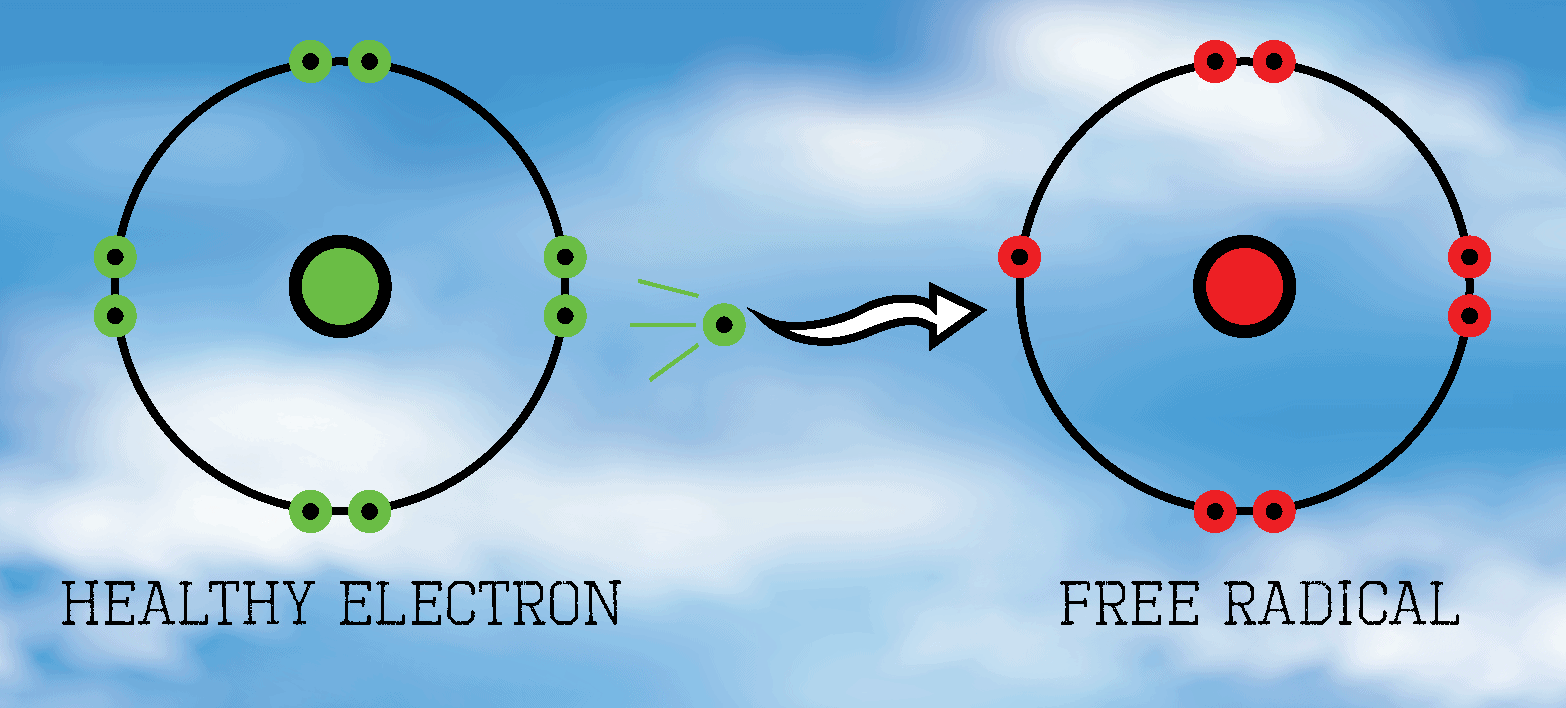
define free radical. Are the reactive?
a molecule with an unpaired electron and they are reactive.
what atoms have an expanded octect
atoms with d orbitals
nonmetals from period 3 (row) or higher