Orgo Reactions - Alkene, Alkyene, Ethers, Hydrid, Alcholos Diels Alder
1/117
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
118 Terms
In heat light reactions what are the numbers of electrons that tell you the reaction?
2,6,10, and dis = heat
2,6,10, and con = light
4,8,12, and dis = light
4,8,12 and con = heat
What are the Aromatic rules?
Cyclohexane structure
No sp3 (single) bonds
Count the number of pi bonds
What are the electron numbers that coorelate to whether a structure is Aromatic vs. Antiaromatic?
2,6,10,14 = aromatic
4,8,12,16 = antiaromatic
What are the Meta directors?
F, Cl, Br, I, NO2, SO3H, CN, NR3
What are the Para Directors?
NH2, NR2, OH, OR, R, Benzene ring
What is important to note about the Para directors?
The Para directors have a reaction that shows structures on both the ortho and para positions
How are Meta structures arranged?
Groups are added one space/carbon position apart
How are Para structures arranged?
Groups are added on opposite sides of the ring (two spaces/carbon positions apart)
How are the Ortho structures arranged?
Groups are added adjacent (there are no directors)
When you see an Alkene structure what happens?
The double bond is broken to a single bond, and the group is added with a plus/minus sign
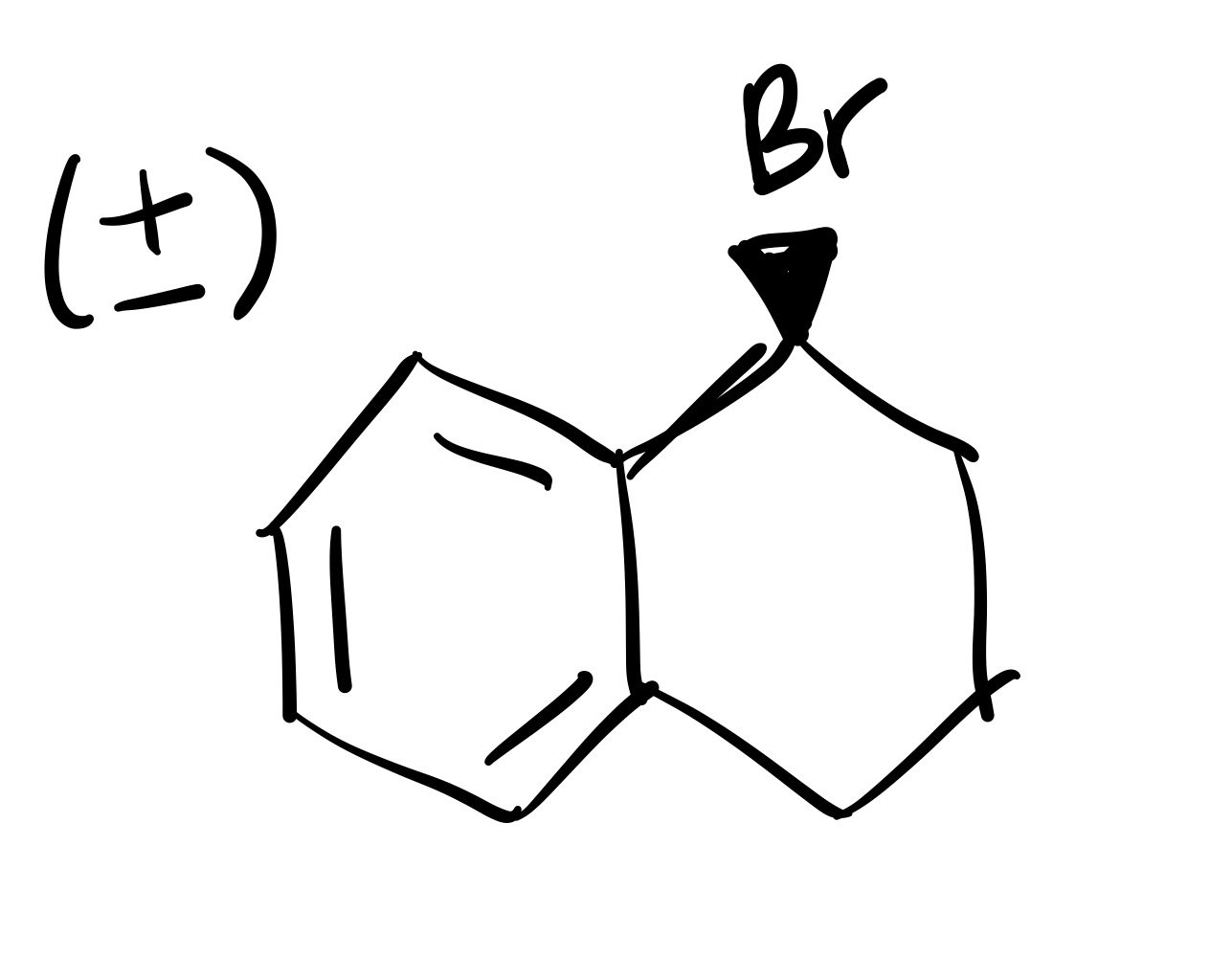
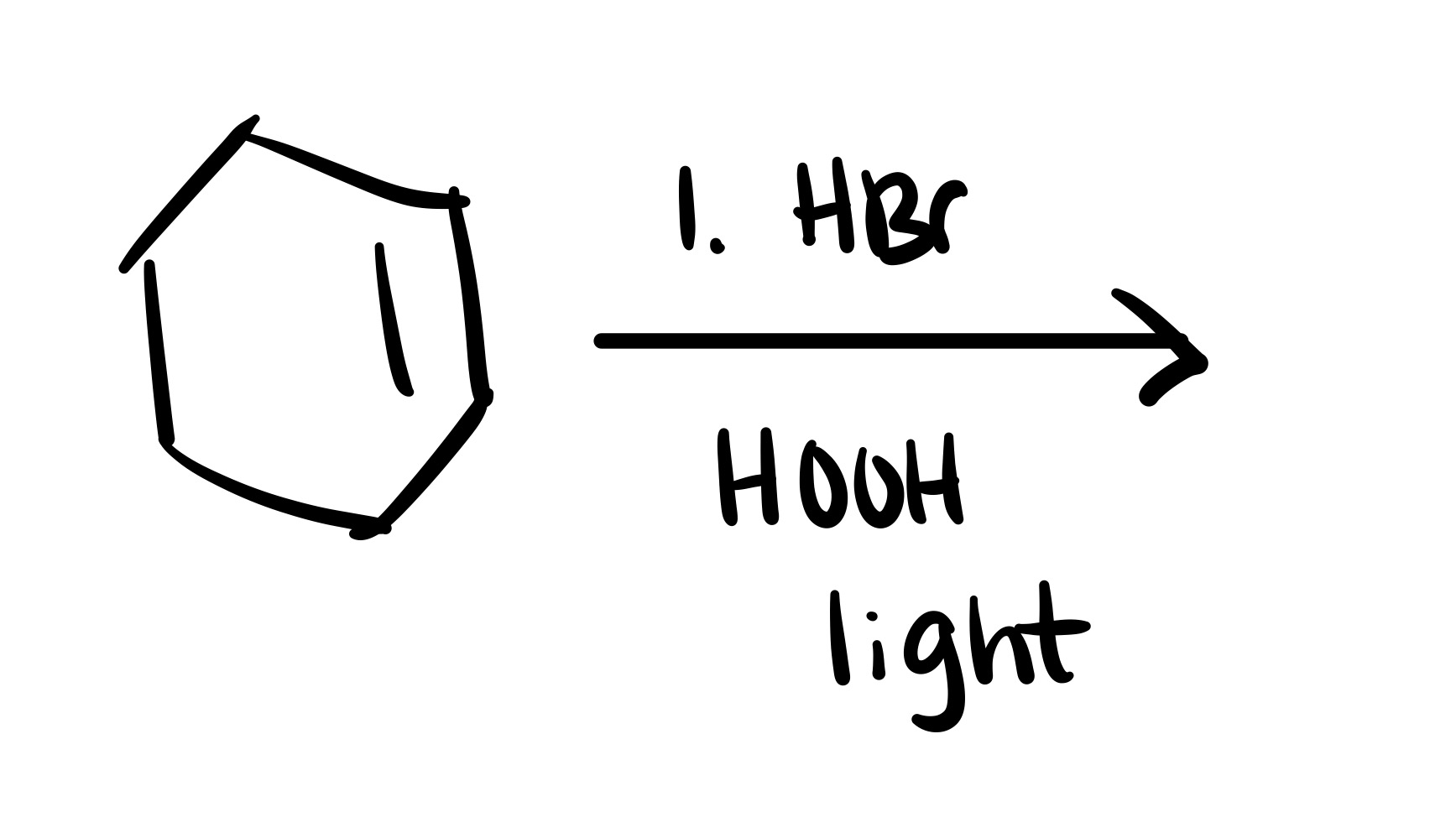
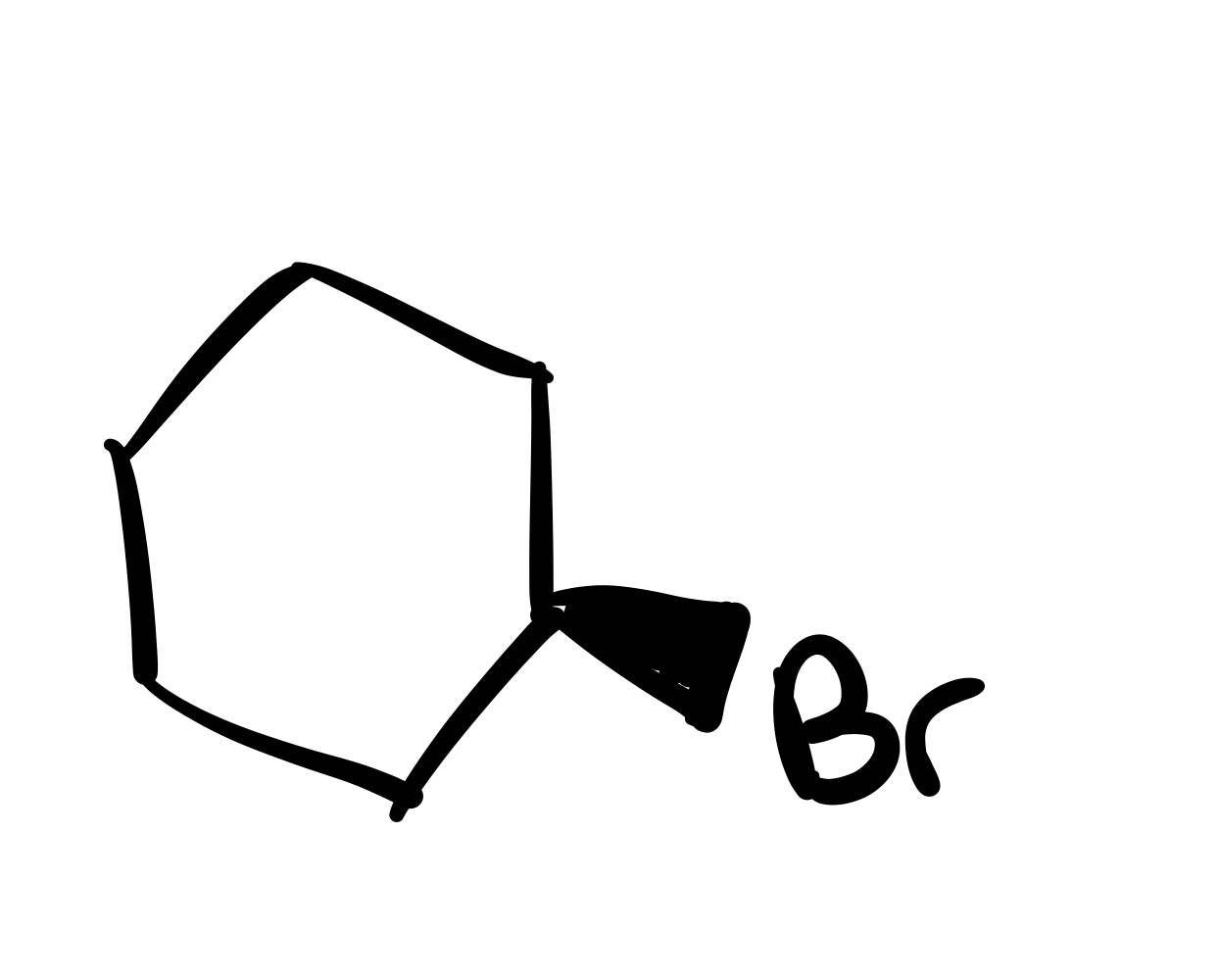
The double bond is broken, and the Br is added (can we wedge or dash). Add plus/minus
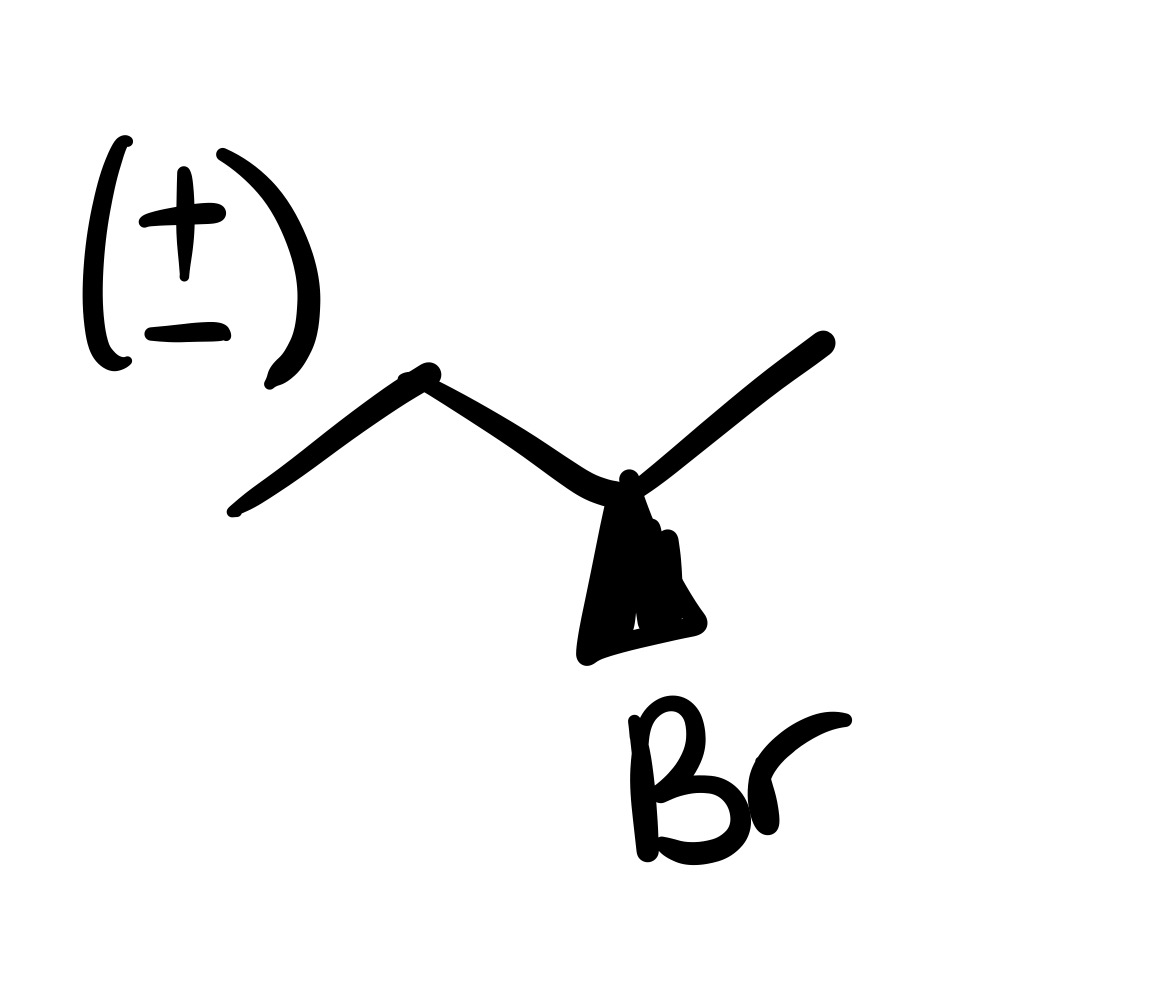
The double bond is broken, and the Br is added to the carbon chain (no dash/wedge)

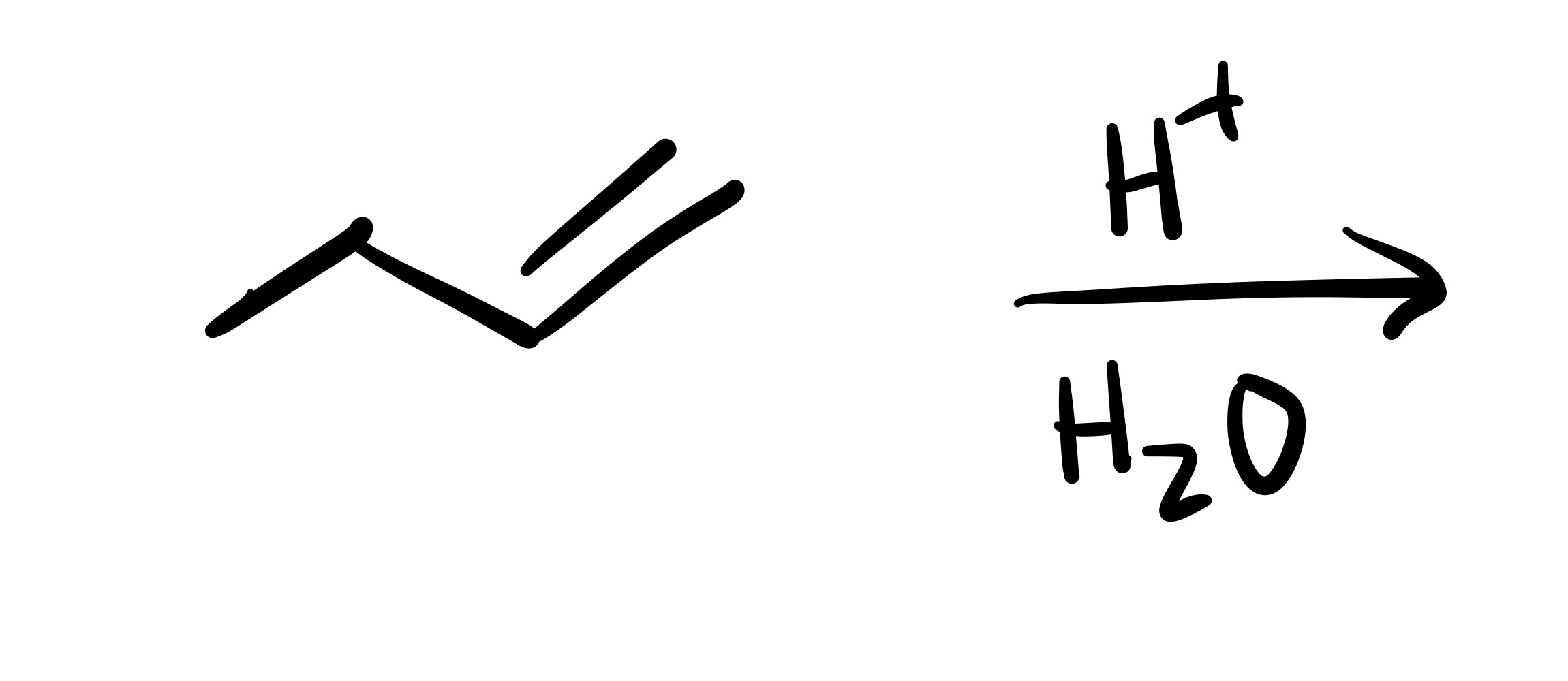
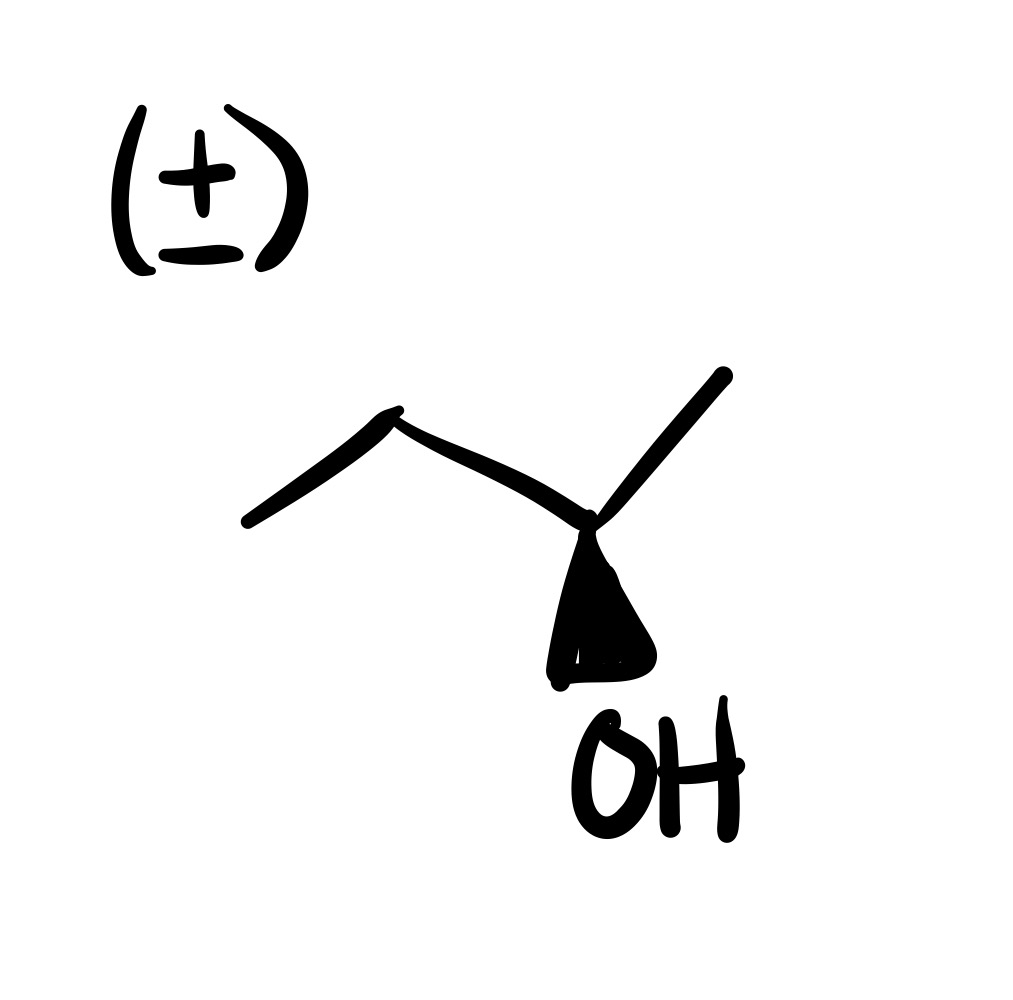
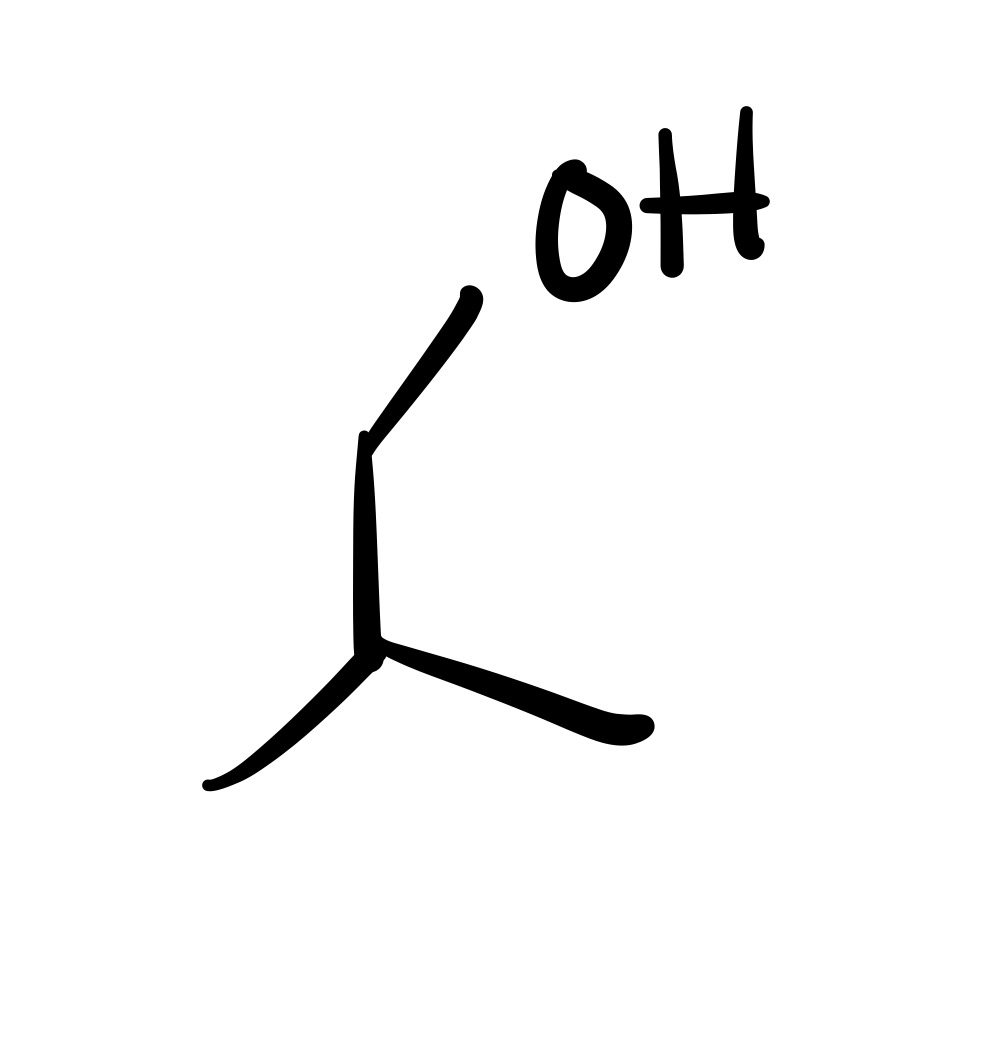
The double bond is broken, and the OH is added (can we wedge/dash). Add plus/minus sign
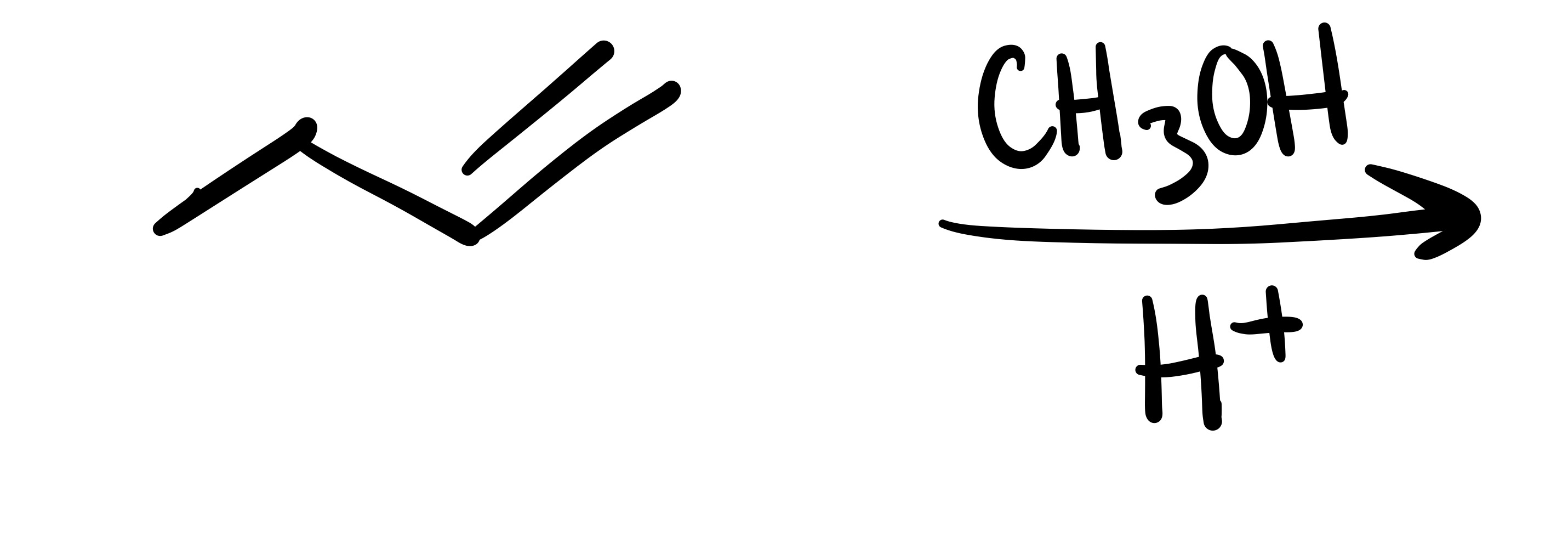
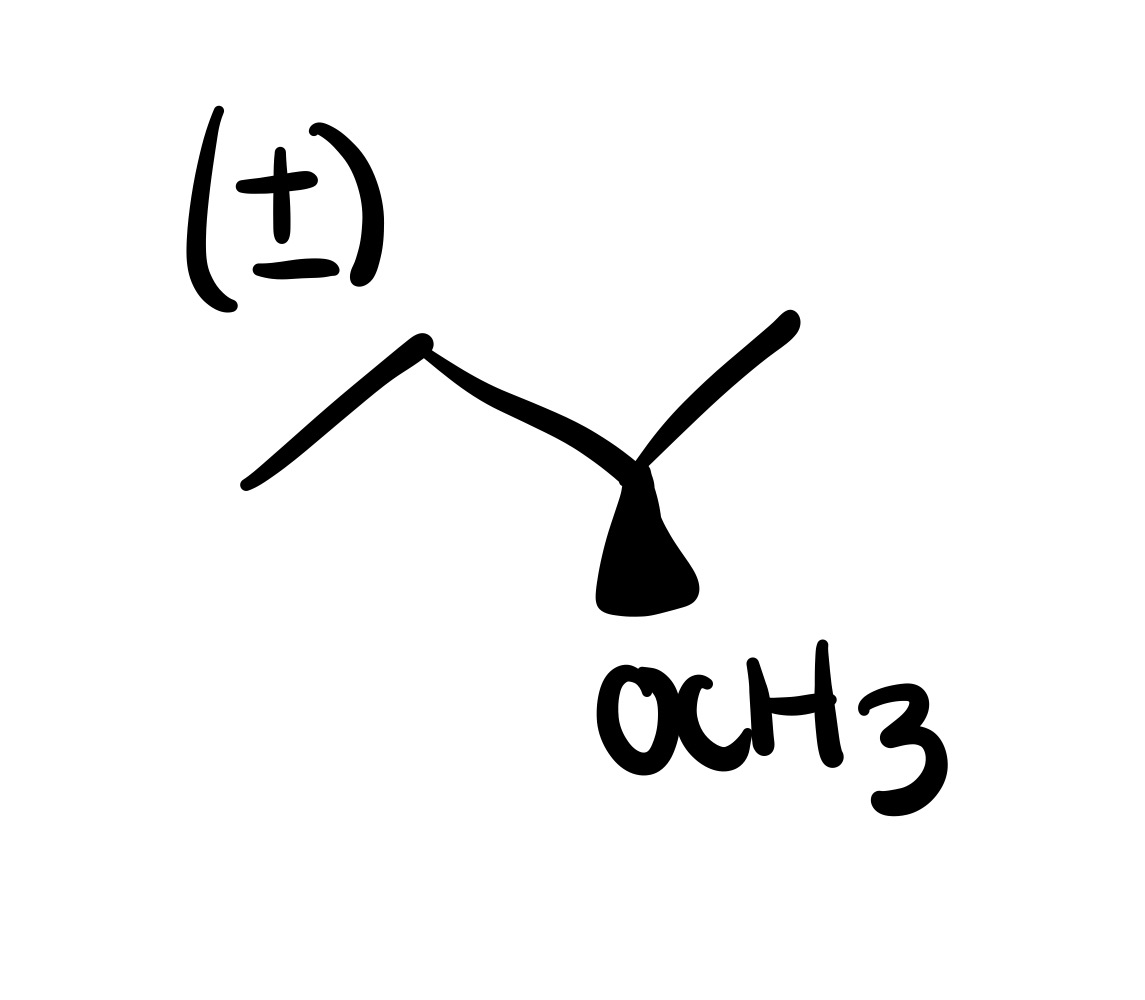
The double bond, and the OCH3 is added (can be wedge or dash). Add plus/minus sign
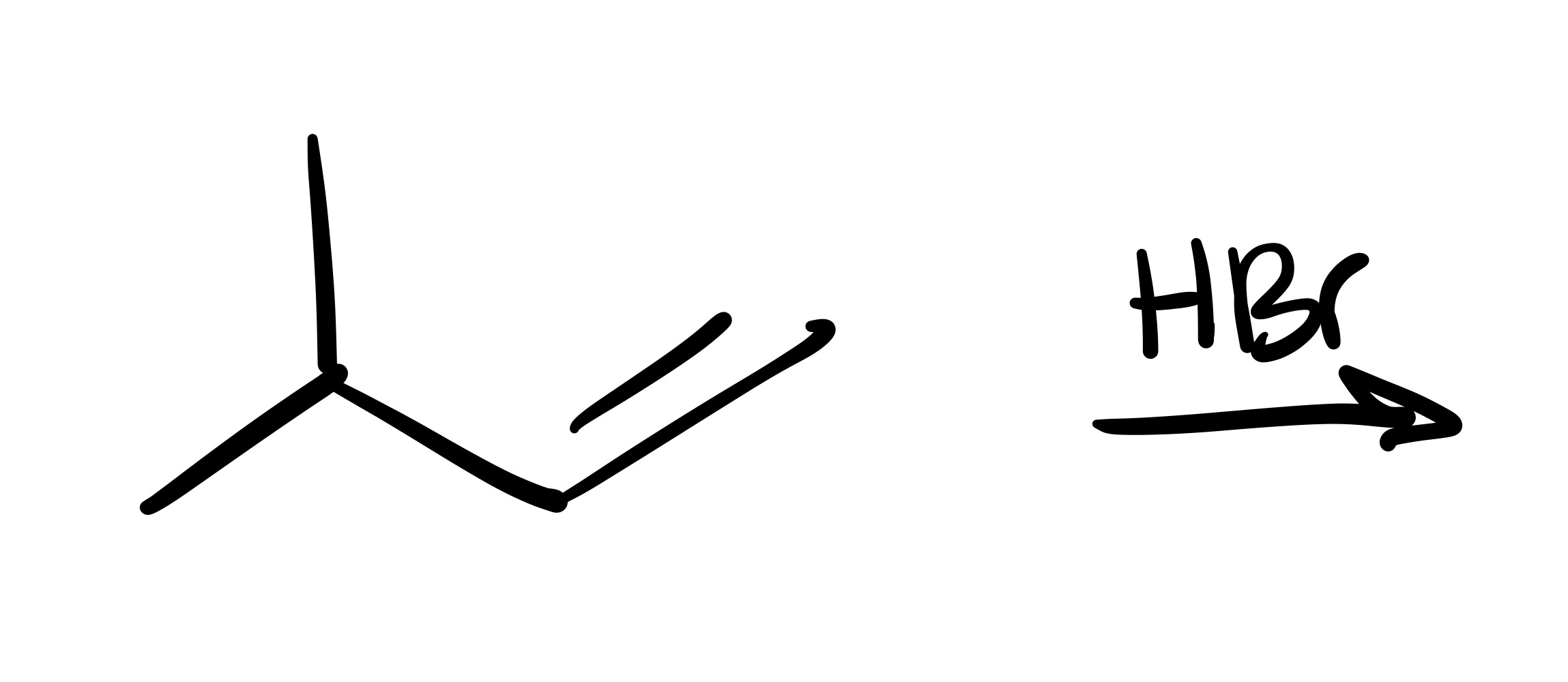
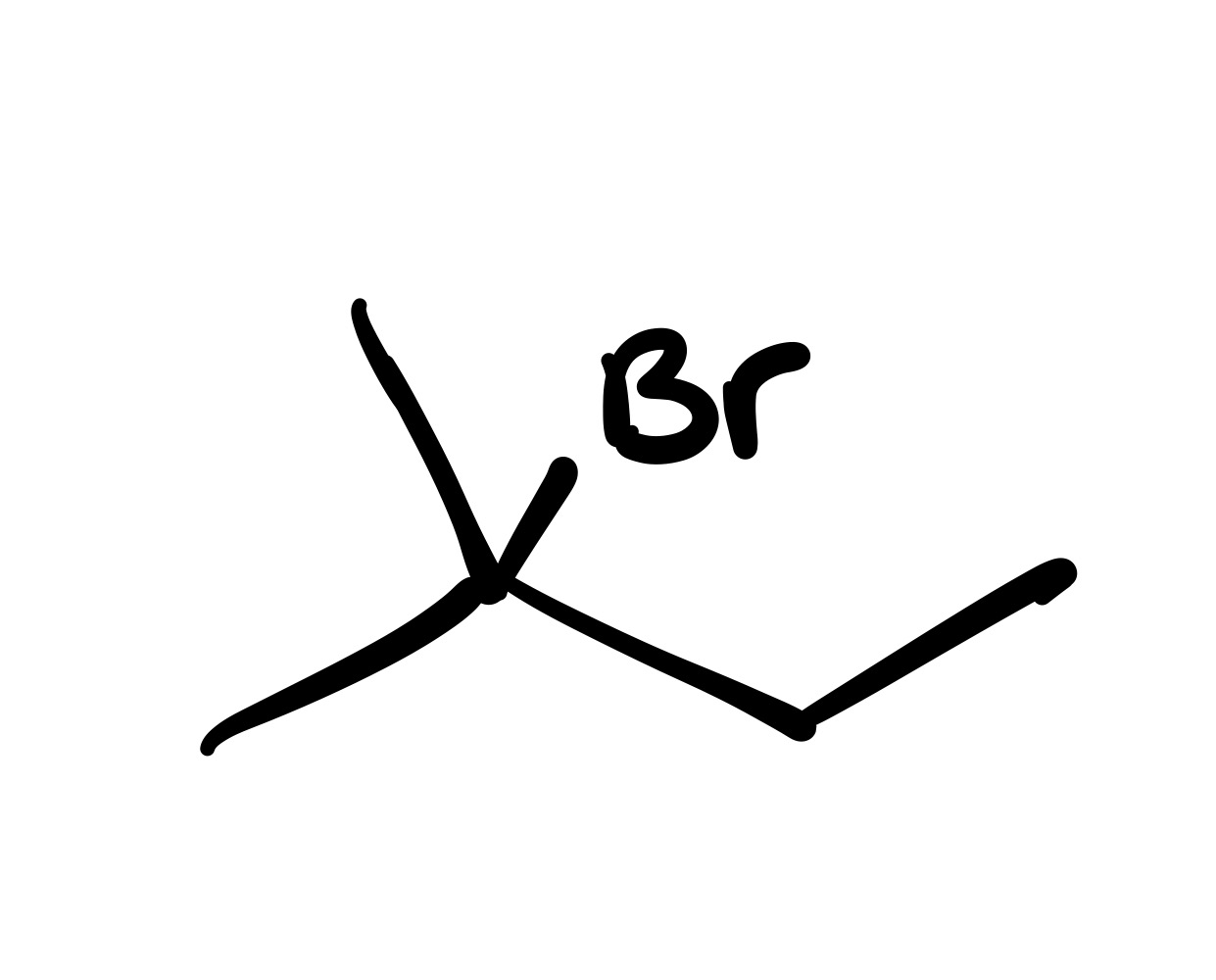
The double bond is broken, and the Br is added to where the methyl group is (no wedge/dash)
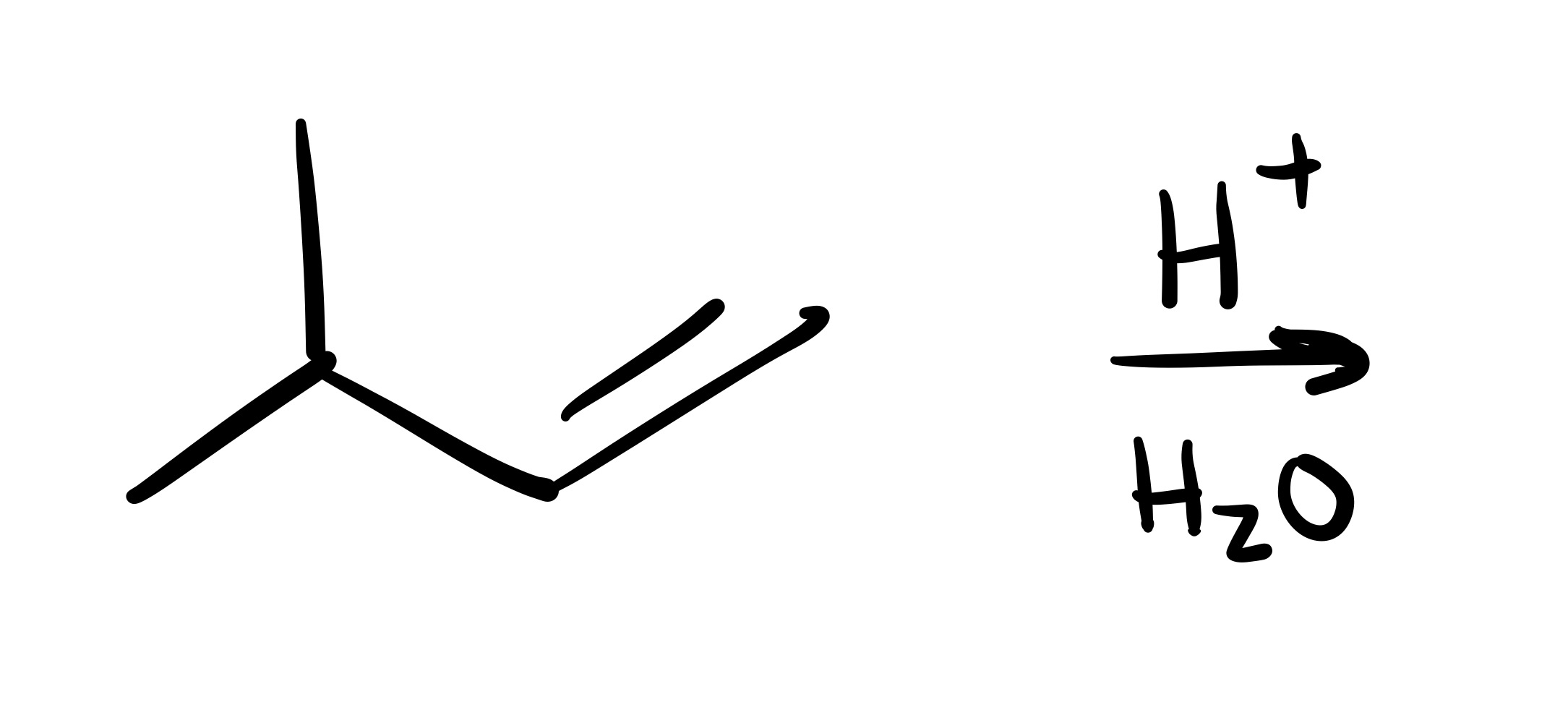
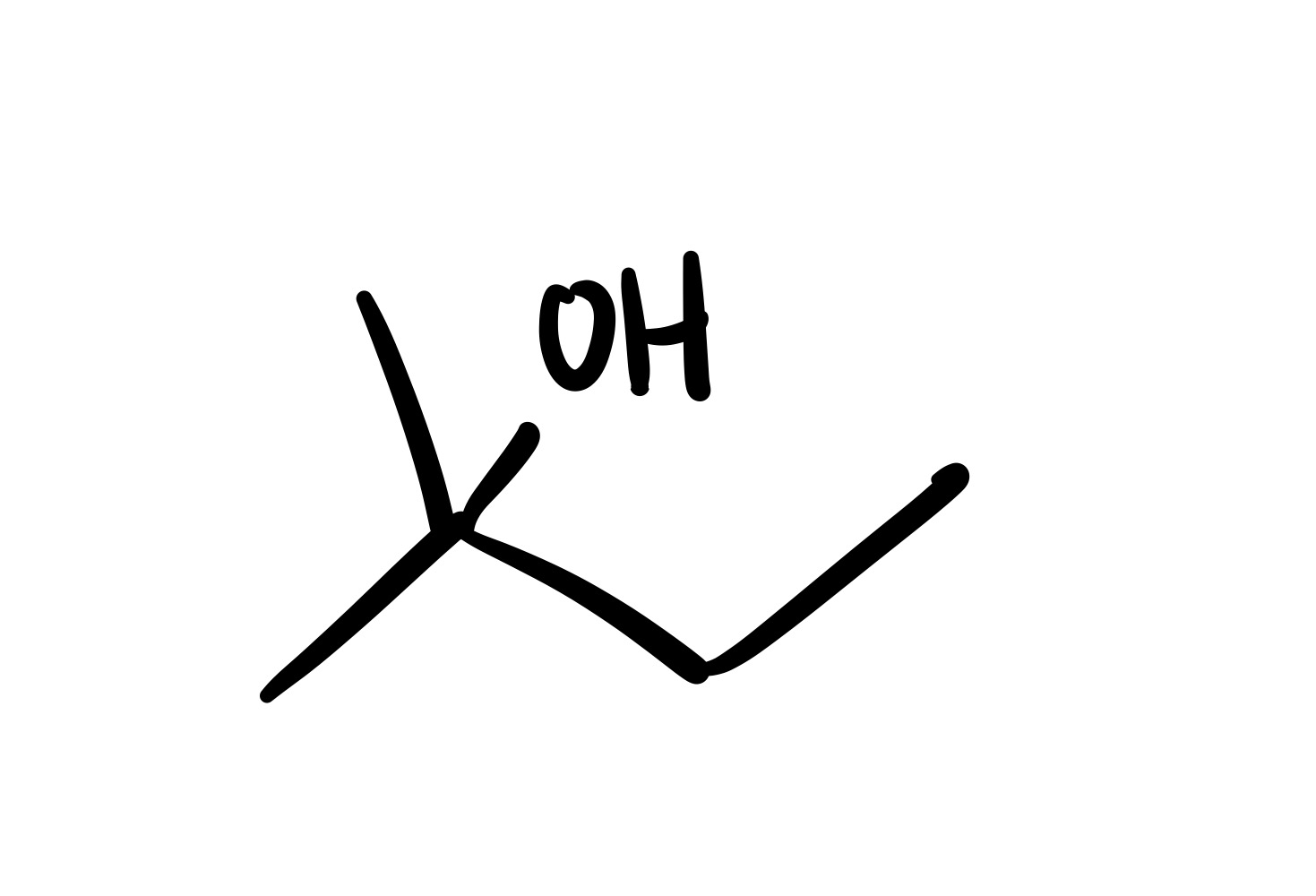
The double bond is broken, and the OH is added to the methyl group (no wedge/dash)
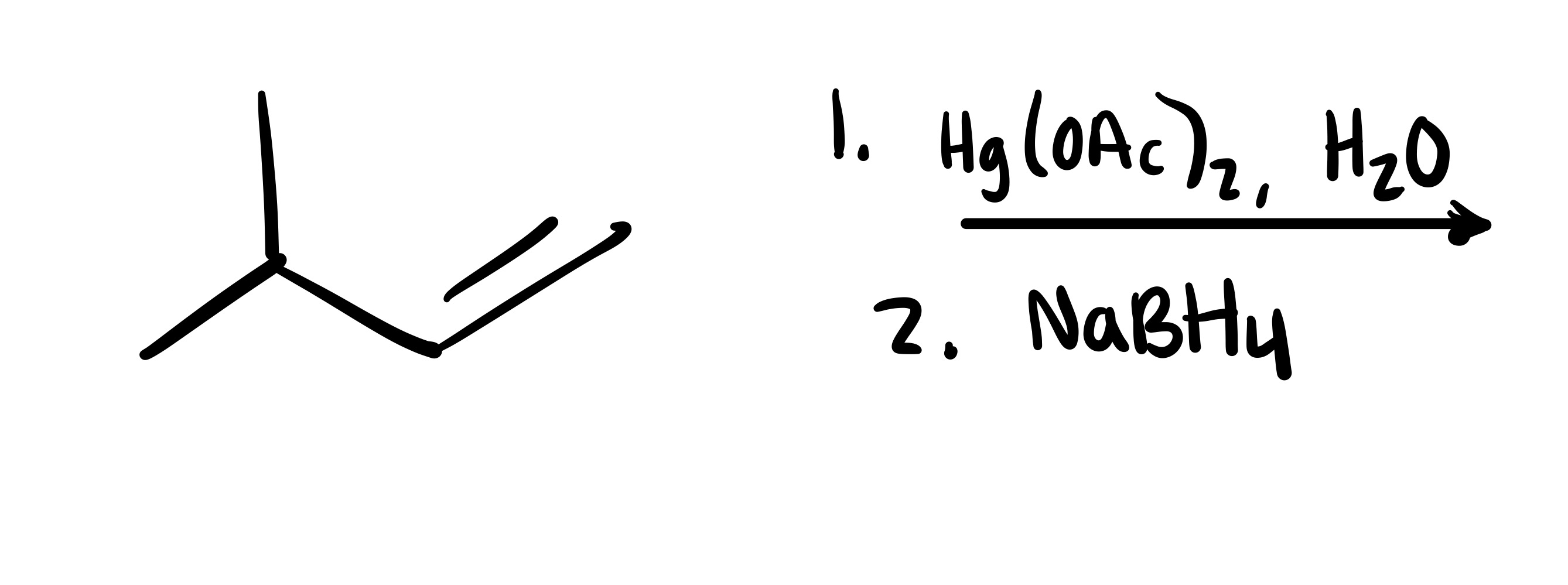
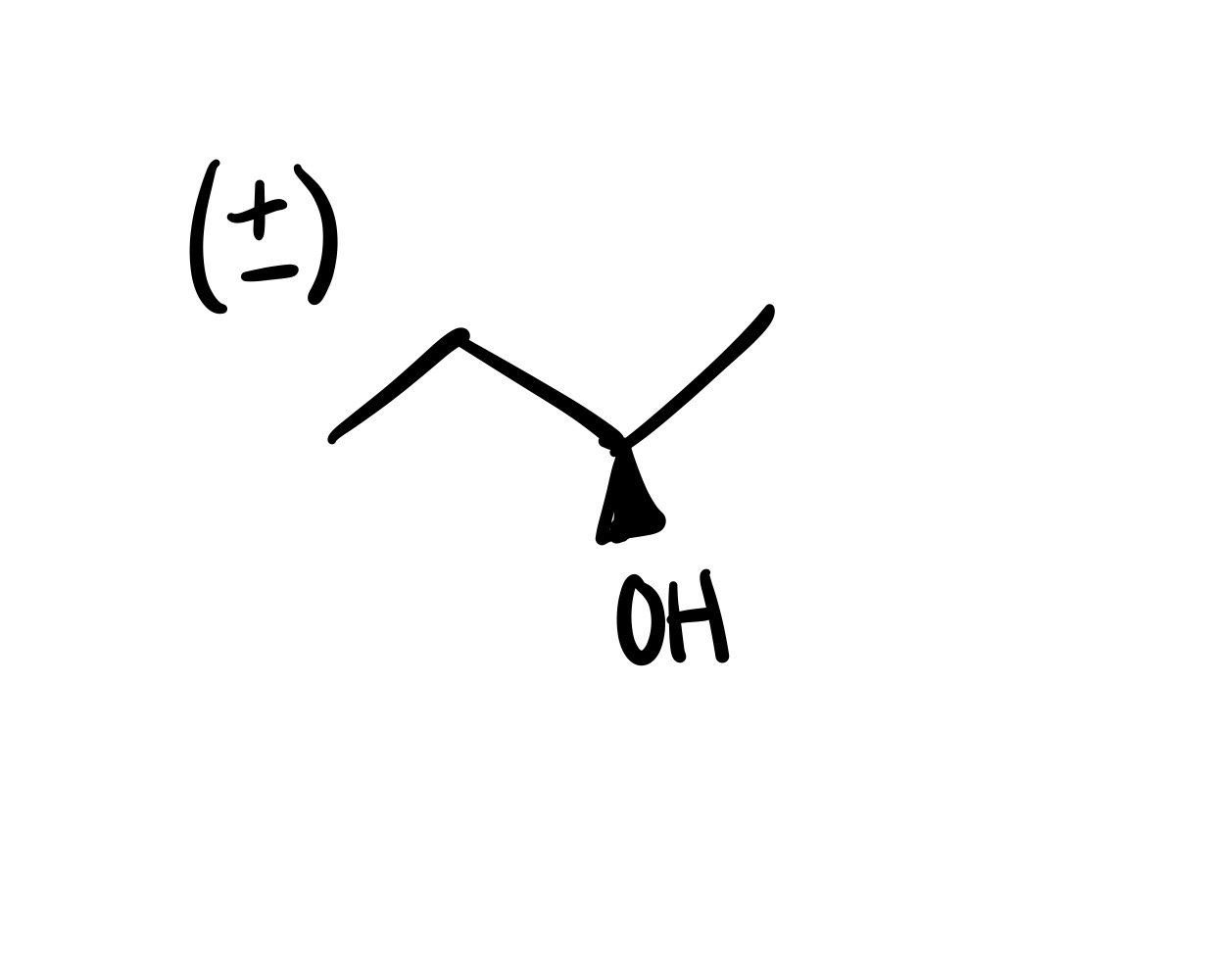
The double bond is broken, and the OH is added (can be wedge or dash). Add plus/minus sign
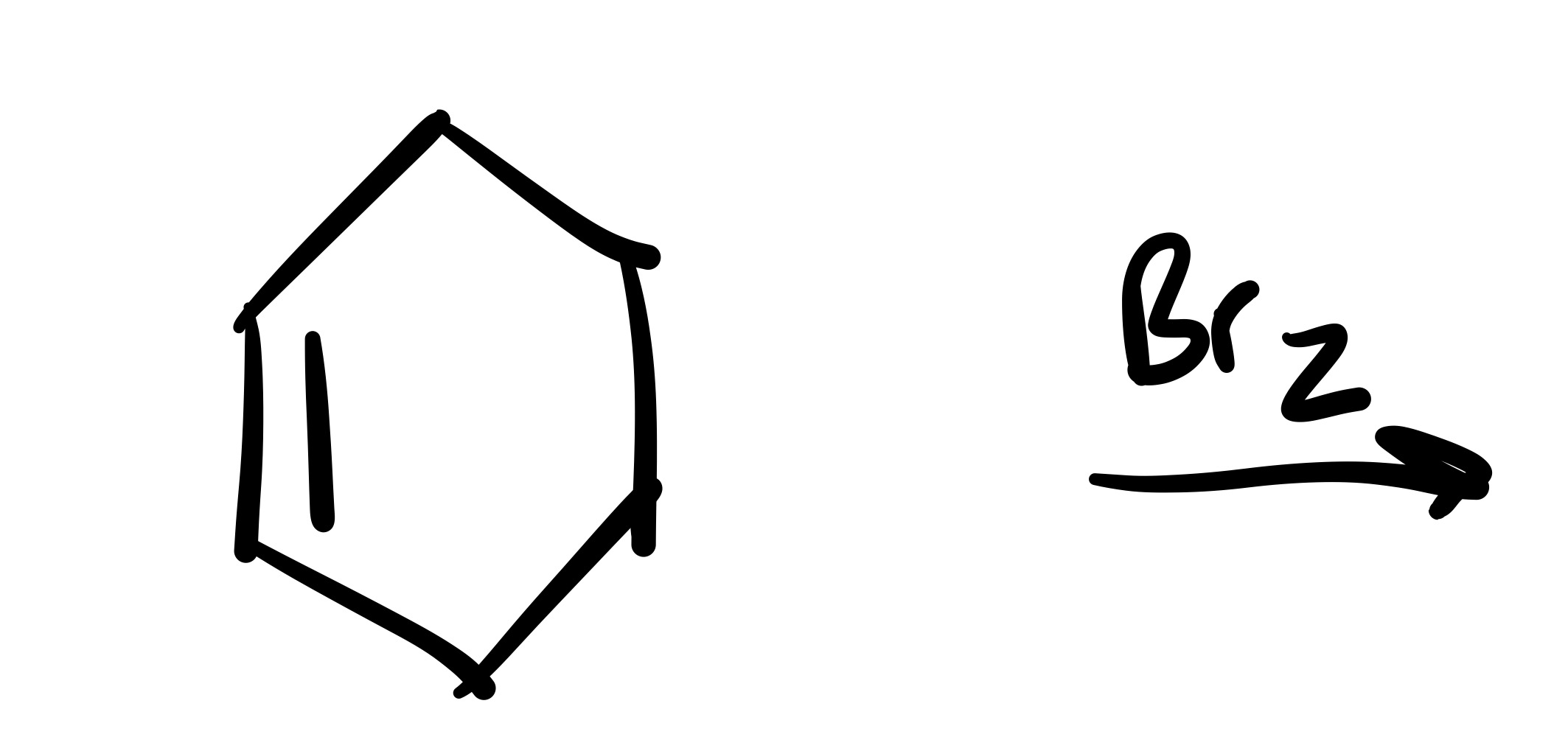
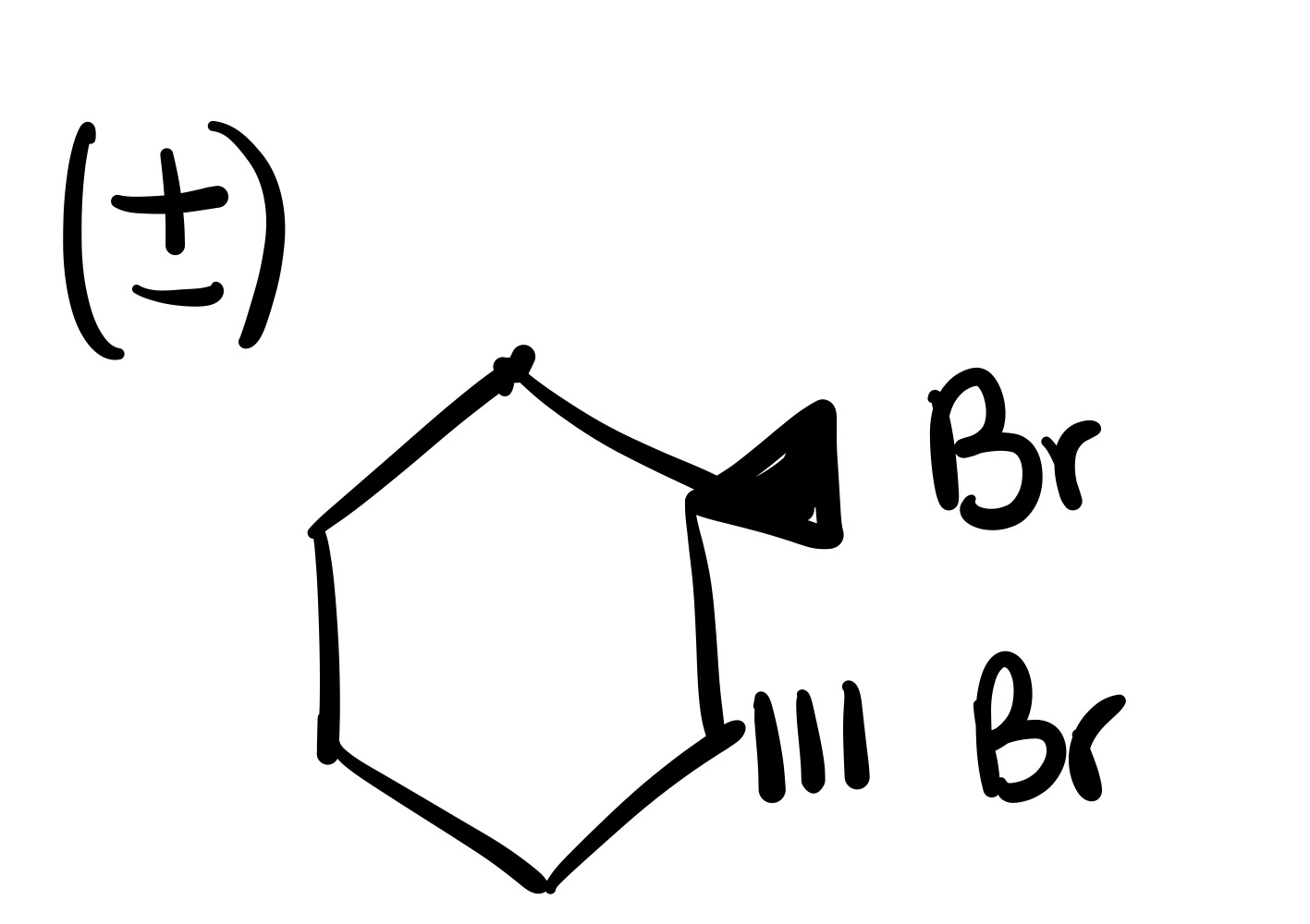
The double bond is broken, and the 2 Br’s are added (one is dash, and one is wedge). Add plus/minus sign
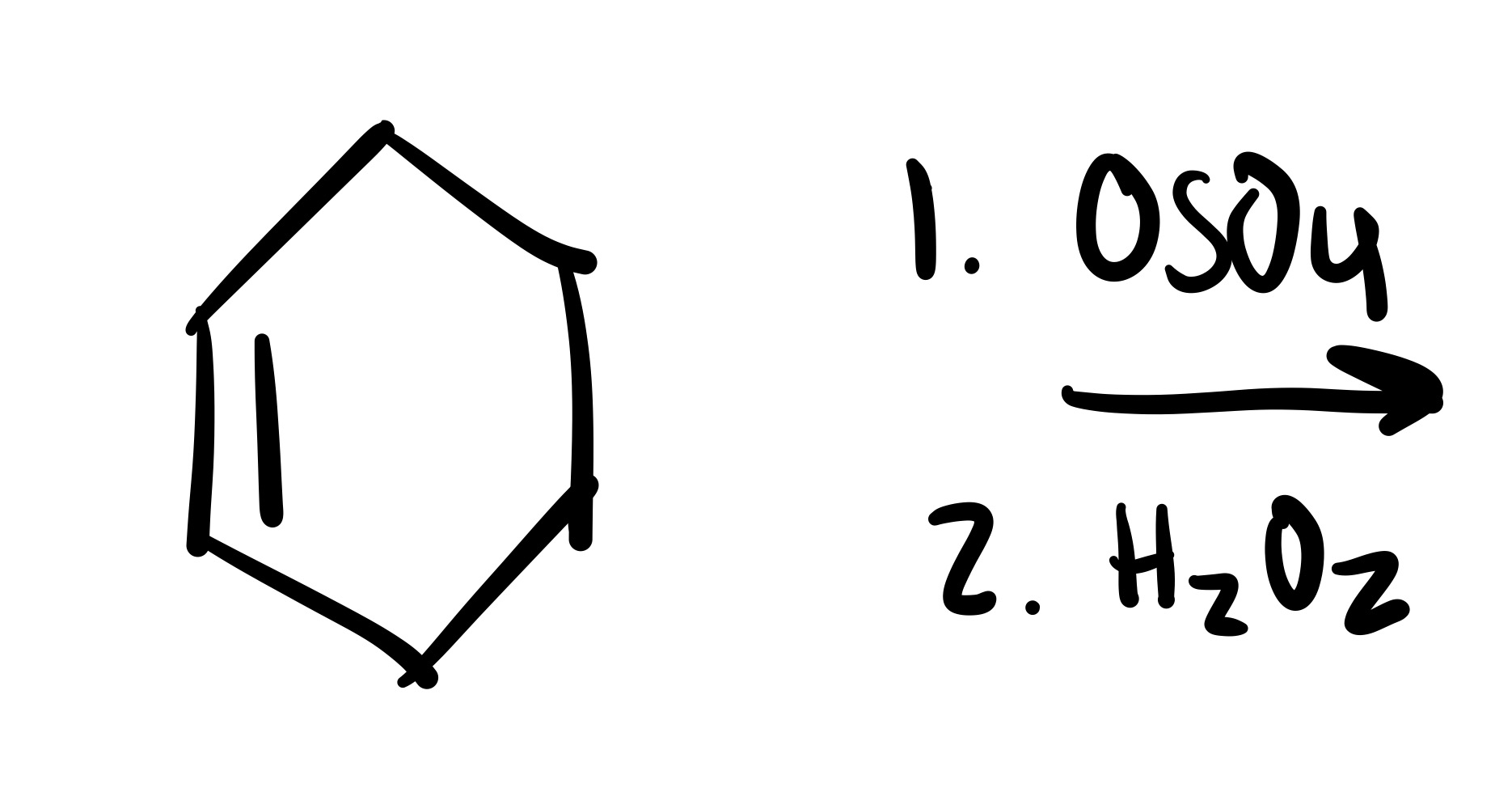
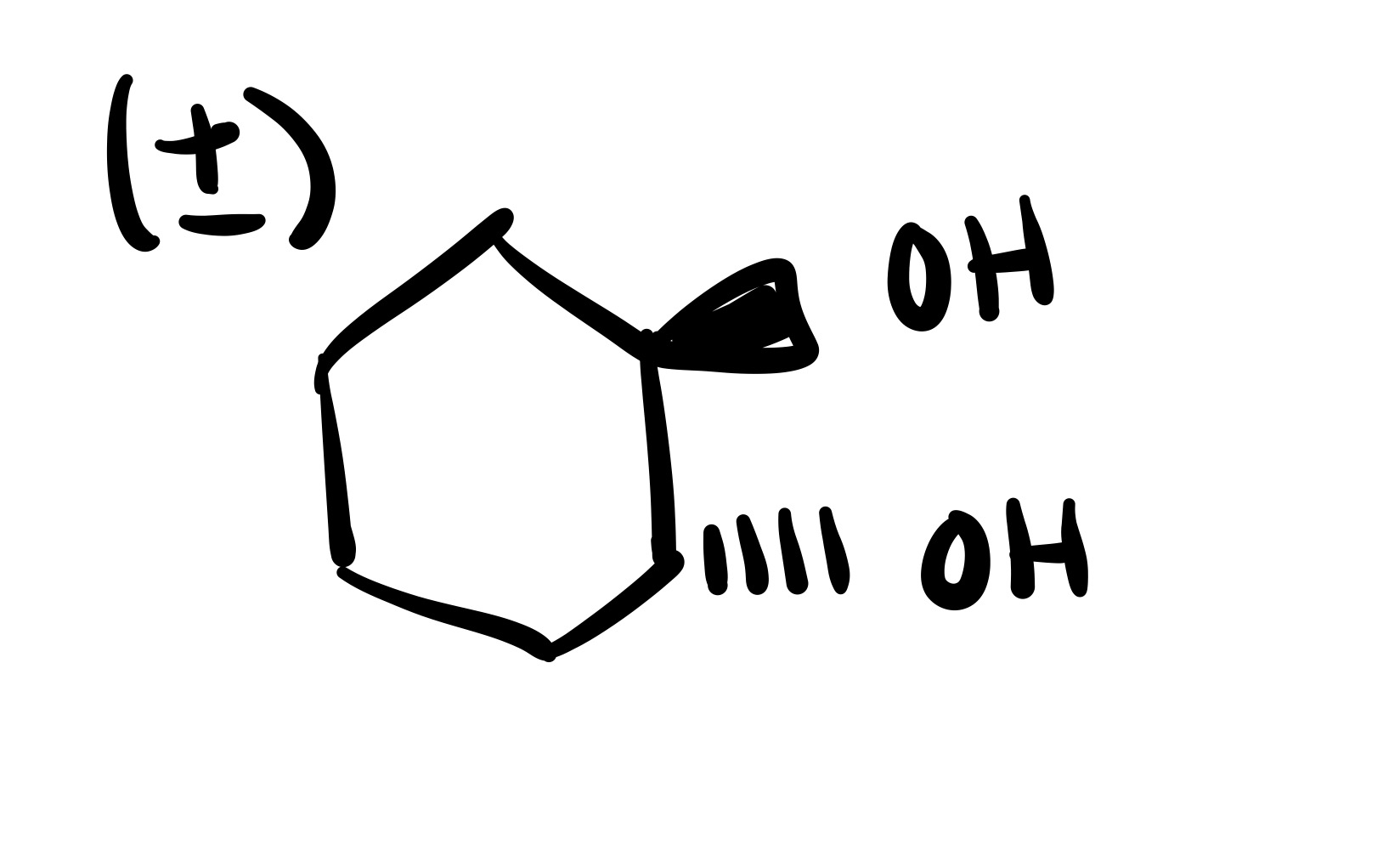
The double bond is broken and the 2 OH’s are added (both are wedge or both are dash). Add plus/minus sign
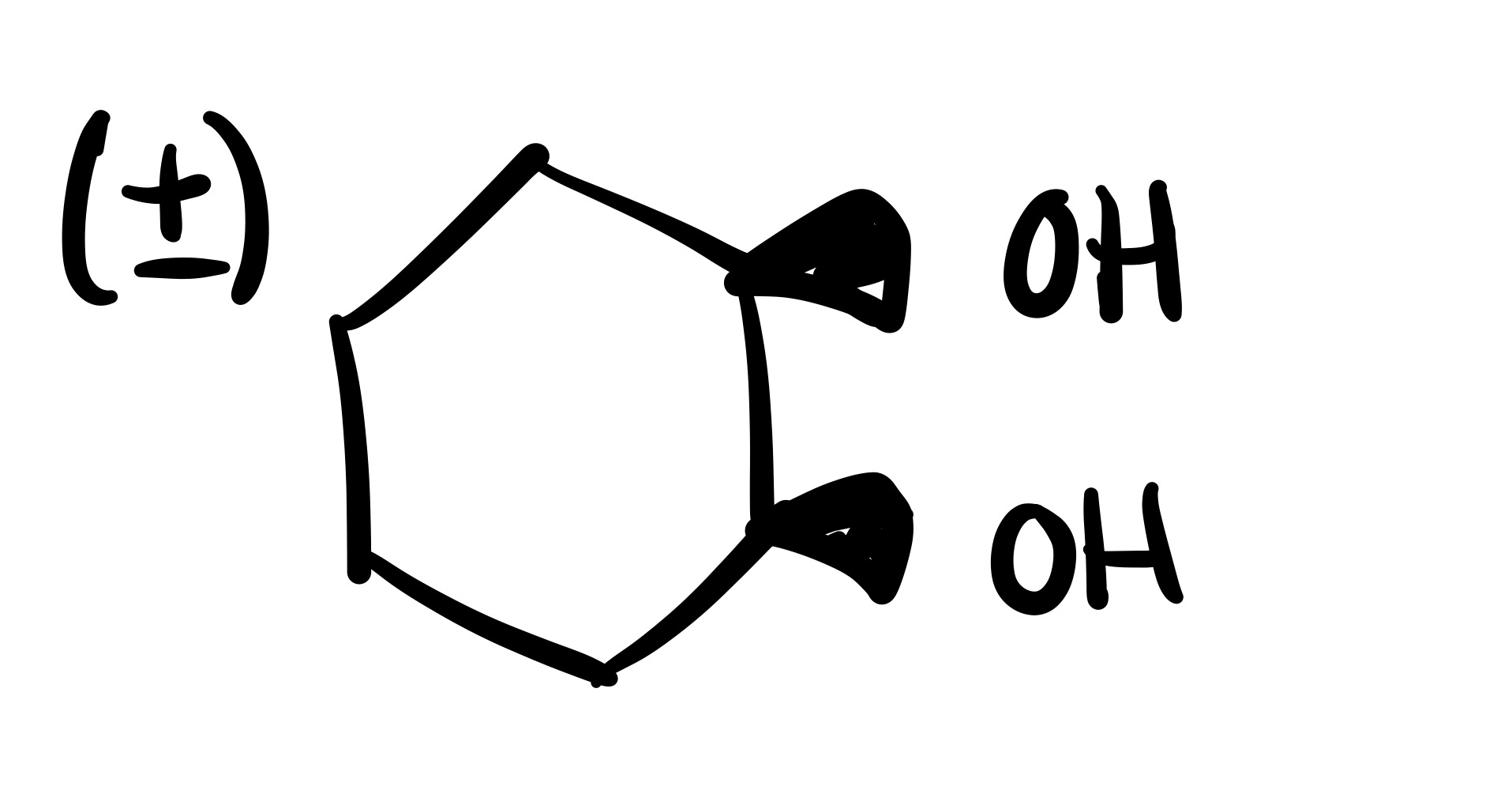
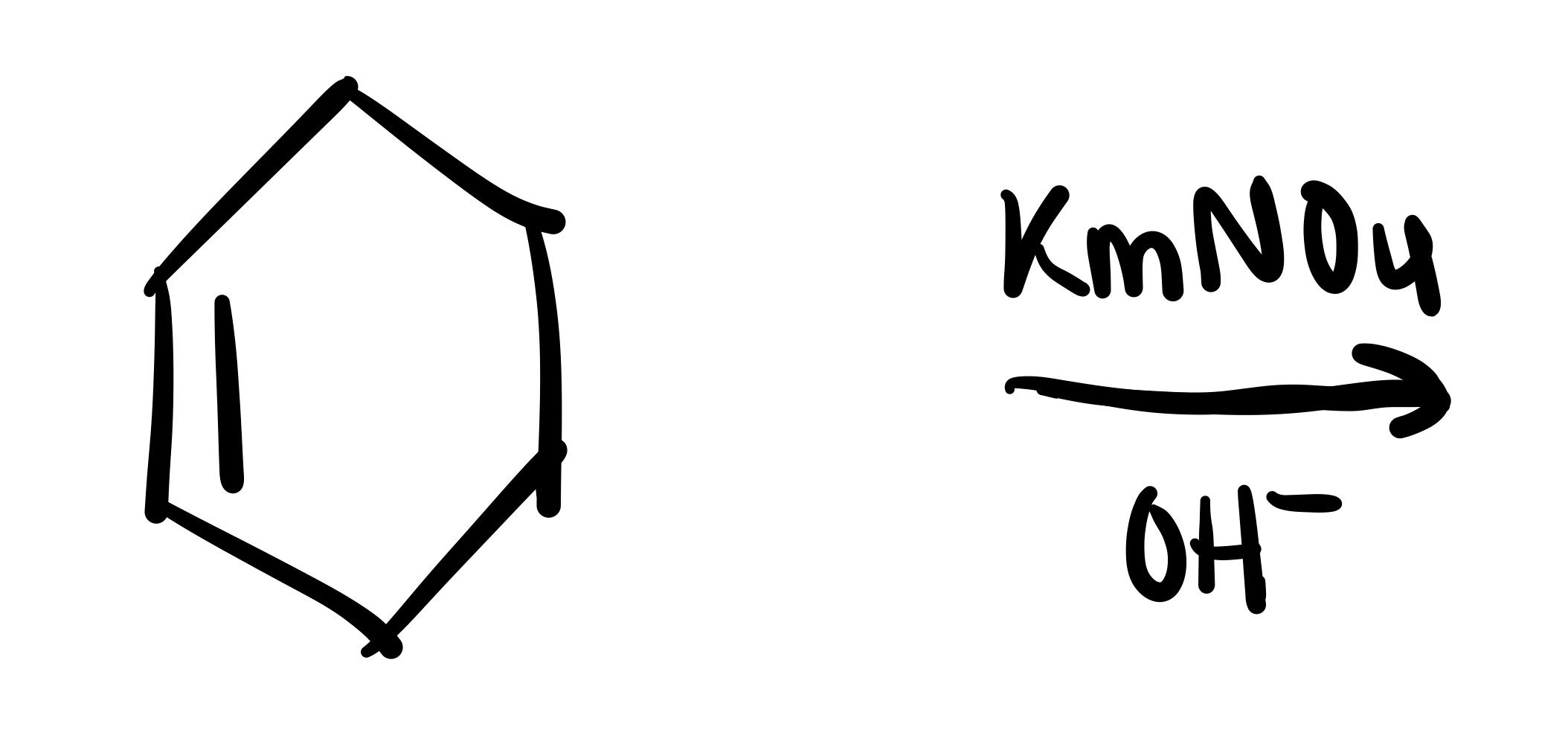
The double bond is broken and the 2 OH’s are added (both are wedge or both are dash). Add plus/minus sign
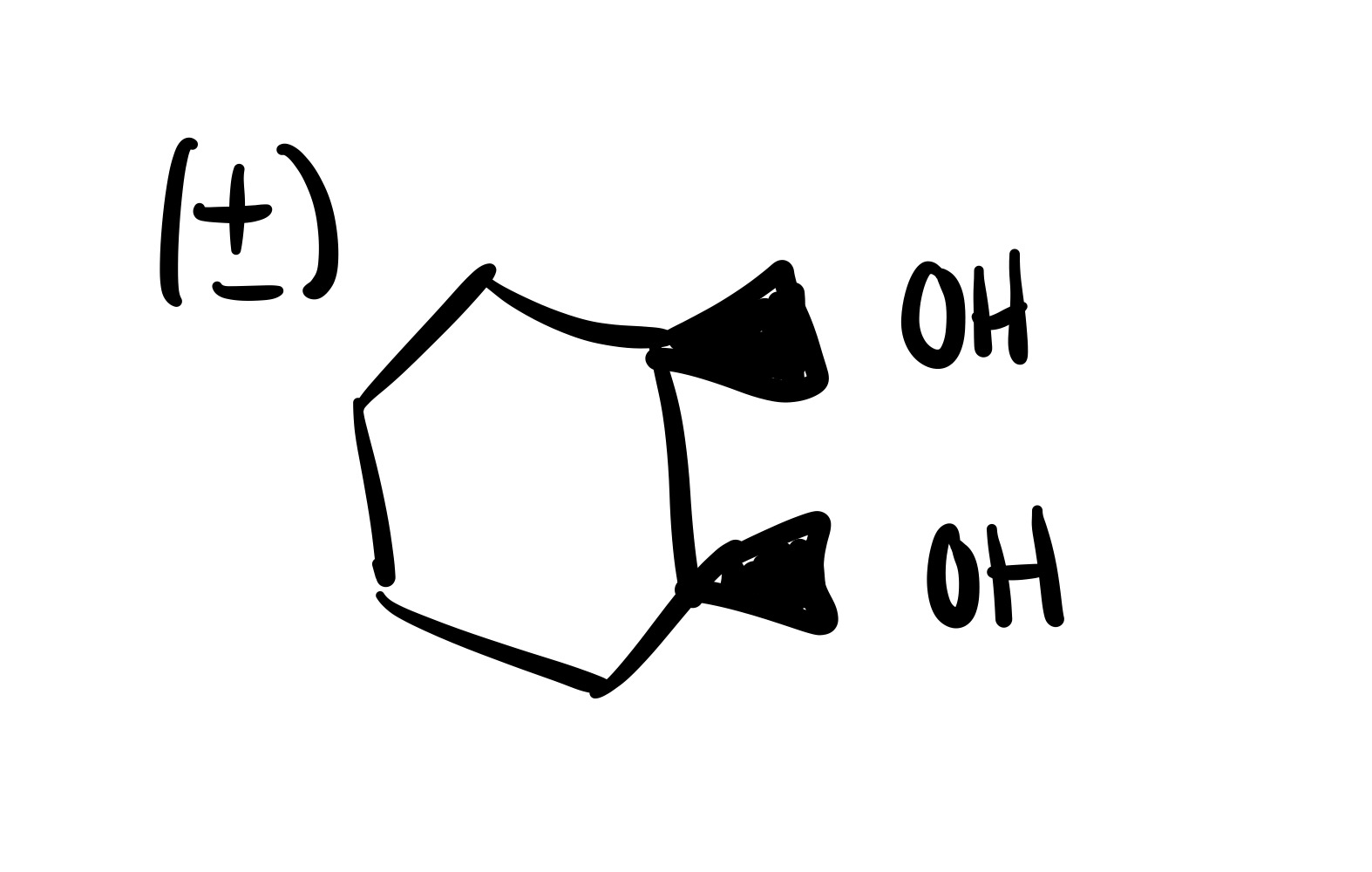
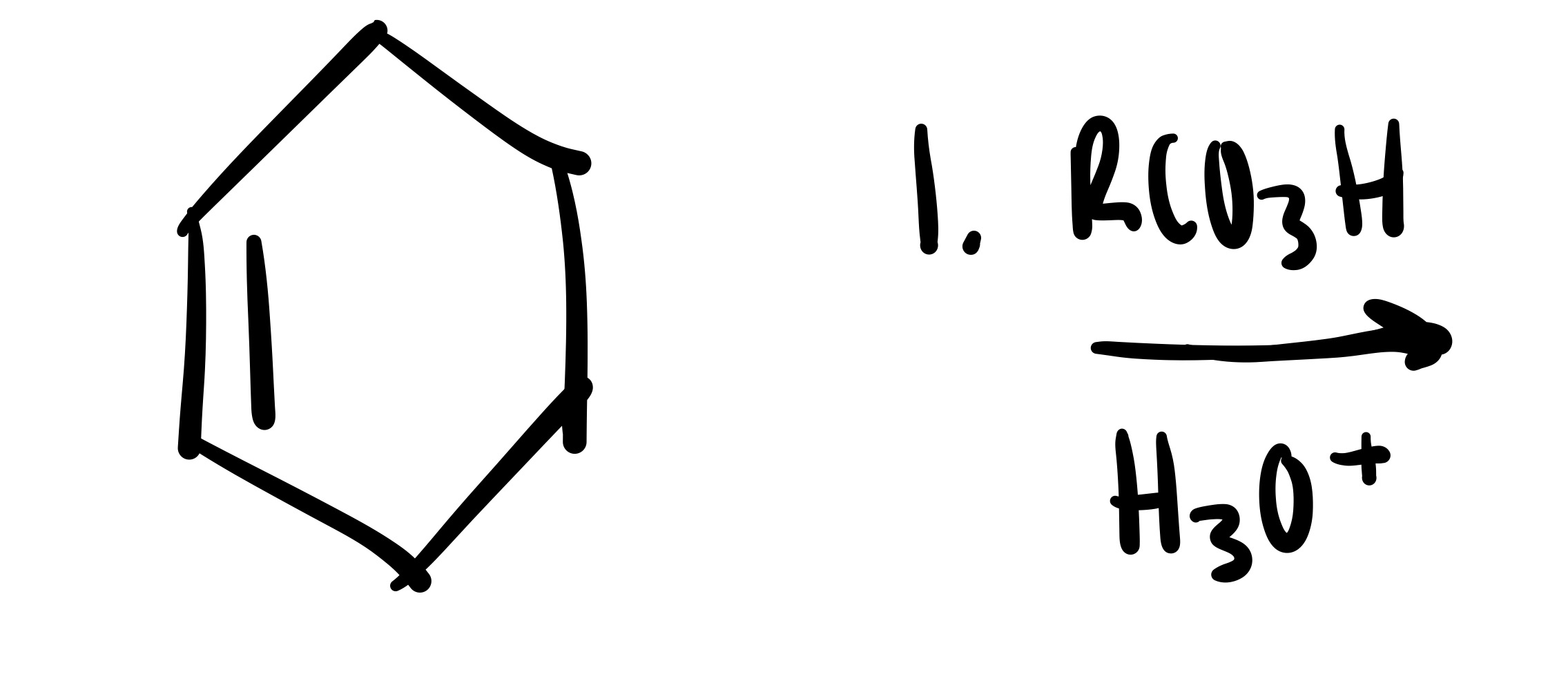
The double bond is broken and the 2 OH’s are added (one is dash, one is wedge). Add plus/minus sign
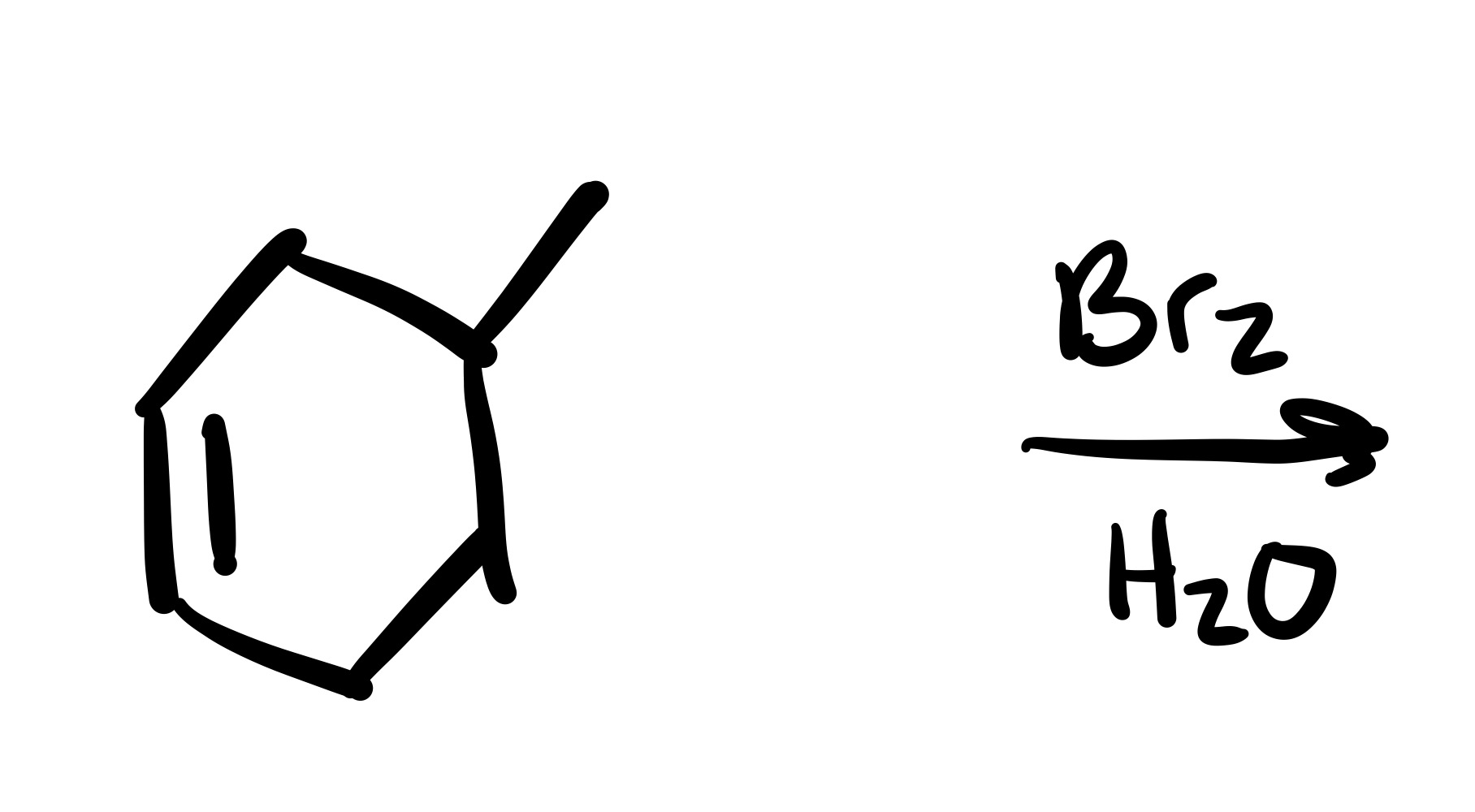
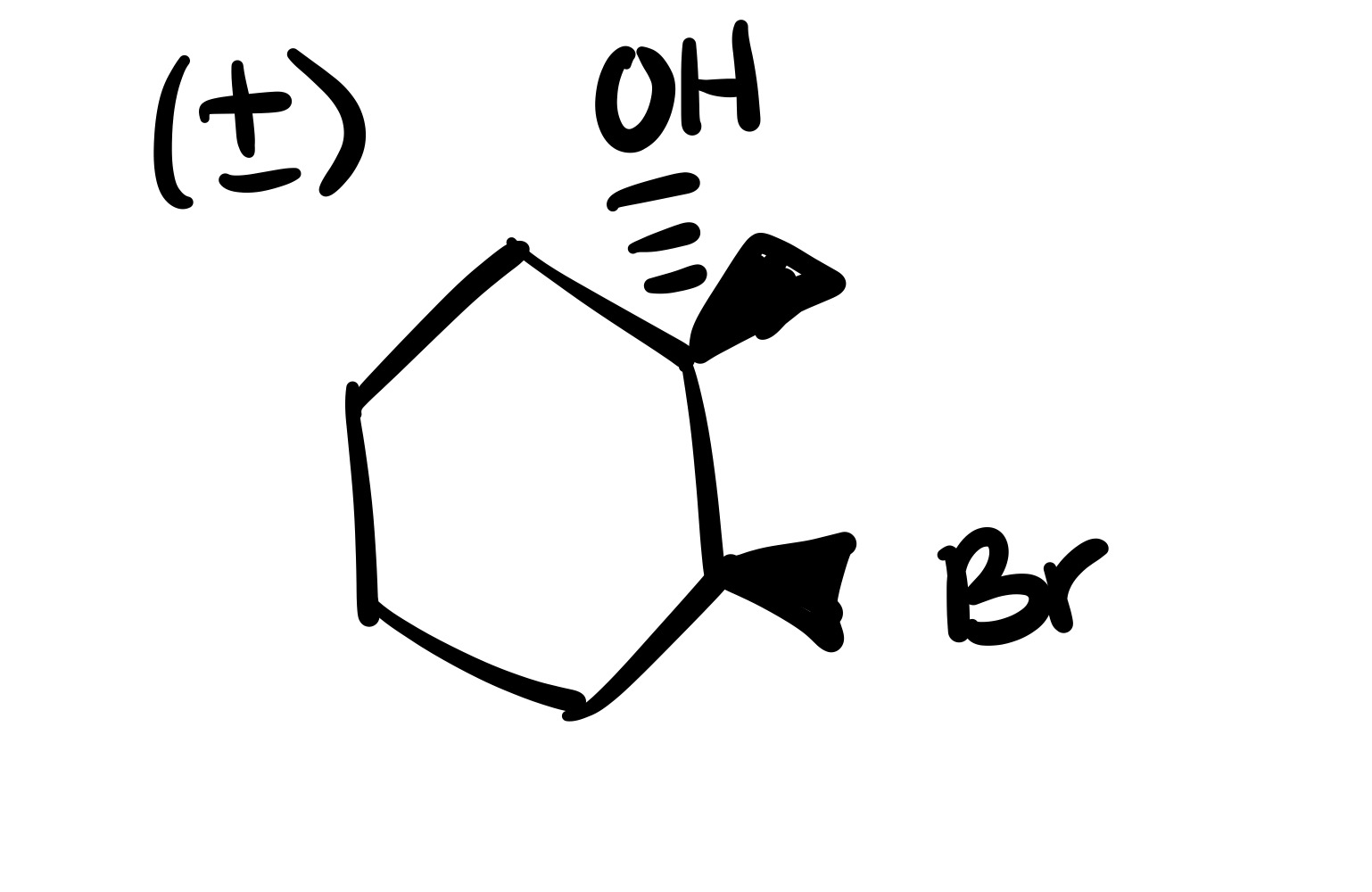
The double bond is broken. The methyl group, and Br added are wedges, and the OH is added to the methyl carbon on a dash (add plus/minus sign).
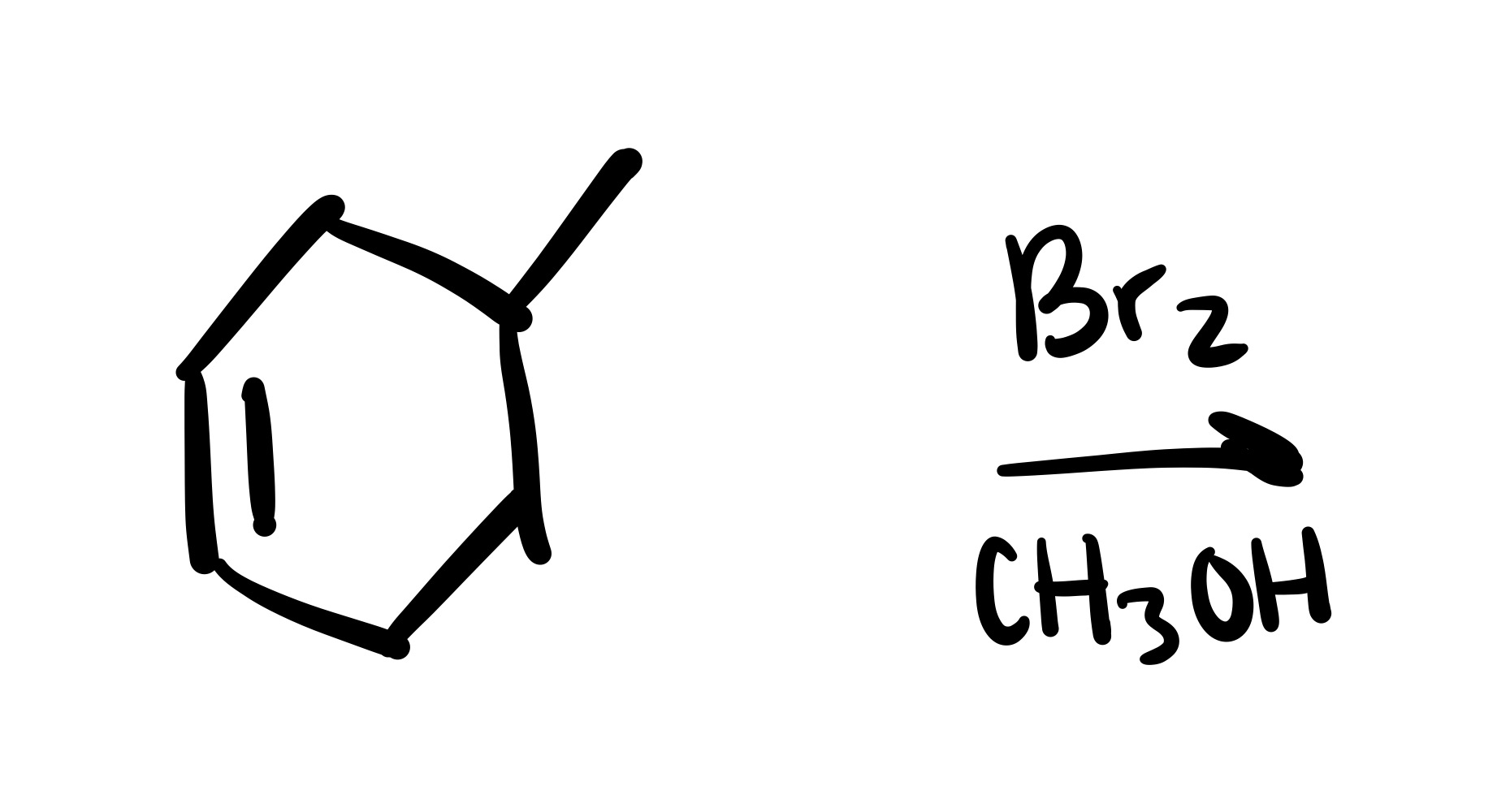
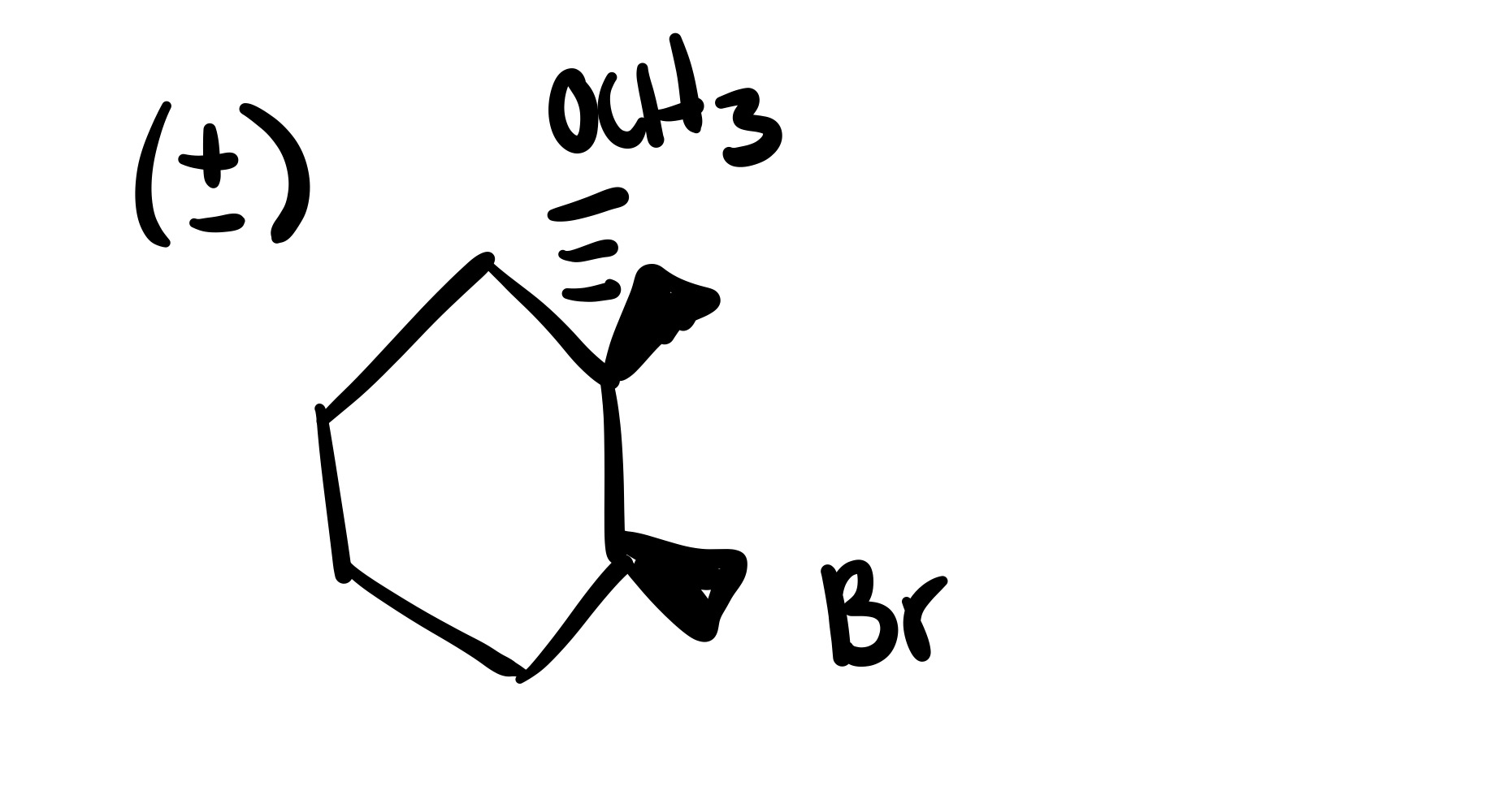
The double bond is broken. The methyl group, and Br added are wedges, and the OCH3 is added to the methyl carbon on a dash (add plus/minus sign).
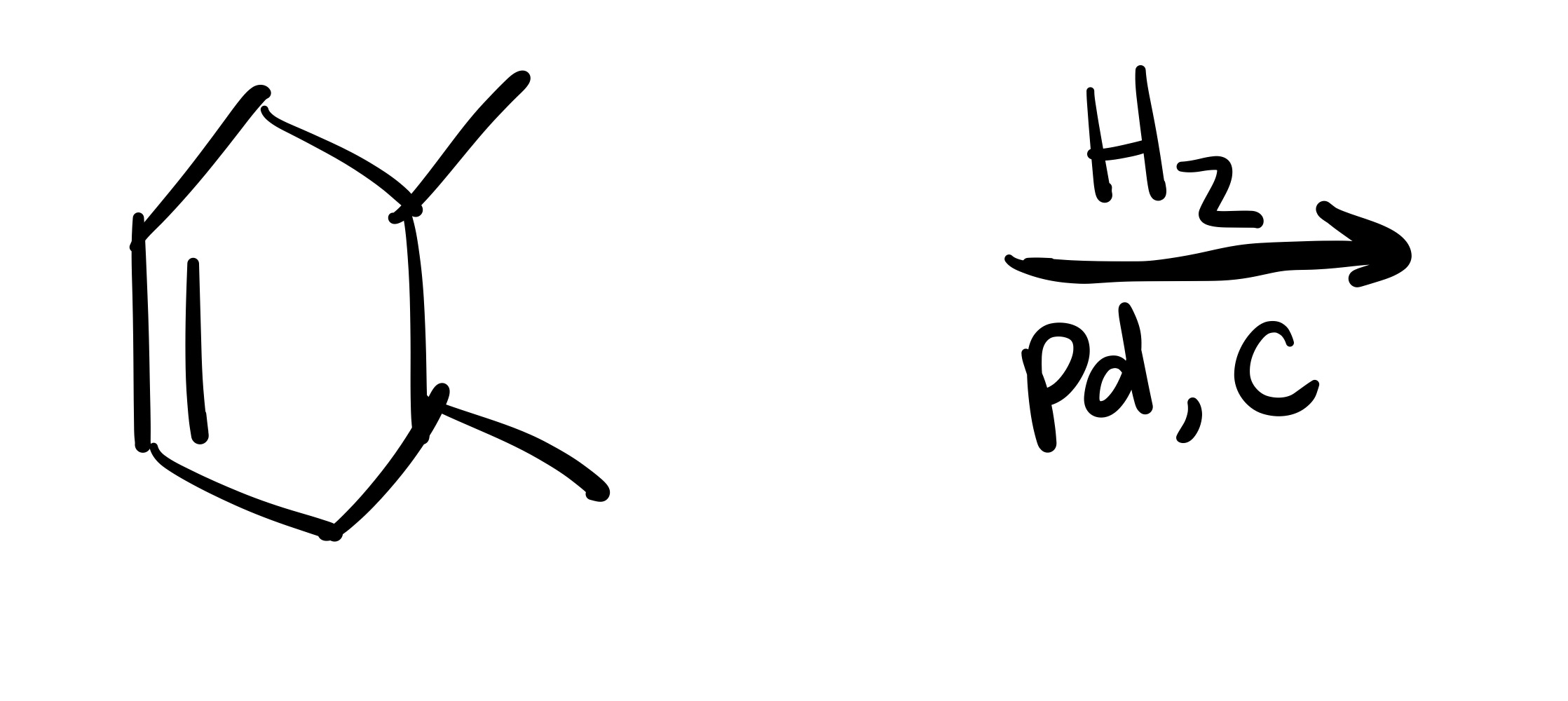
The double bond is broken. The 2 methyl groups are wedges
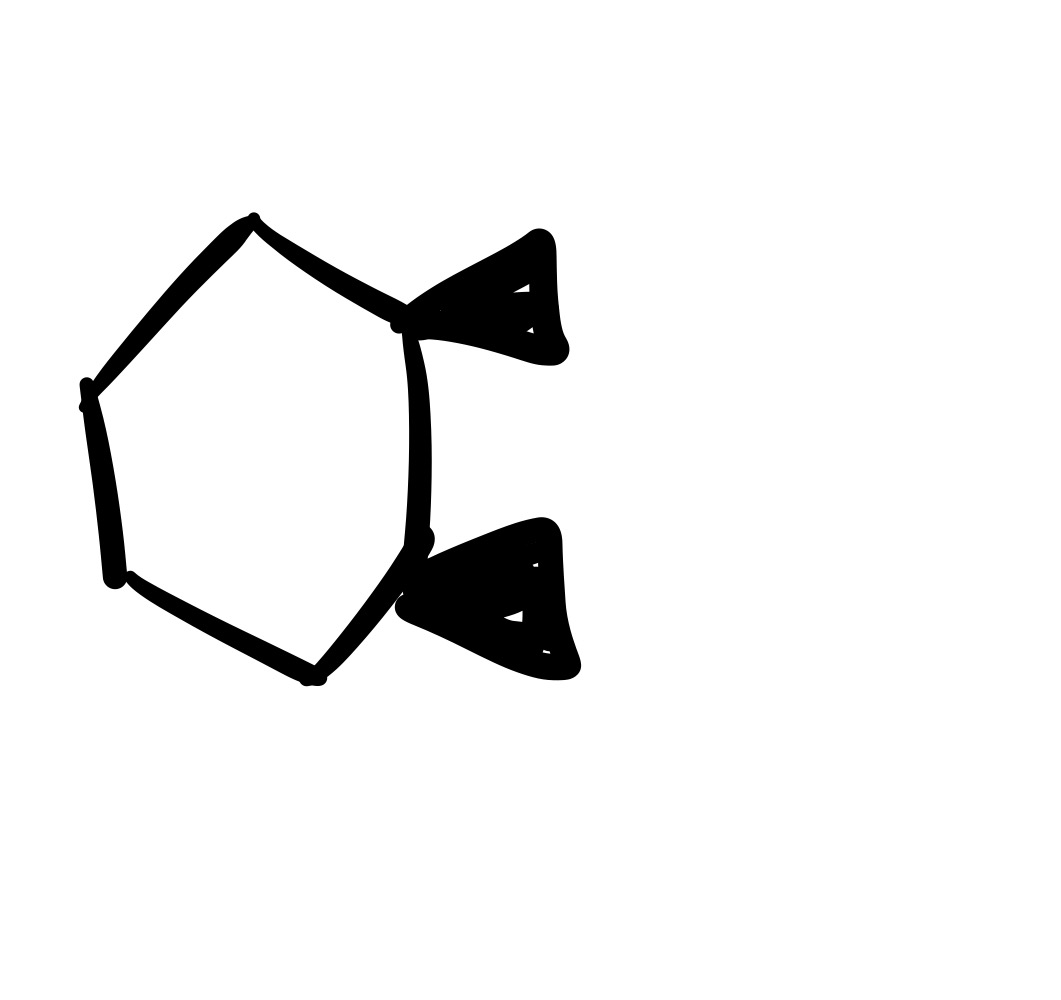
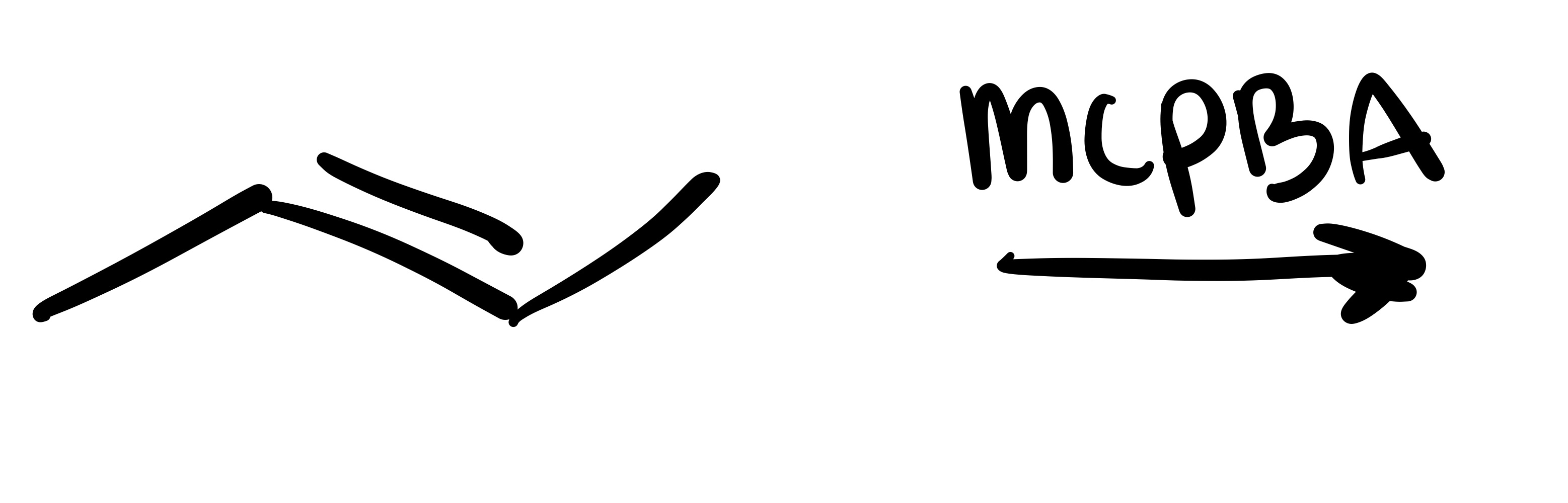
The double bond is broken, and forms an epioxide w/ O
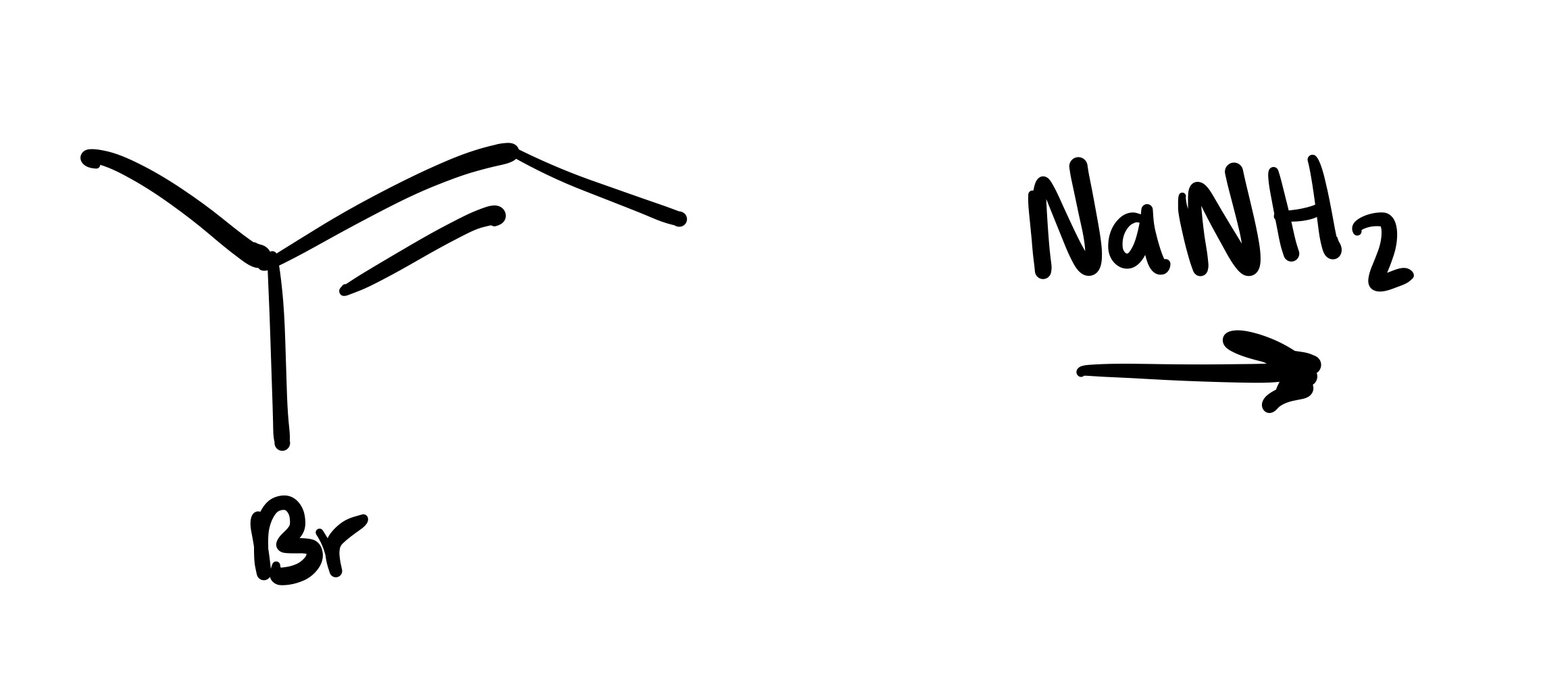
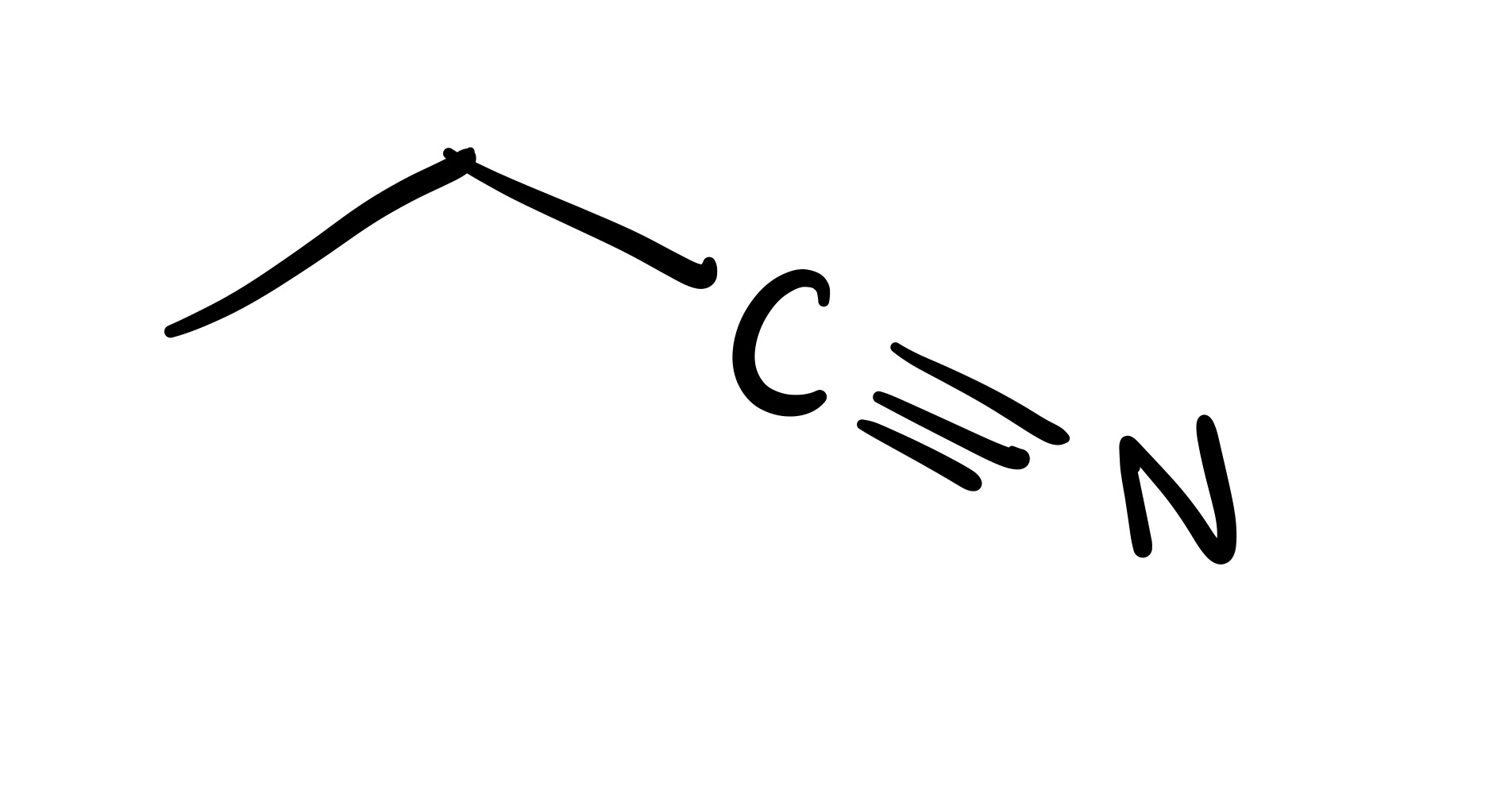
The double bond is broken, and a triple bond is formed (into a straight line)
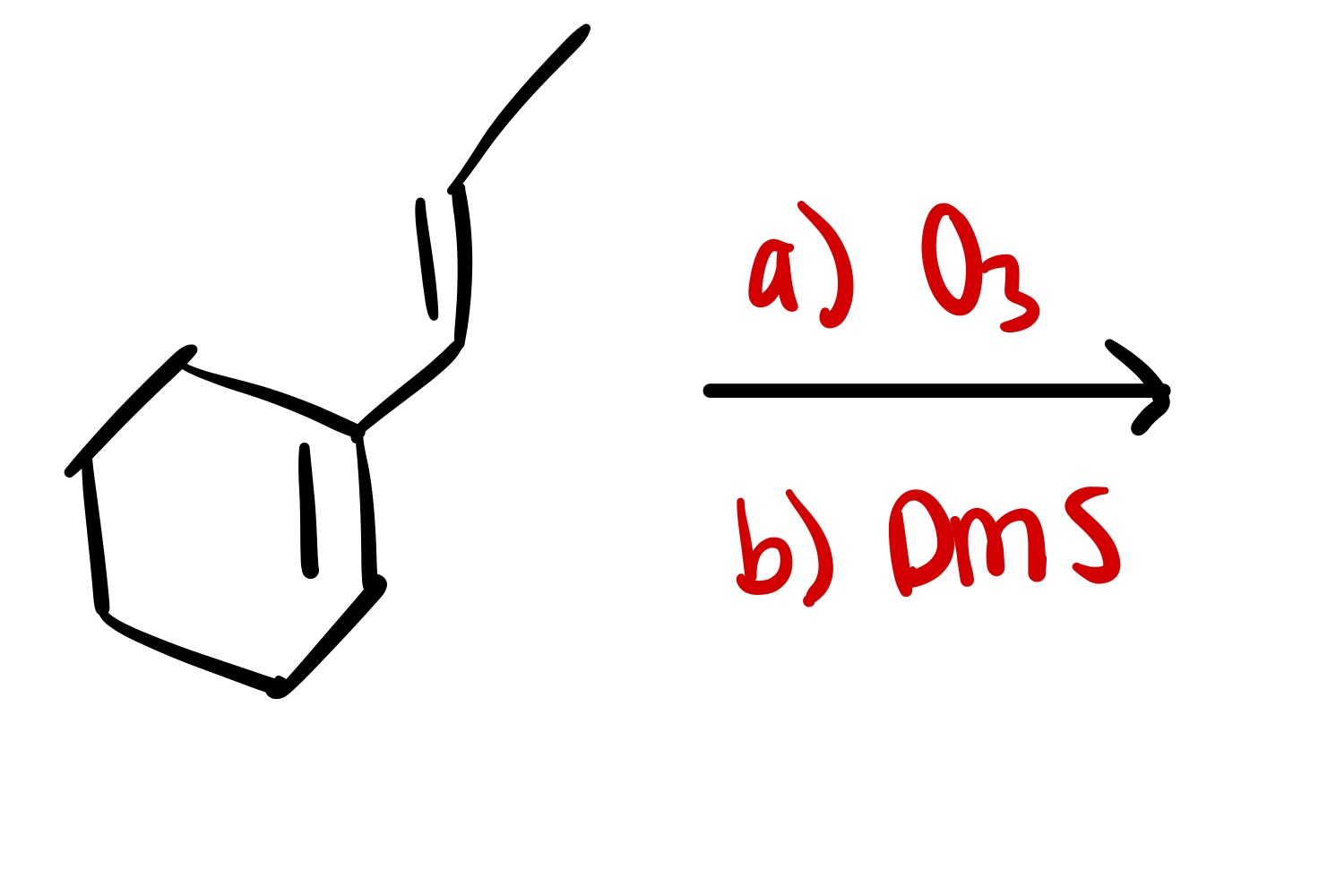
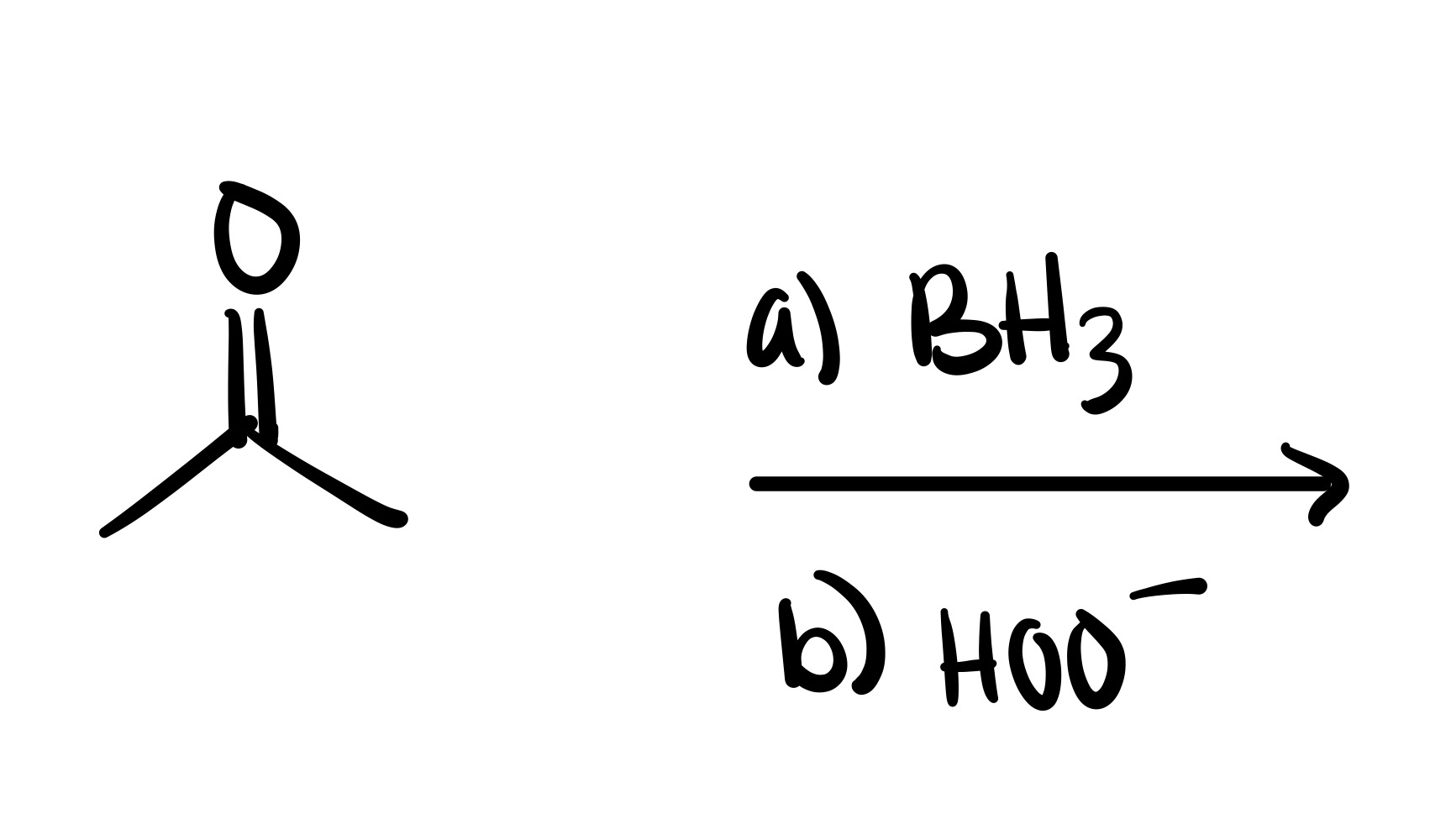
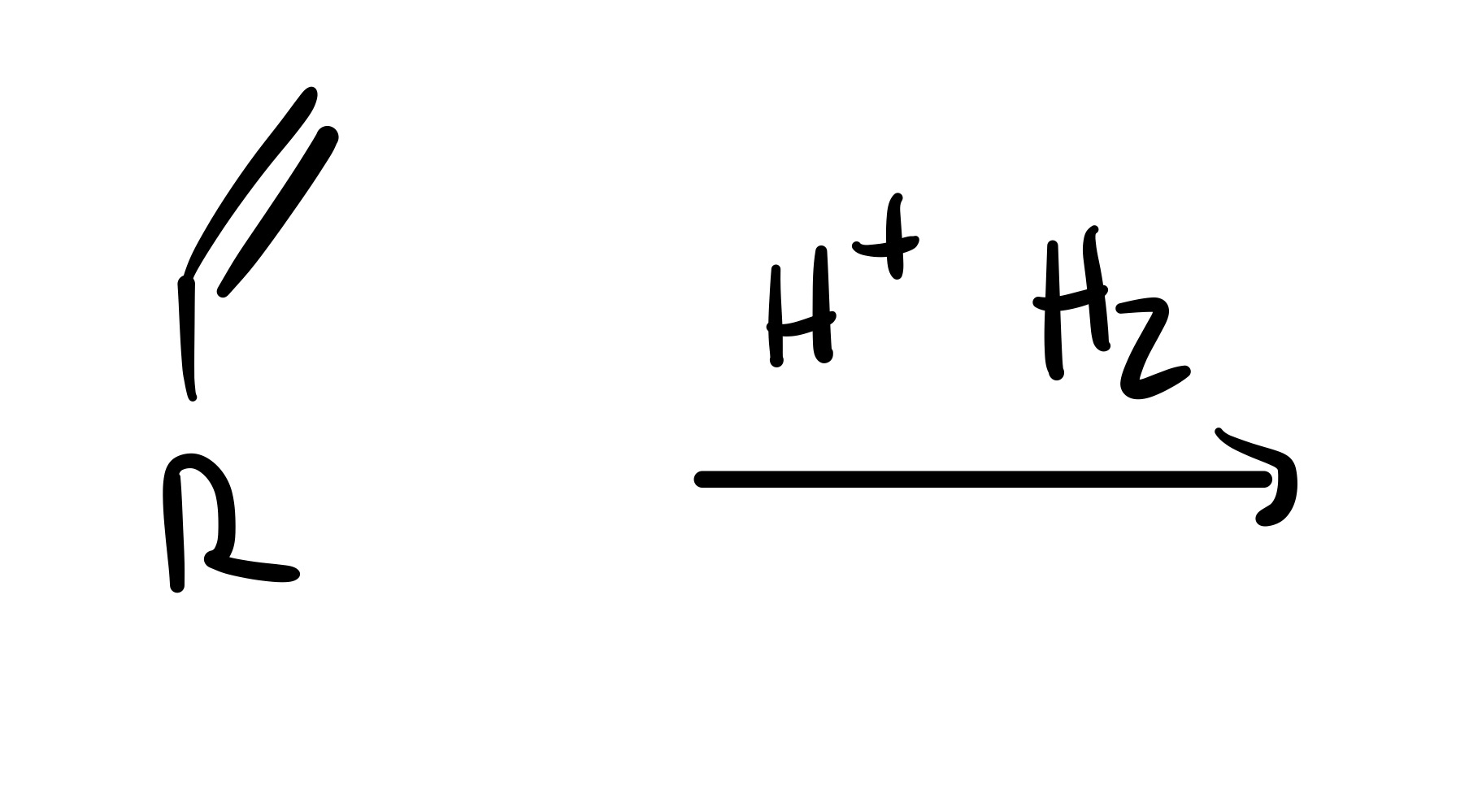
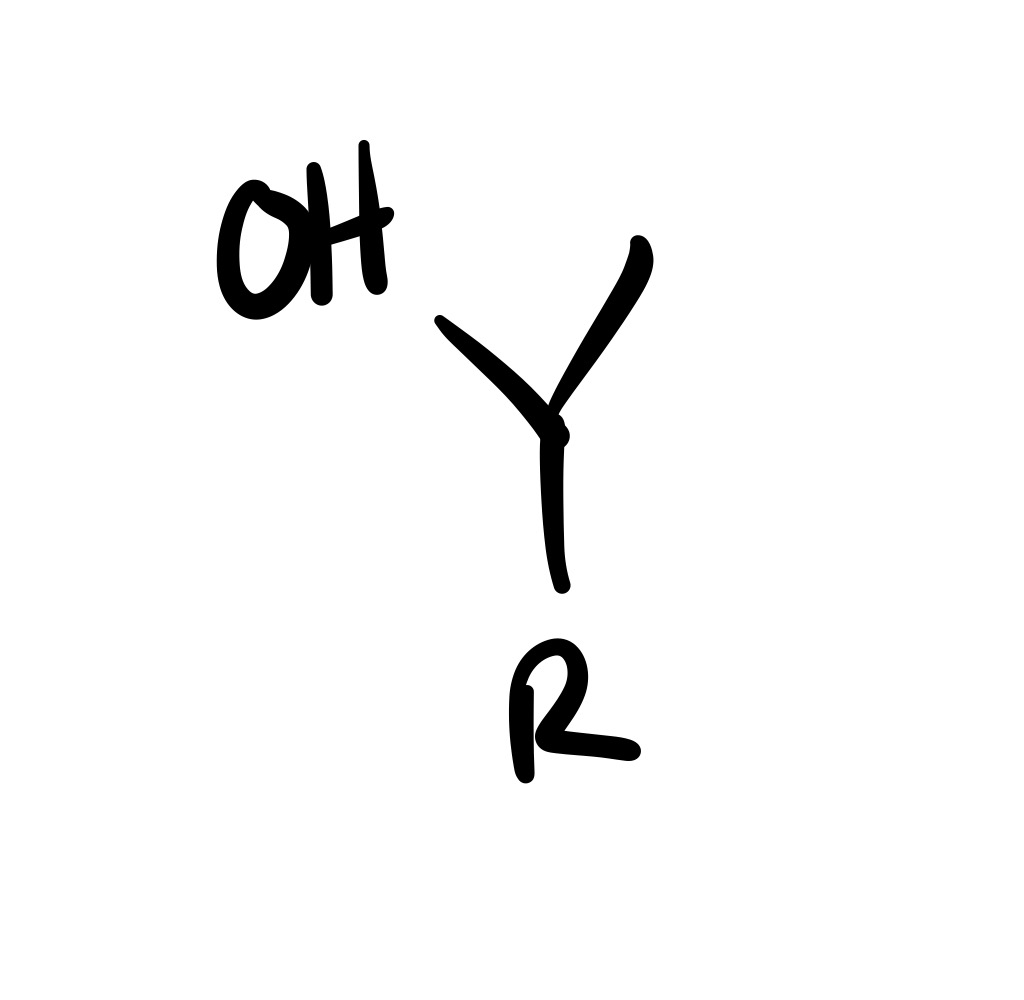
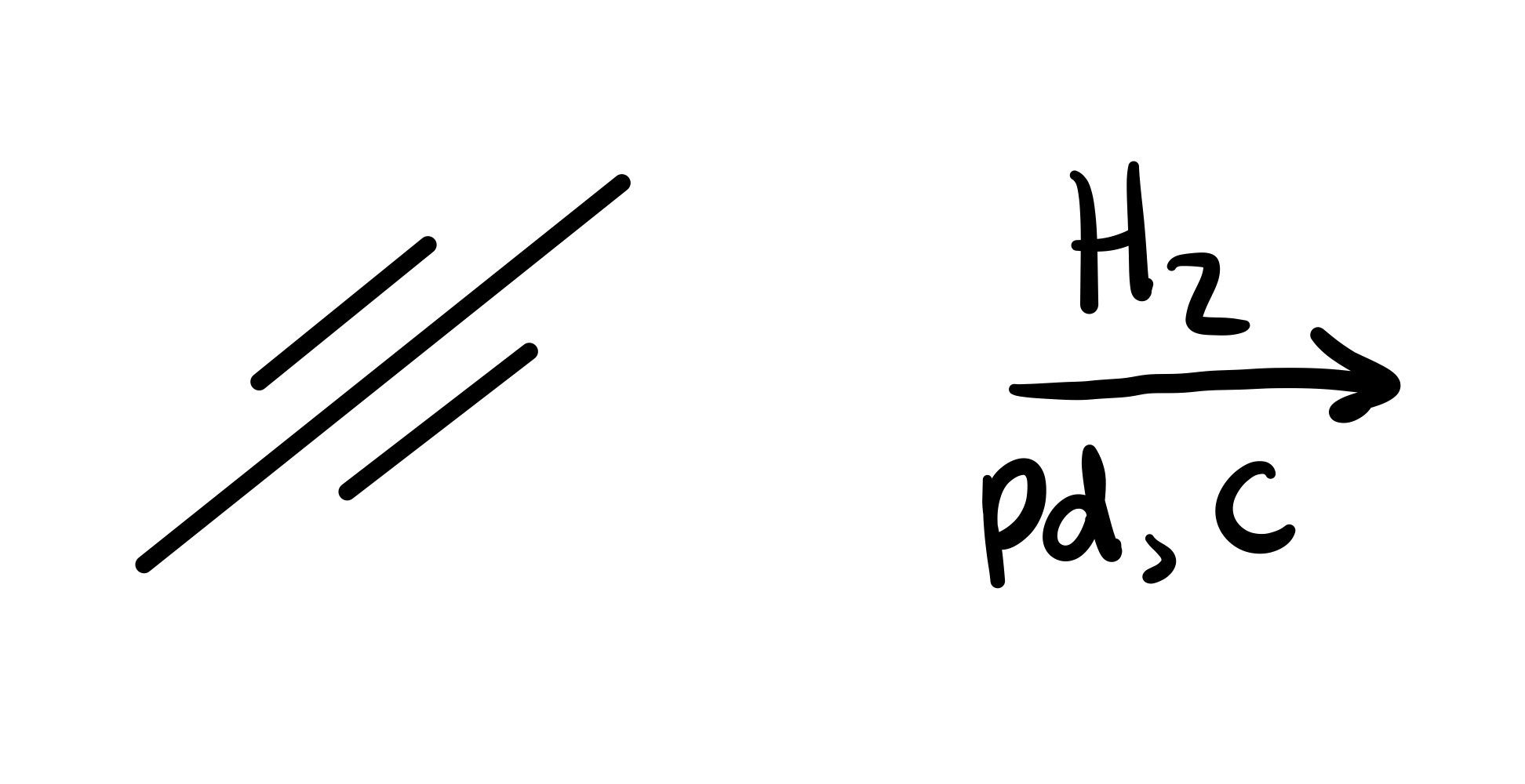
The triple bond is broken (all single bonds)
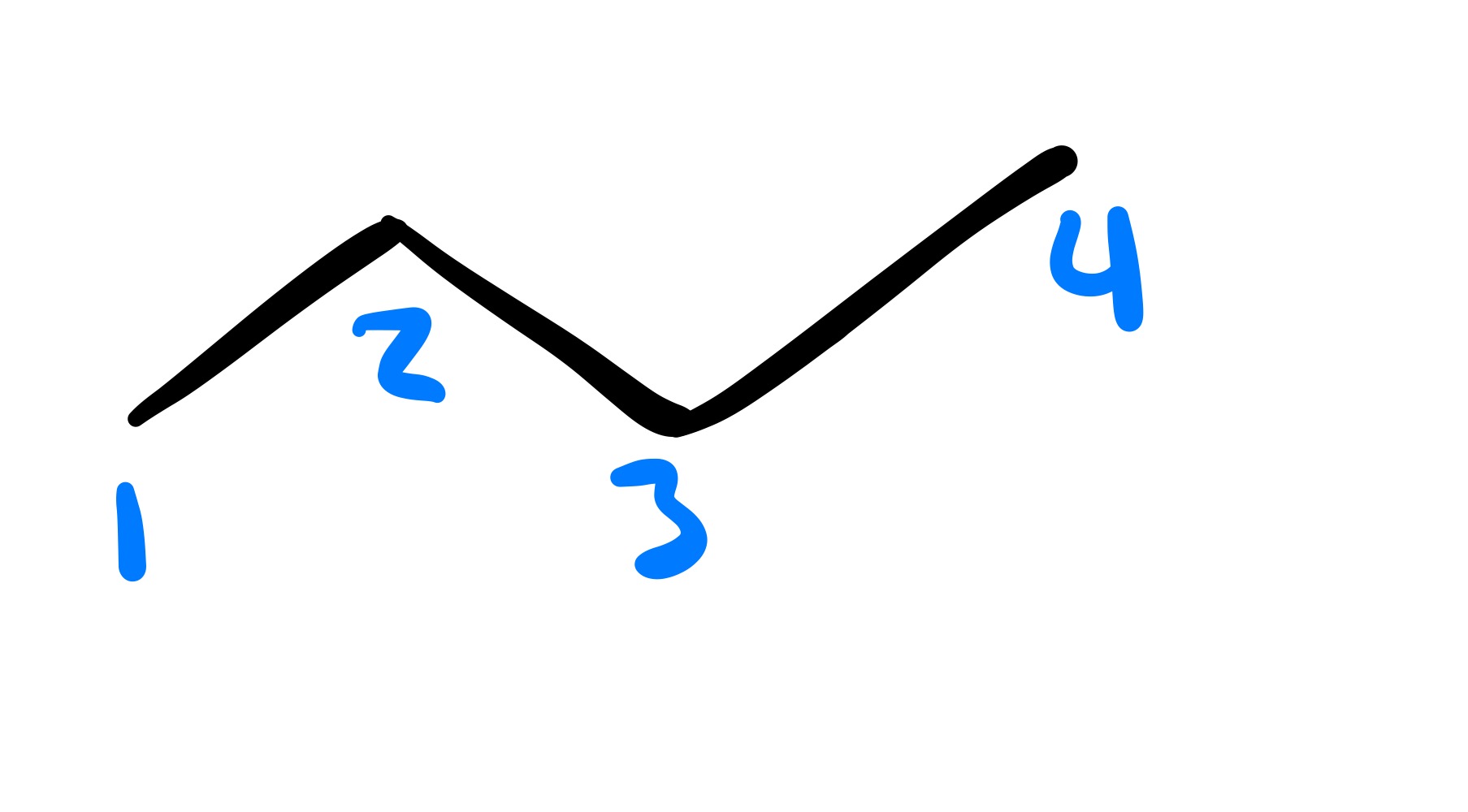
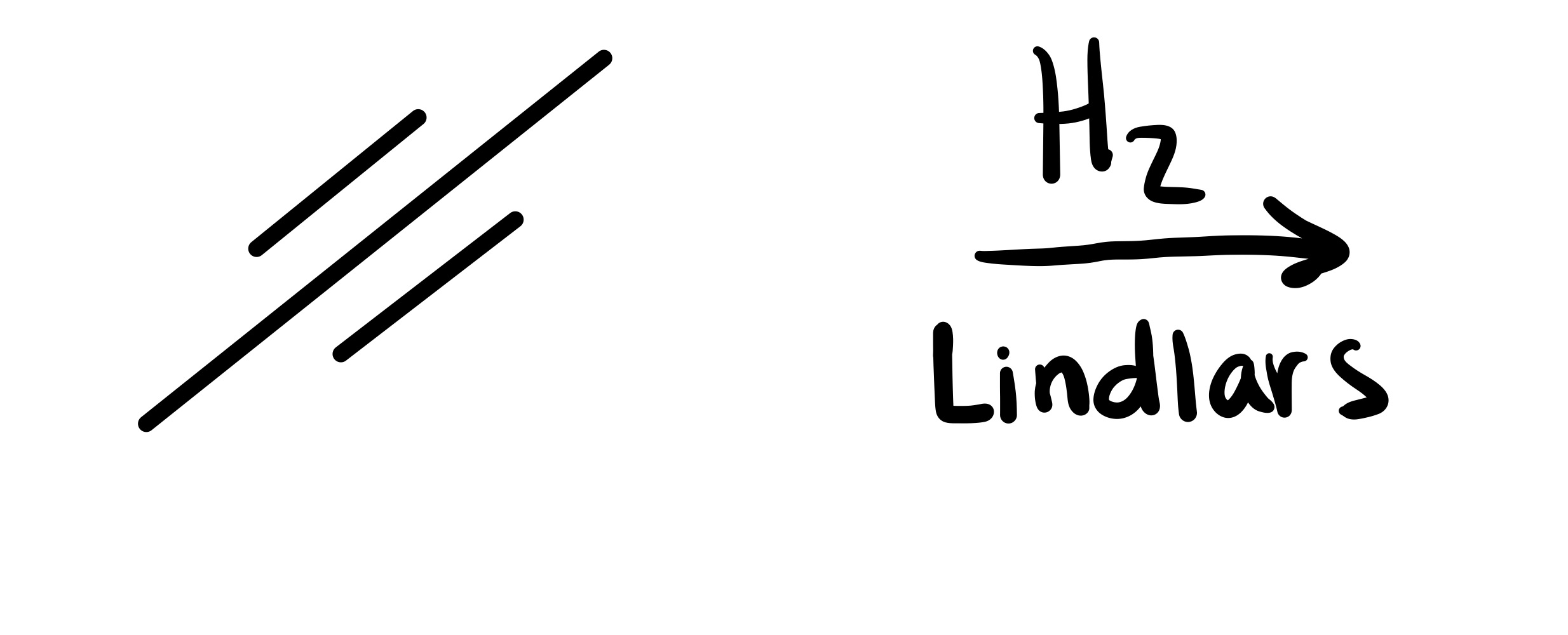
The triple bond is broken to form a double bond. Has to be Cis
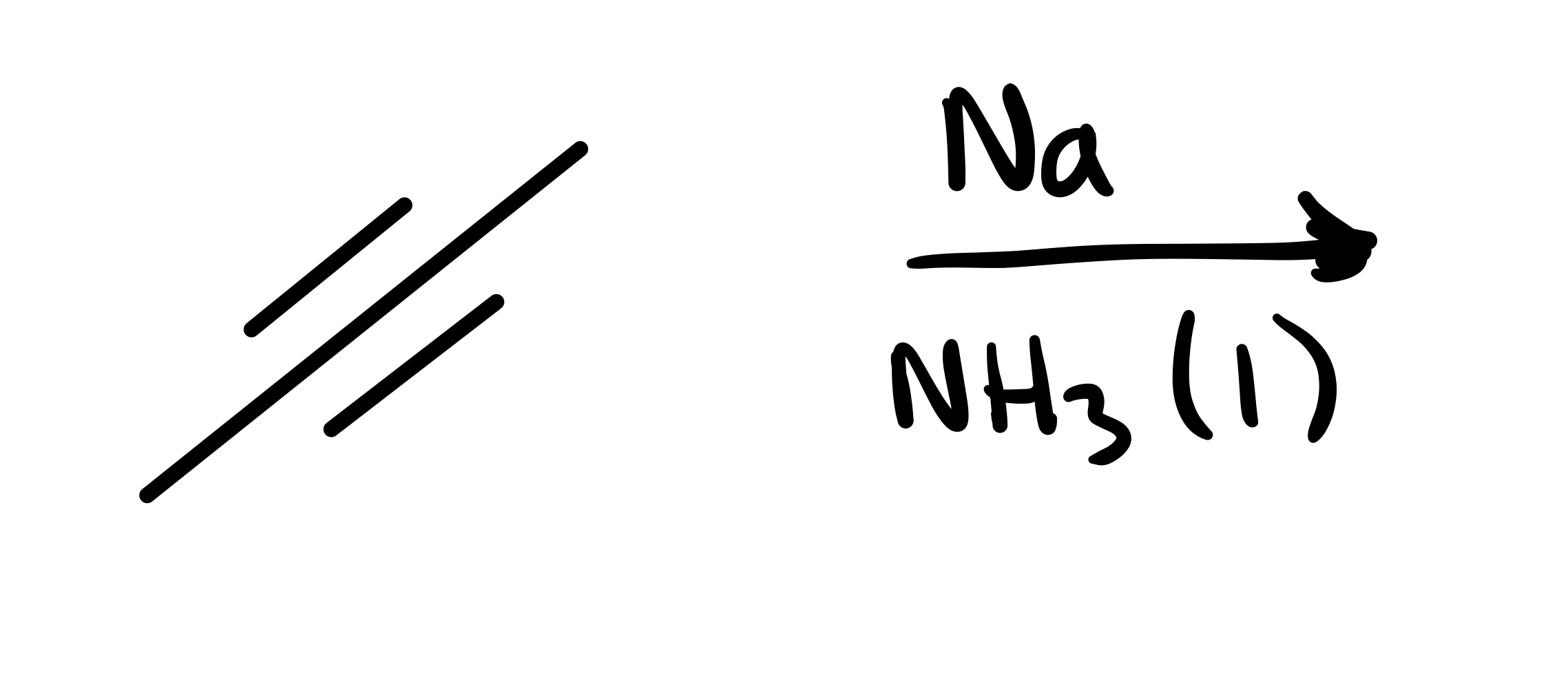
The triple bond is broken to form a double bond. Has to be Trans
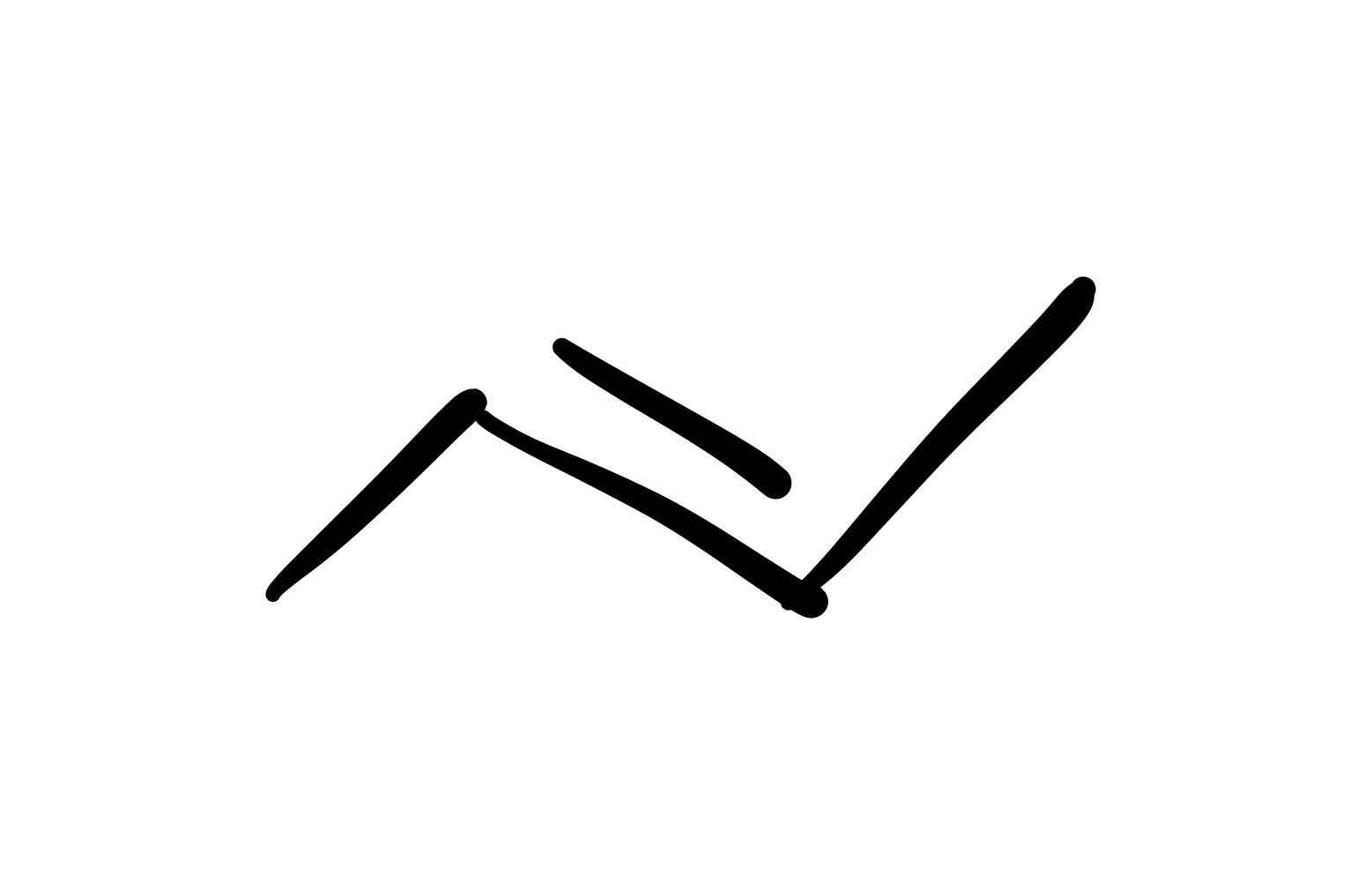
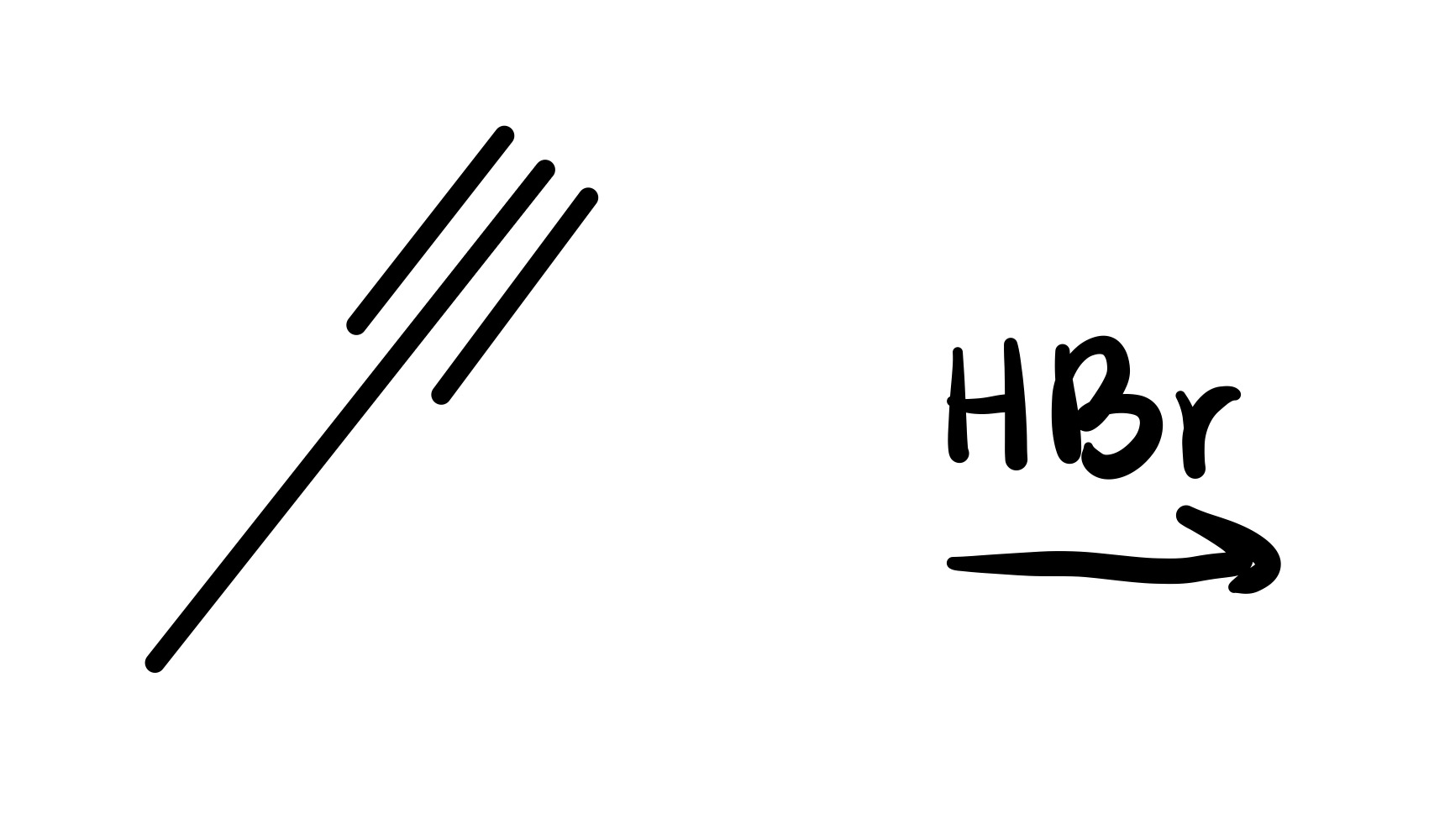
The triple bond is broken (only single bond carbon chain). And the 2 Br groups are added on the same carbon
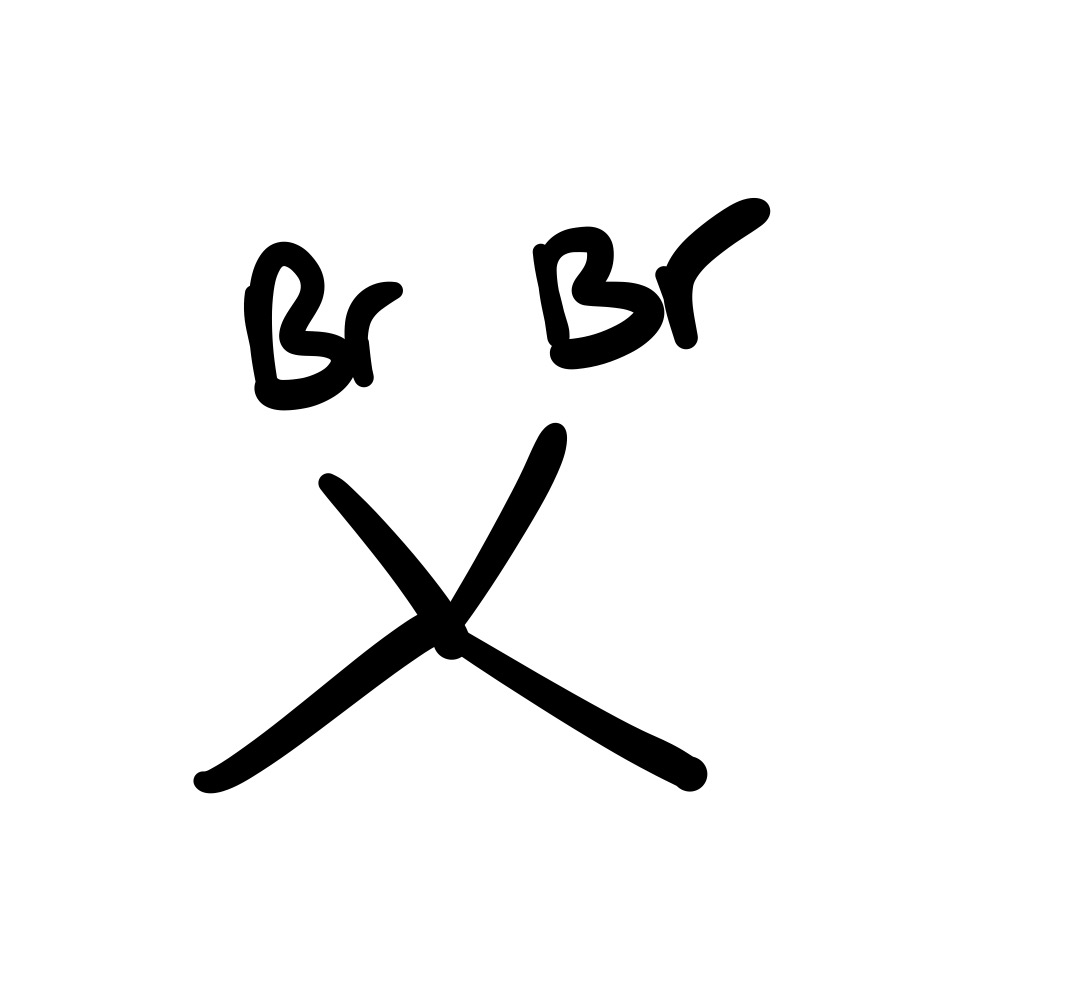
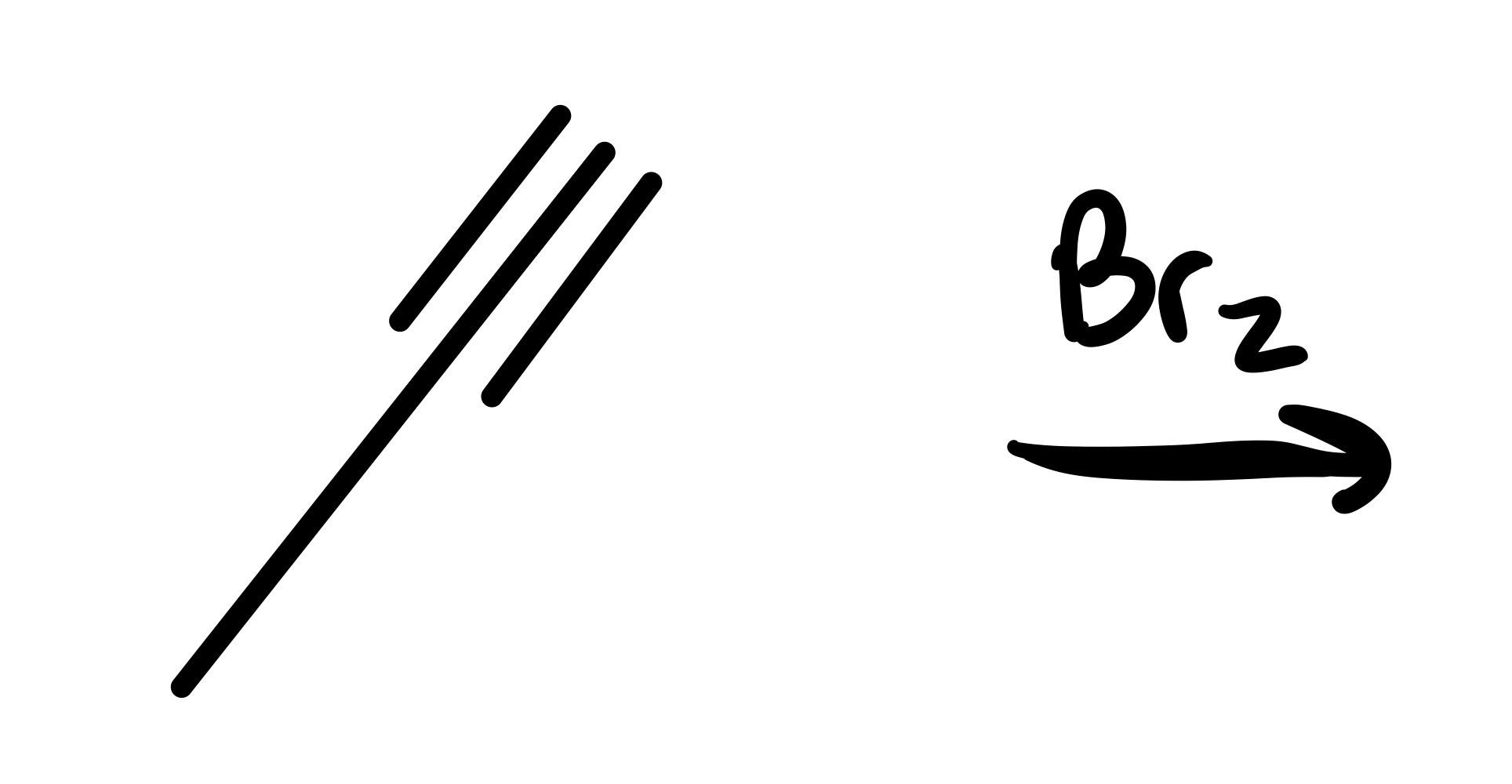
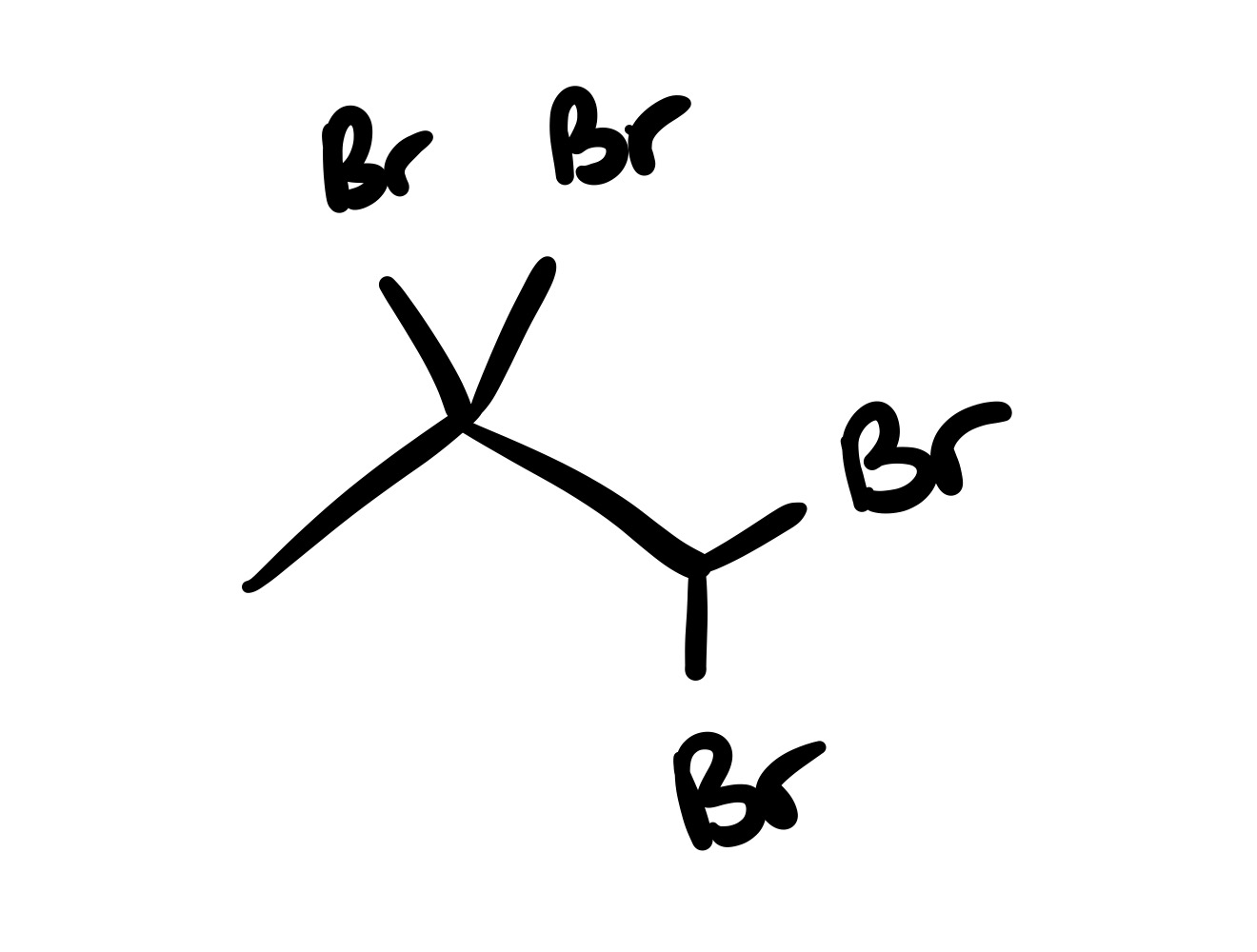
The triple bond is broken (only single bond carbon chain). And the 4 Br groups are added (two Br chains on one carbon)
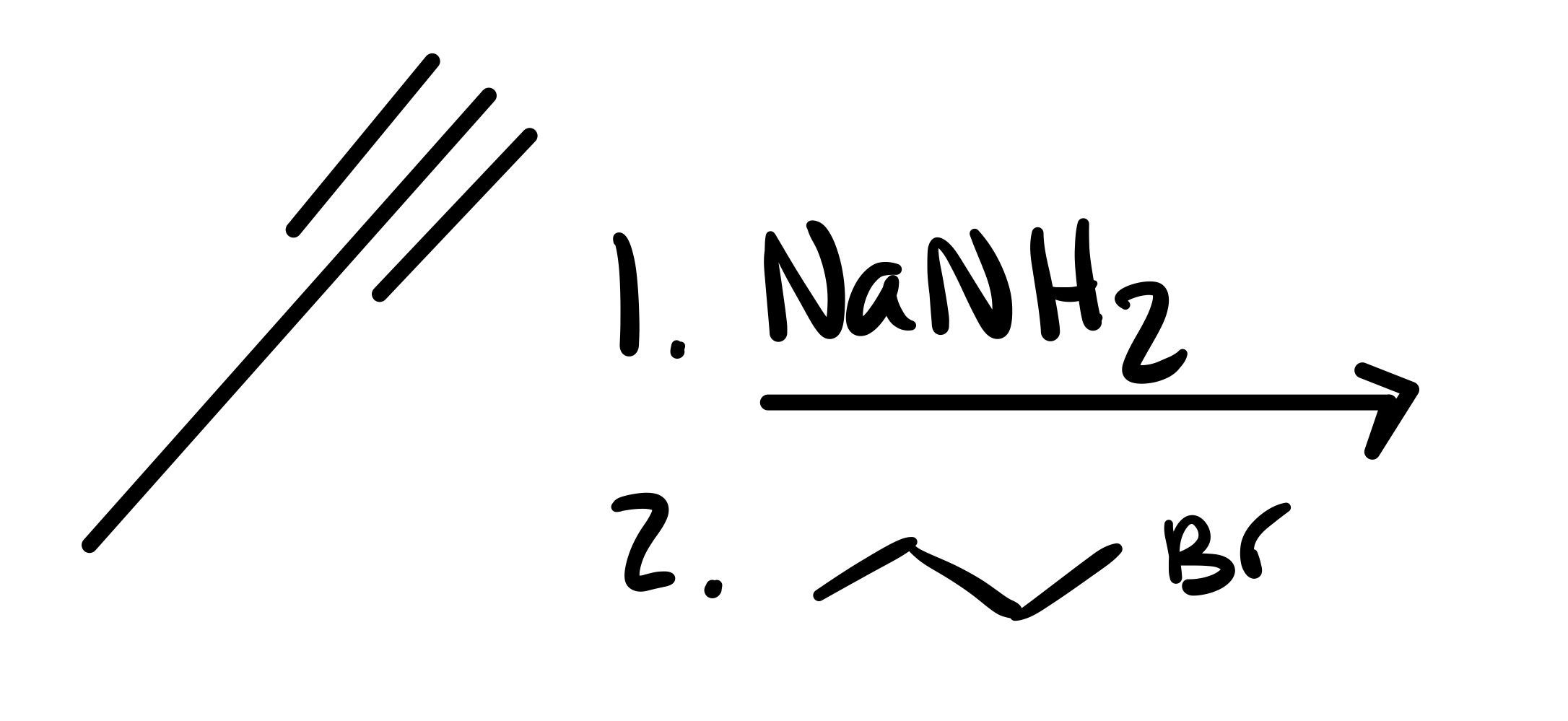
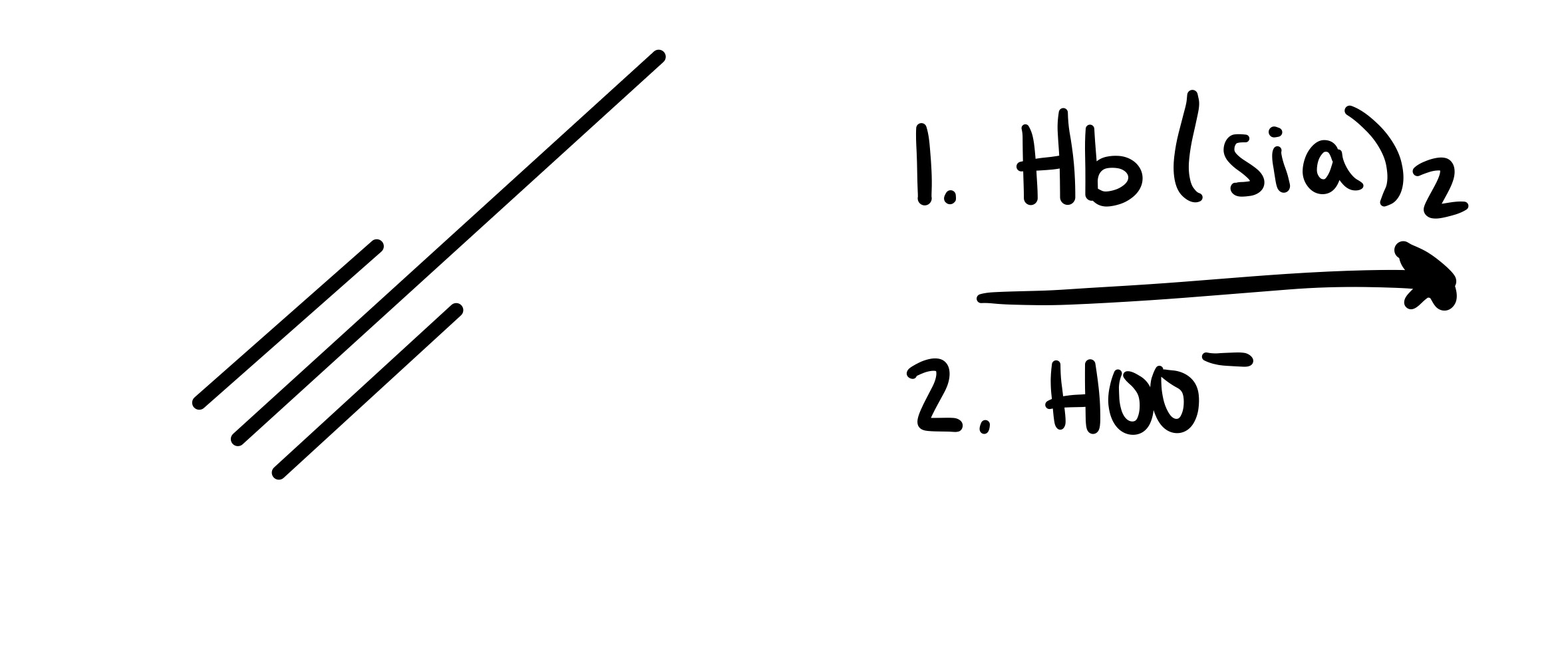
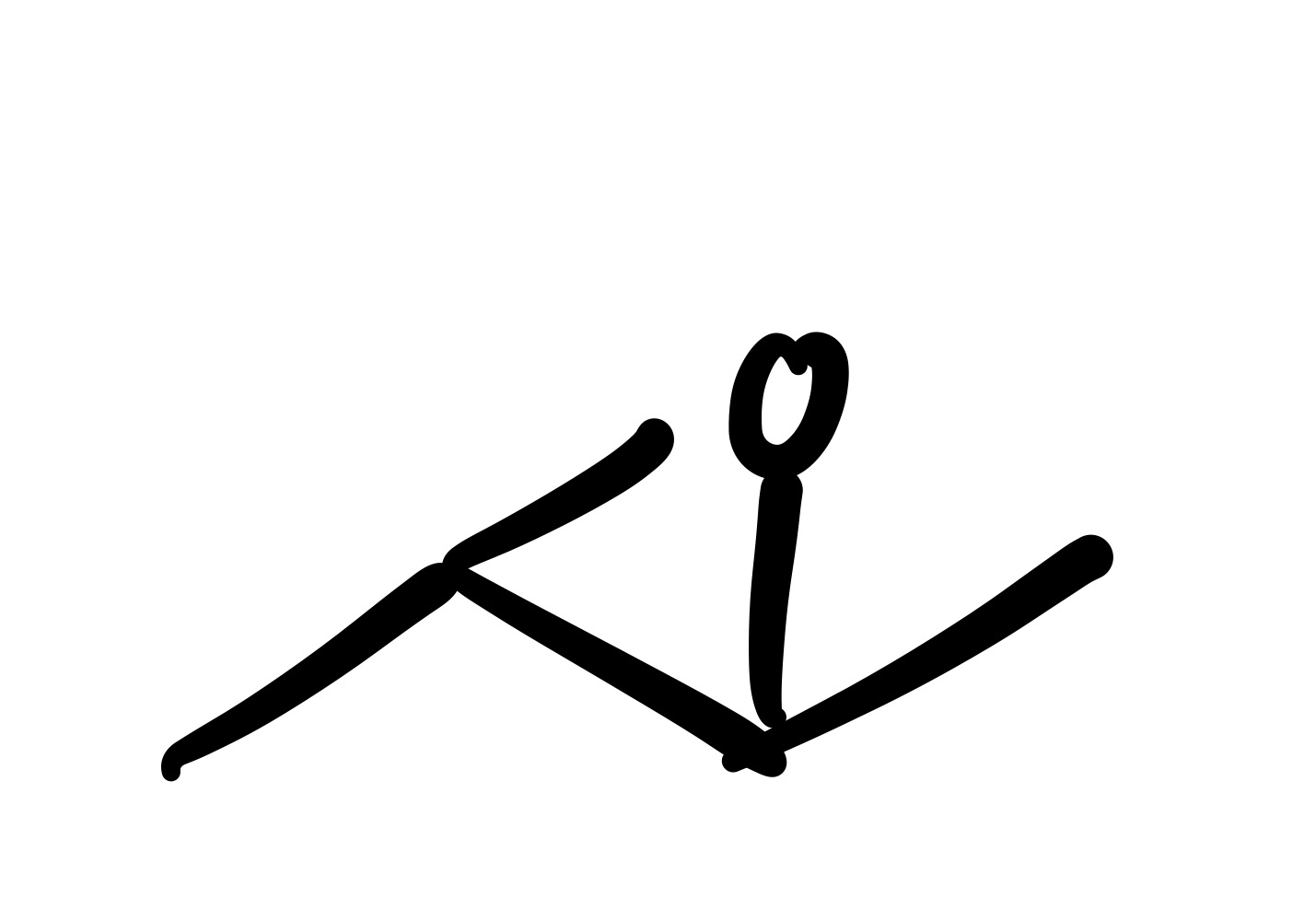
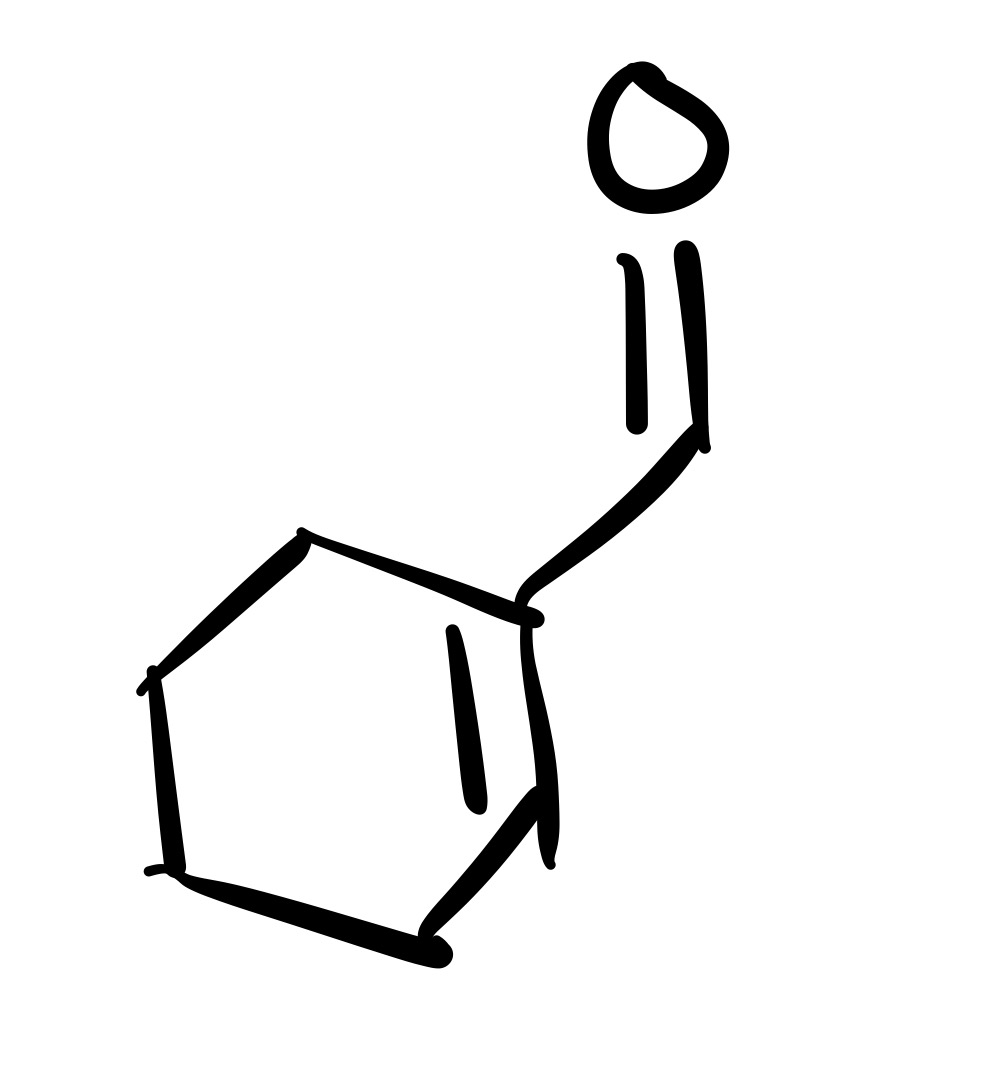
The triple bond is broken. The O is added as a double bond, and the H as a single bond on the carbon chain
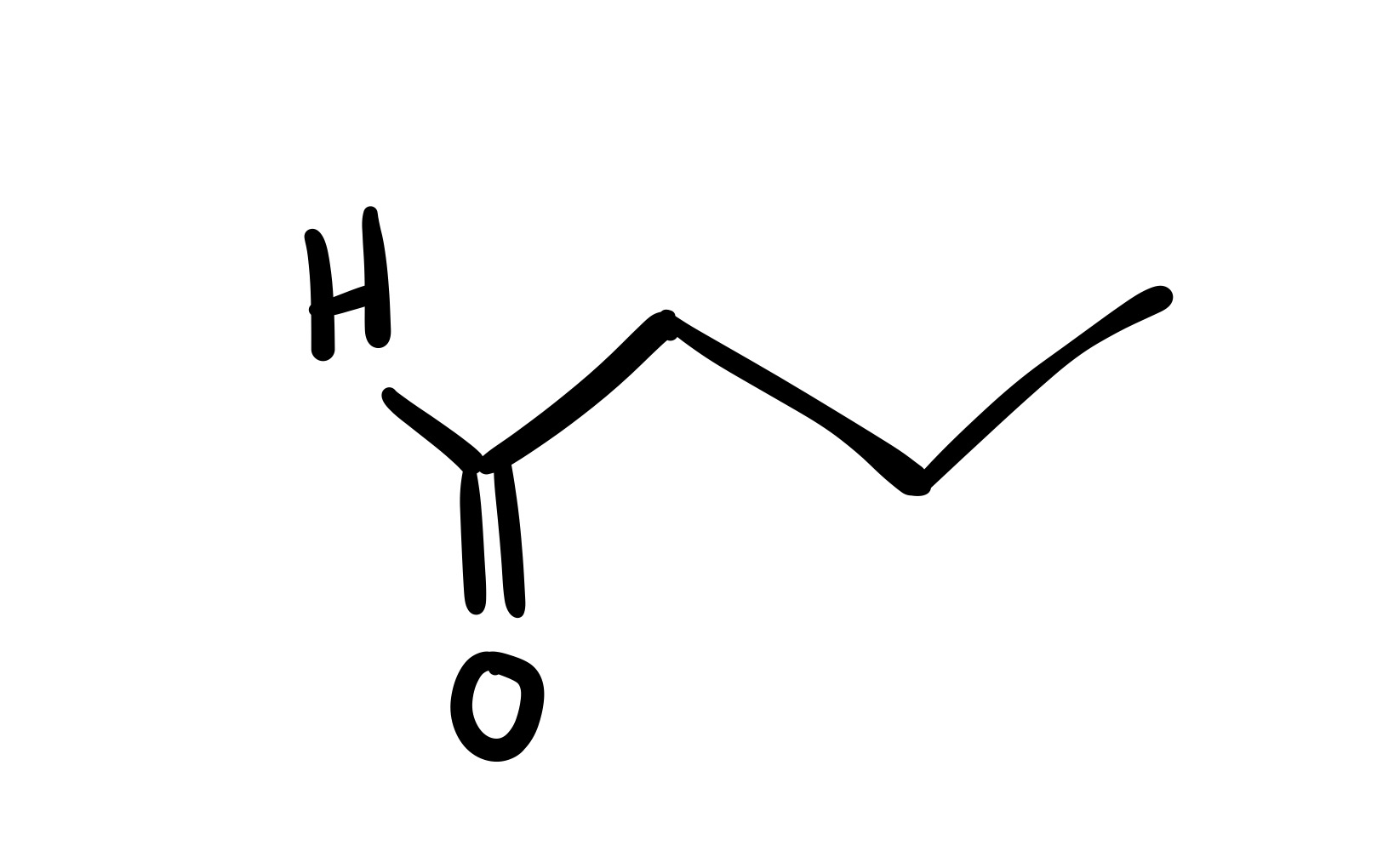
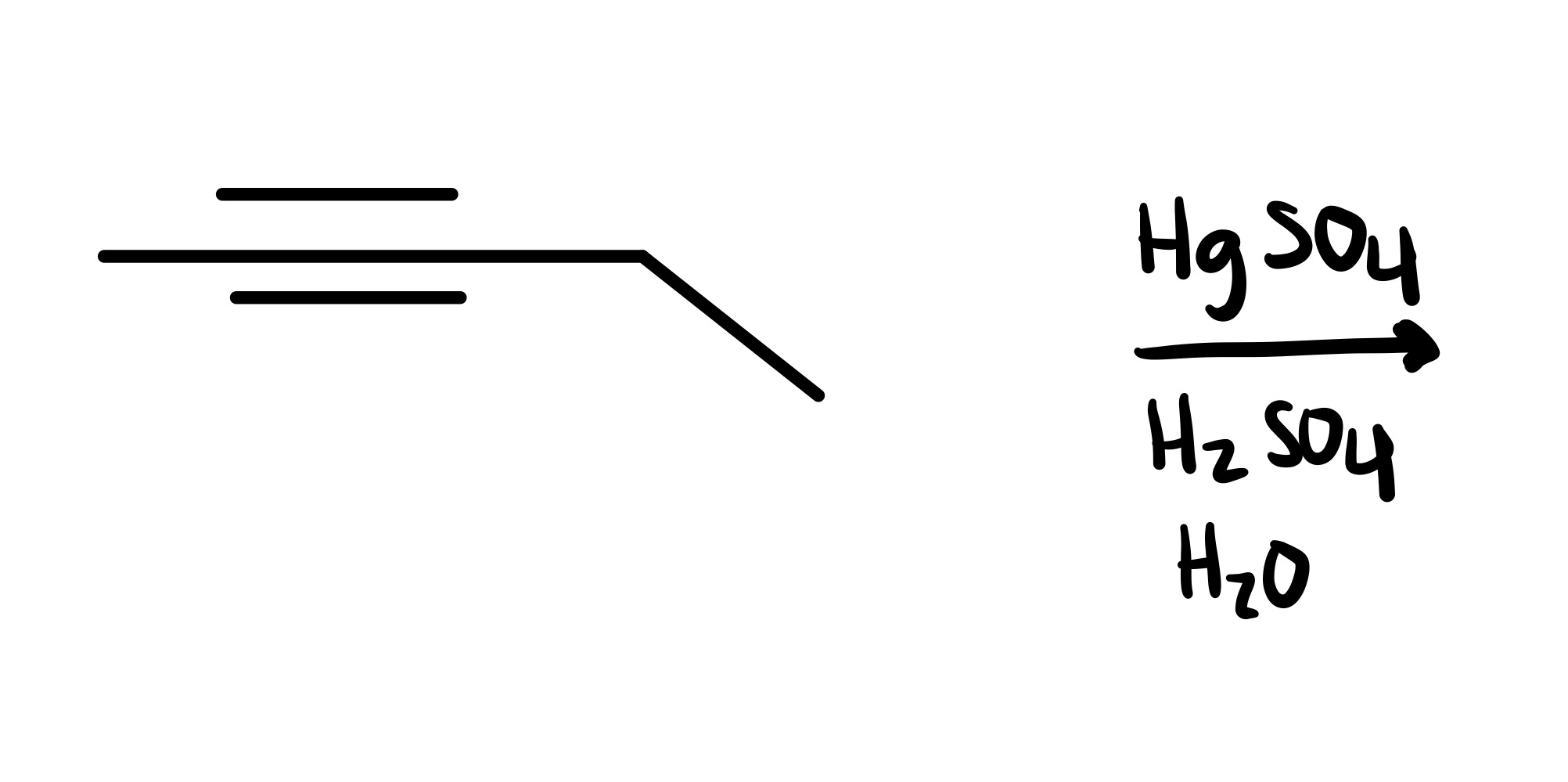
The triple bond is broken, and the O is added as a double bond on the carbon chain
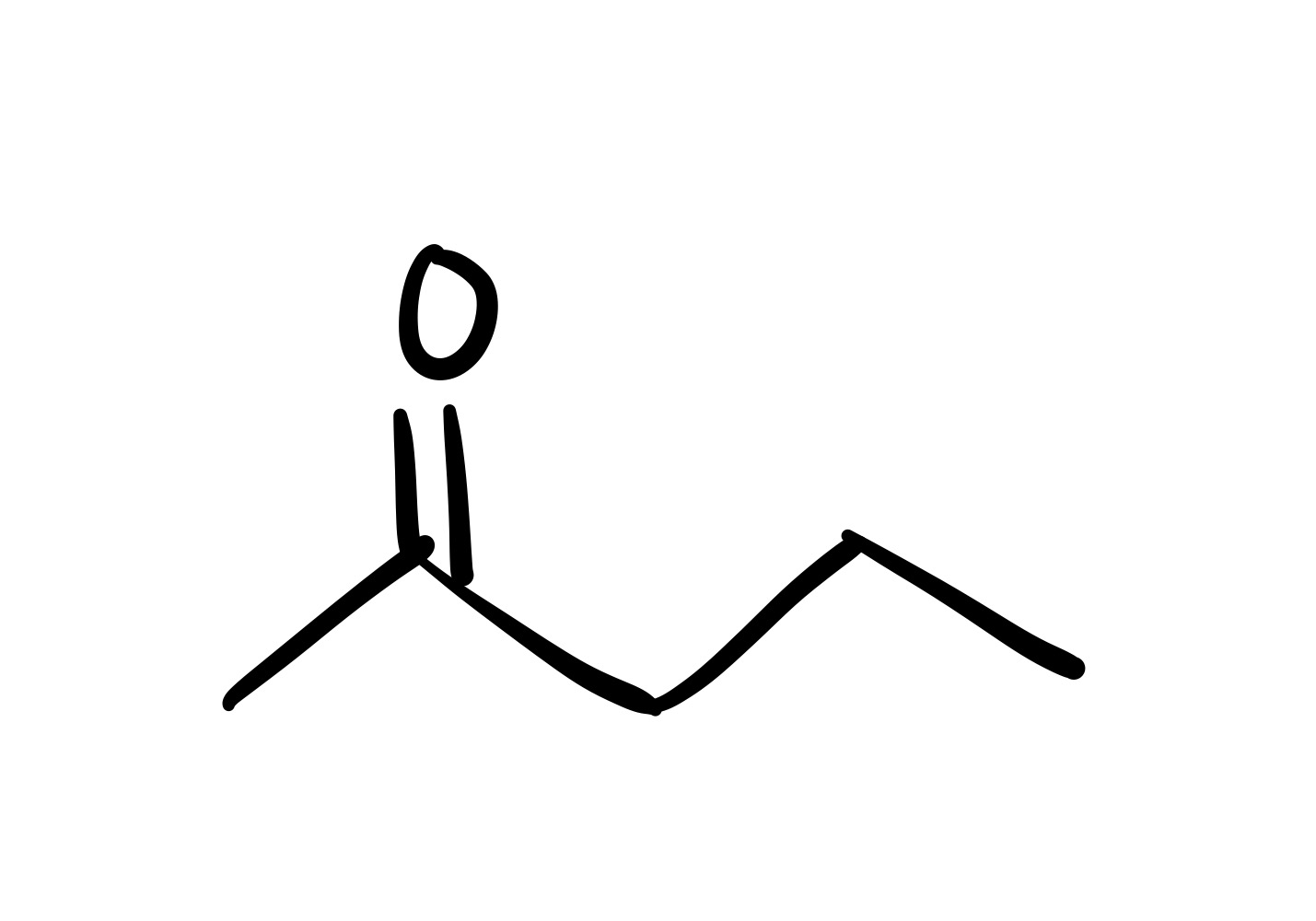
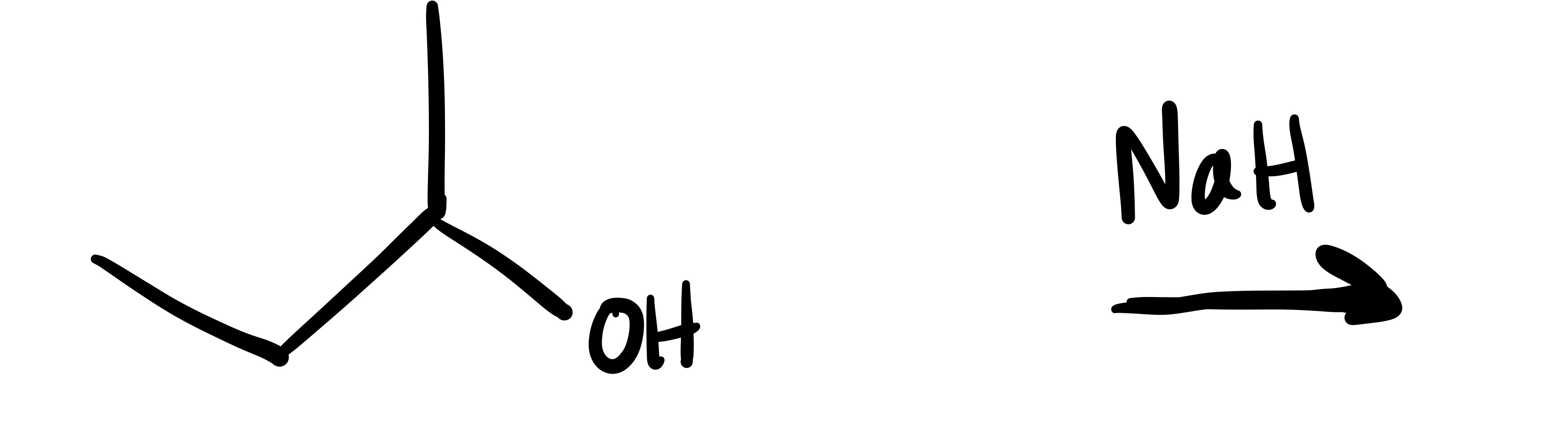
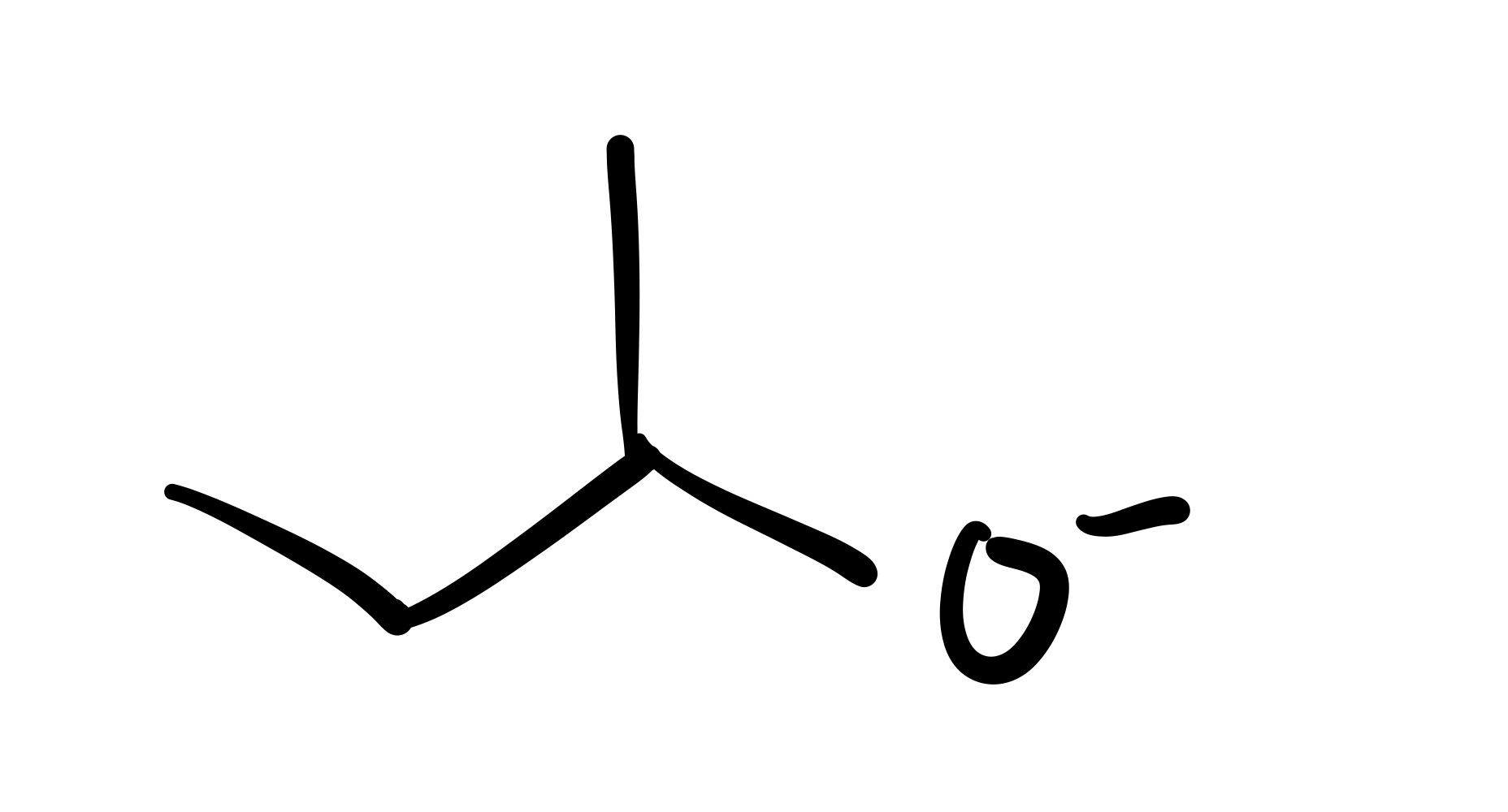
The H leaves the OH bonds (the O has a negative charge)
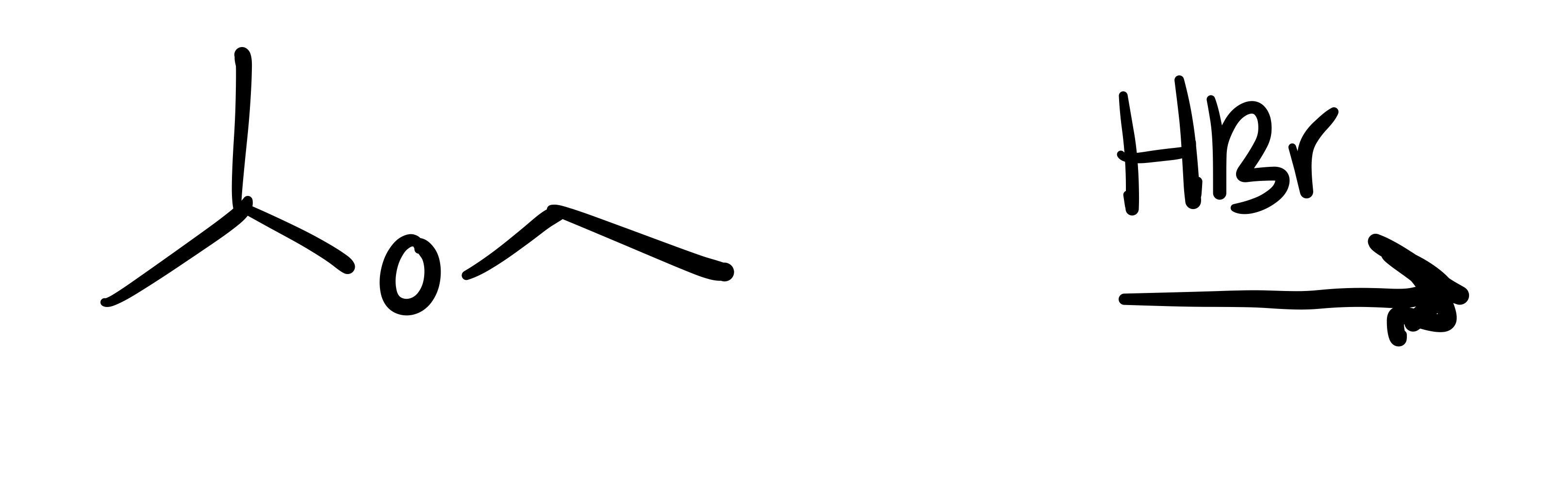
The O leaves the carbon chain and makes a new chain with an H attached.
Where the O leaves, the Br attaches
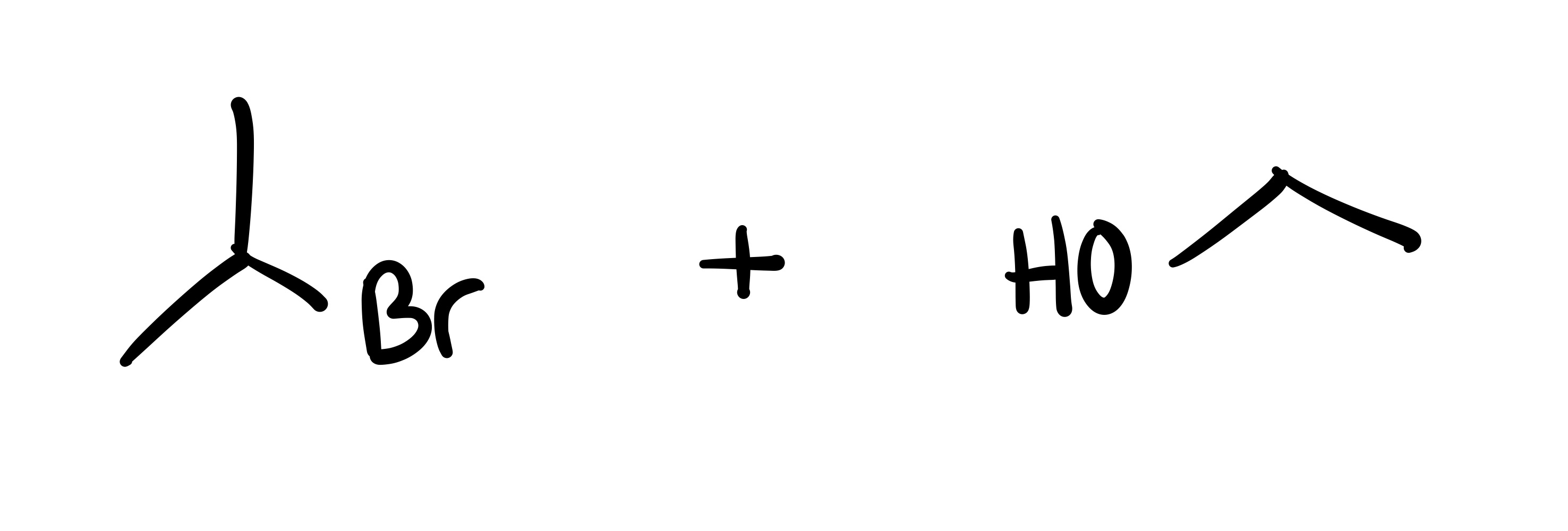
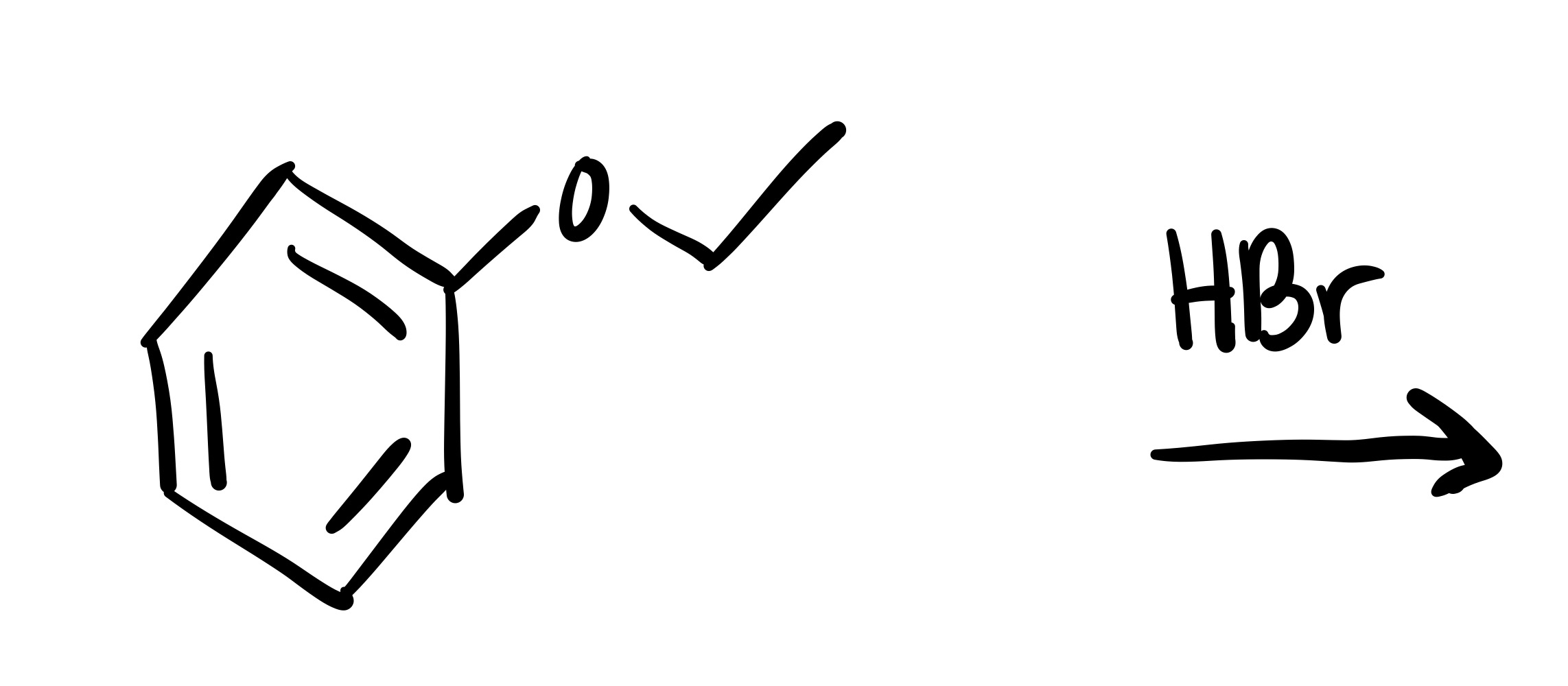
The carbon chain attached to the O leaves, and the Br adds to it
The H attaches to the O
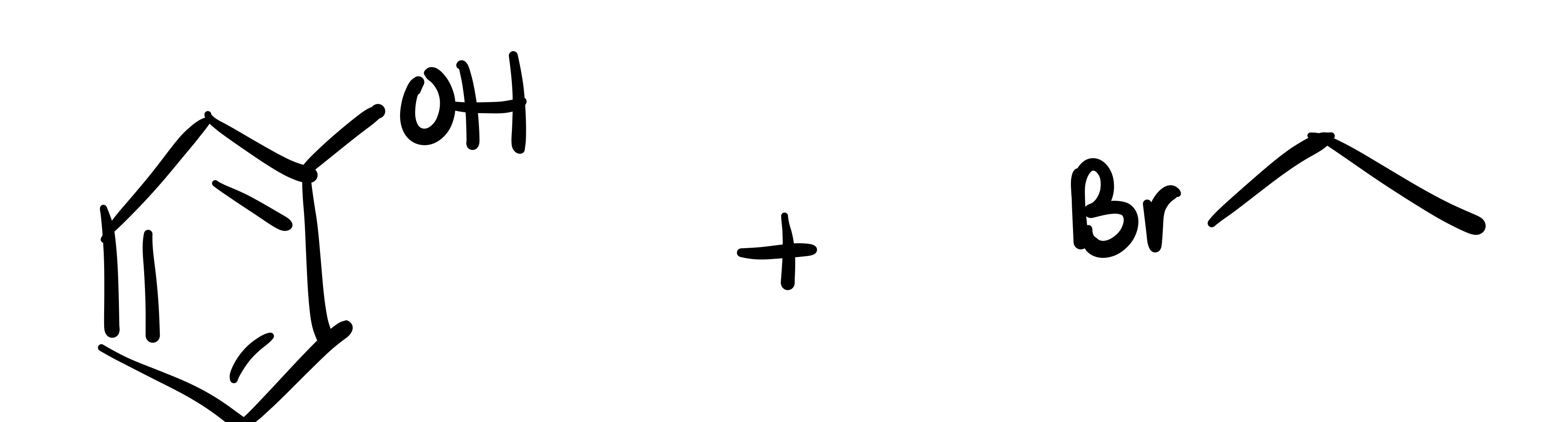
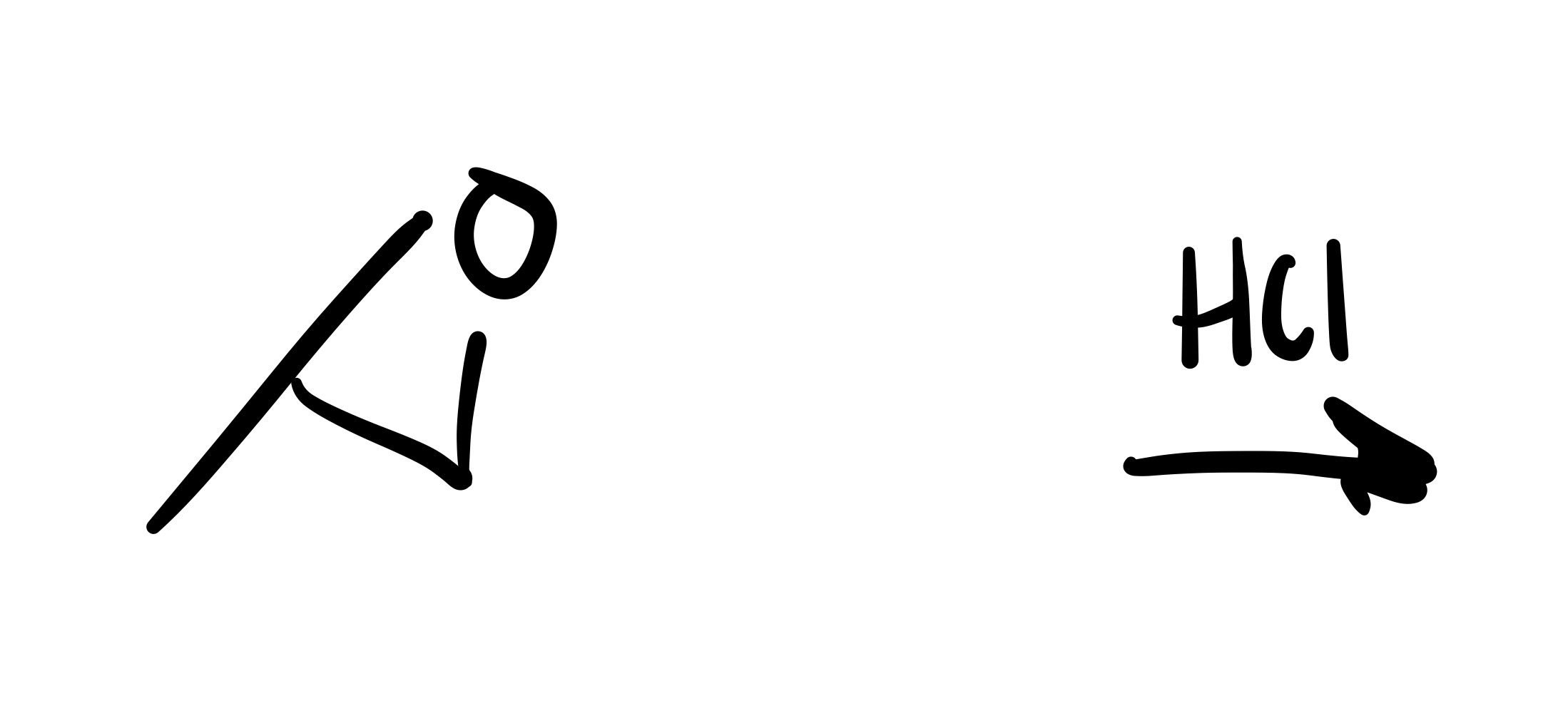
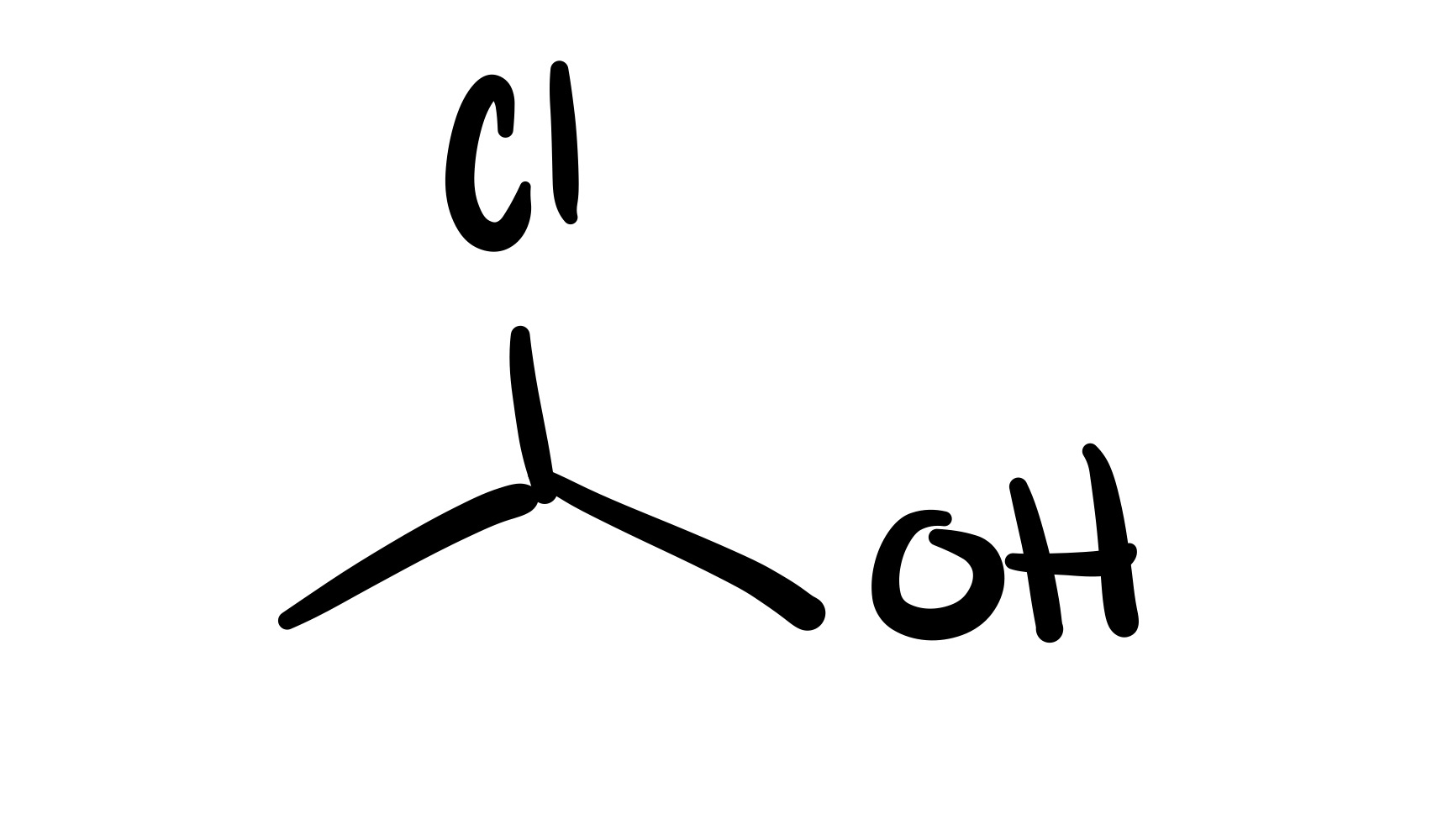
The epioxide breaks, and an OH and Cl attaches to the carbon chain
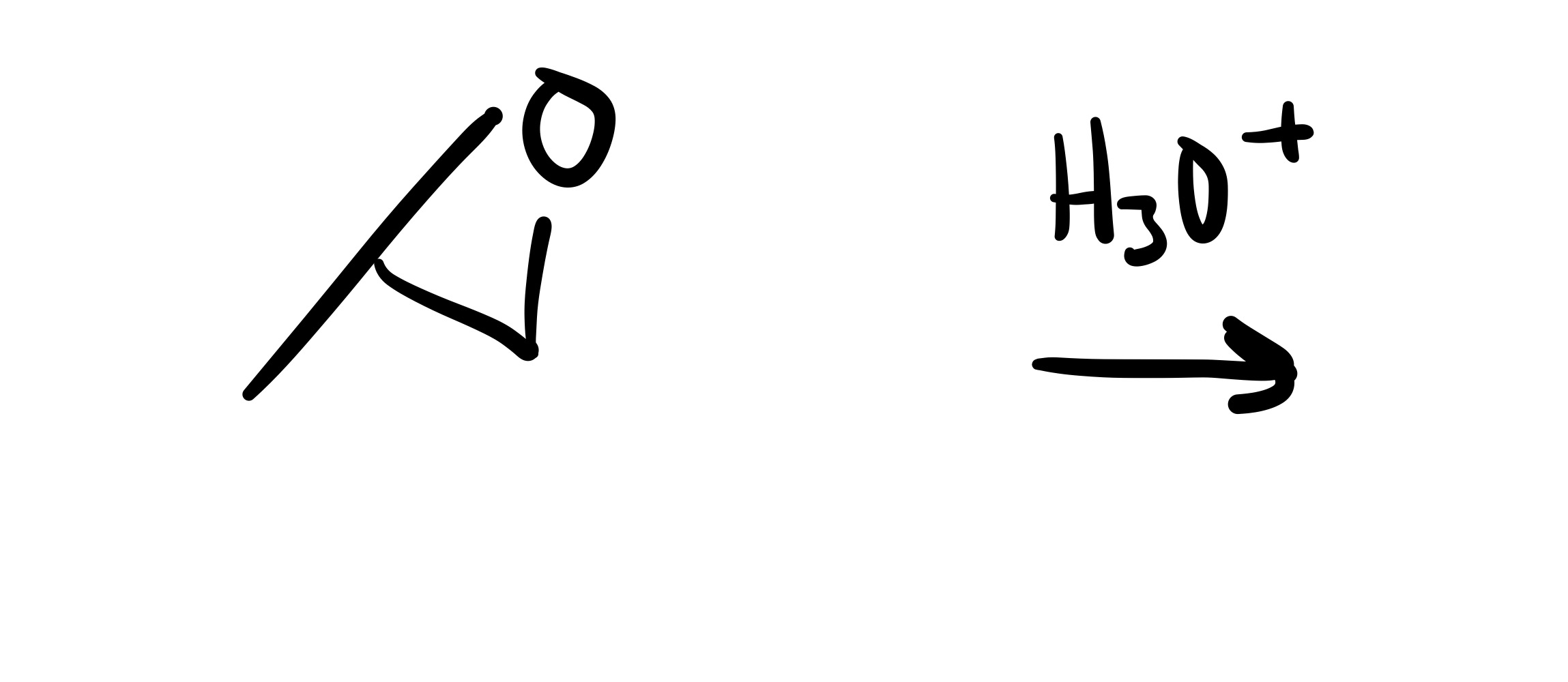
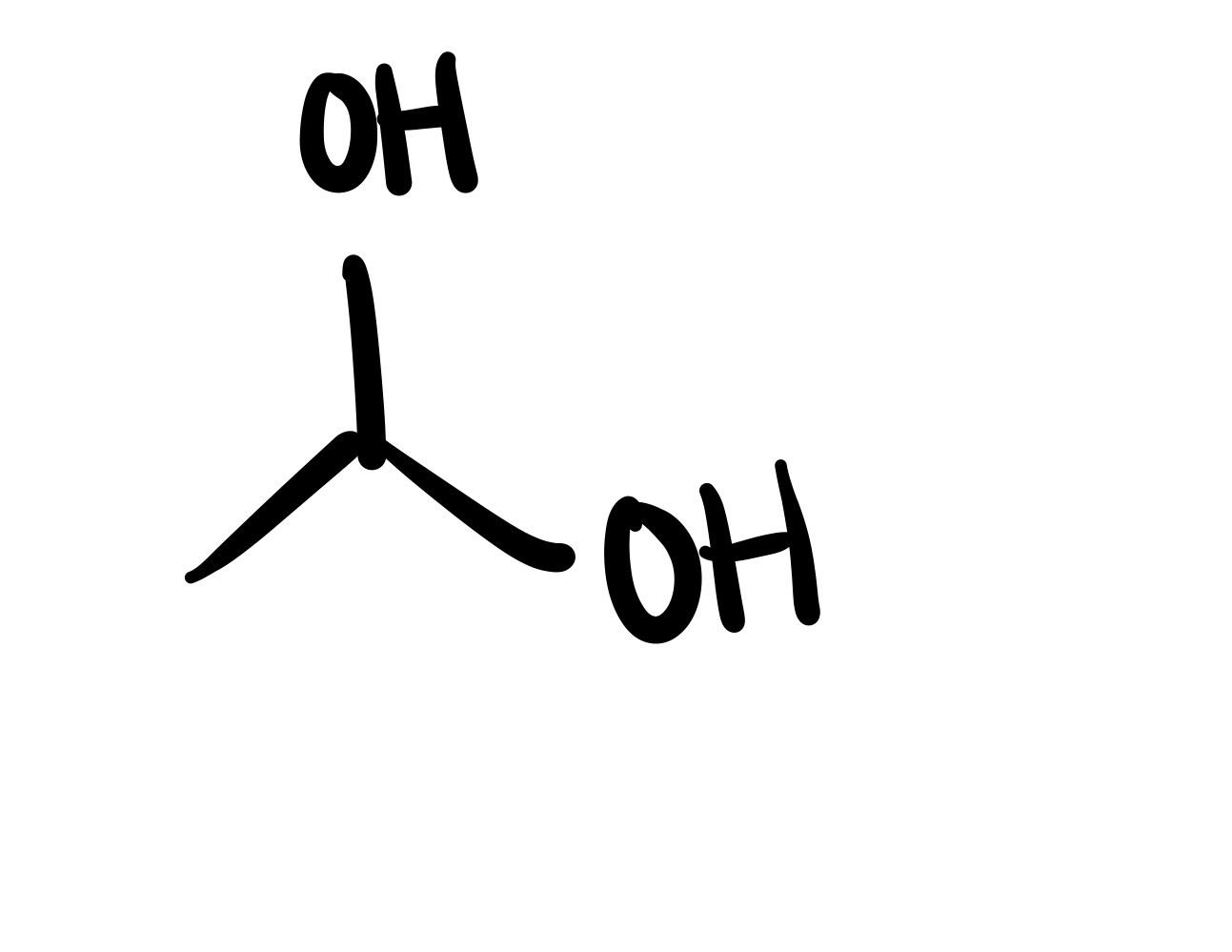
The epioxide breaks, and the 2 OH’s attach to the carbon chain (on separate carbons)
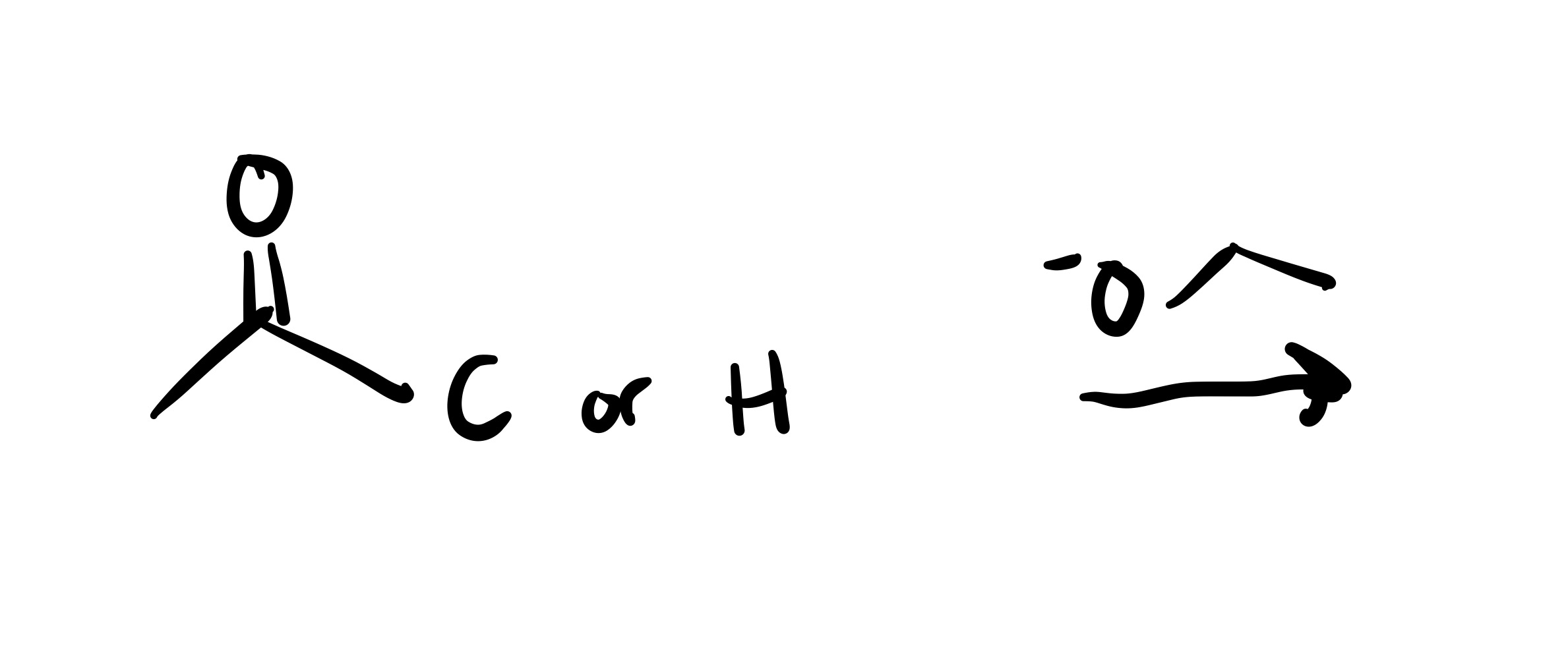
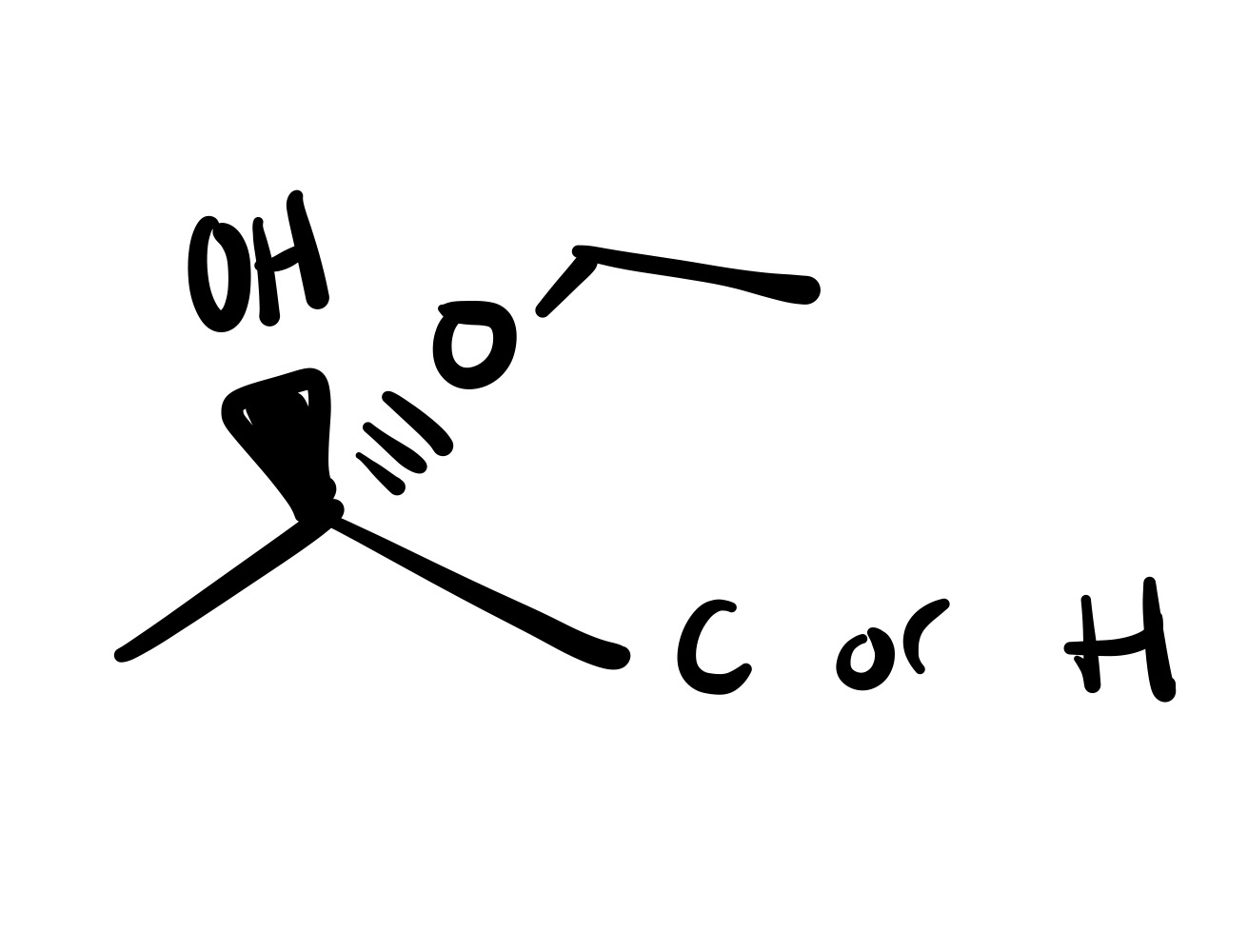
The double bond is broken, and the H attaches to the O (on a wedge). The O carbon chain attaches on the same carbon (as a dash)
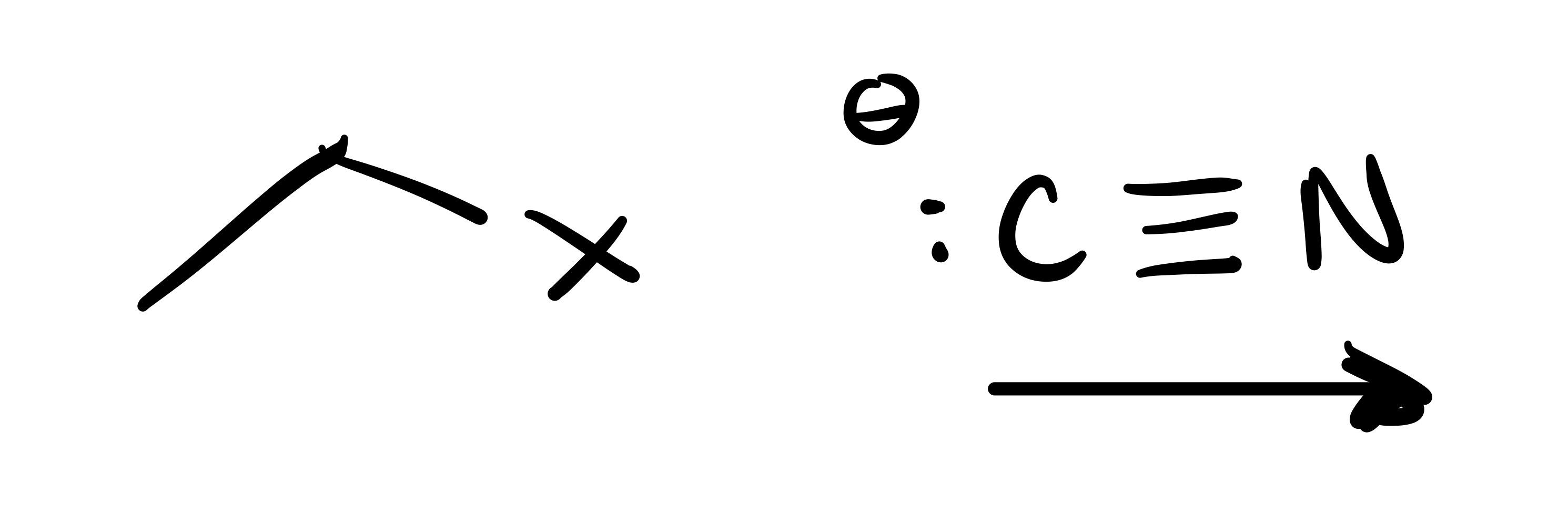
The leave in group leaves the carbon chain, and the bullet structure attaches
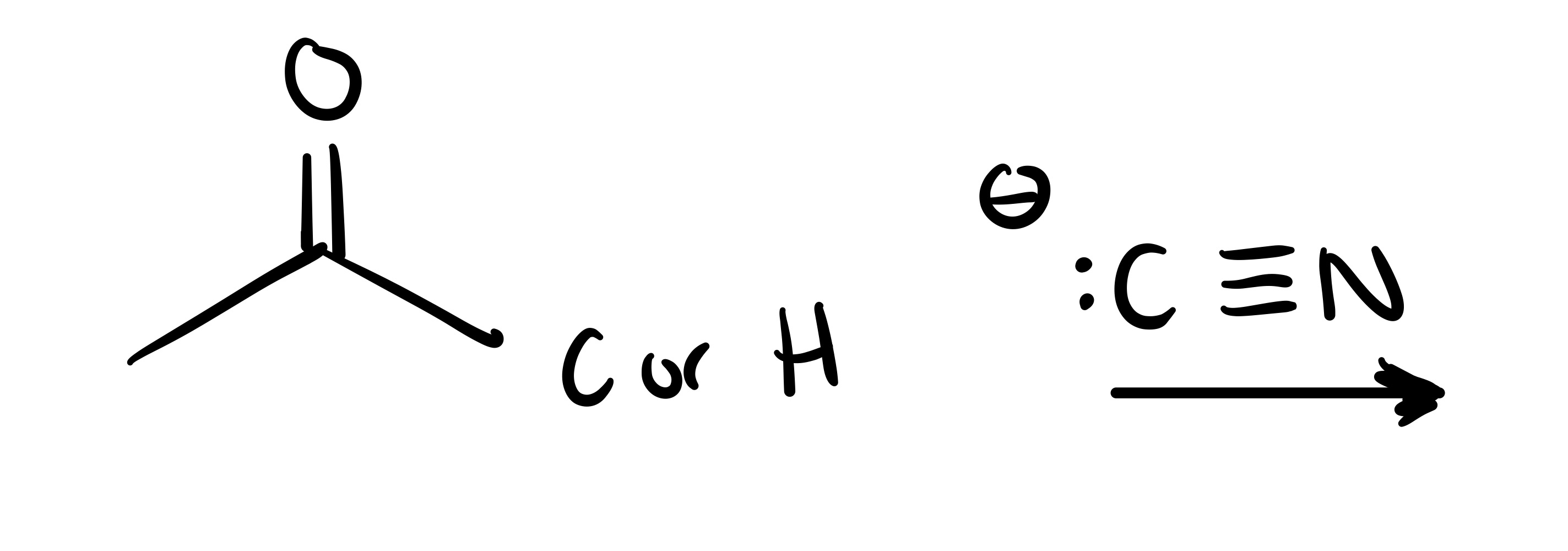
The double bond O breaks, and the H attaches to the O. On the same carbon the bullet shape attaches
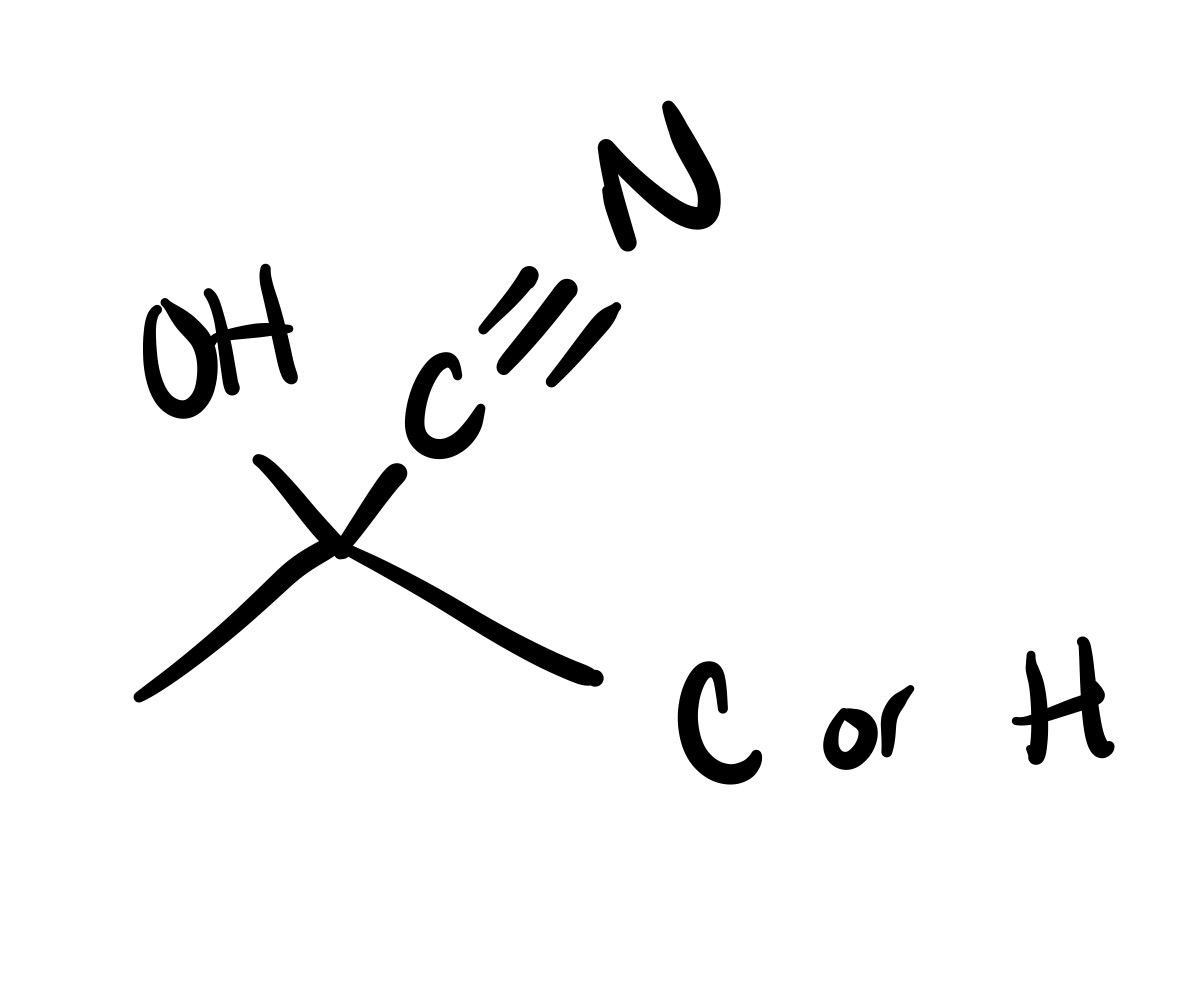
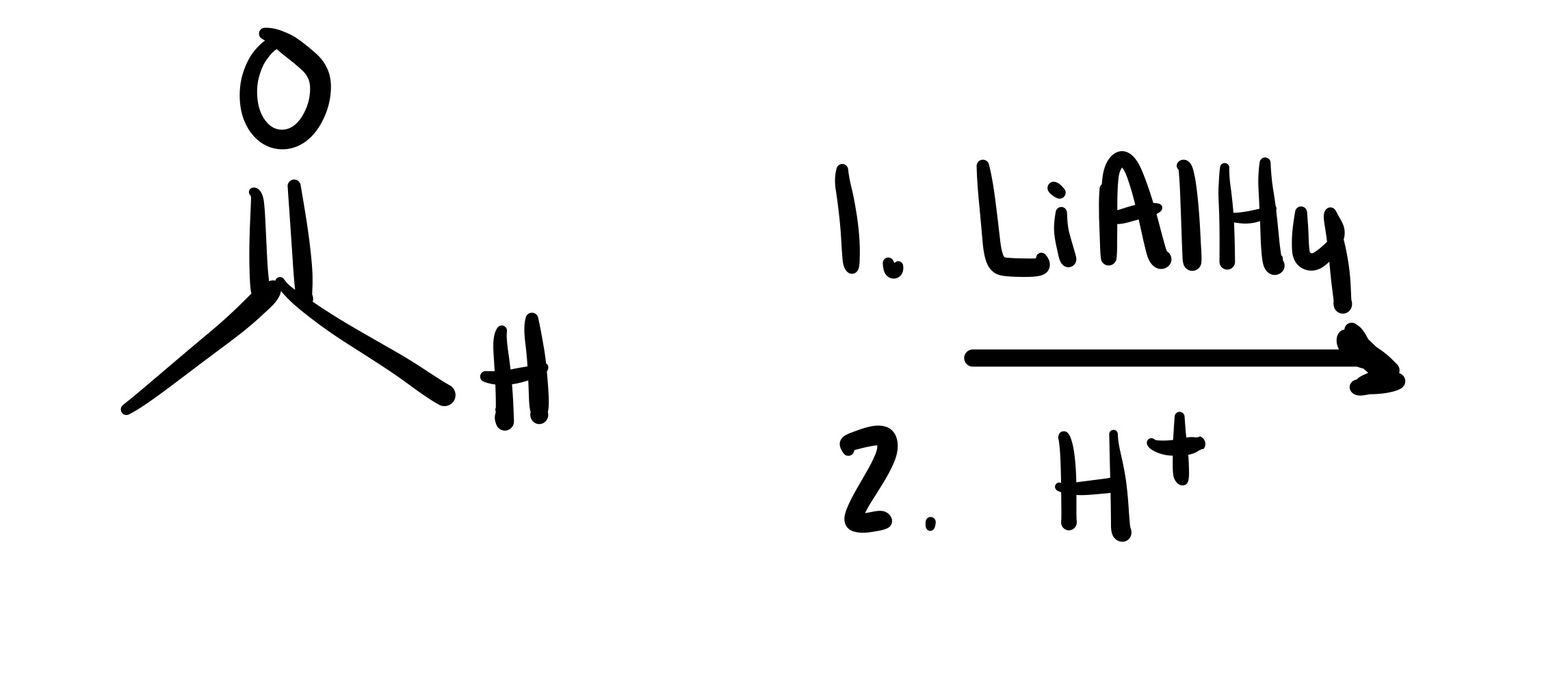
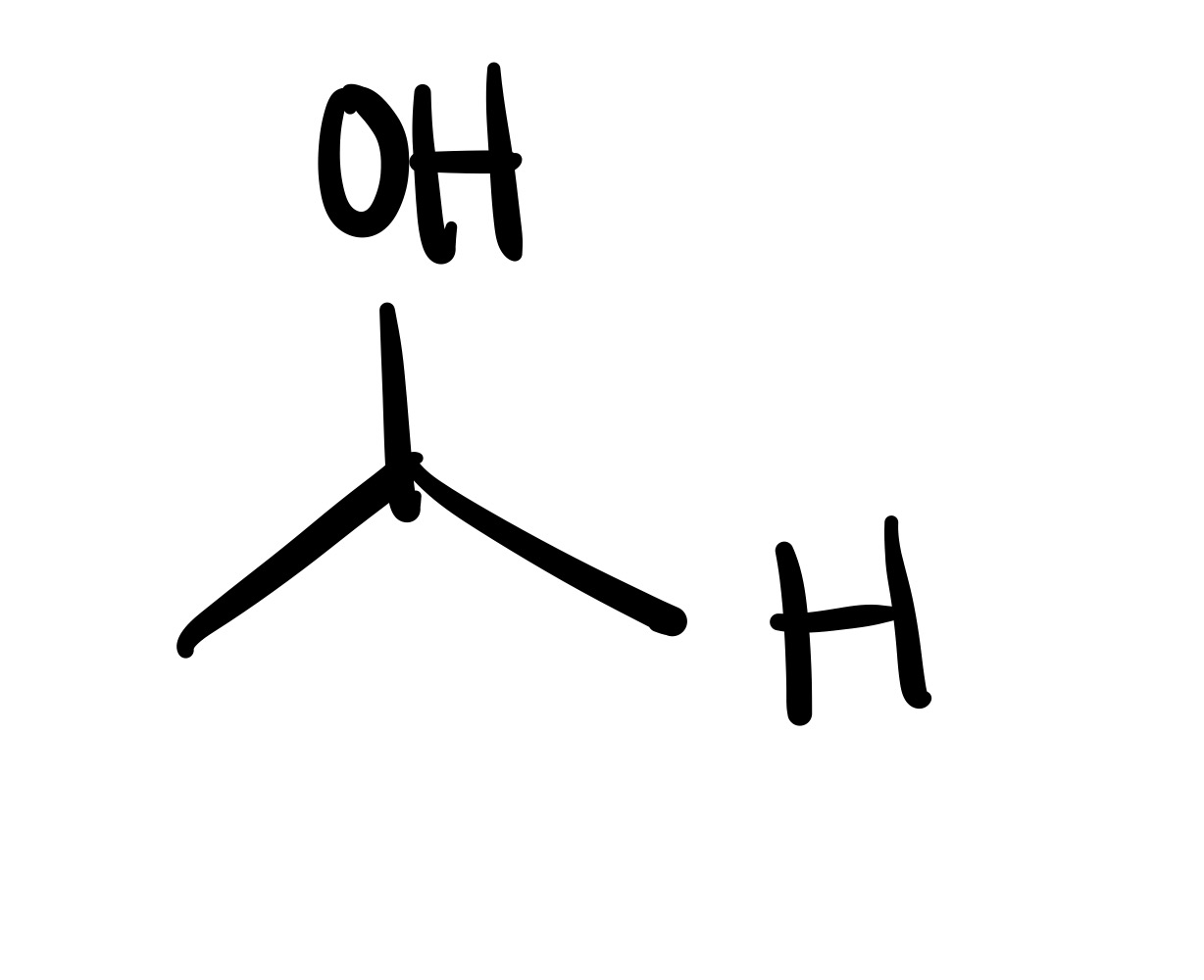
The double O bond breaks, and an H attaches to the O
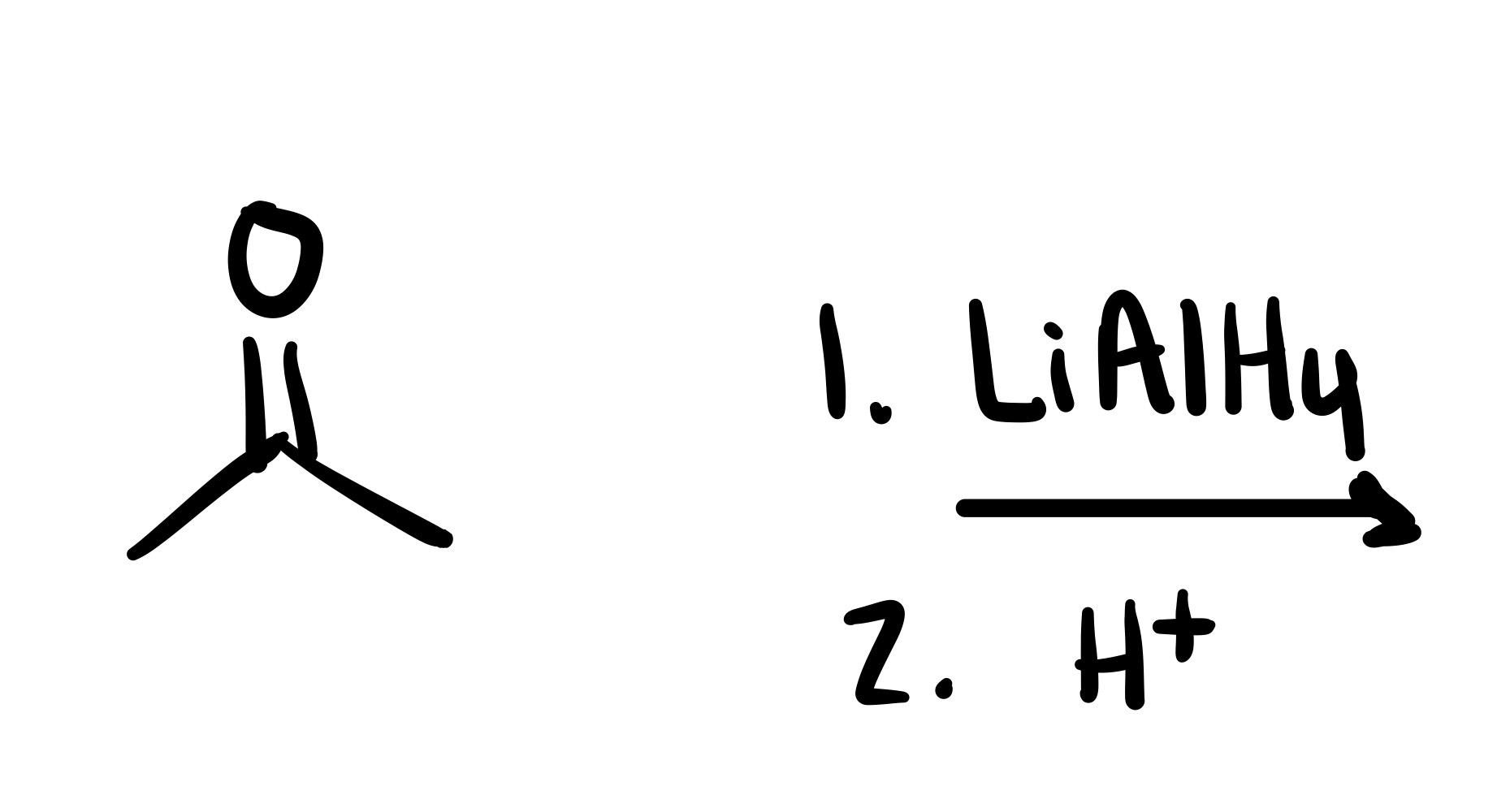
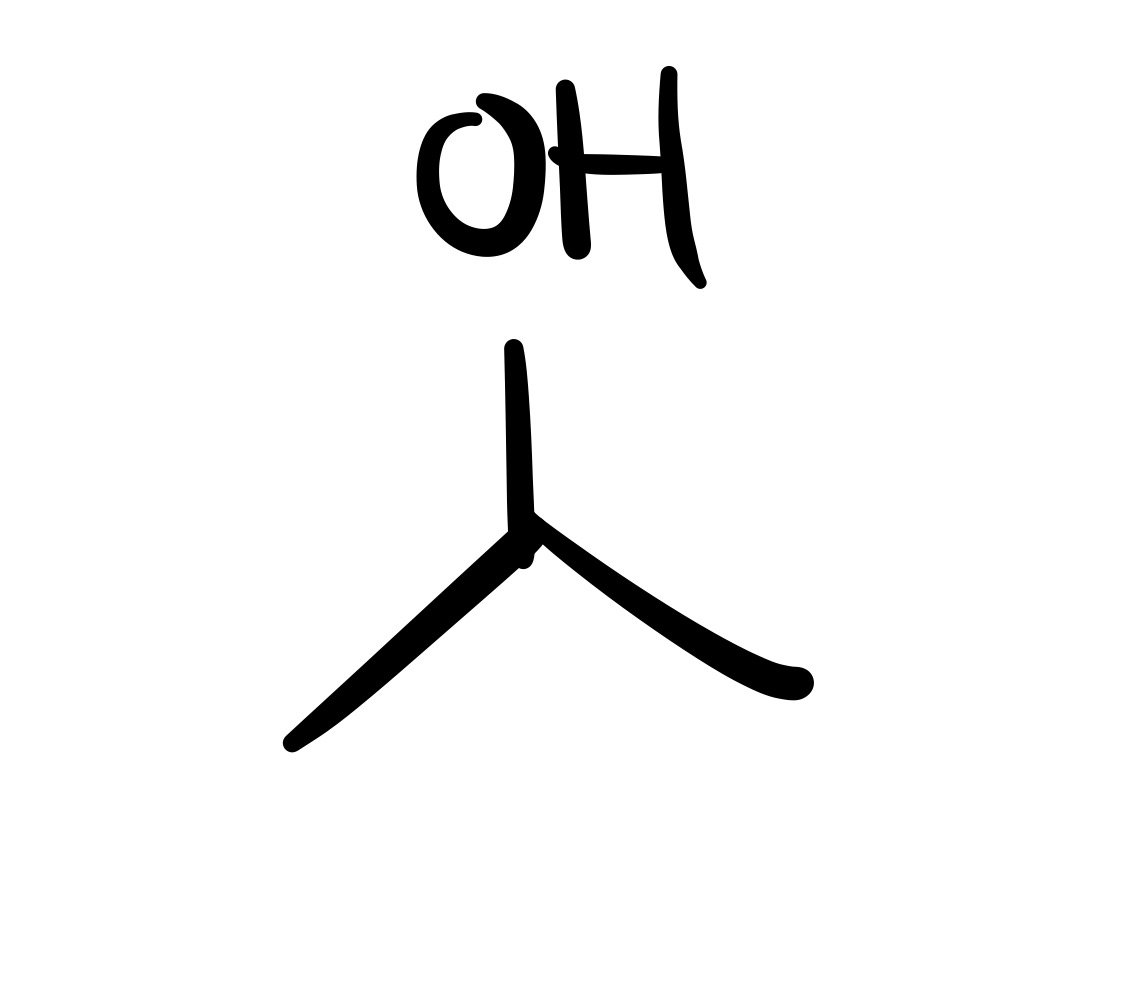
The double O bond breaks, and an H attaches to the O
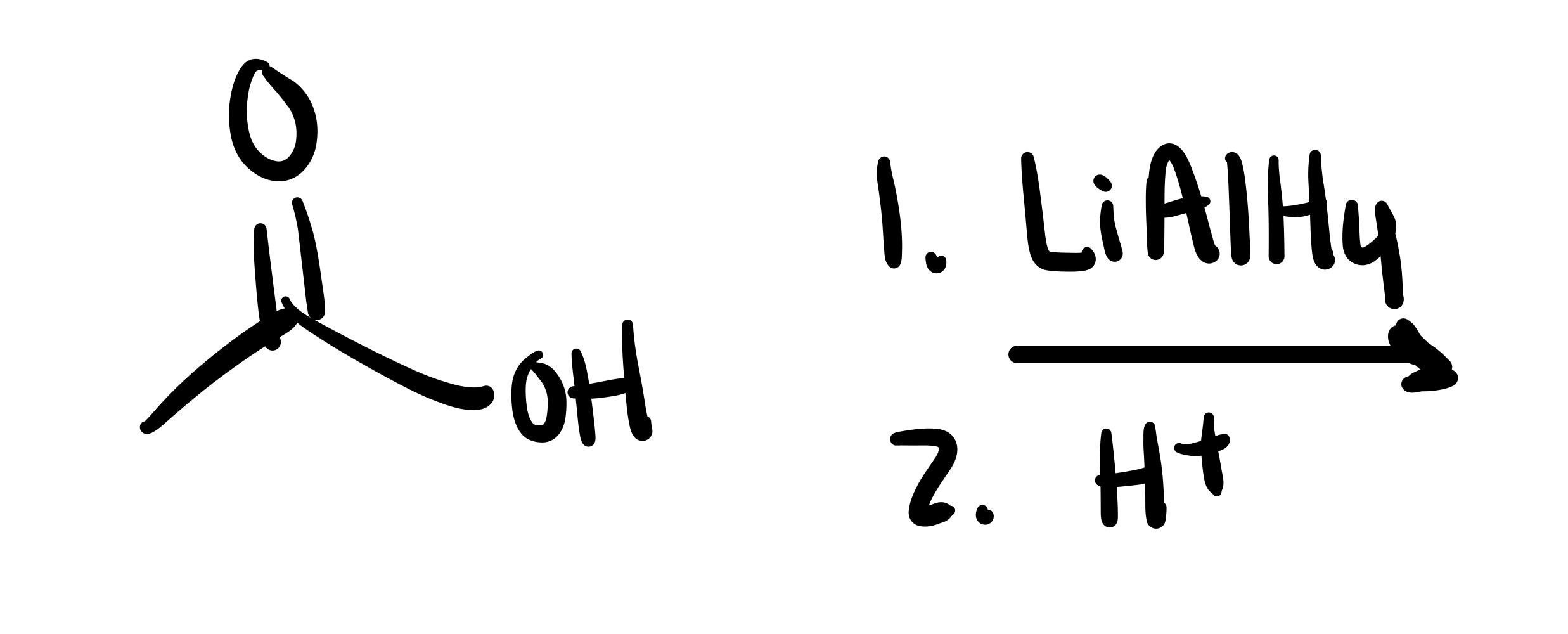
The double O bond breaks, and an H attaches to the O. On the OH, the O leaves (only an h remains)
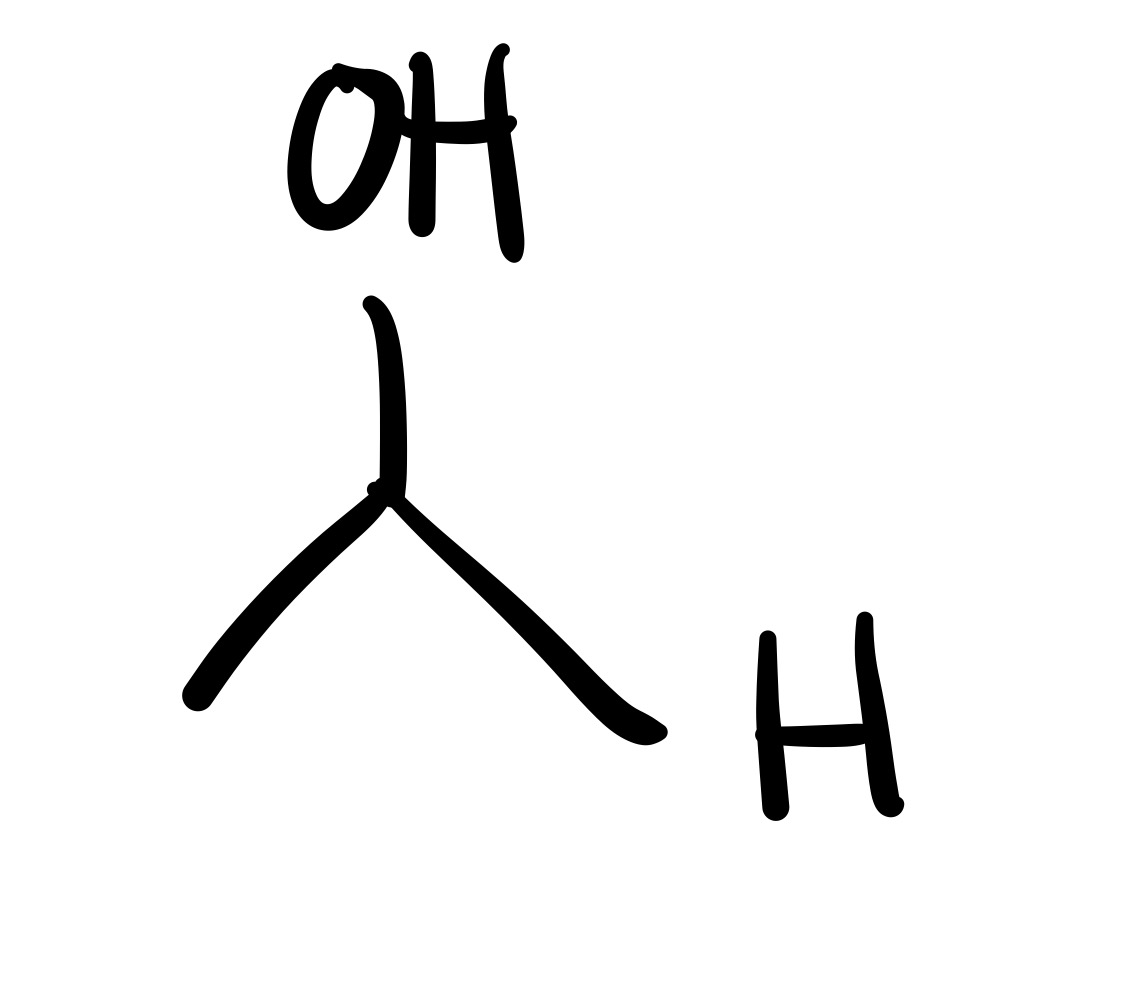
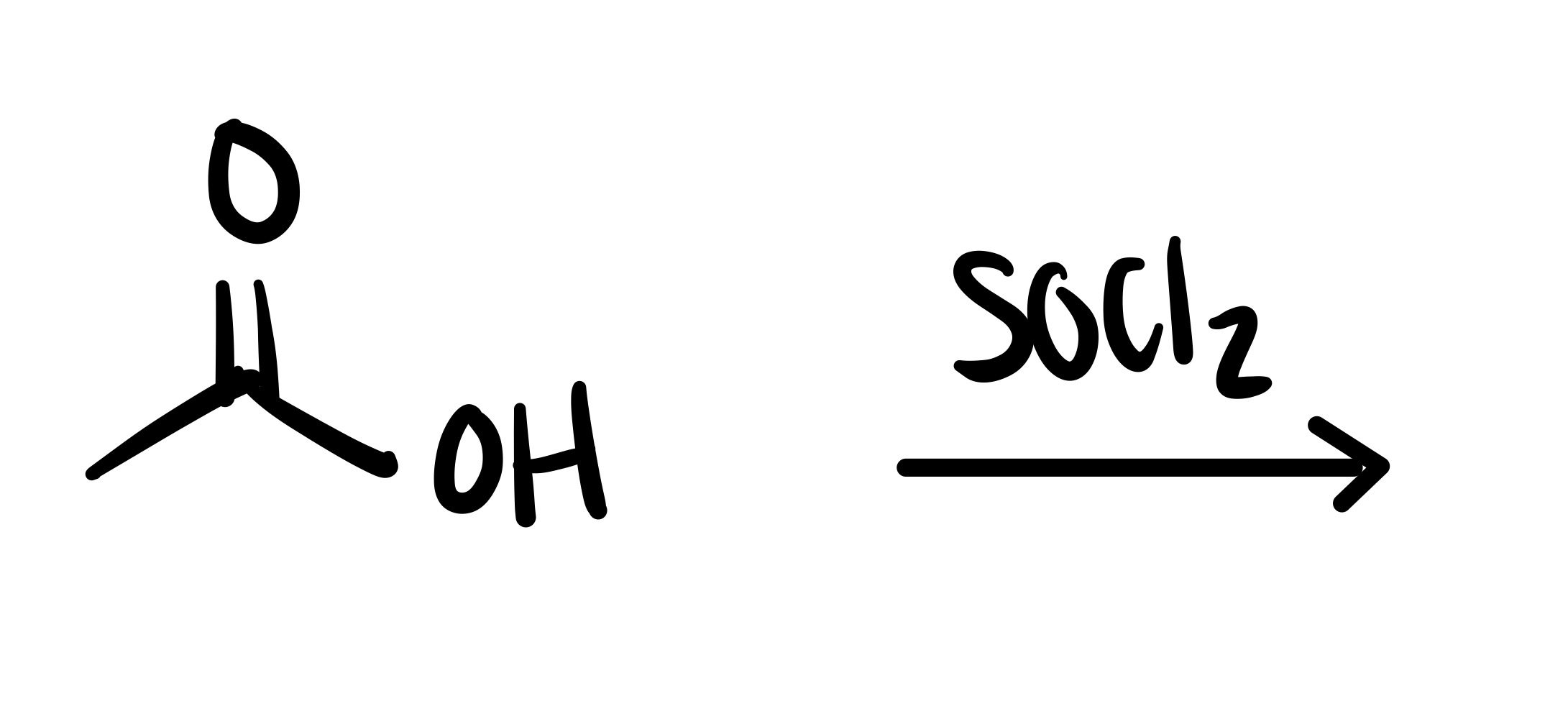
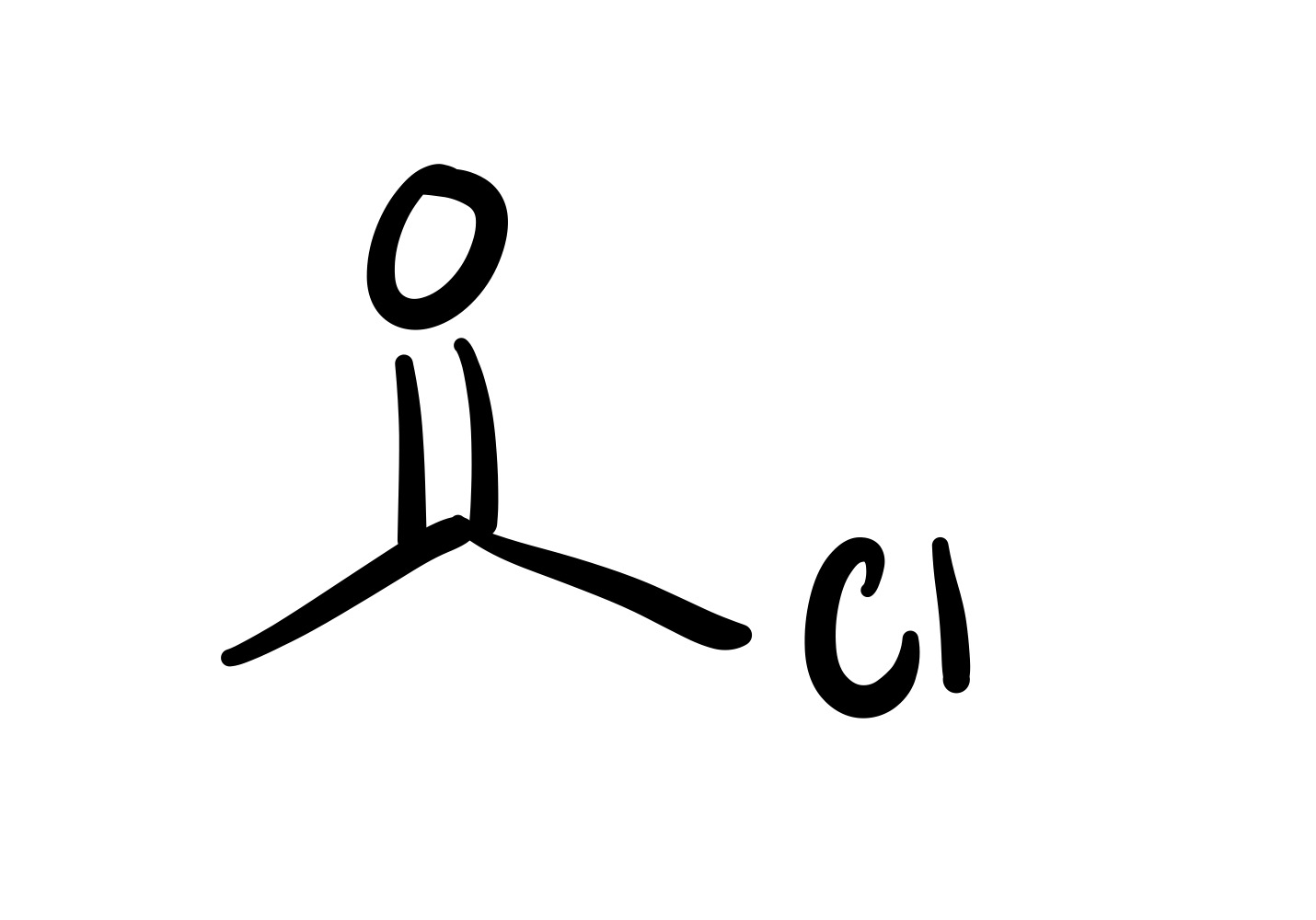
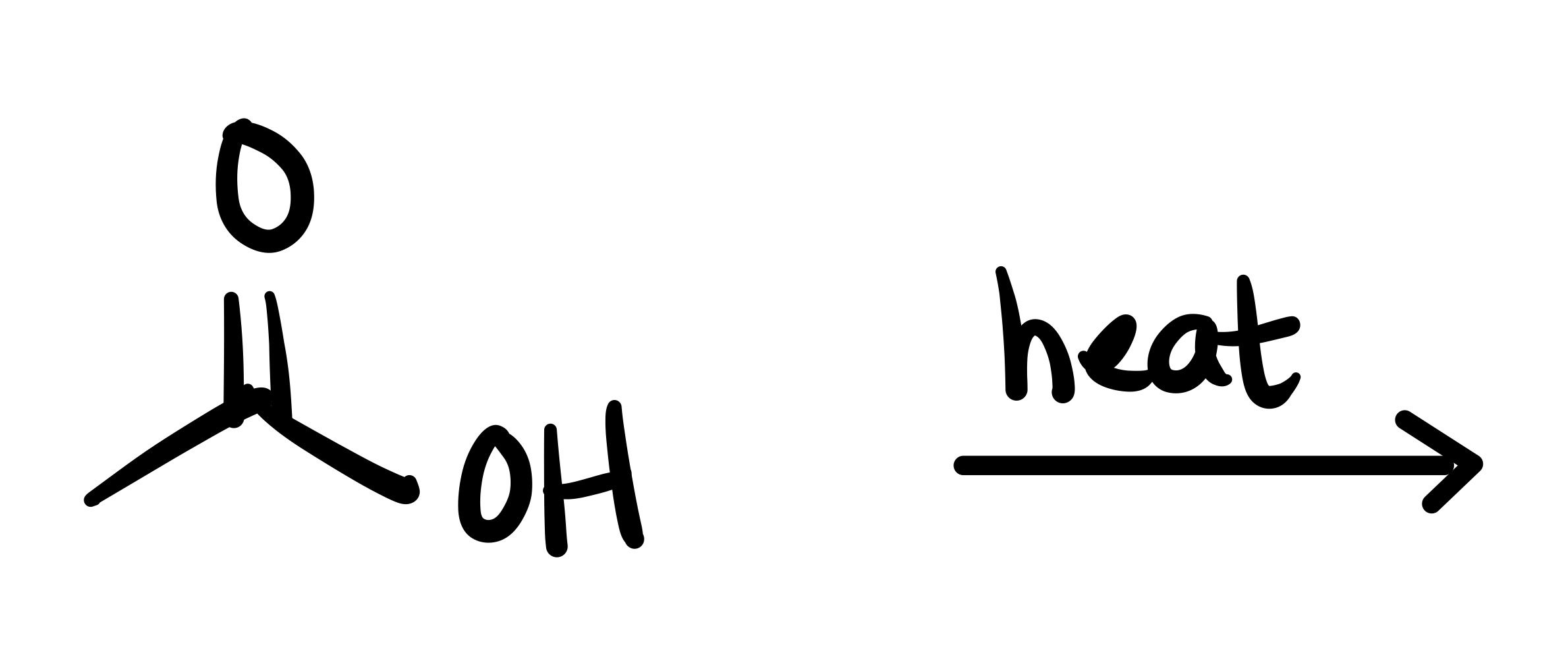
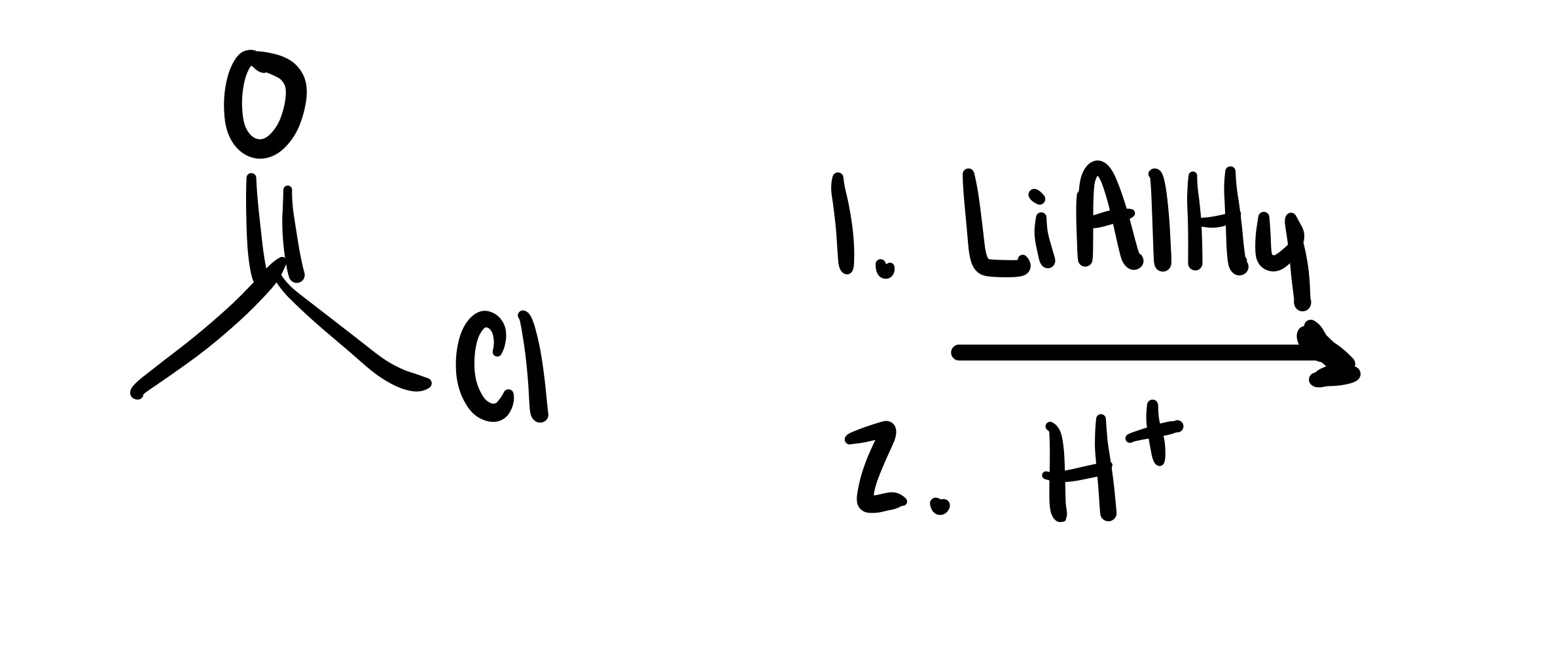
The double O bond breaks, and an H attaches to the O. On the OH, the O leaves (only an h remains)
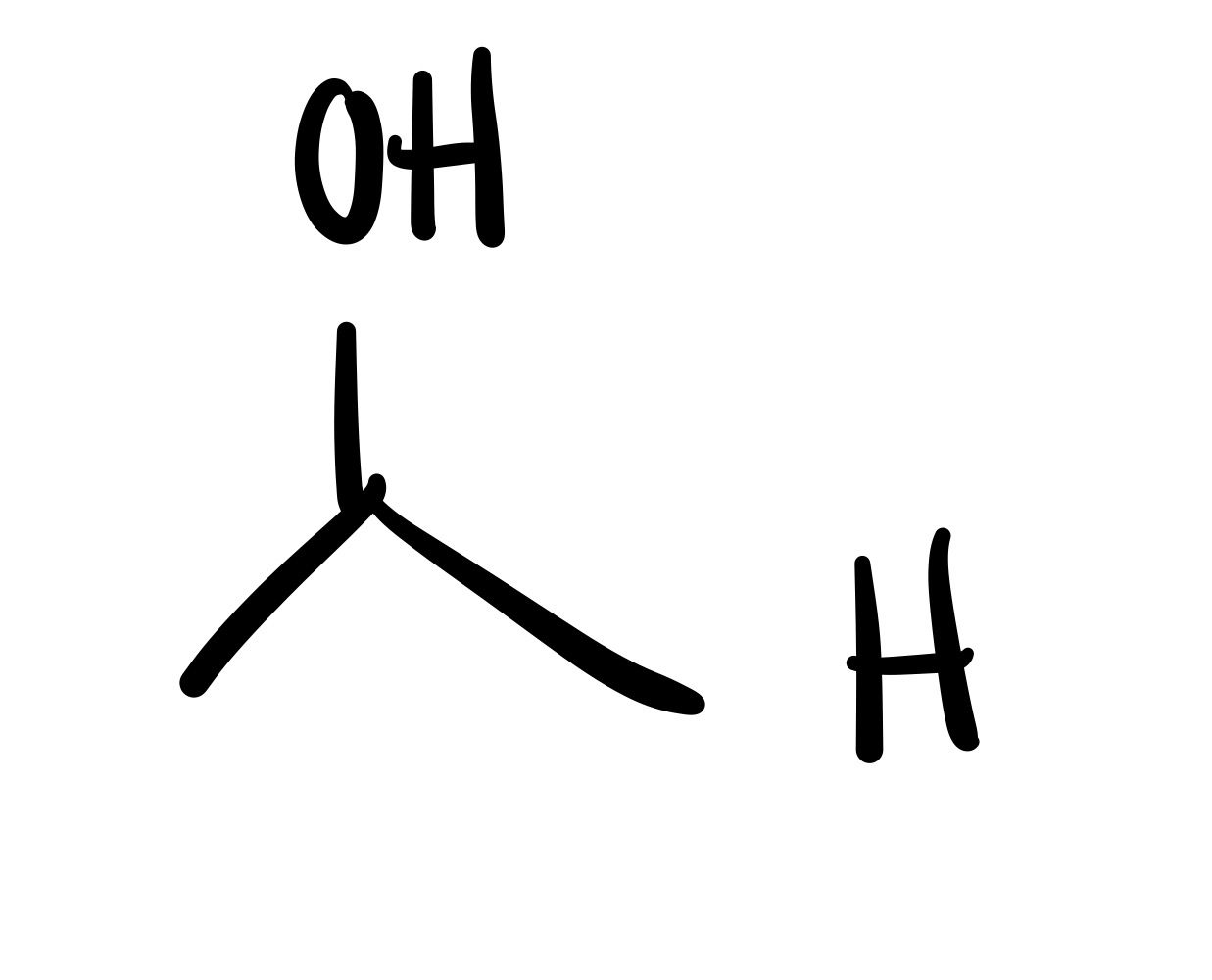
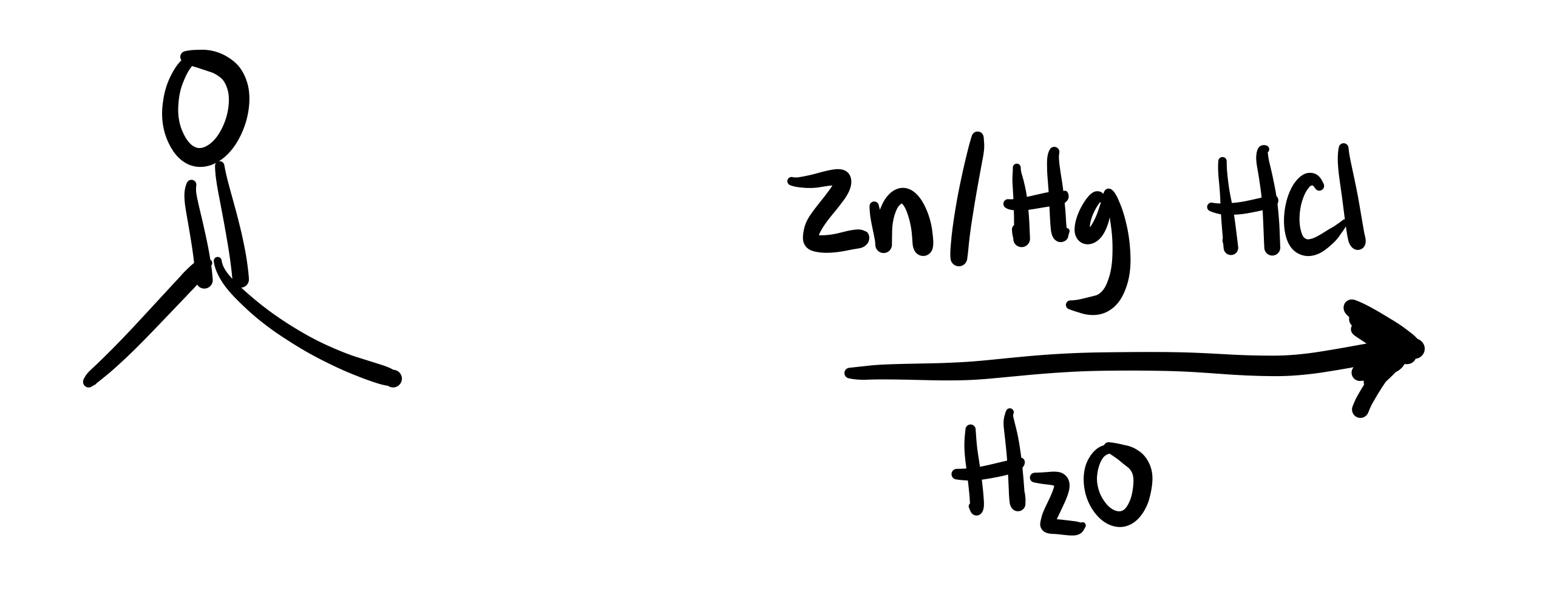
The O double bond leaves the carbon chain

The OH leaves the carbon chain, and the substituent adds
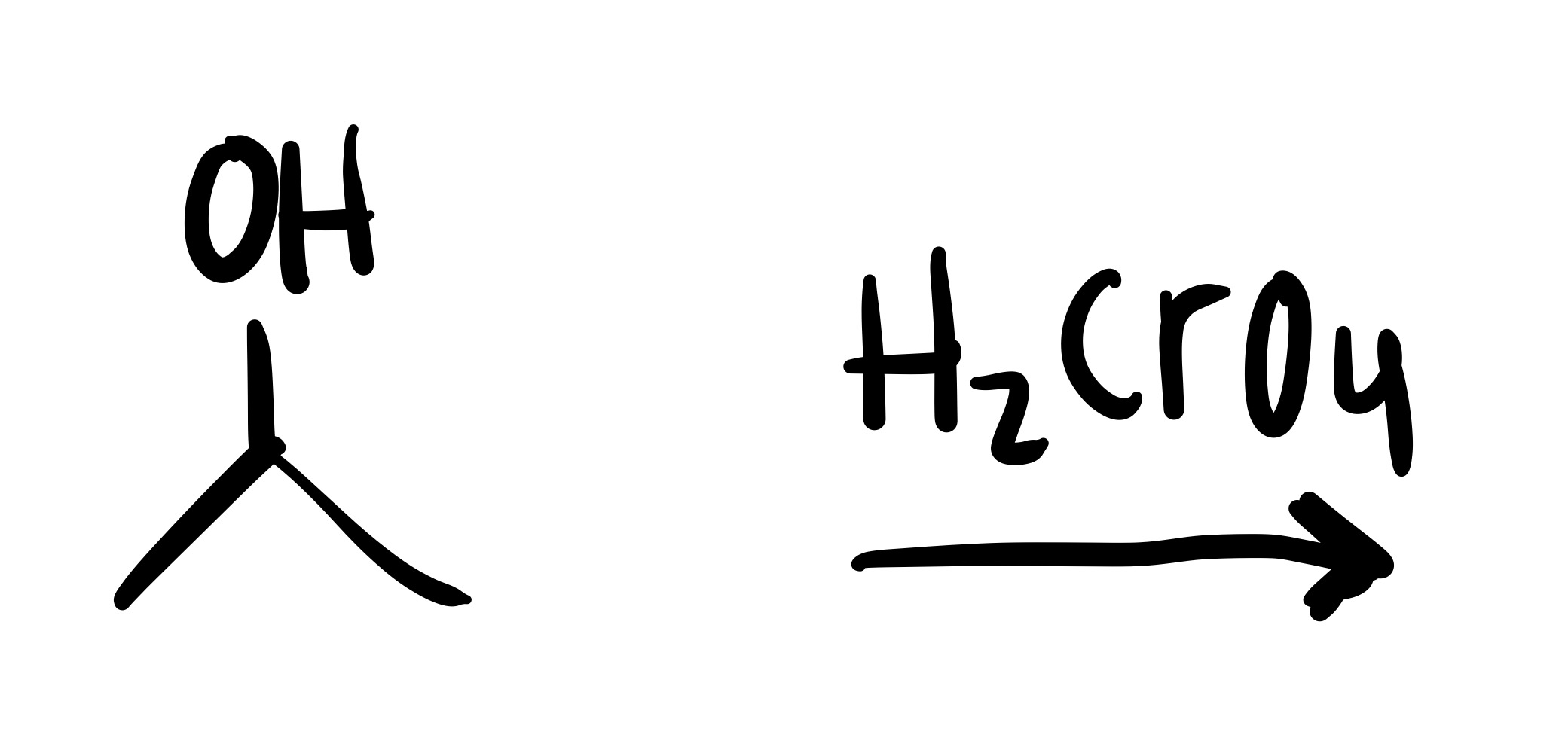
The H leaves the O, and a double bond is formed to the O attached to the carbon chain

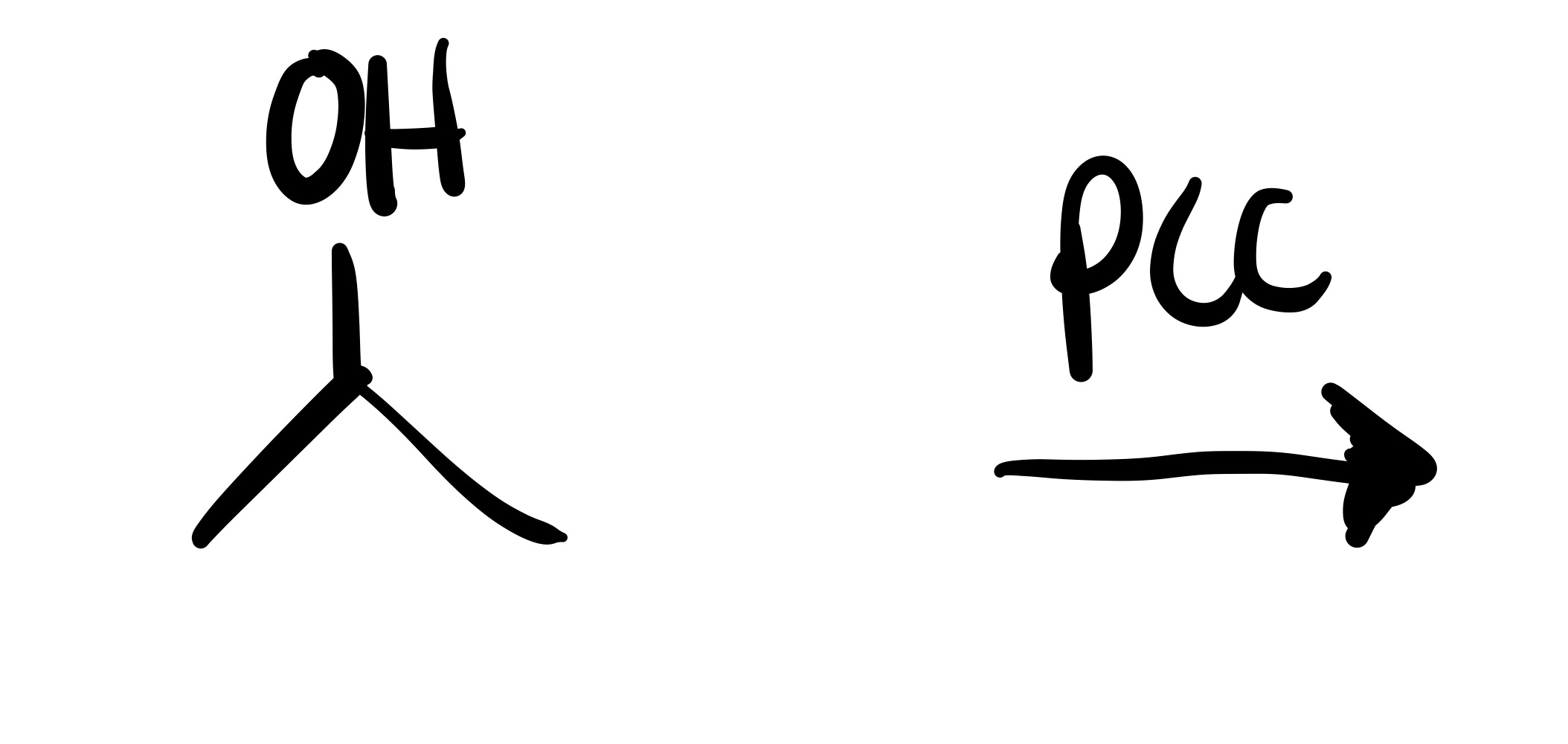
The H leaves the O, and a double bond is formed to the O attached to the carbon chain

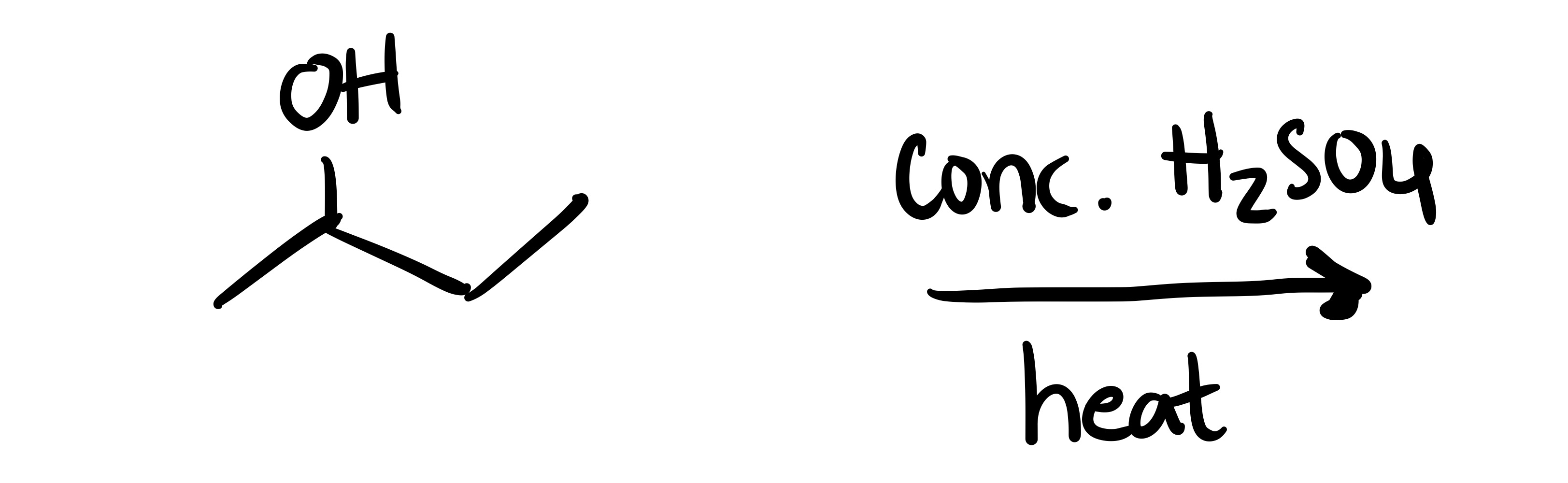
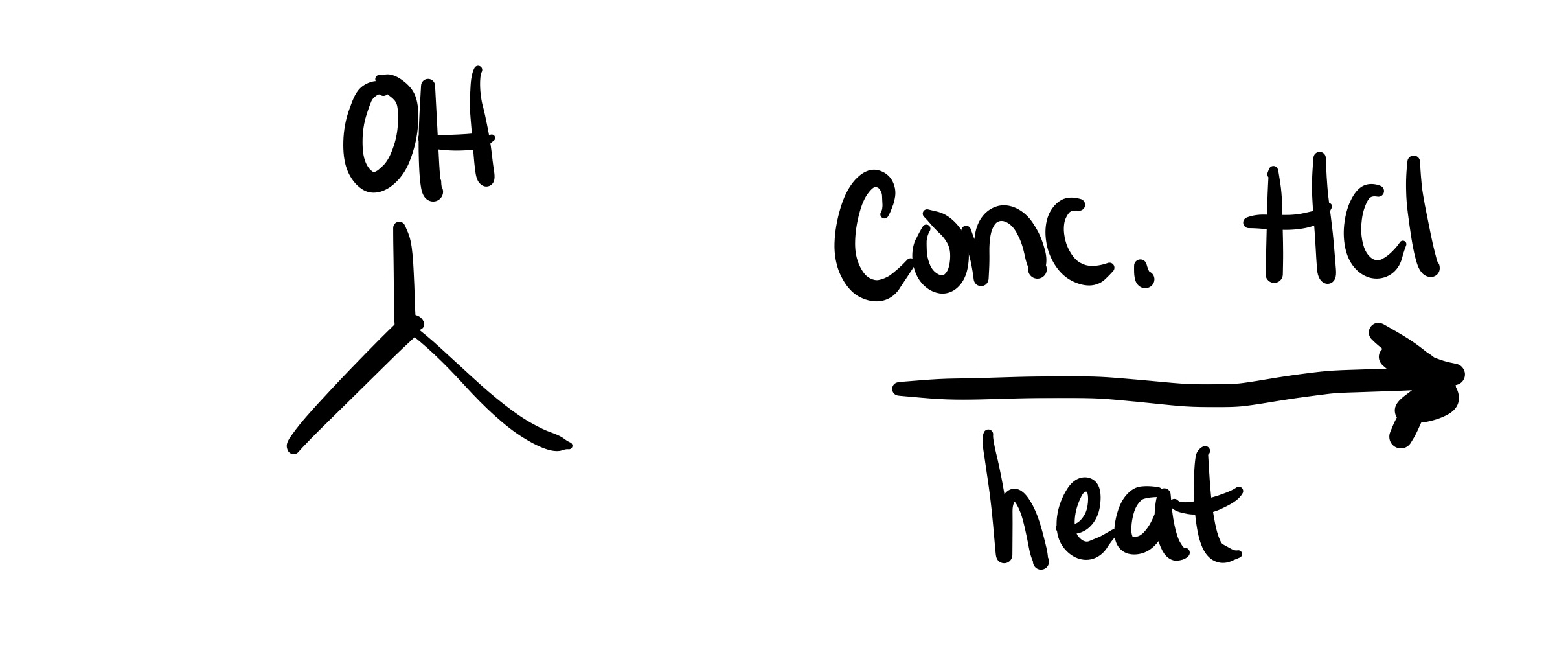
The OH leaves, and a double bond is formed on the carbon chain (Alkene)
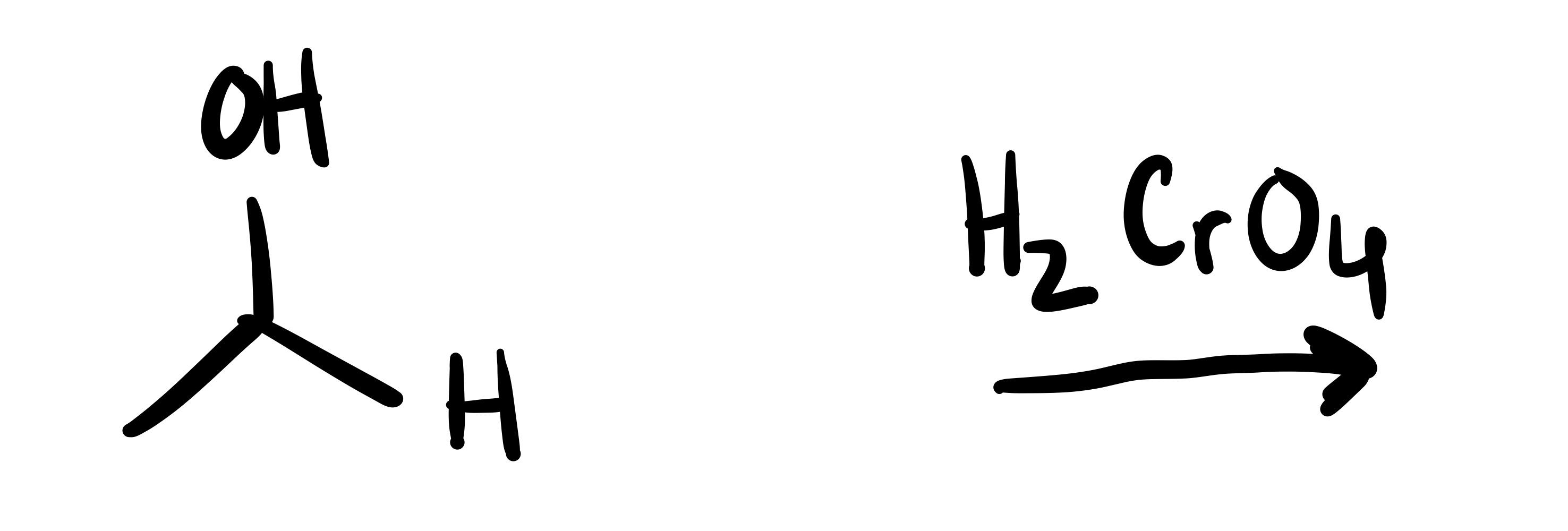
The H leaves the OH group, and a double bond forms to the O and carbon chain. And an O adds to the H group
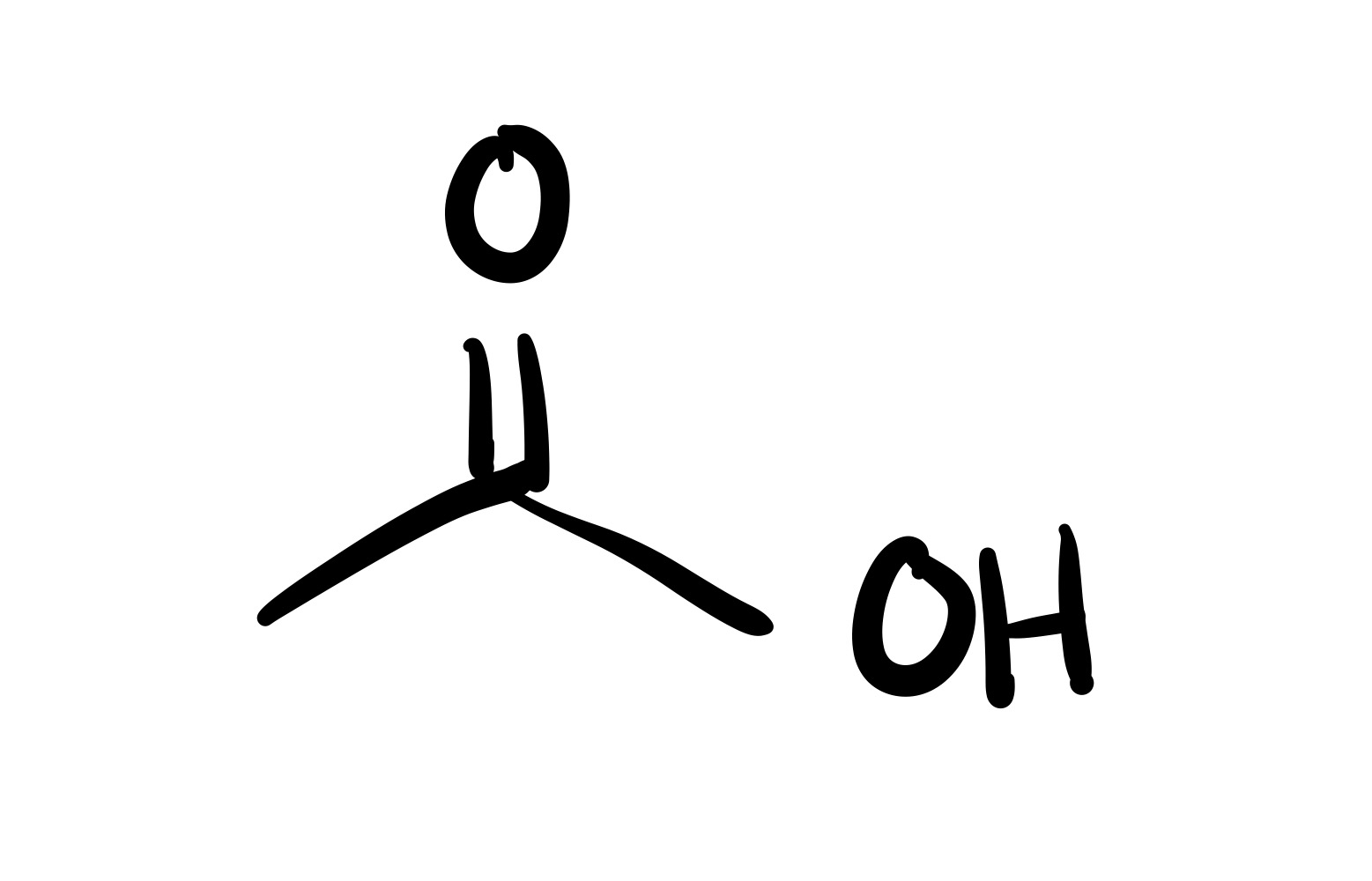
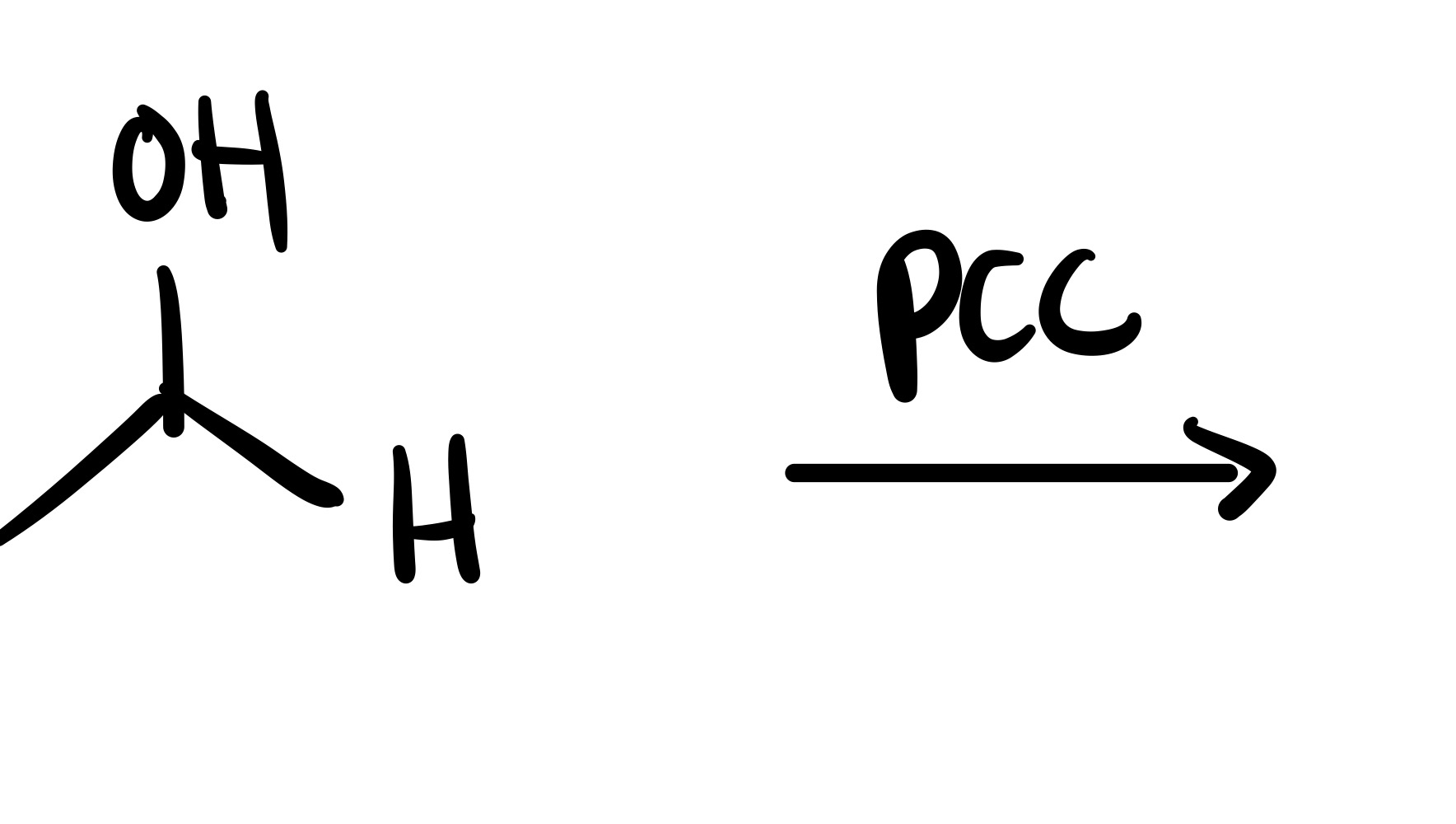

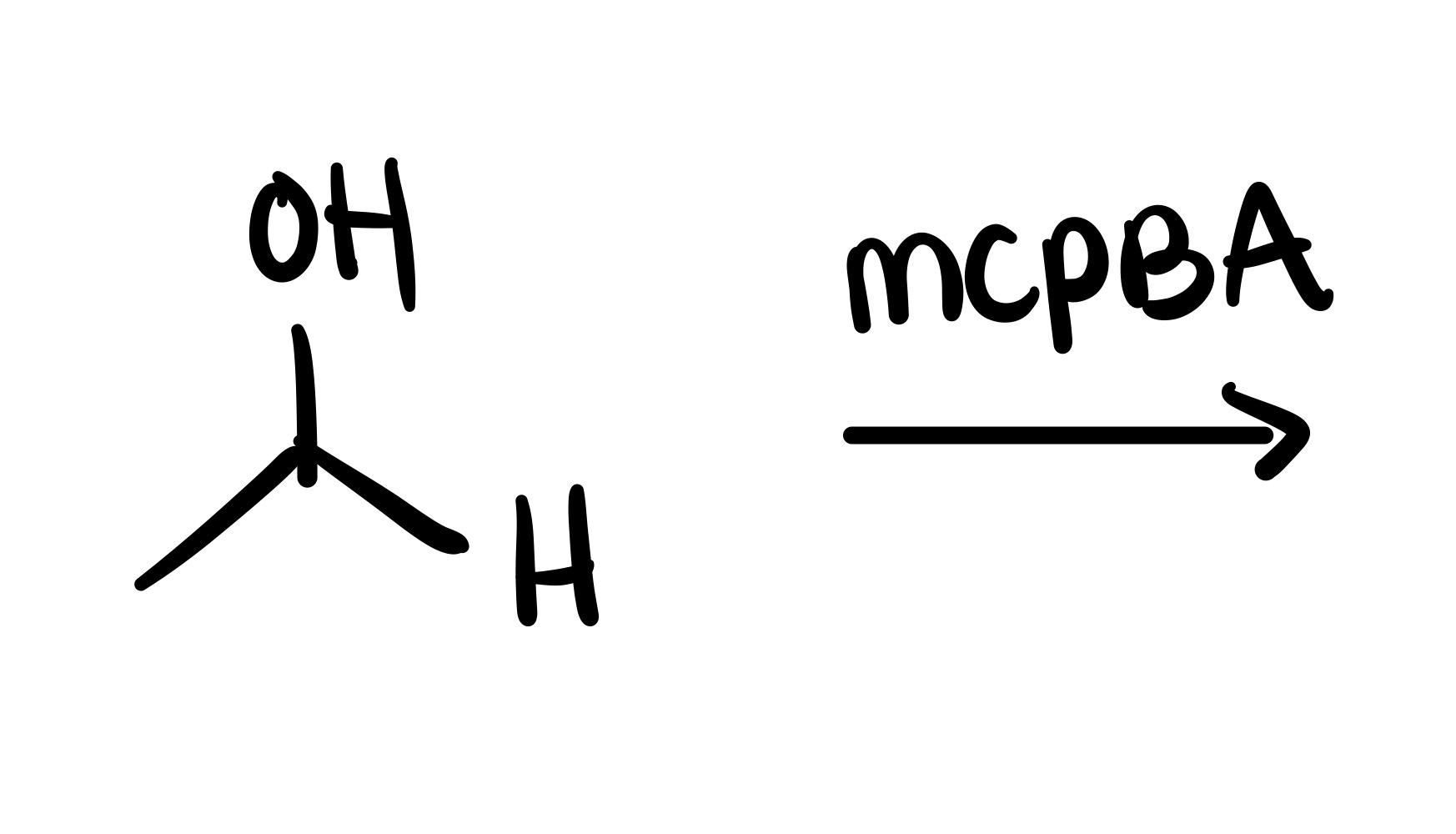
The H leaves the OH, and a double bond forms to the remaining O. And an O adds to the H
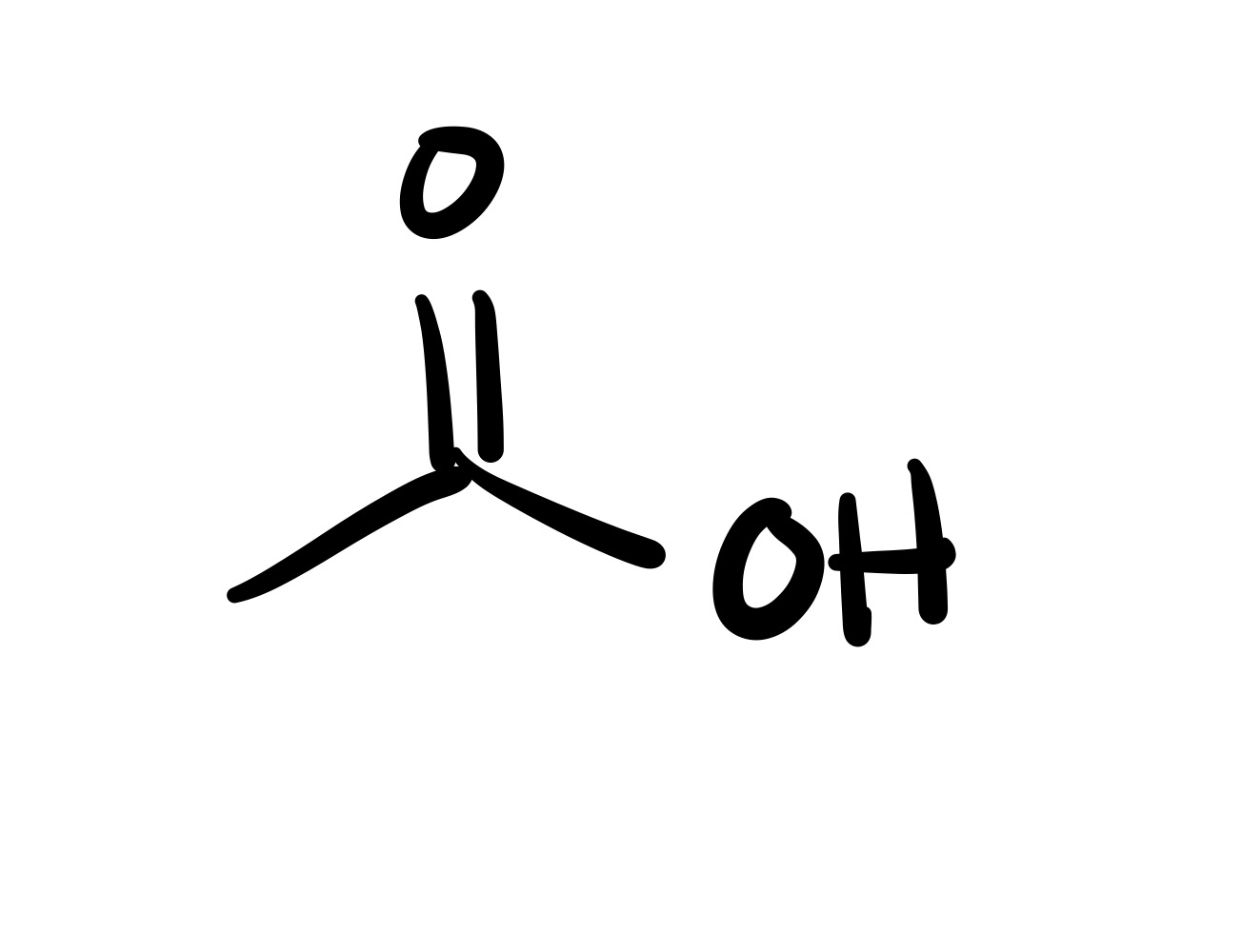
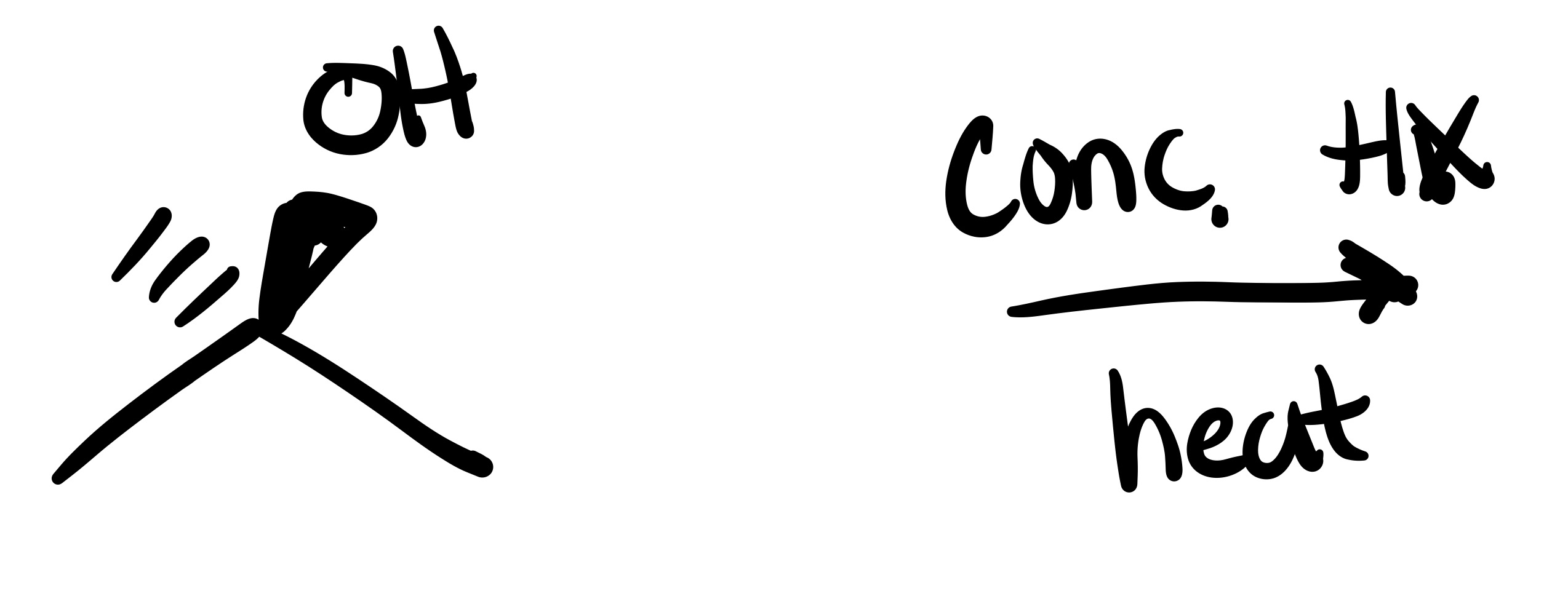
The OH leaves, and the substituent adds to where it was on the carbon chain
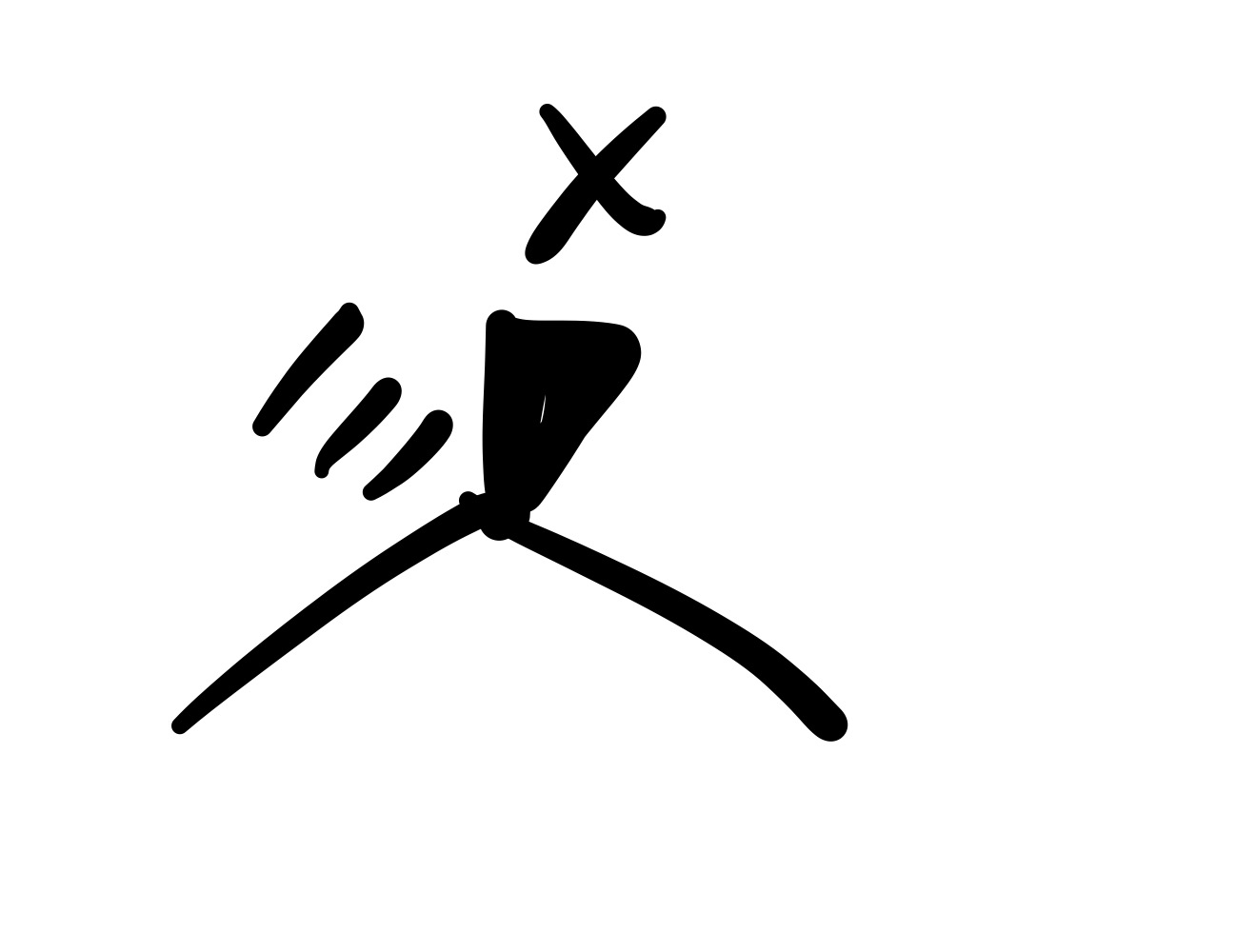
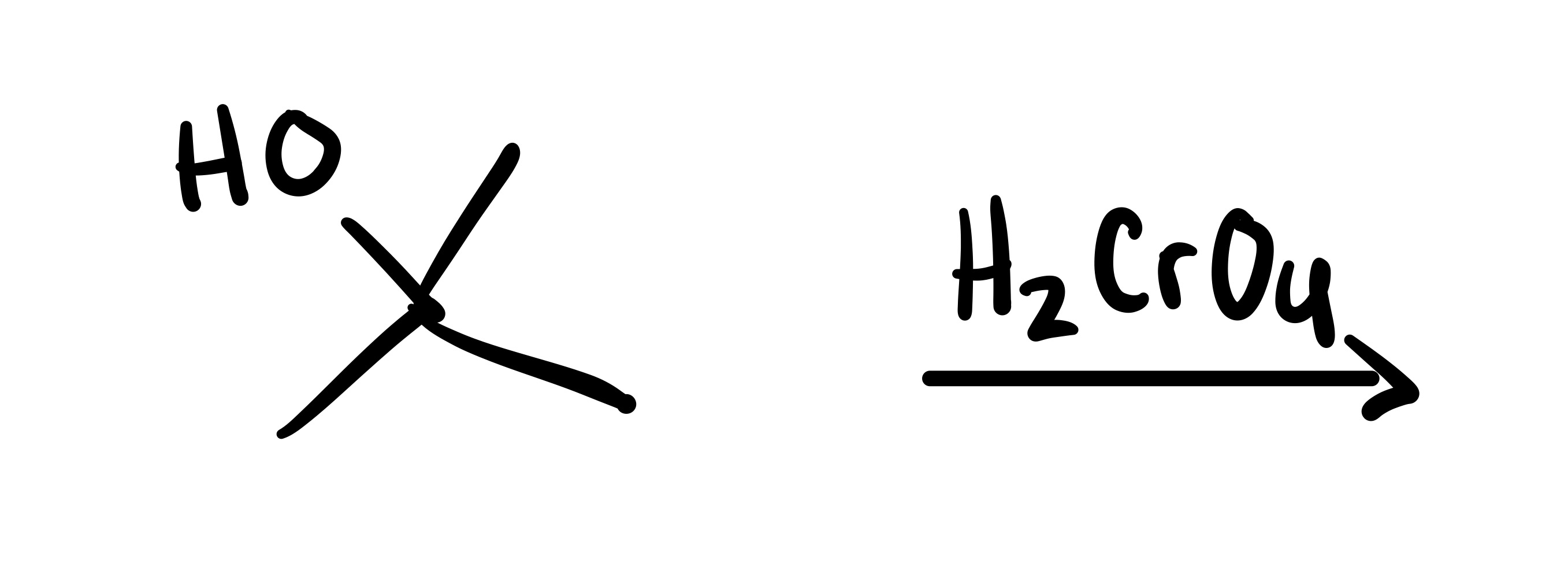
No reaction
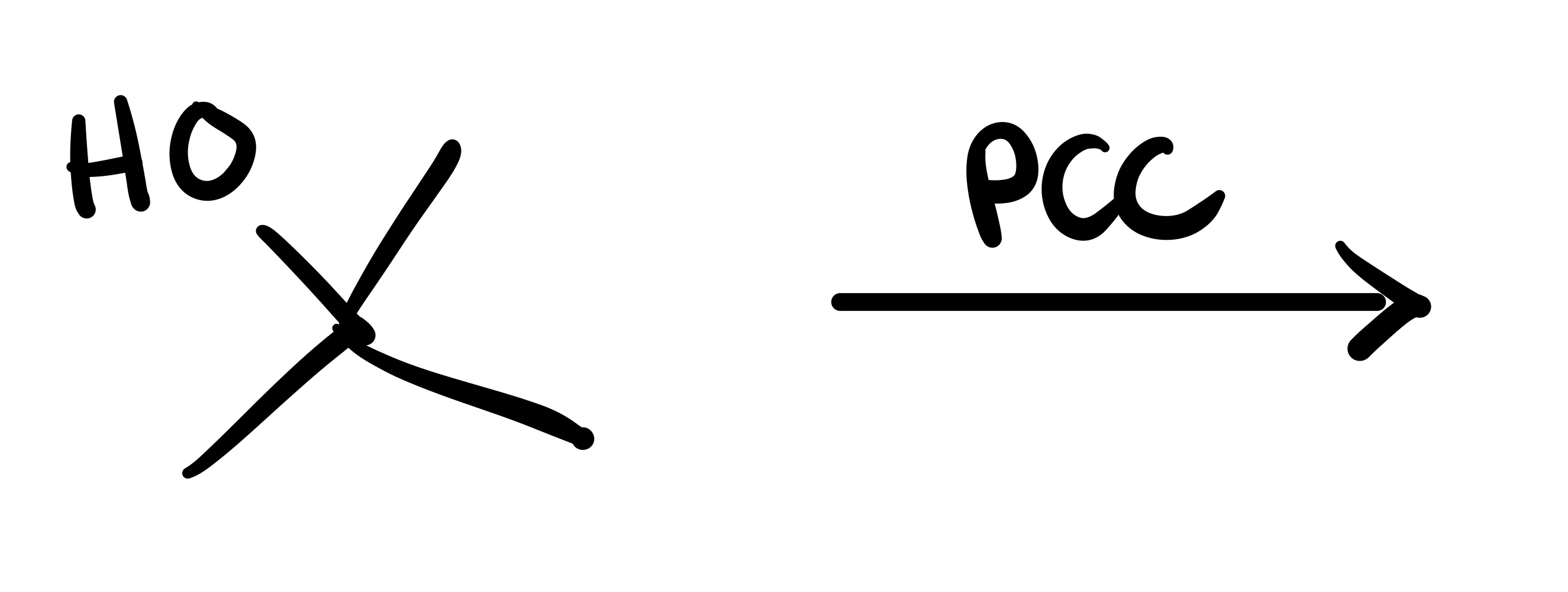
No reaction
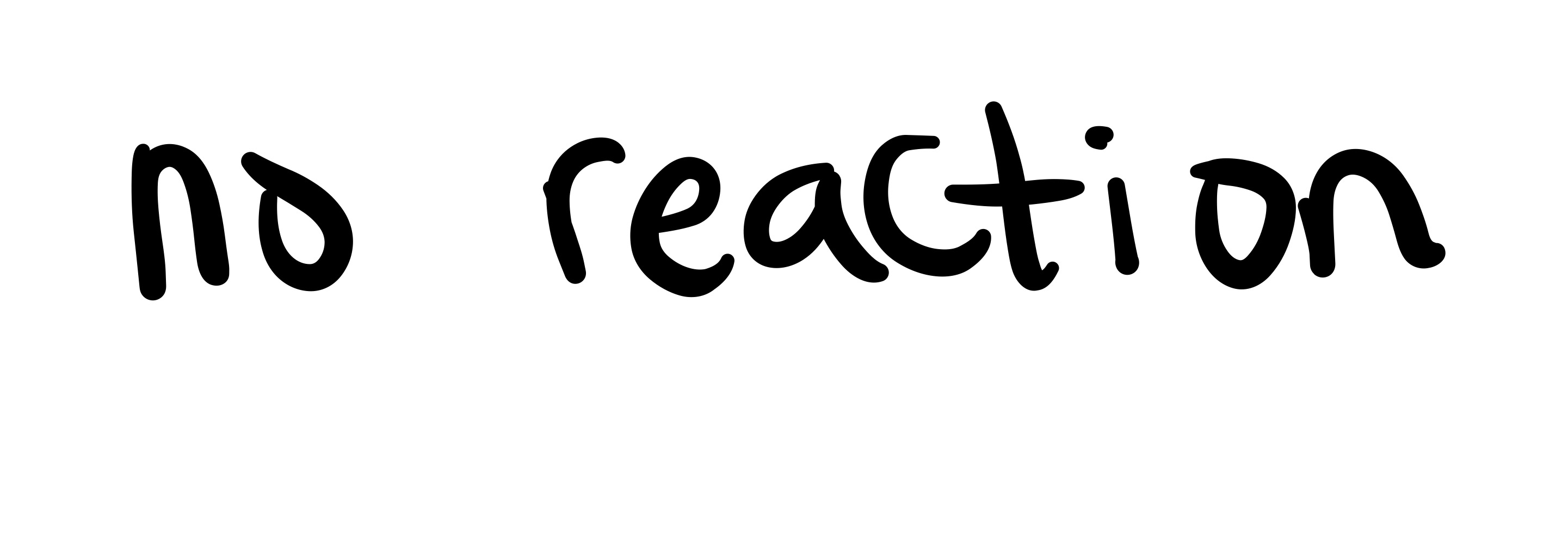
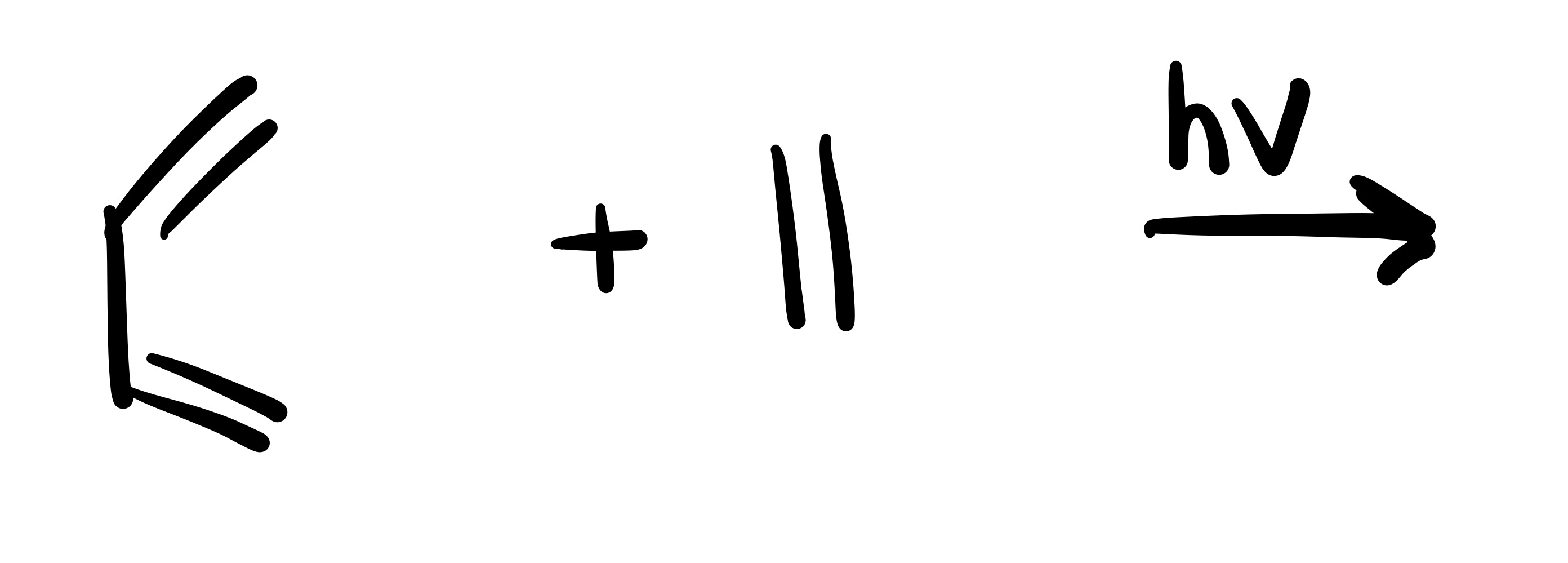
The diene adds to the dienophile (one pi bond in cyclohexane)
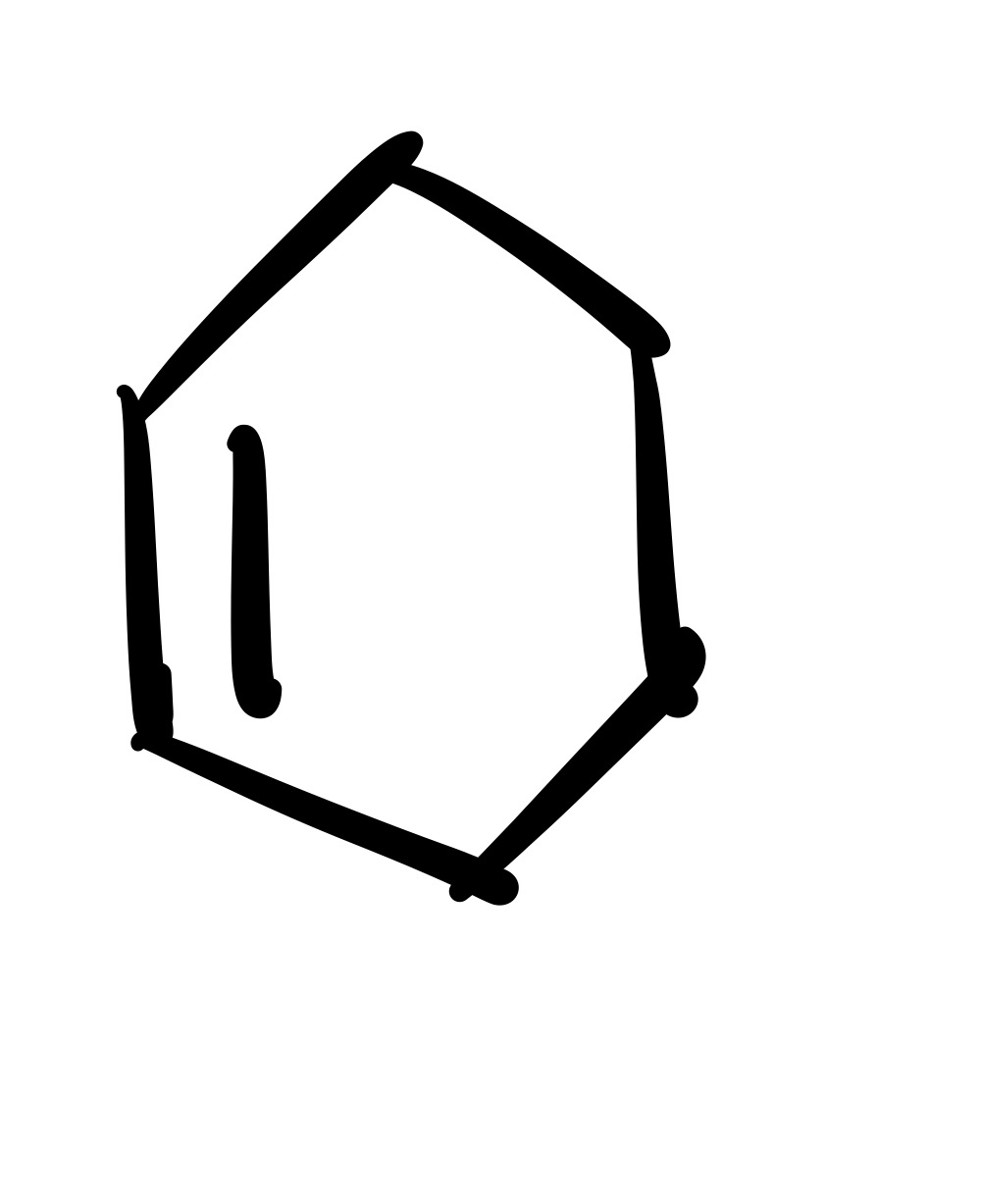
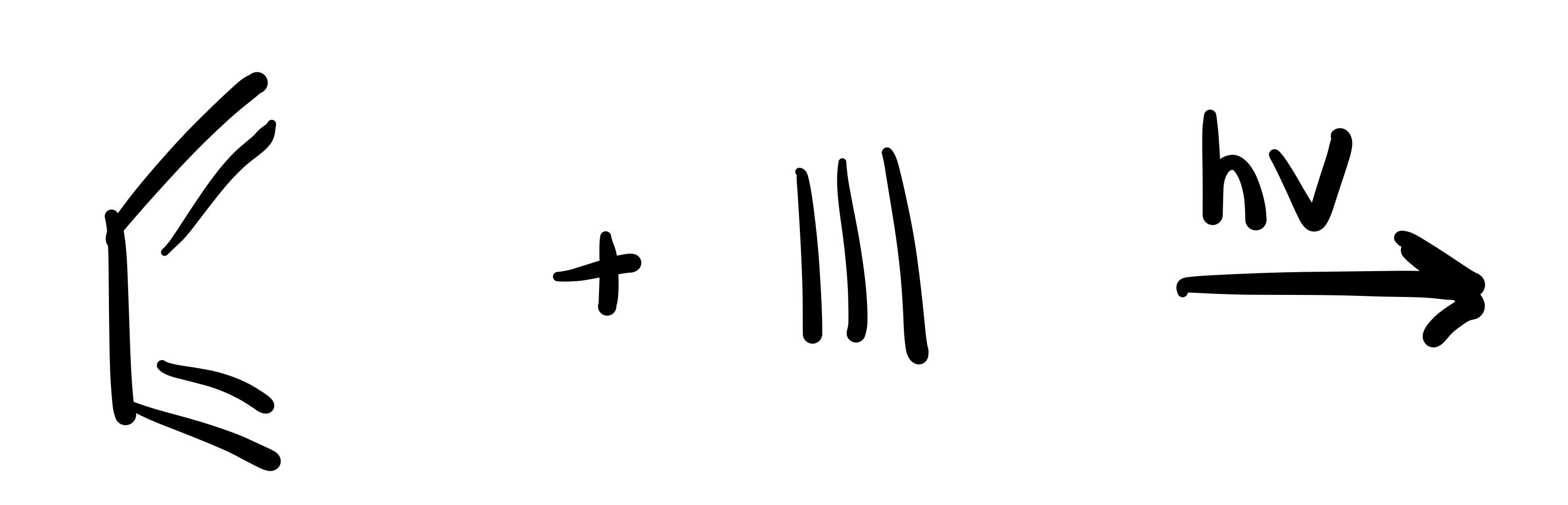
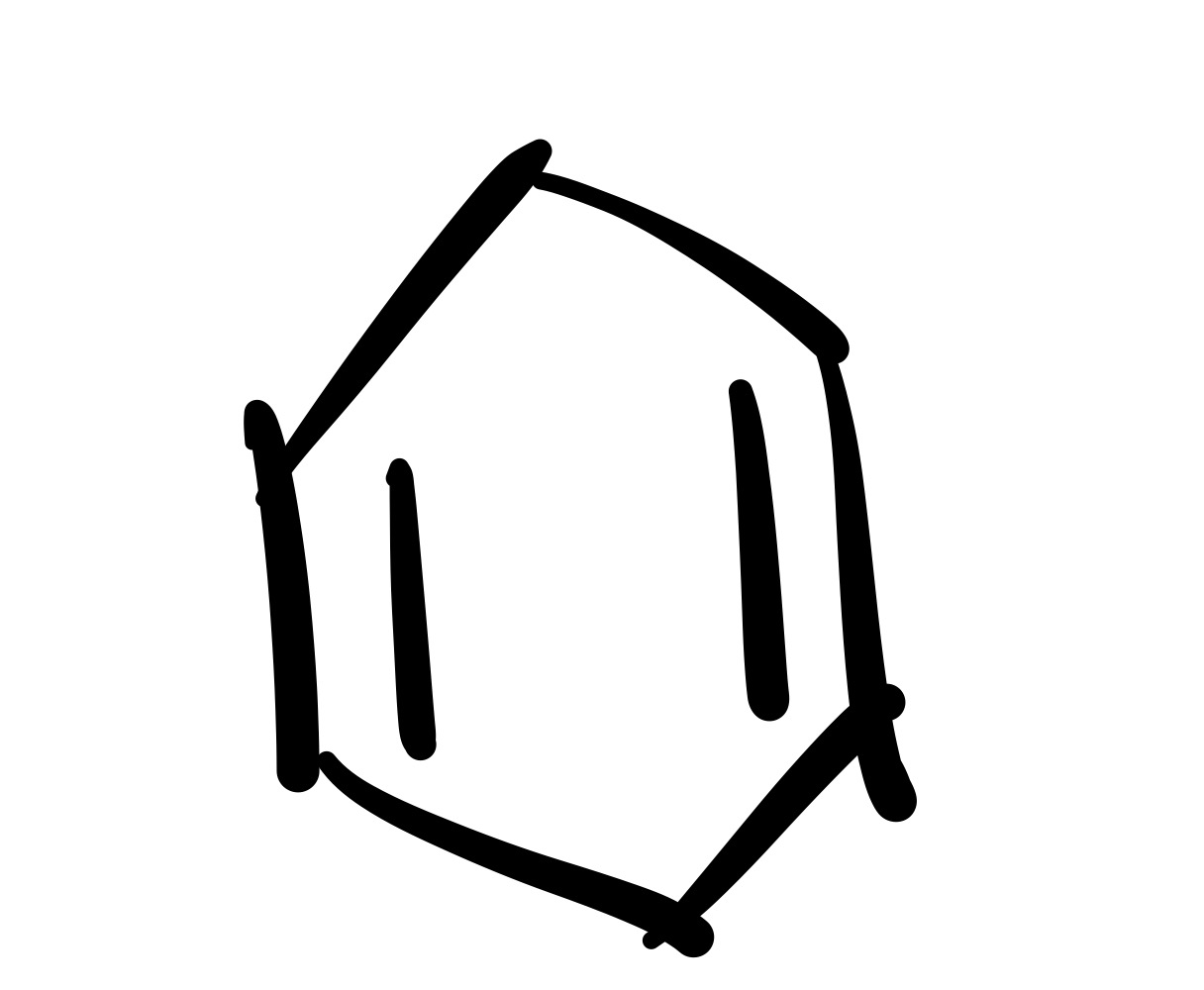
The diene adds to the dieneophile (two pi bonds in cylohexane)
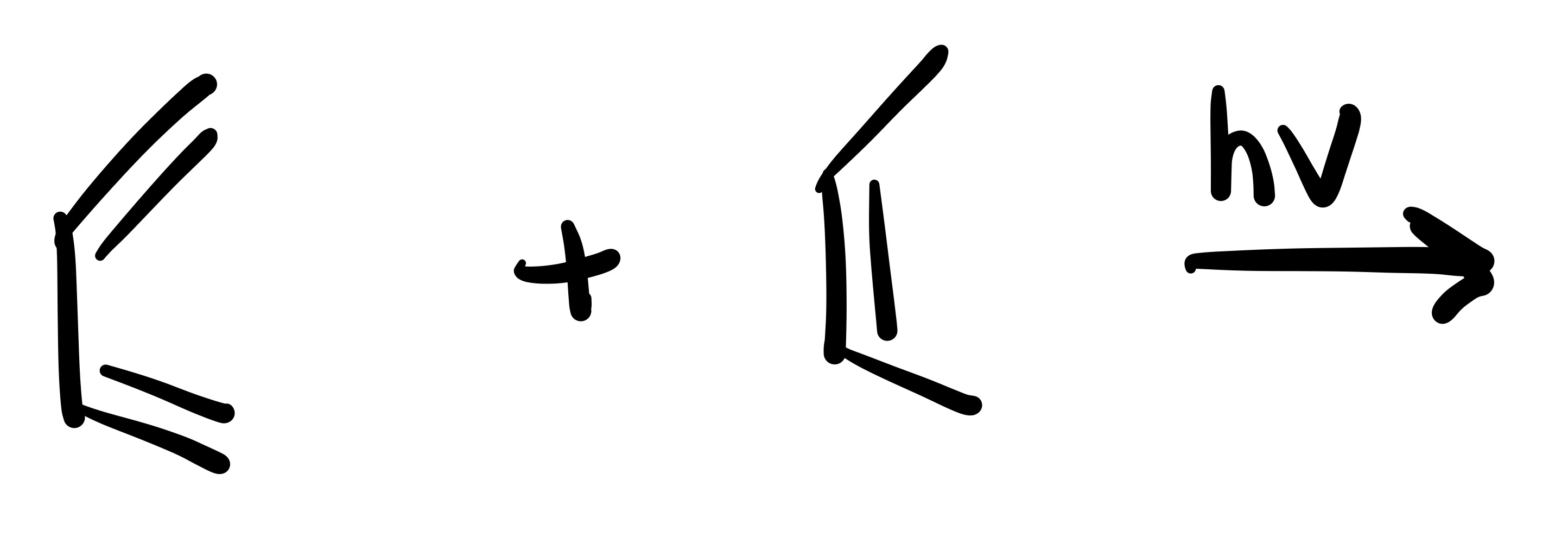
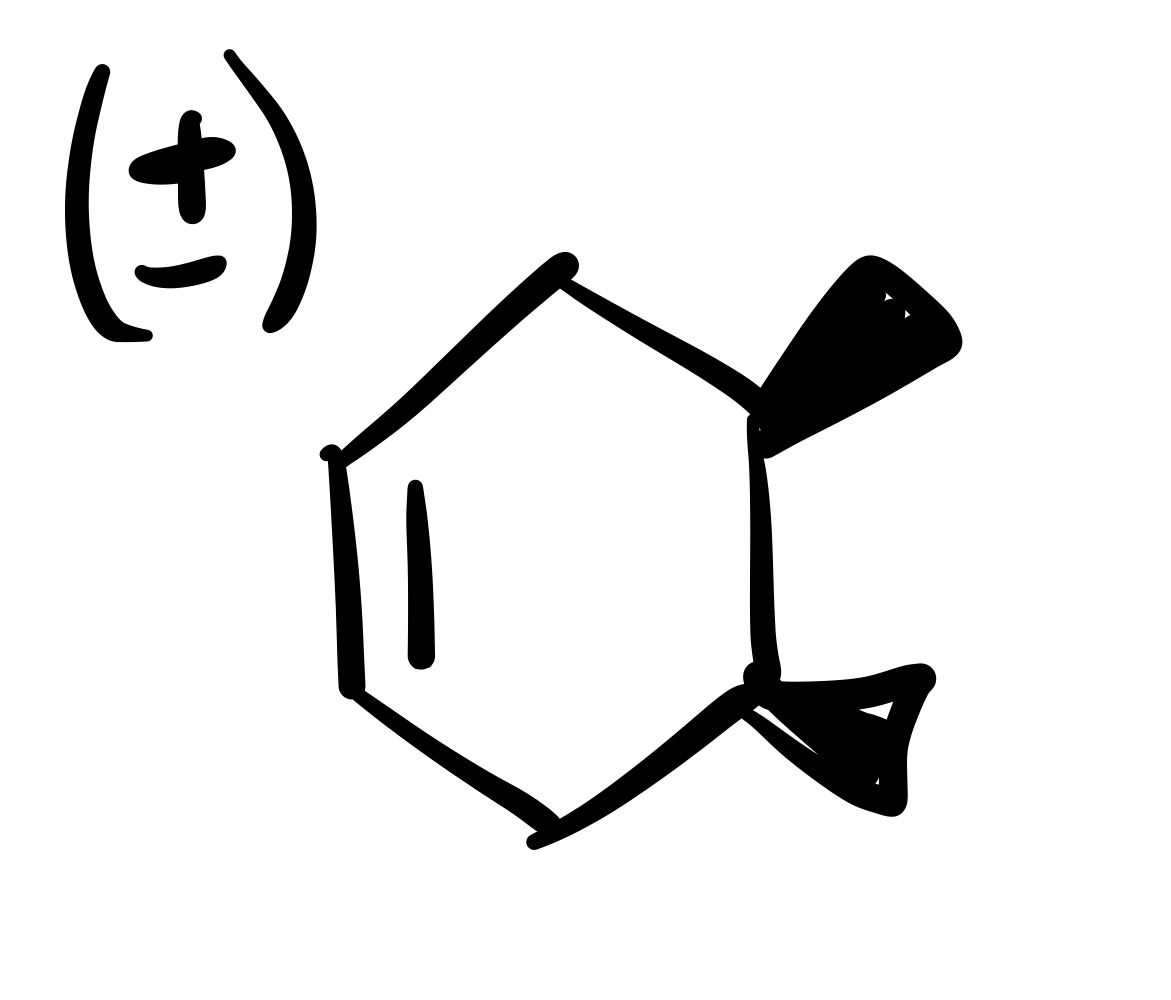
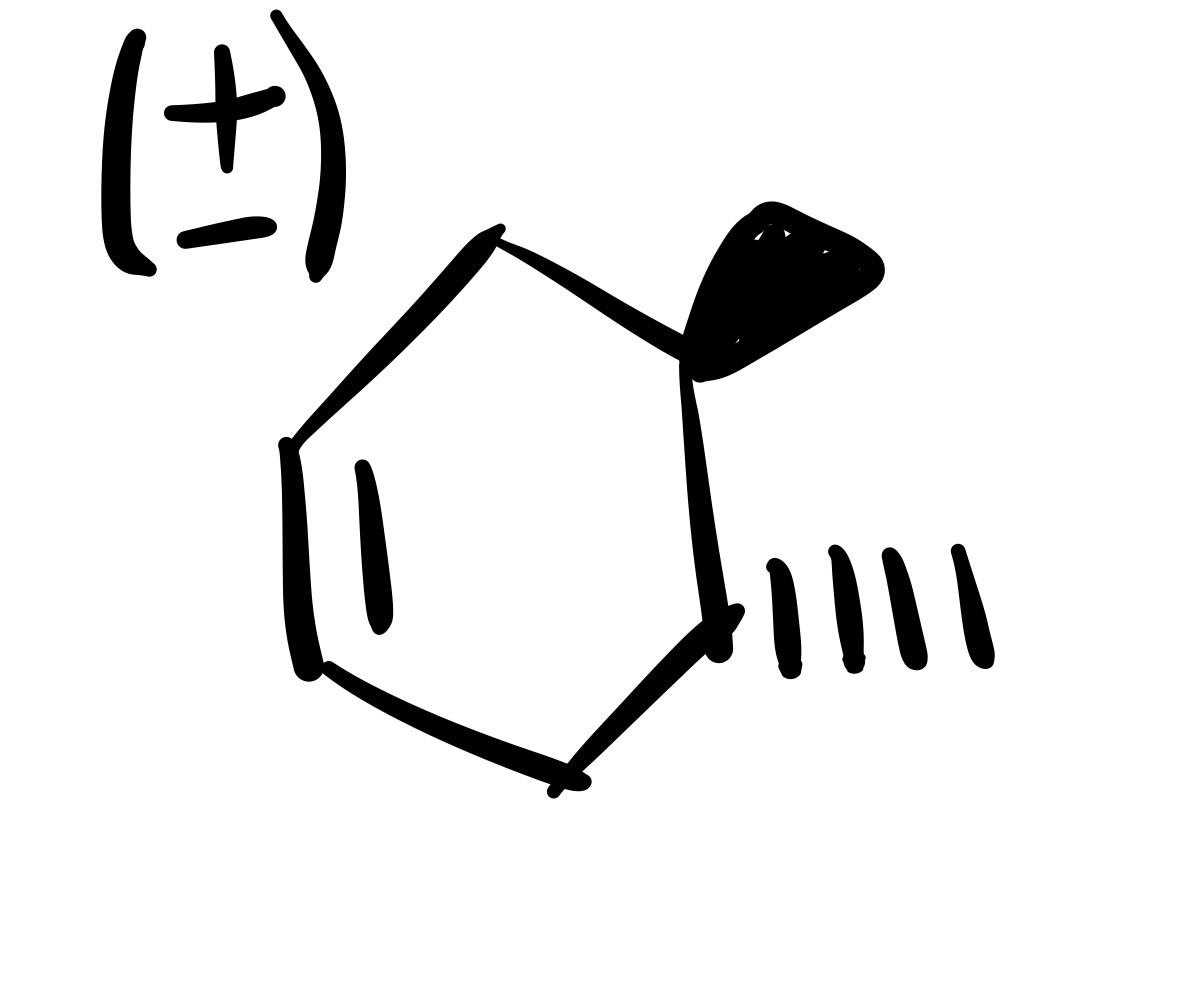
The diene adds to the dienophile. The two methyl’s are added (cis). Add plus/ minus sign
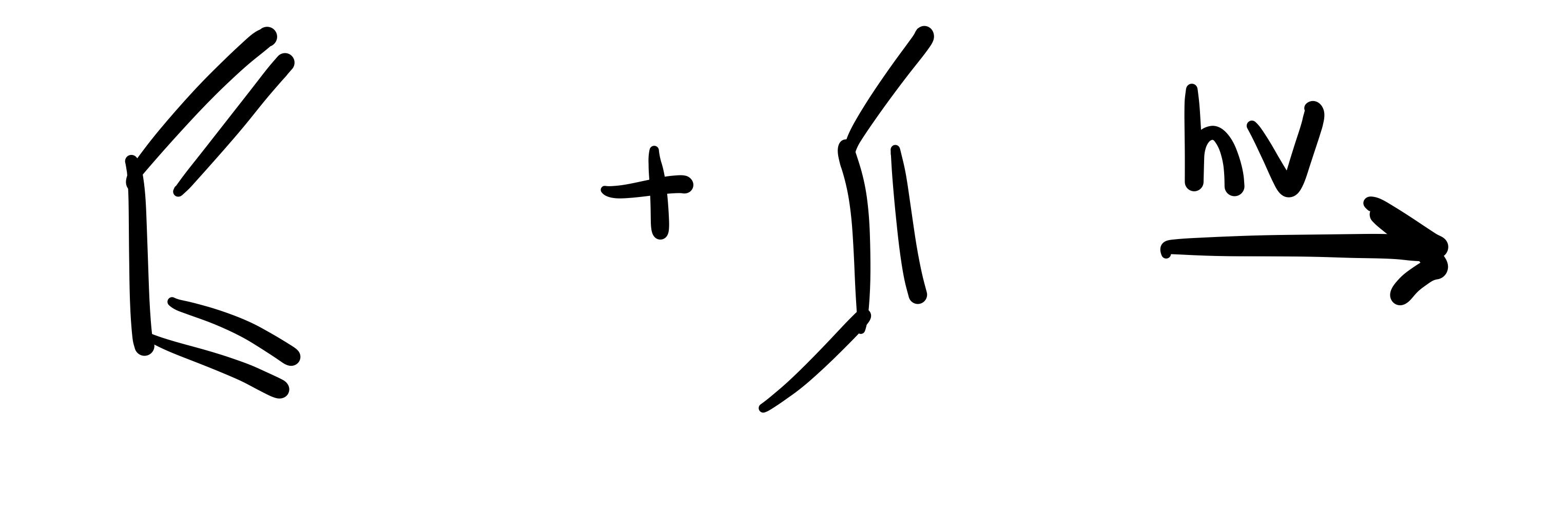
The diene adds to the dienophile. The two methyl’s are added (cis). Add plus/ minus sign
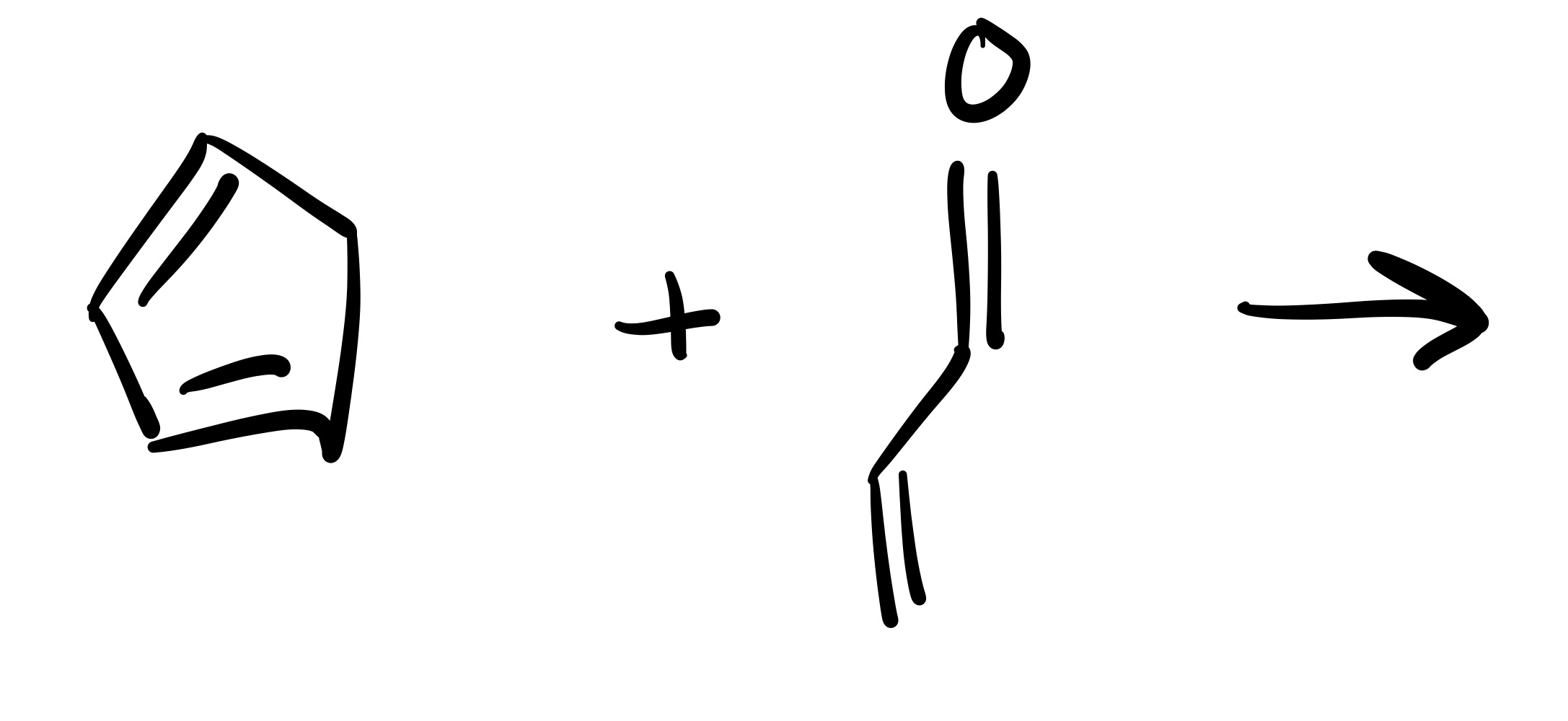
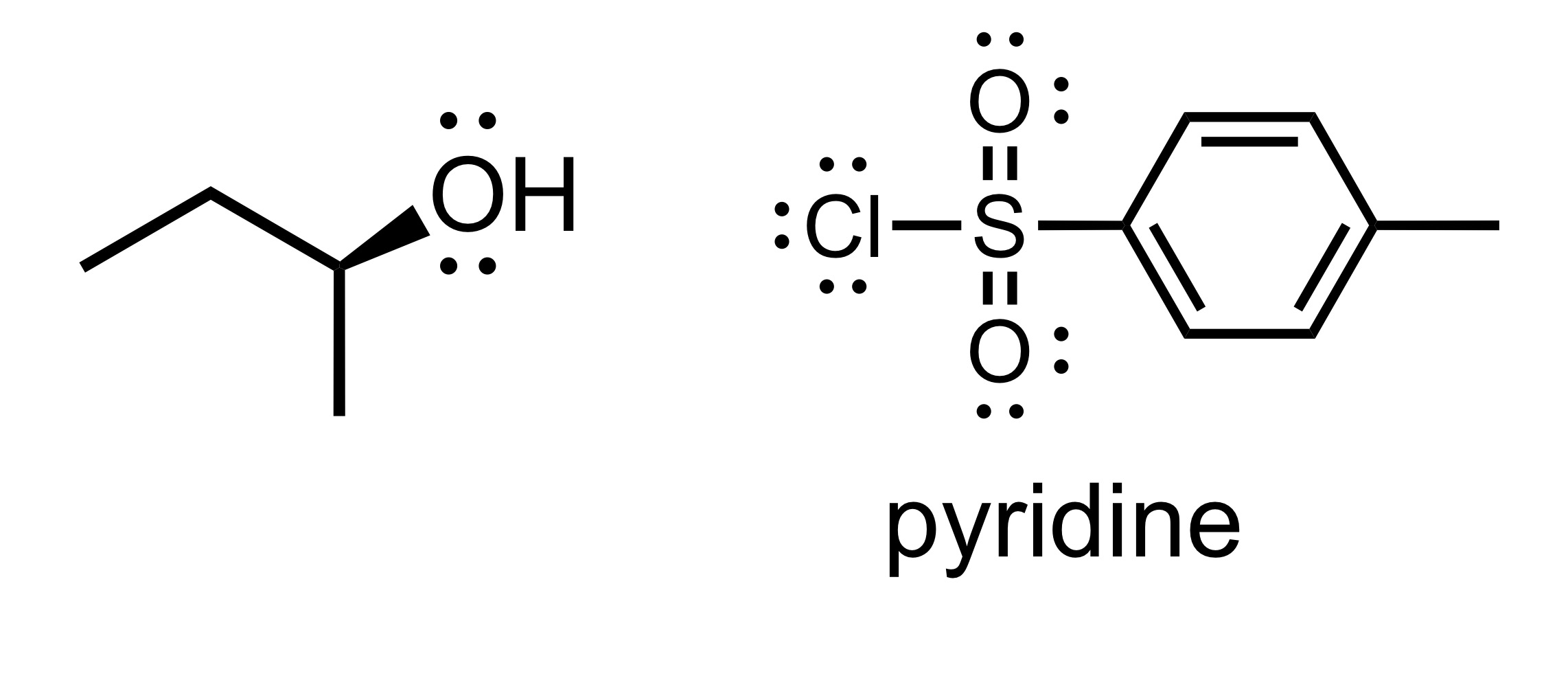
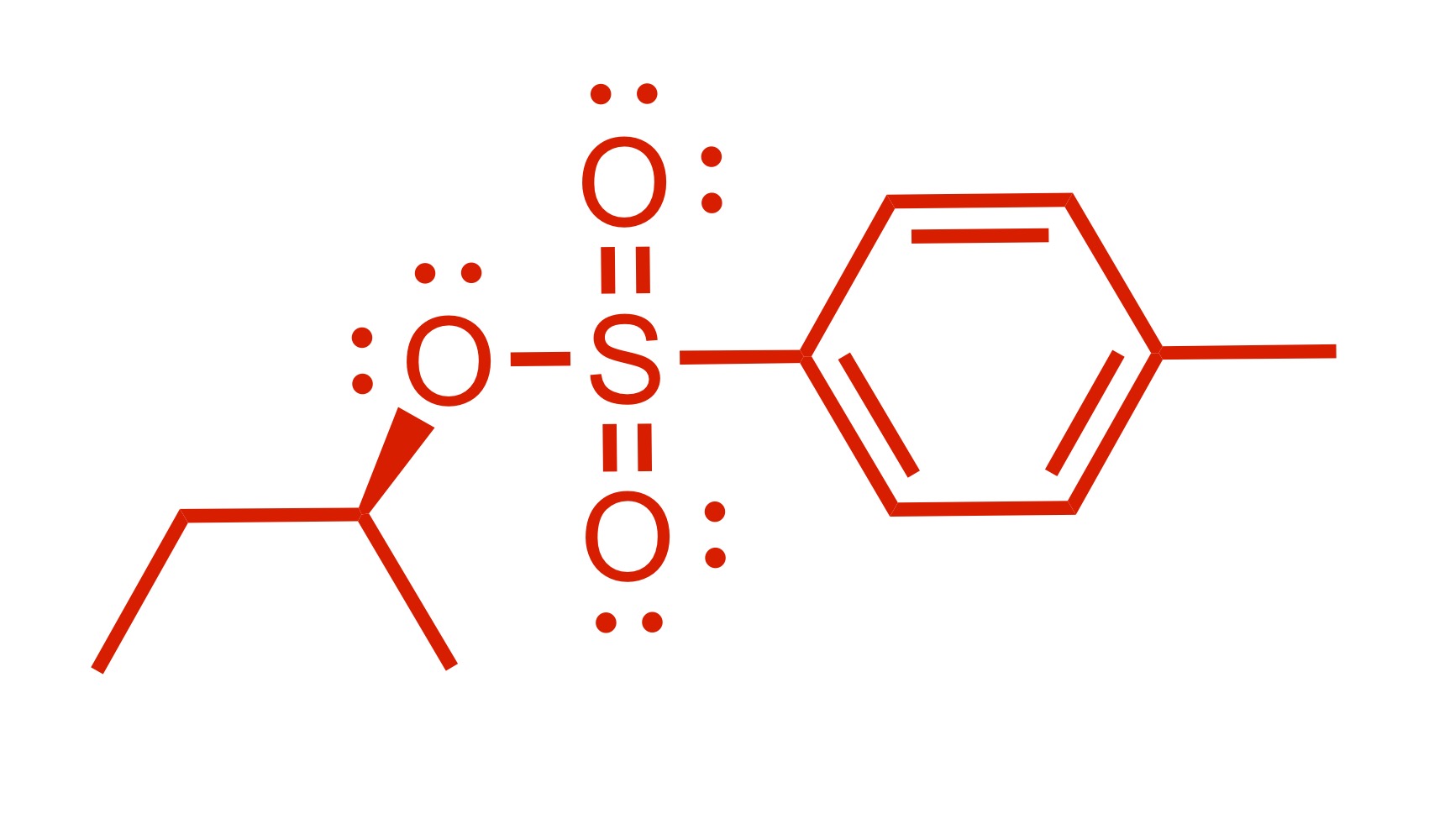
(Substitution) The H leaves the OH, and the Cl leaves the pyridine. The Pyridine attaches to the O
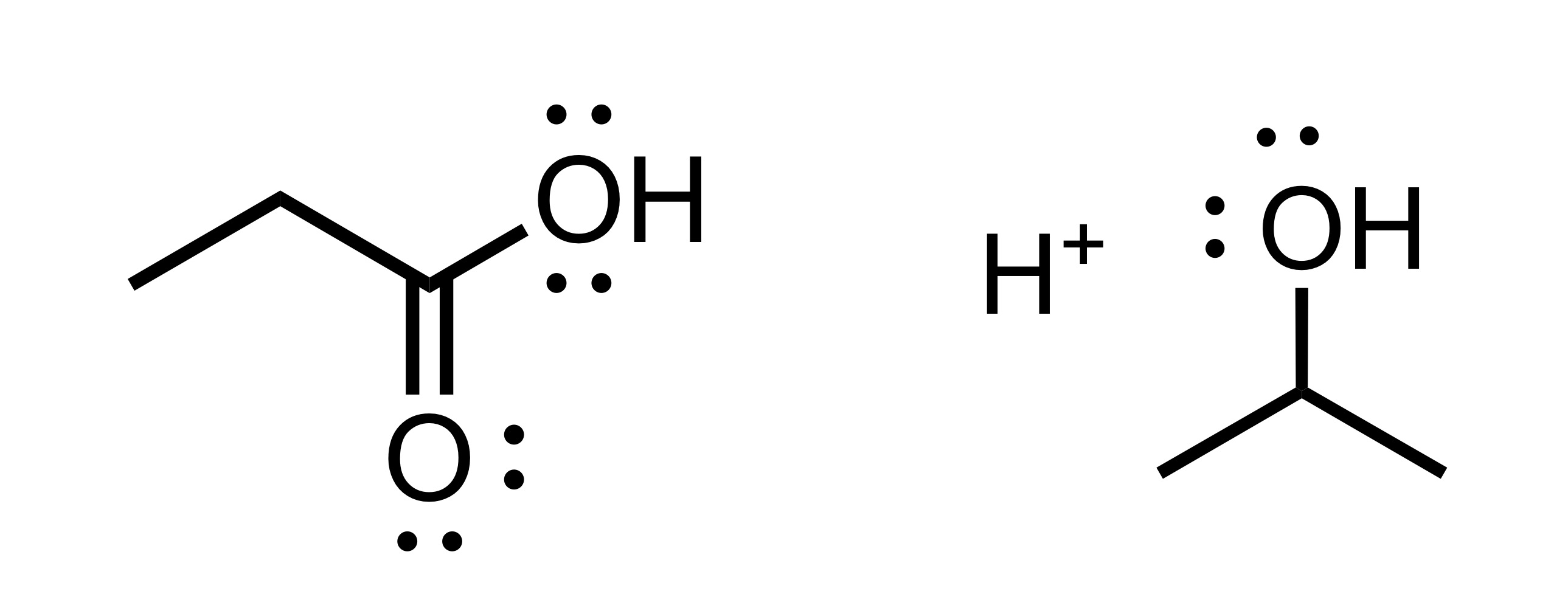
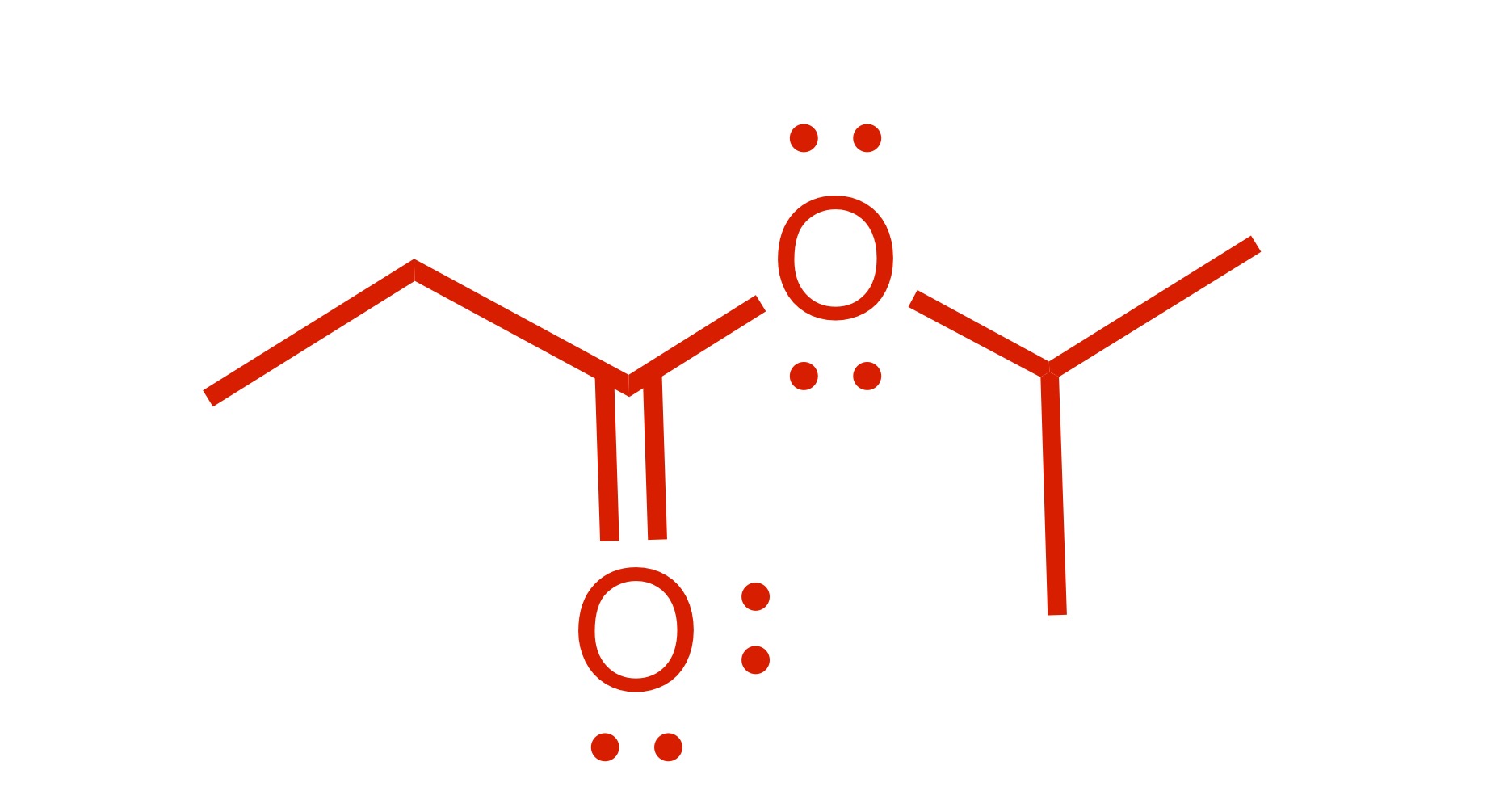
The H leaves the OH group on the carbon chain. And the OH leaves on the other group, which allows the carbon chain to attach to the O
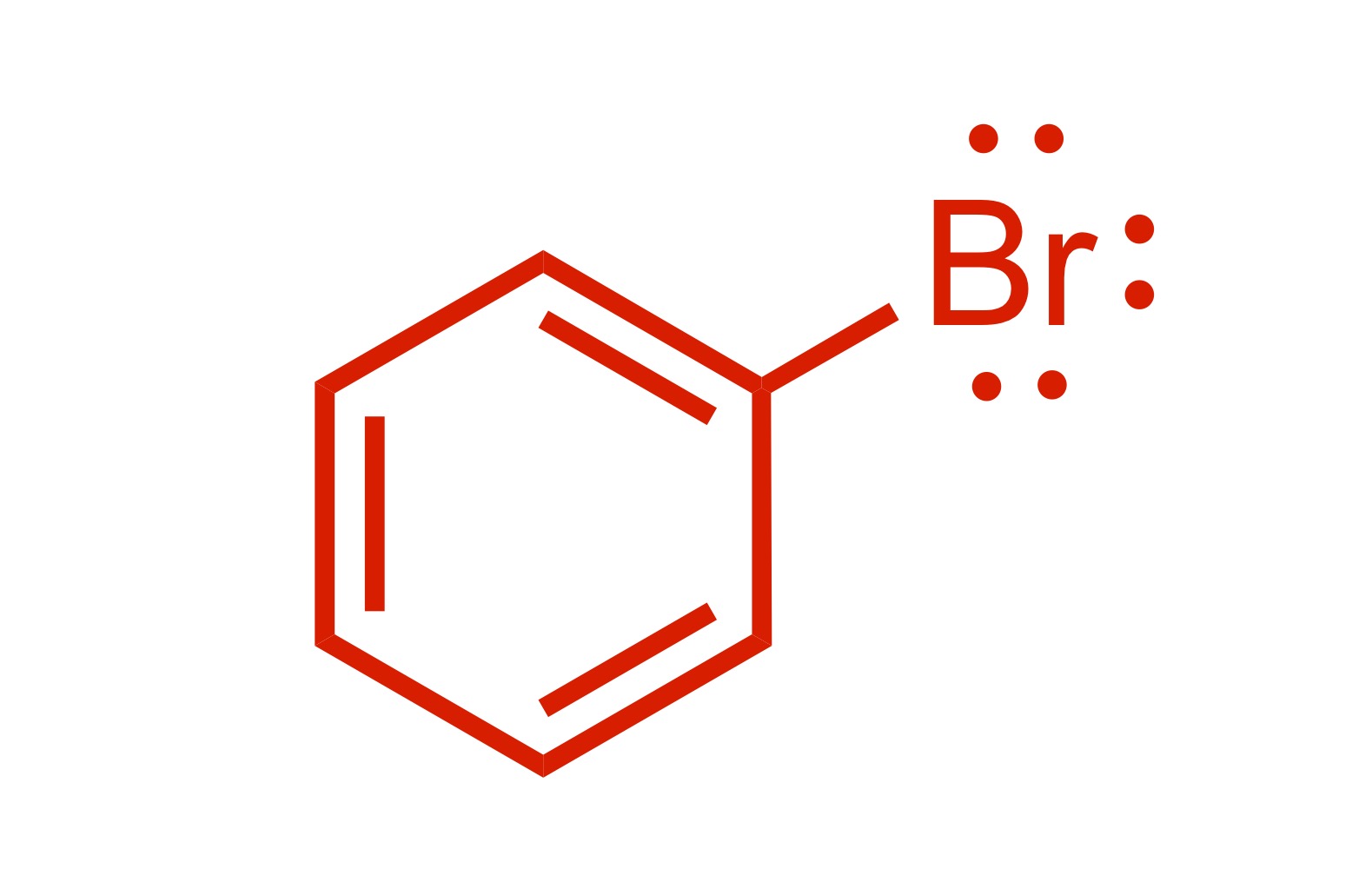
1 Br group attaches to the cyclohexane (with lone pairs)
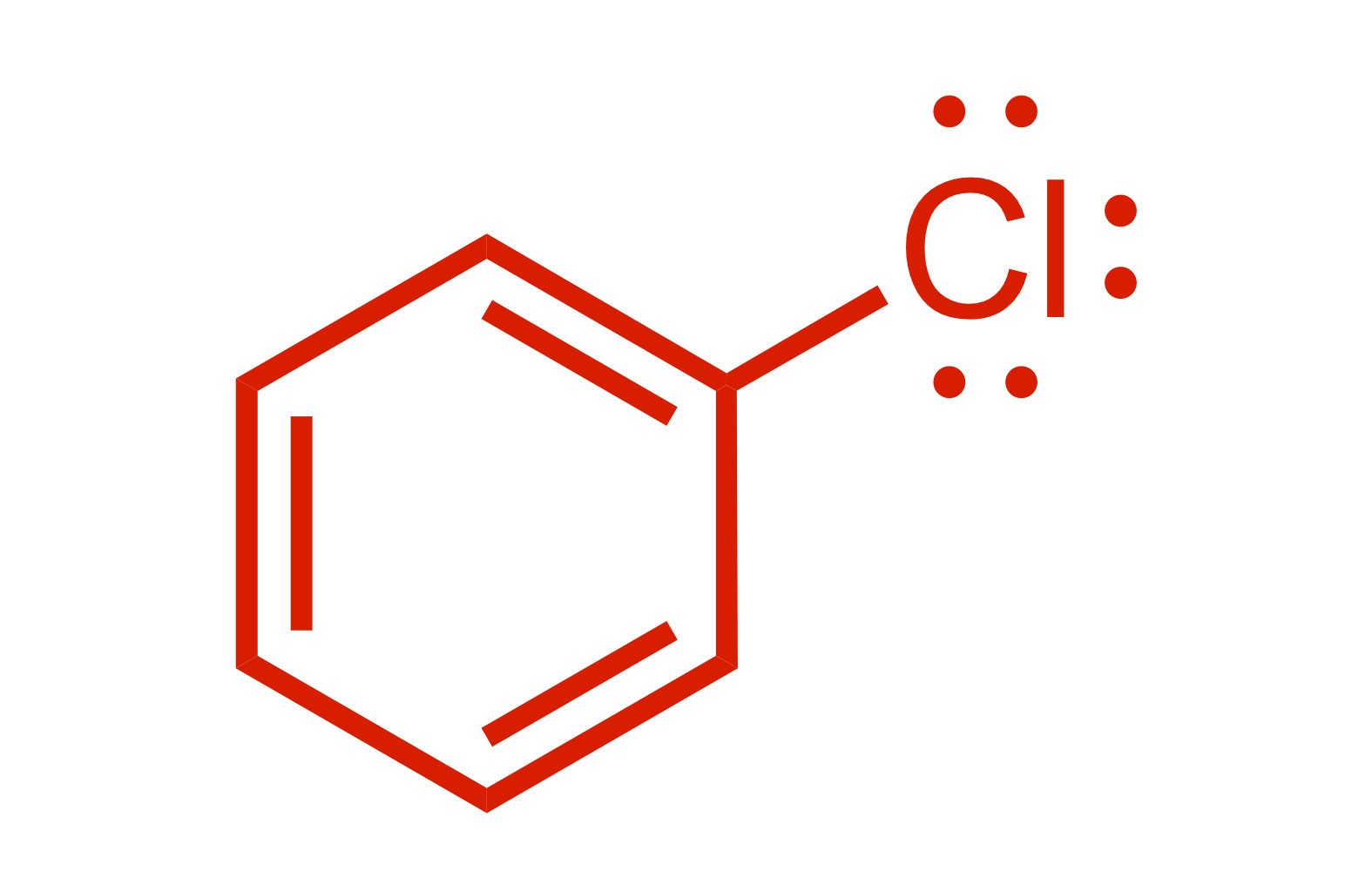
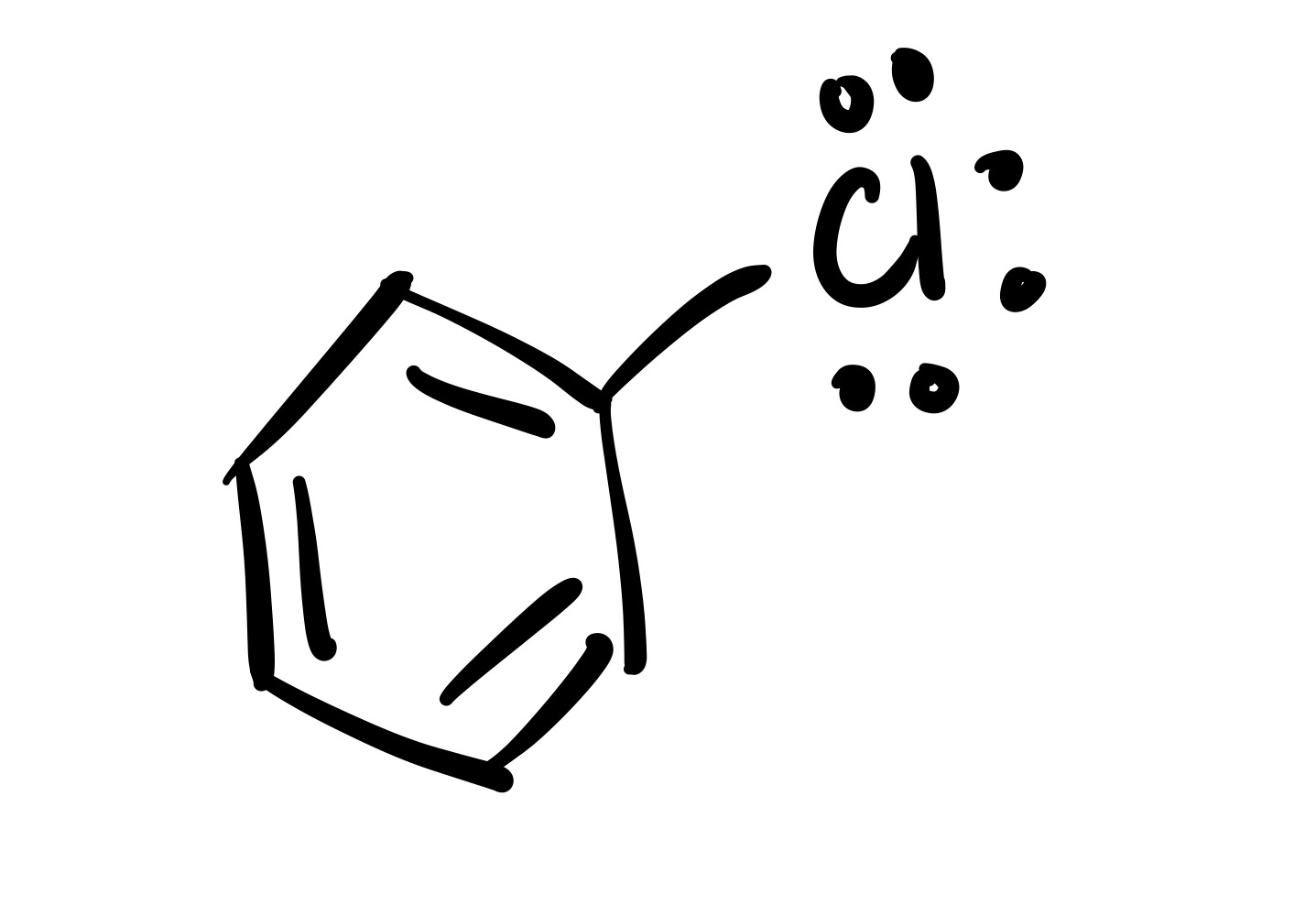
1 Cl group attaches to the cyclohexane (with lone pairs)
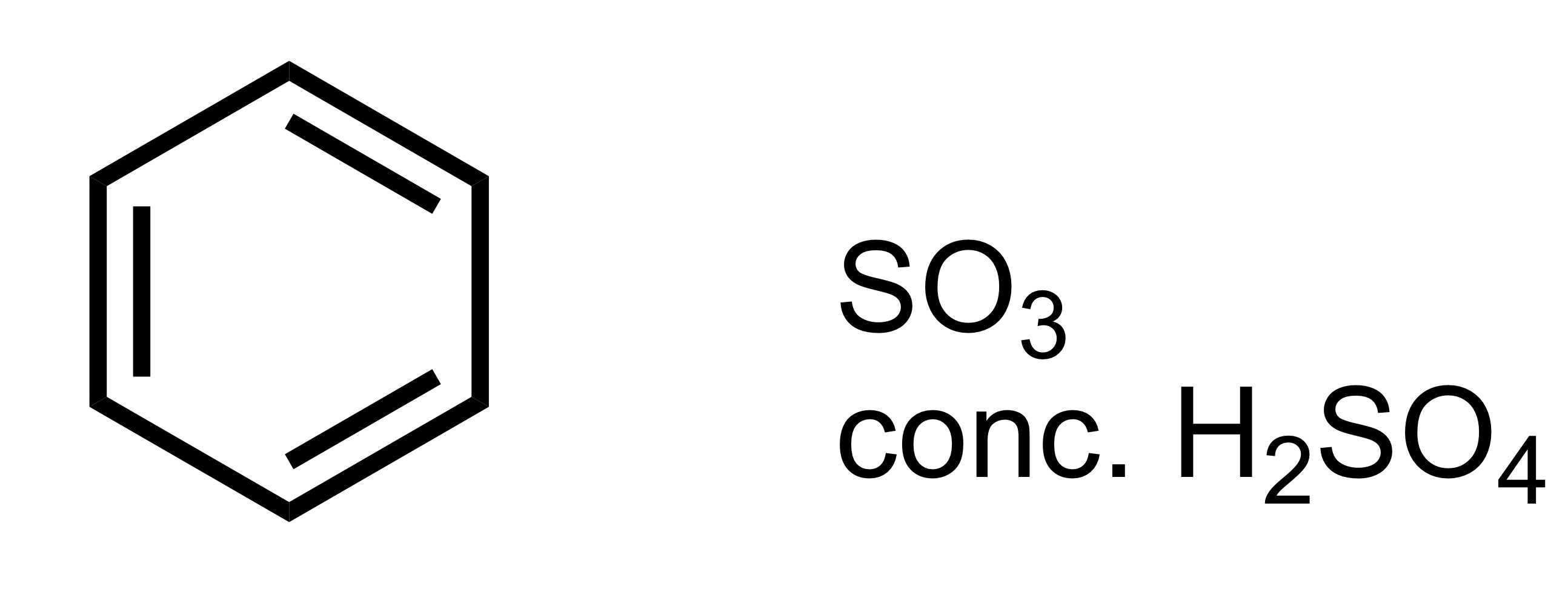
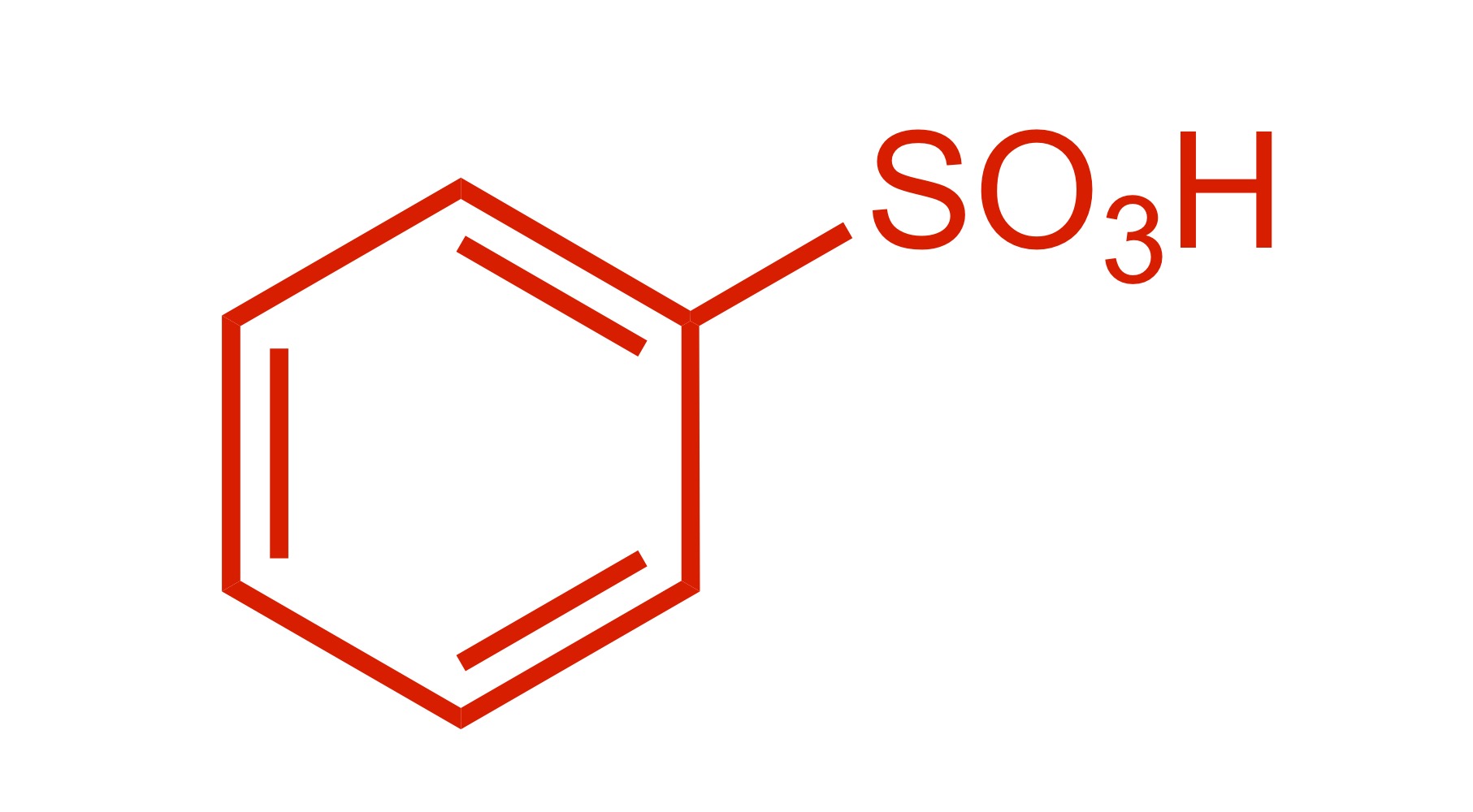
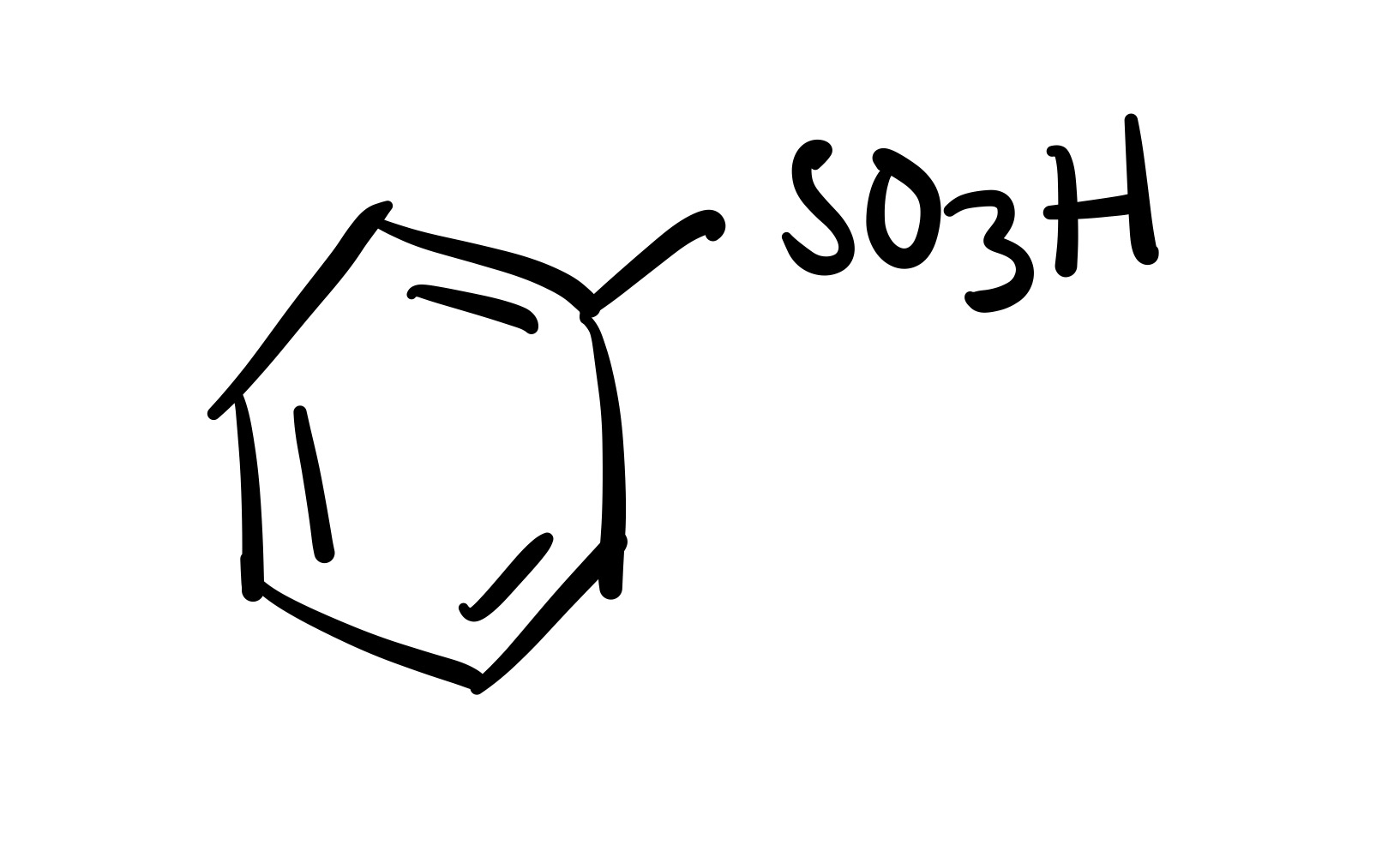
SO3H attaches to the cyclohexane
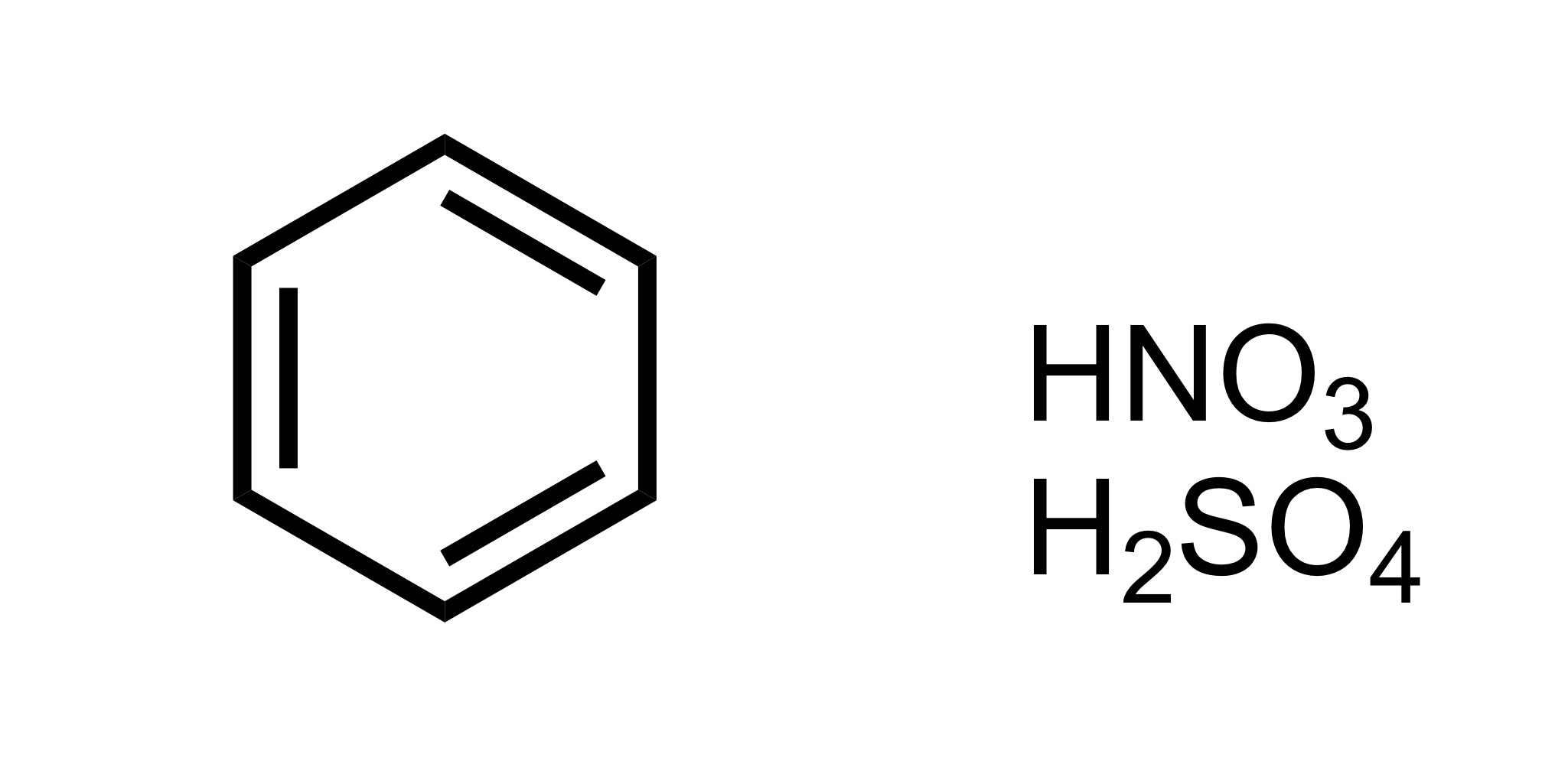
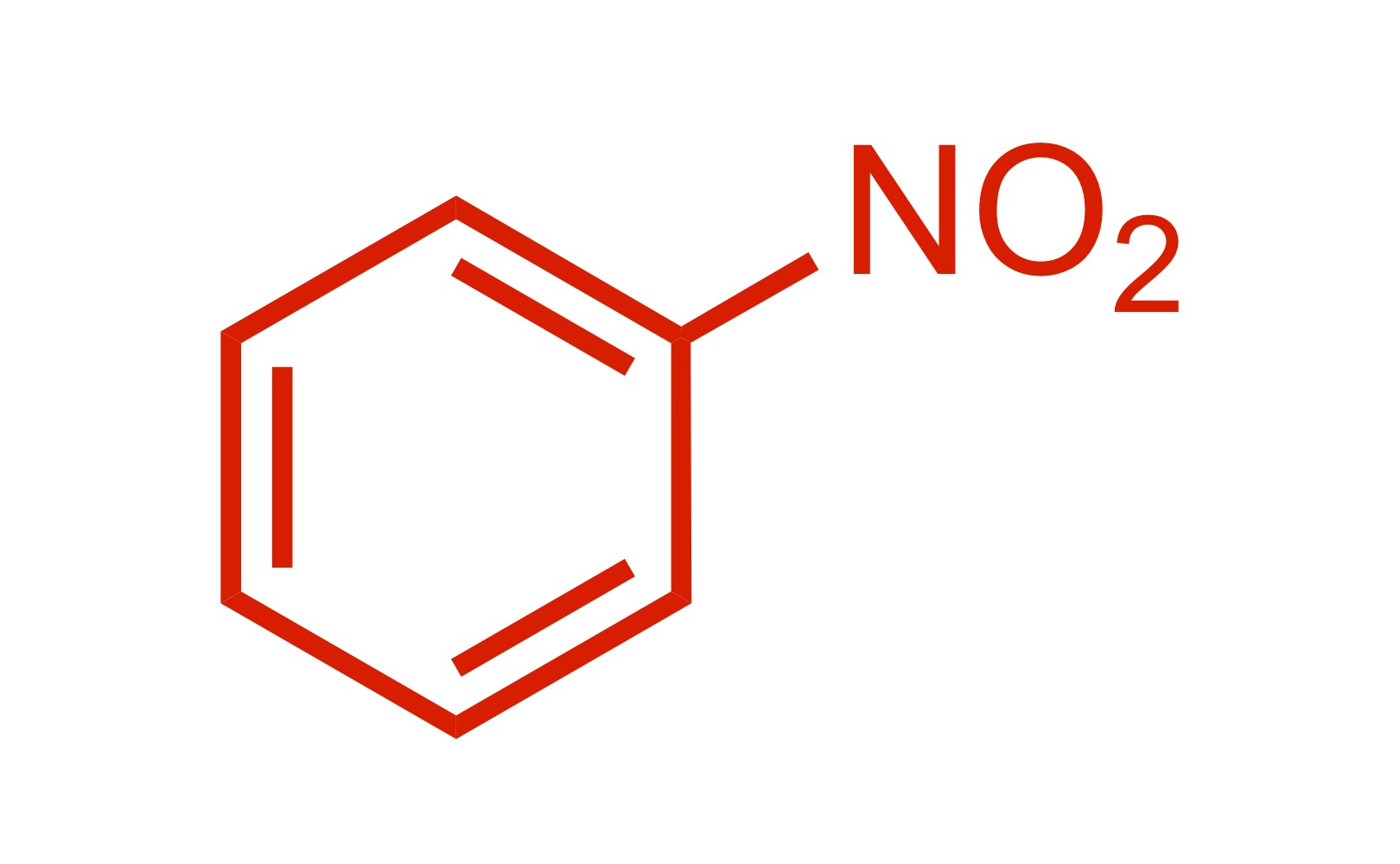
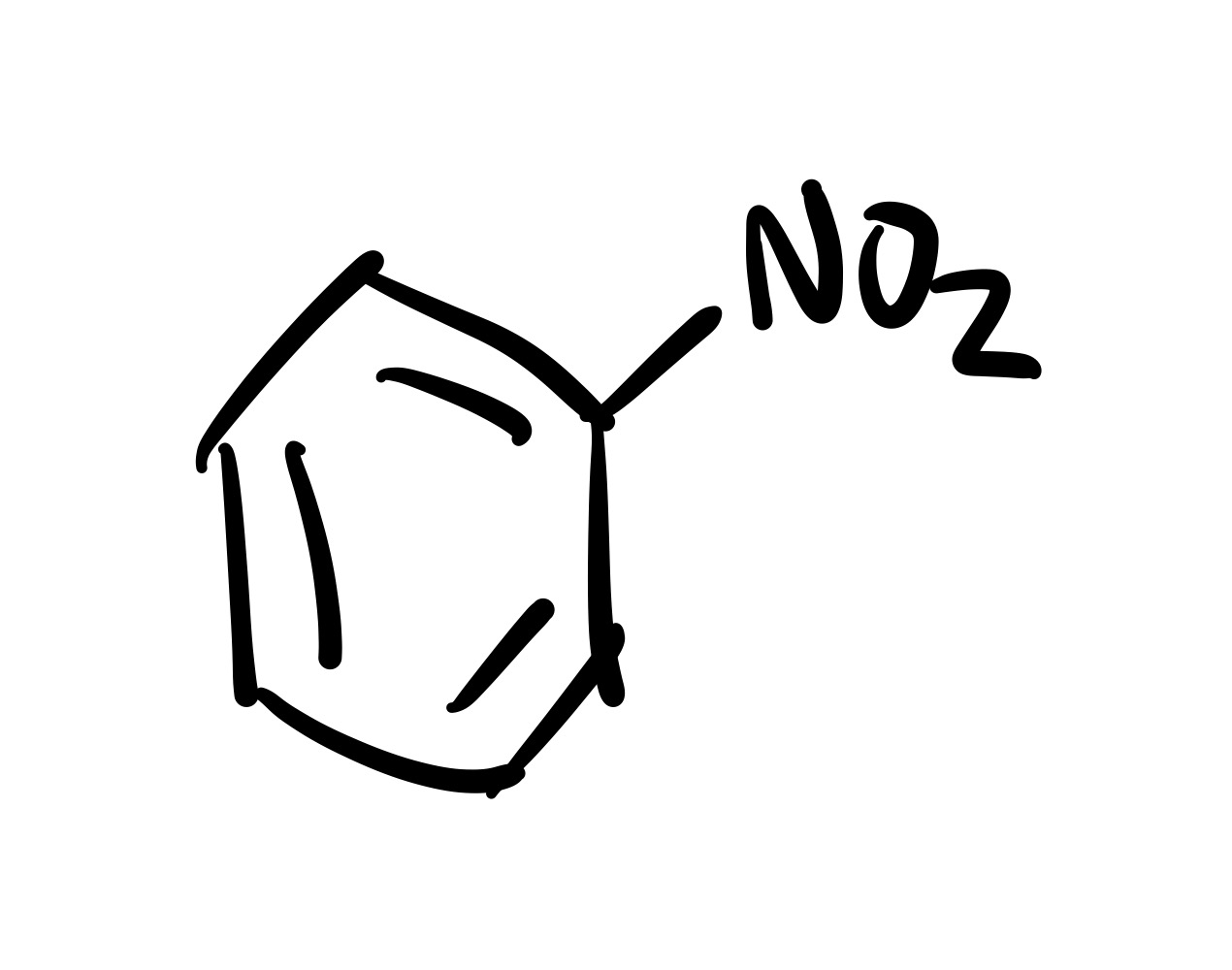
(Ortho) no directors - so the NO2 attaches to the cyclohexane
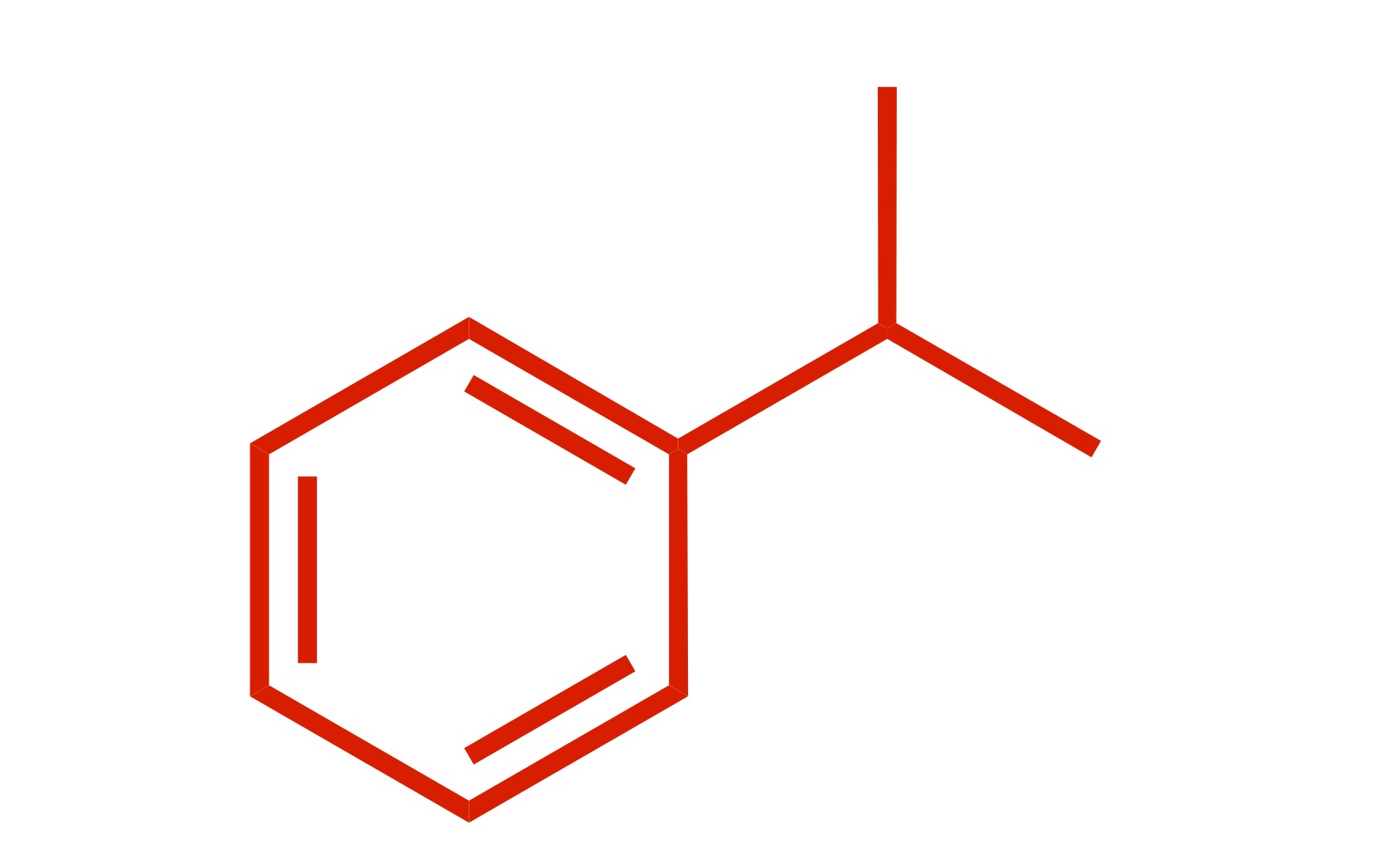
(Ortho) - no directors - the carbon chain attaches to the cyclohexane w/out the Cl
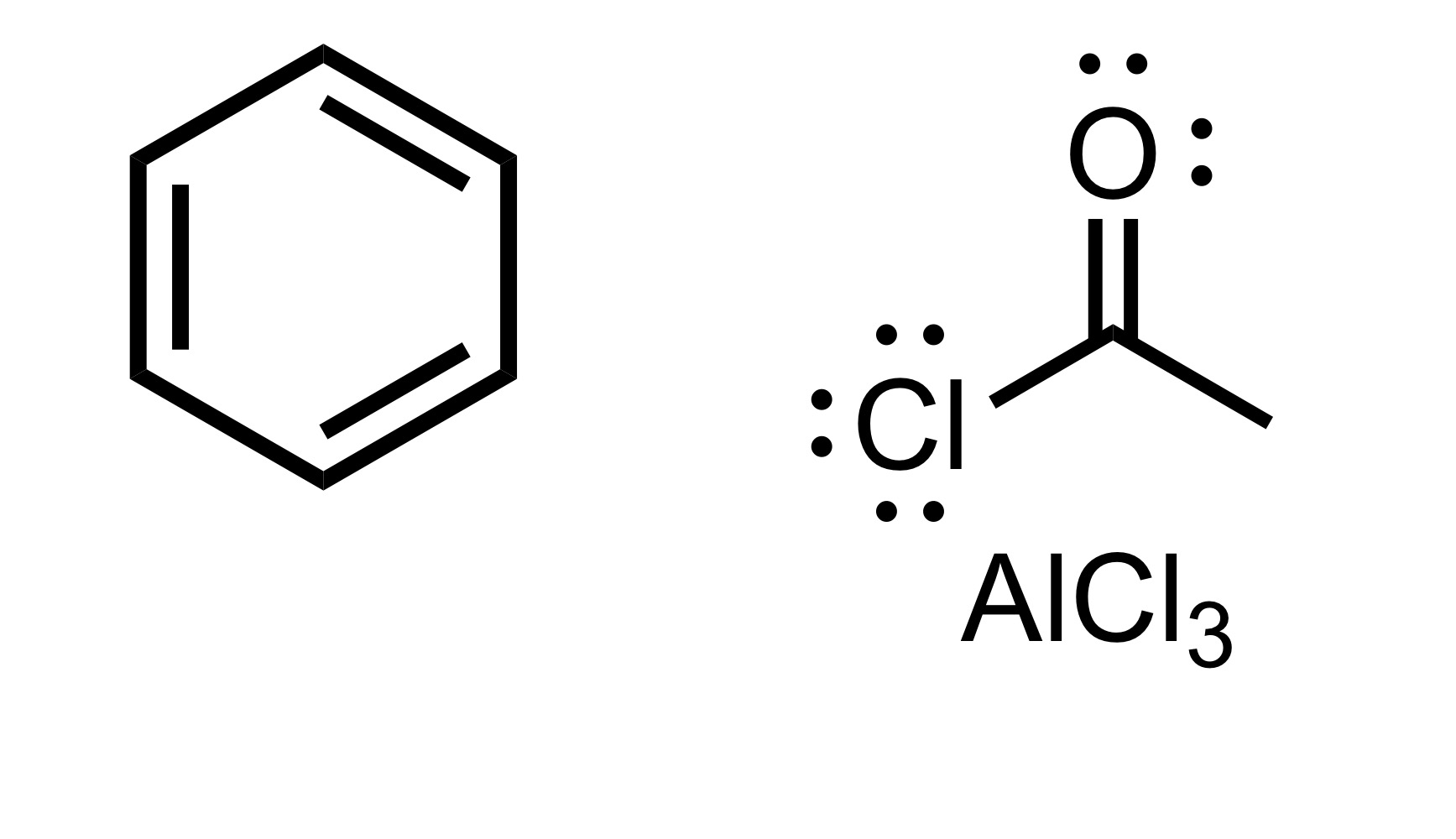
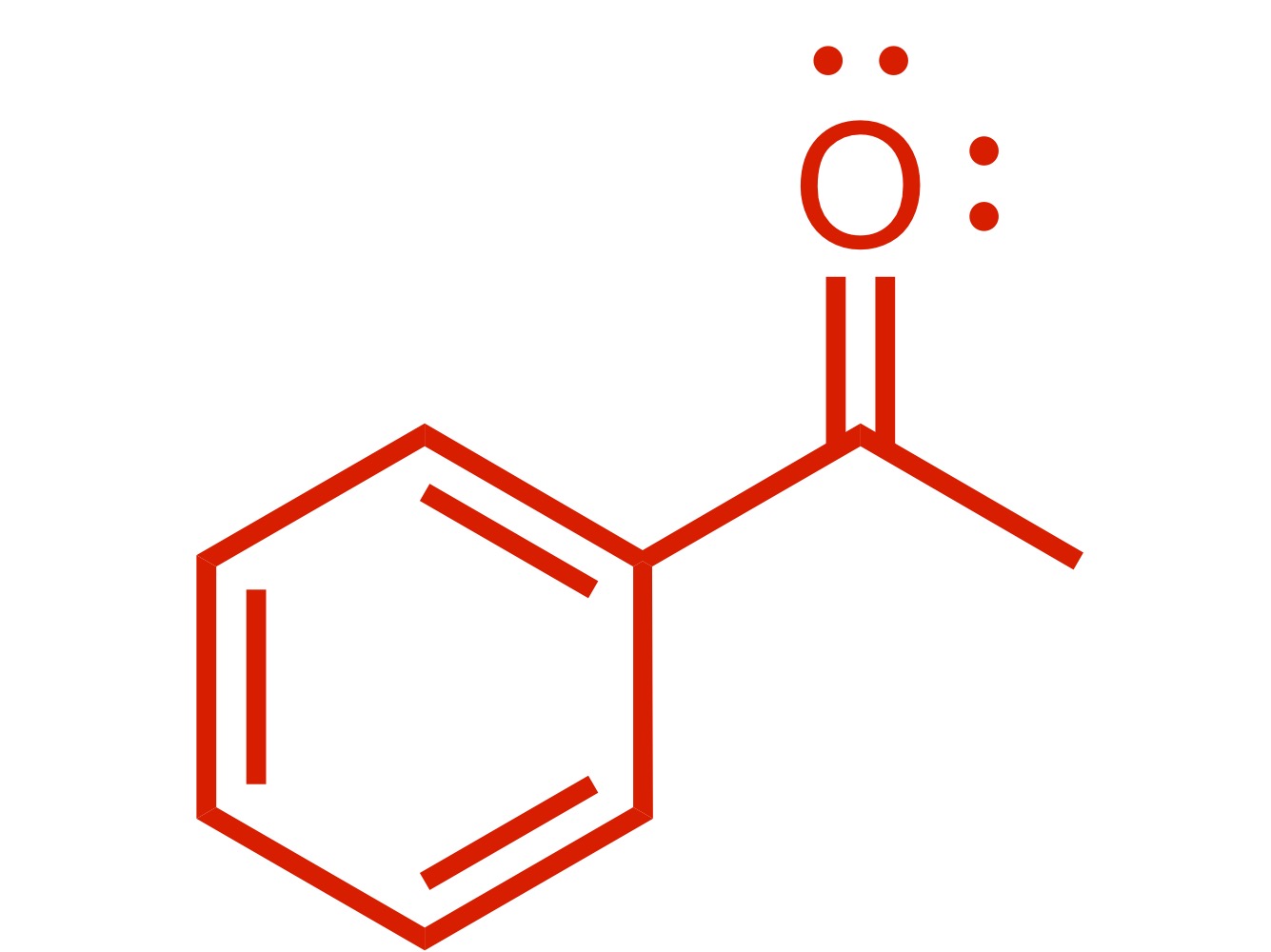
(Ortho) - no directors - the carbon chain attaches to the cyclohexane w/out the Cl
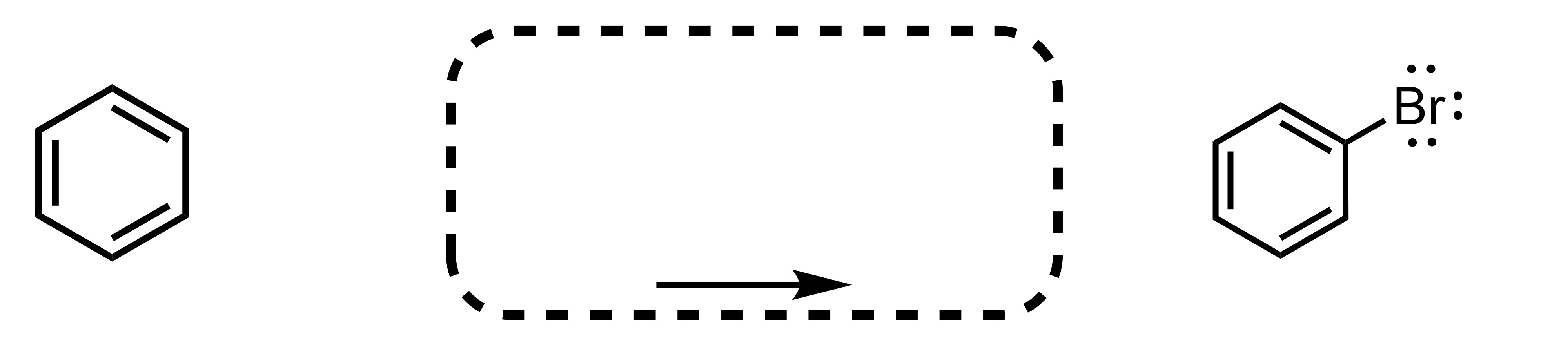
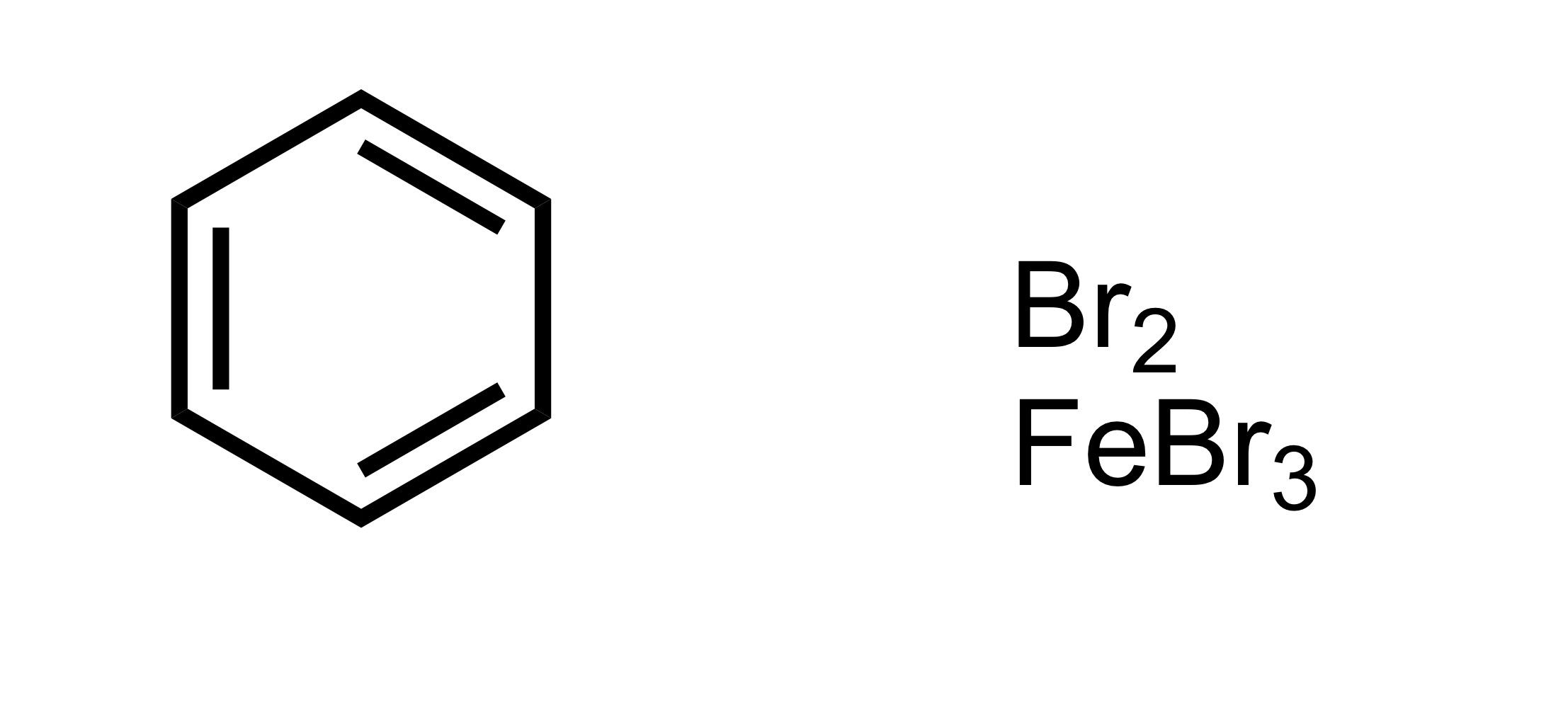
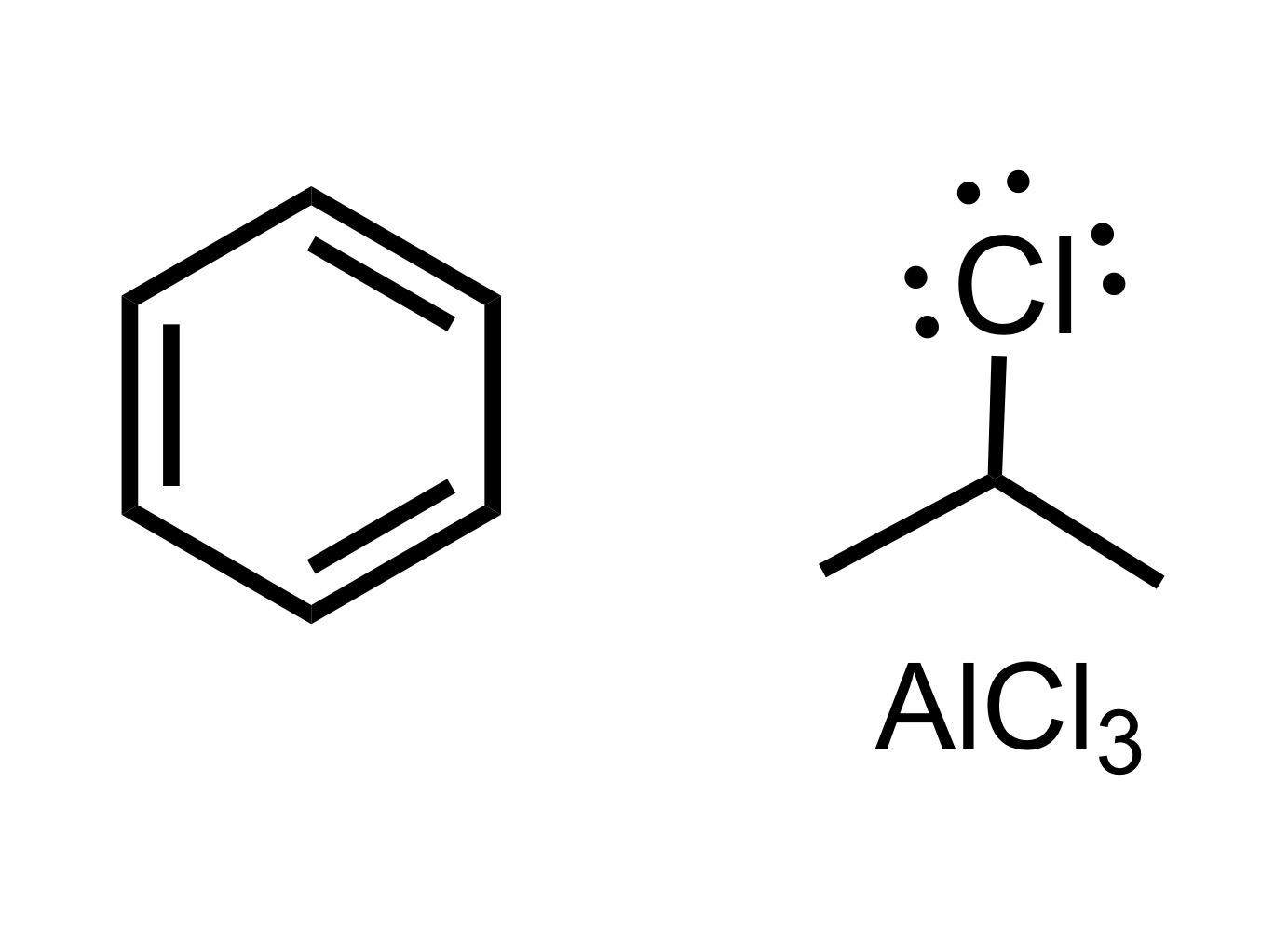
Fill in the missing reagent
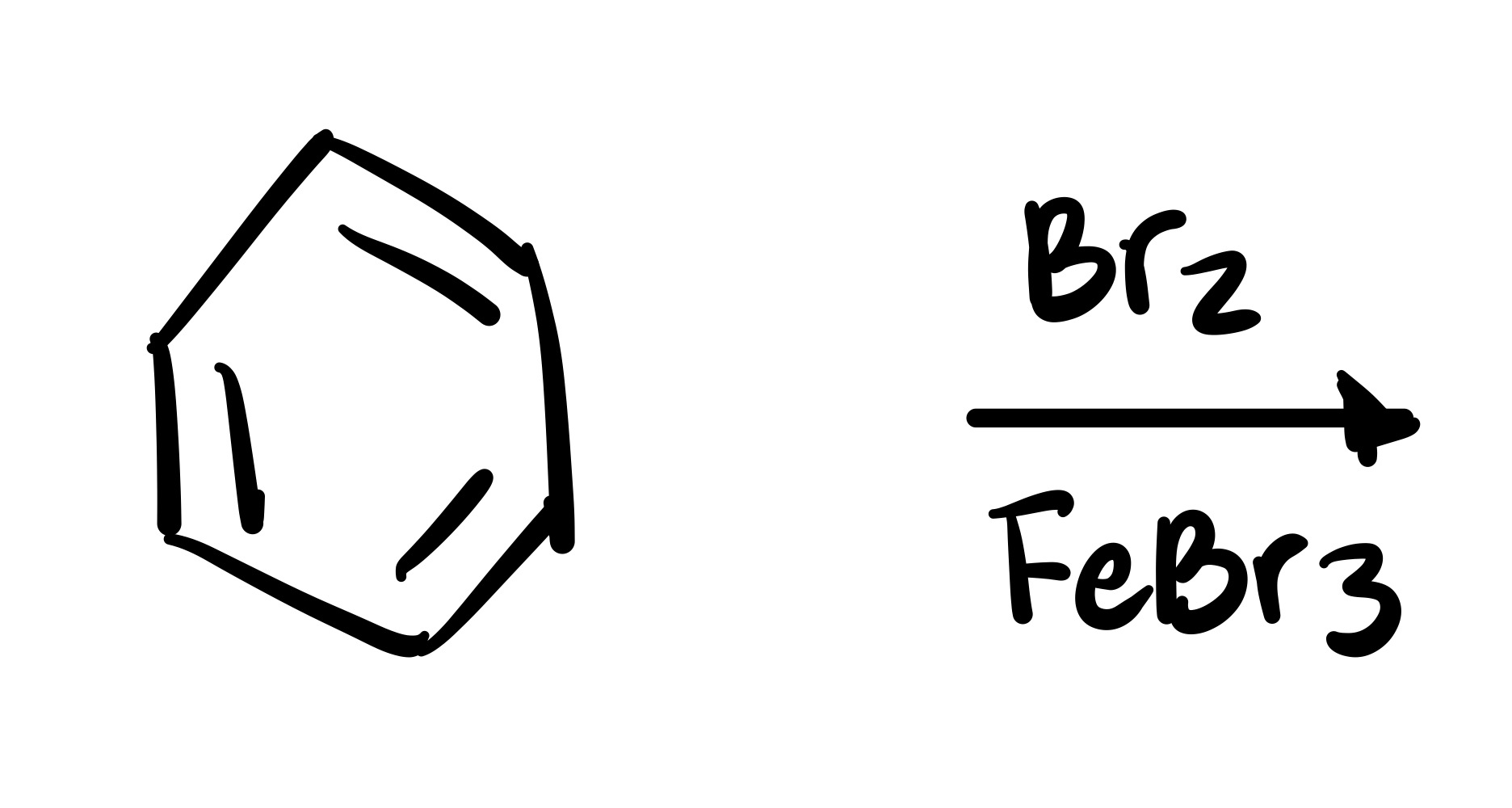
The Br attaches to the cyclohexane - BR2, FeBR3 is the reagent
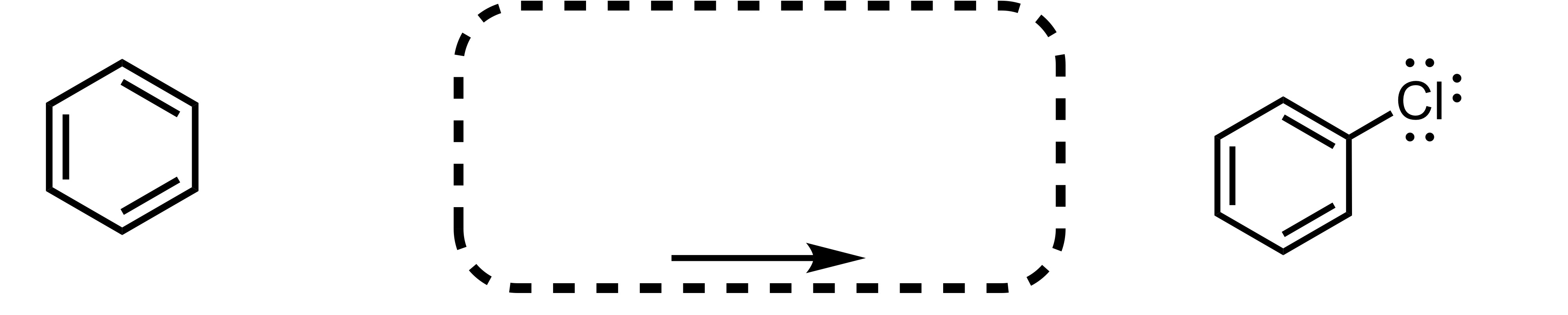
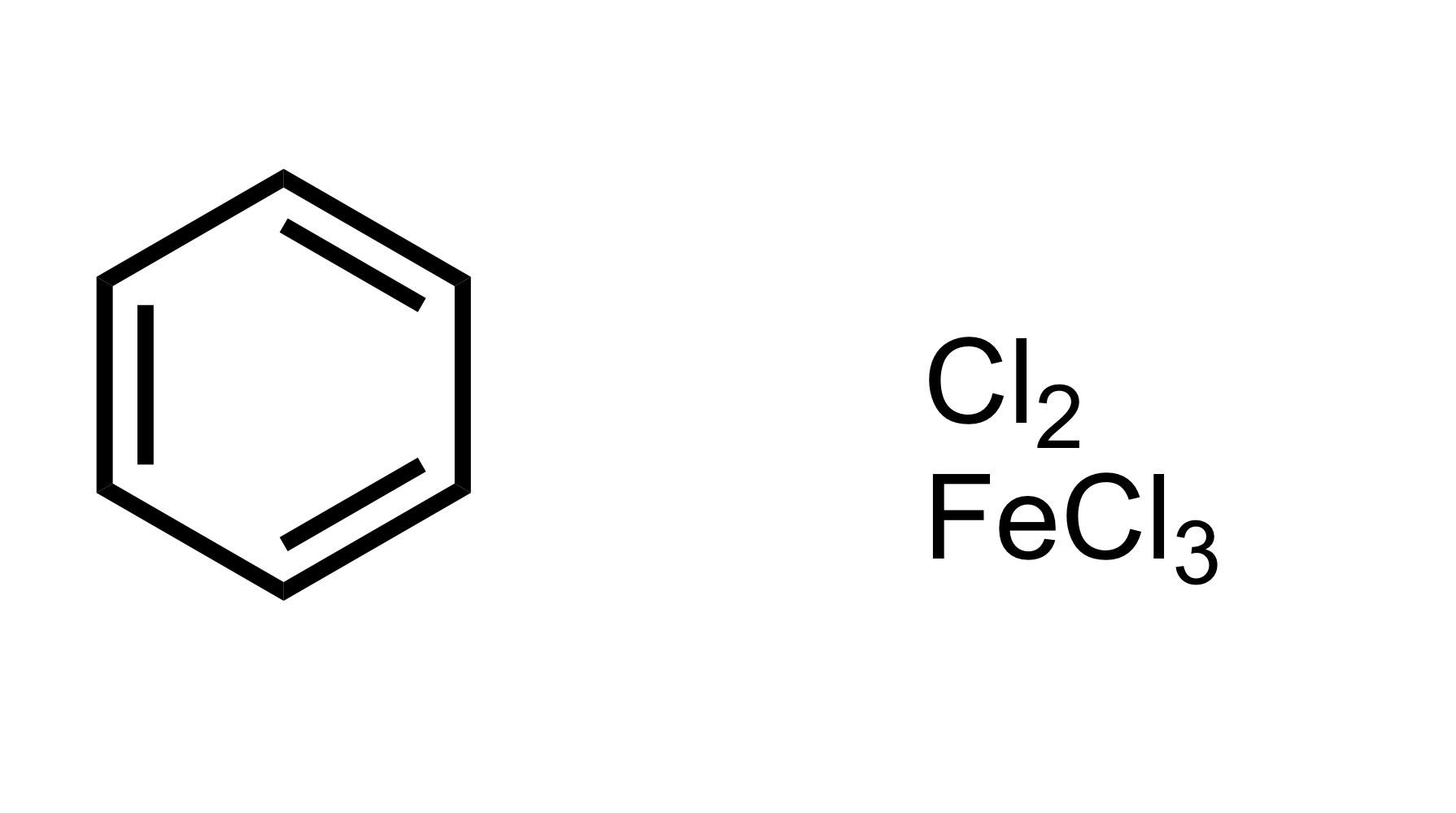
Fill in the missing reagent
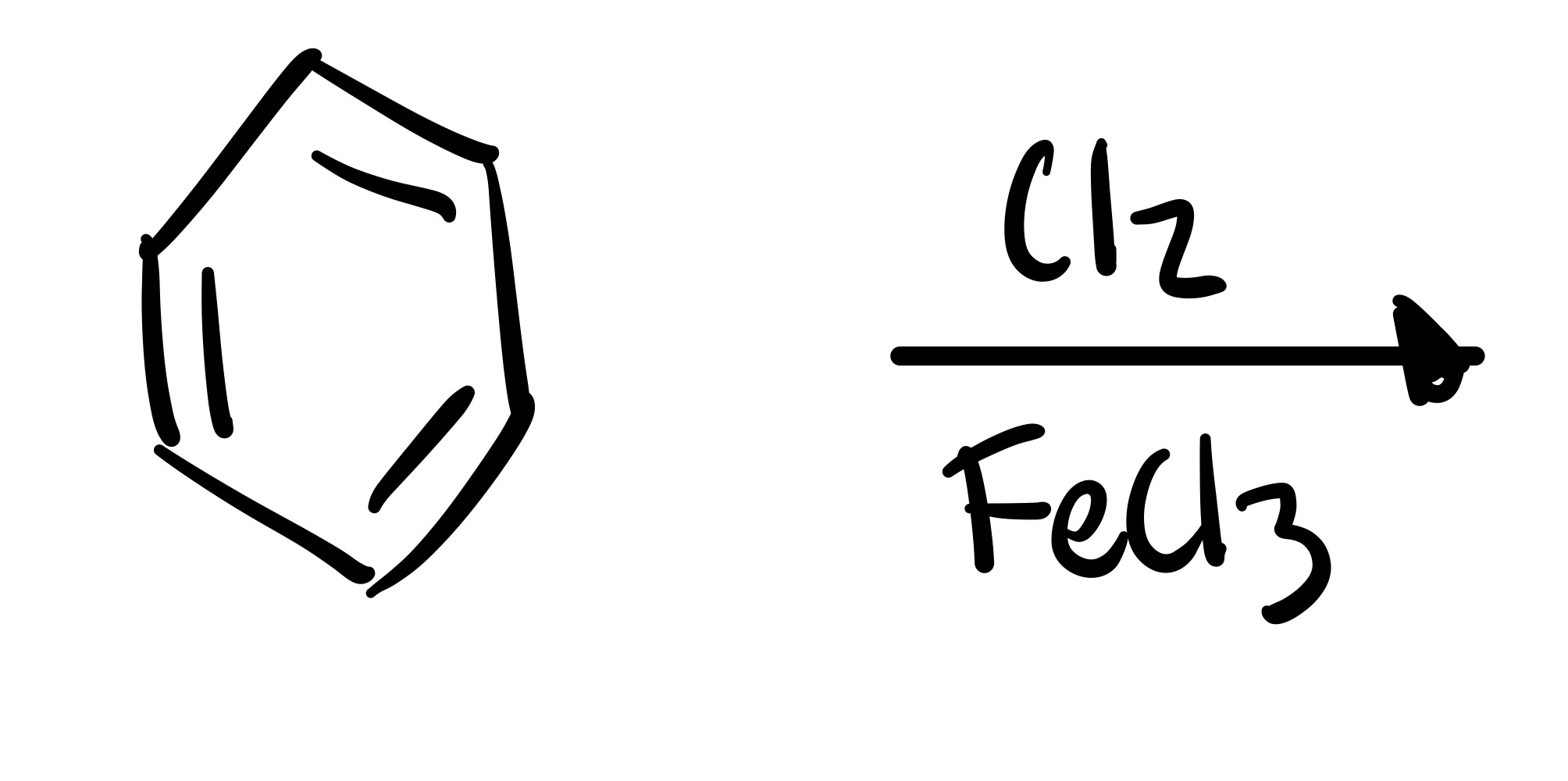
The Cl attaches to the cyclohexane - Cl2 FeCl3 is the reagent

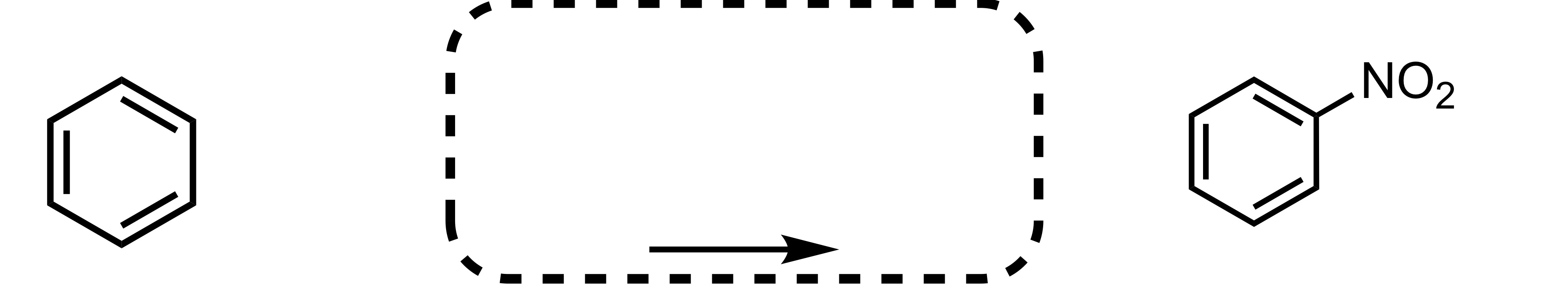
Fill in the missing reagent
NO2 attaches to the cyclohexane - HNO3, H2SO4
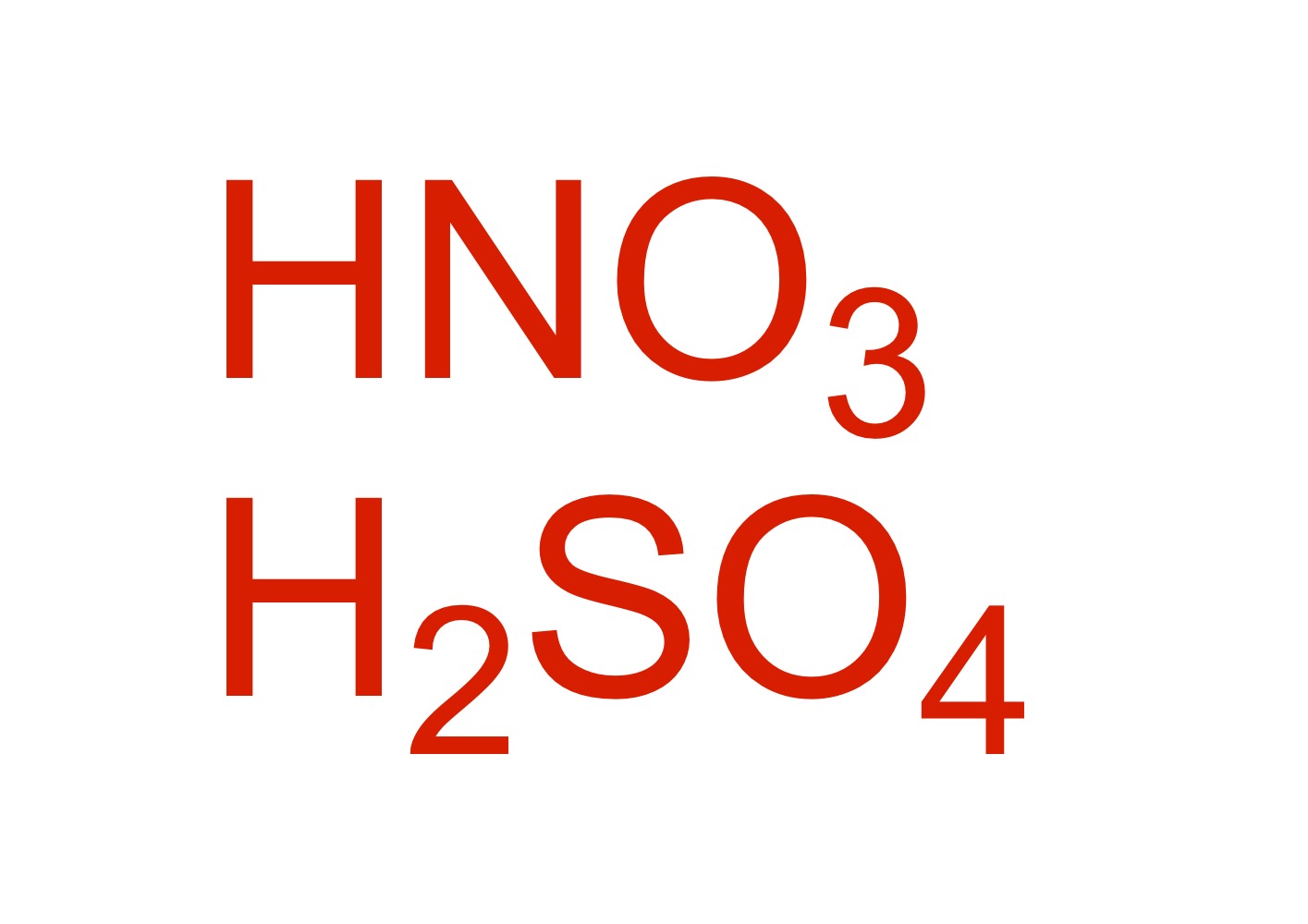
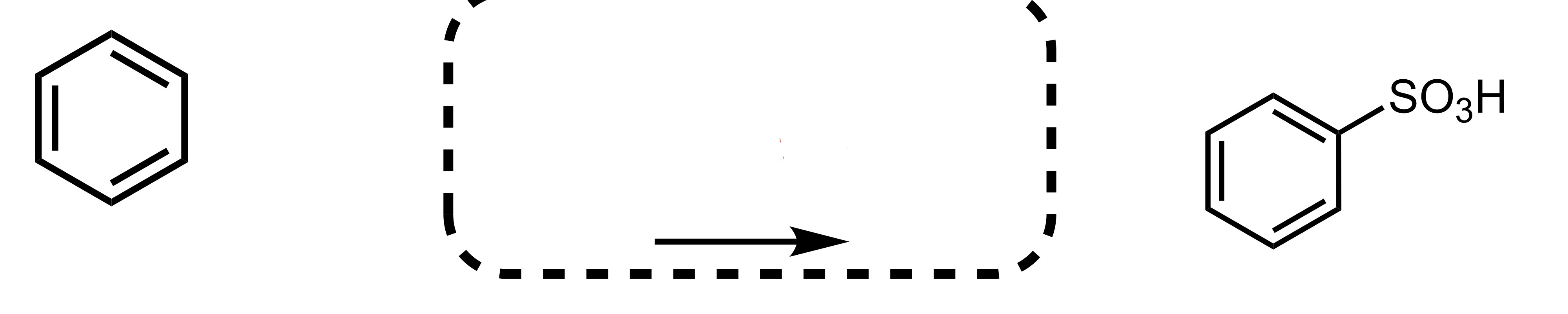
Fill in the missing reagent
SO3H attaches to the carbon chain - SO3, conc. H2SO4 is the reagent
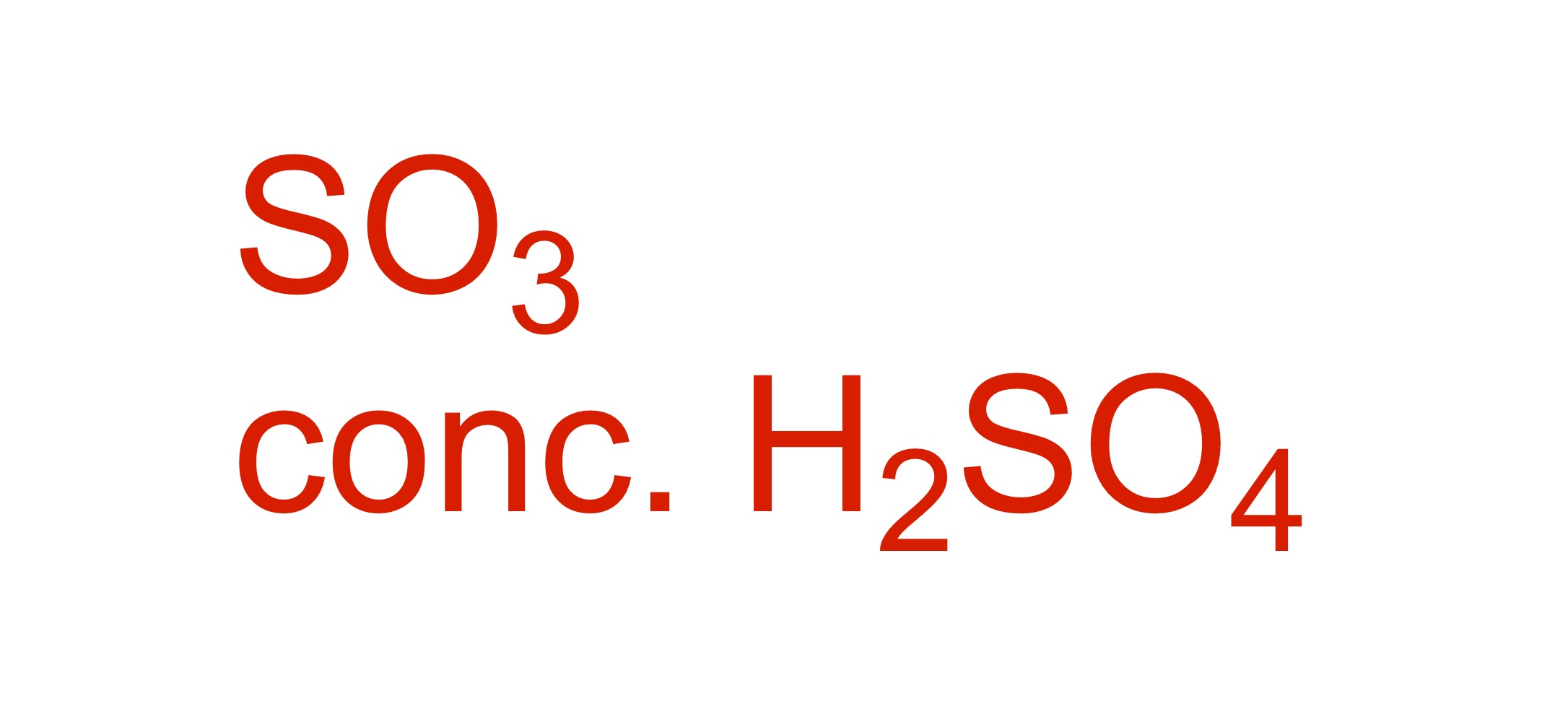
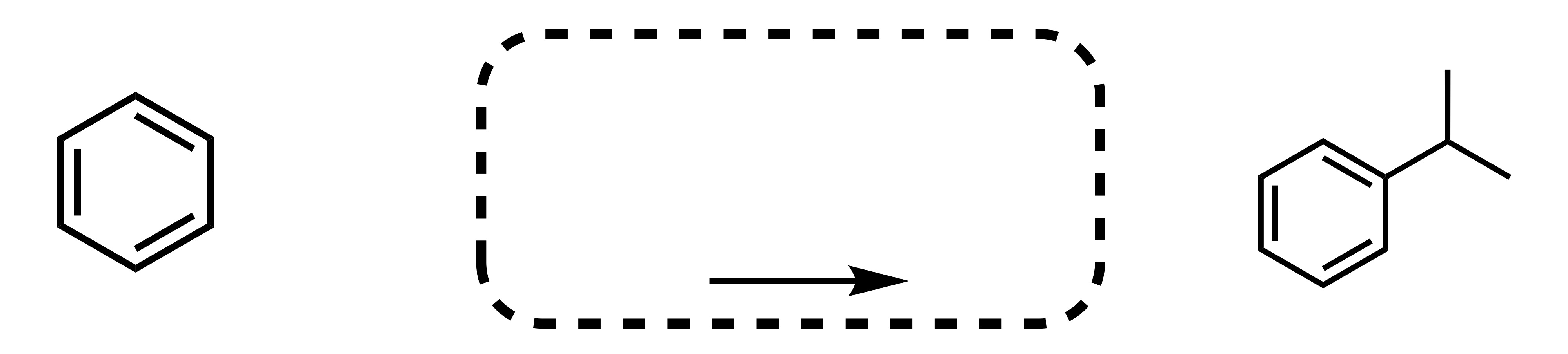
Fill in the missing reagent
Carbon chain attaches to the cyclohexane - (the Cl leaves the carbon chain). The Cl carbon group with AlCl3 is the reagent
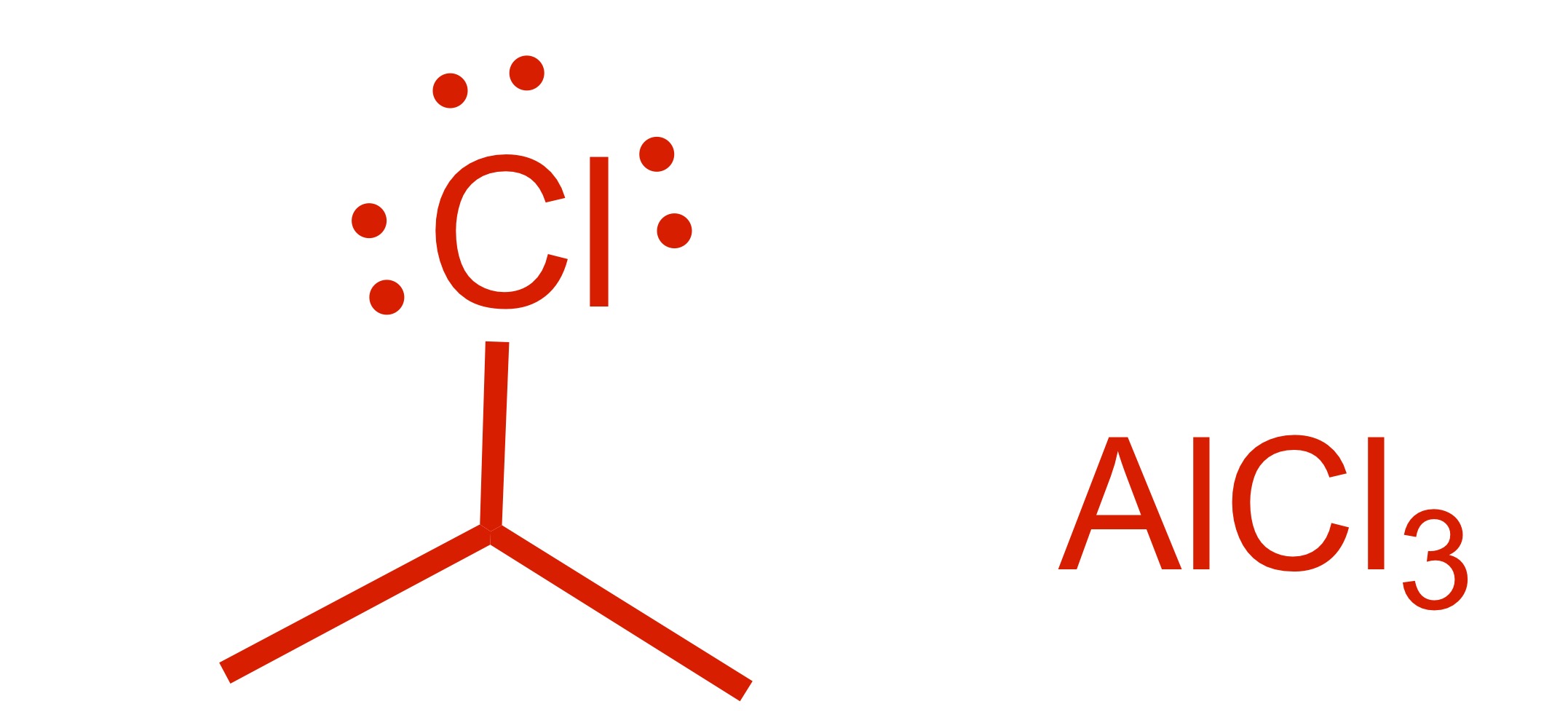
Fill in the missing reagent
Carbon chain attaches to the cyclohexane w/ double O bond - (the Cl leaves the carbon chain). The Cl carbon group w/ attached double O bond, with AlCl3 is the reagent
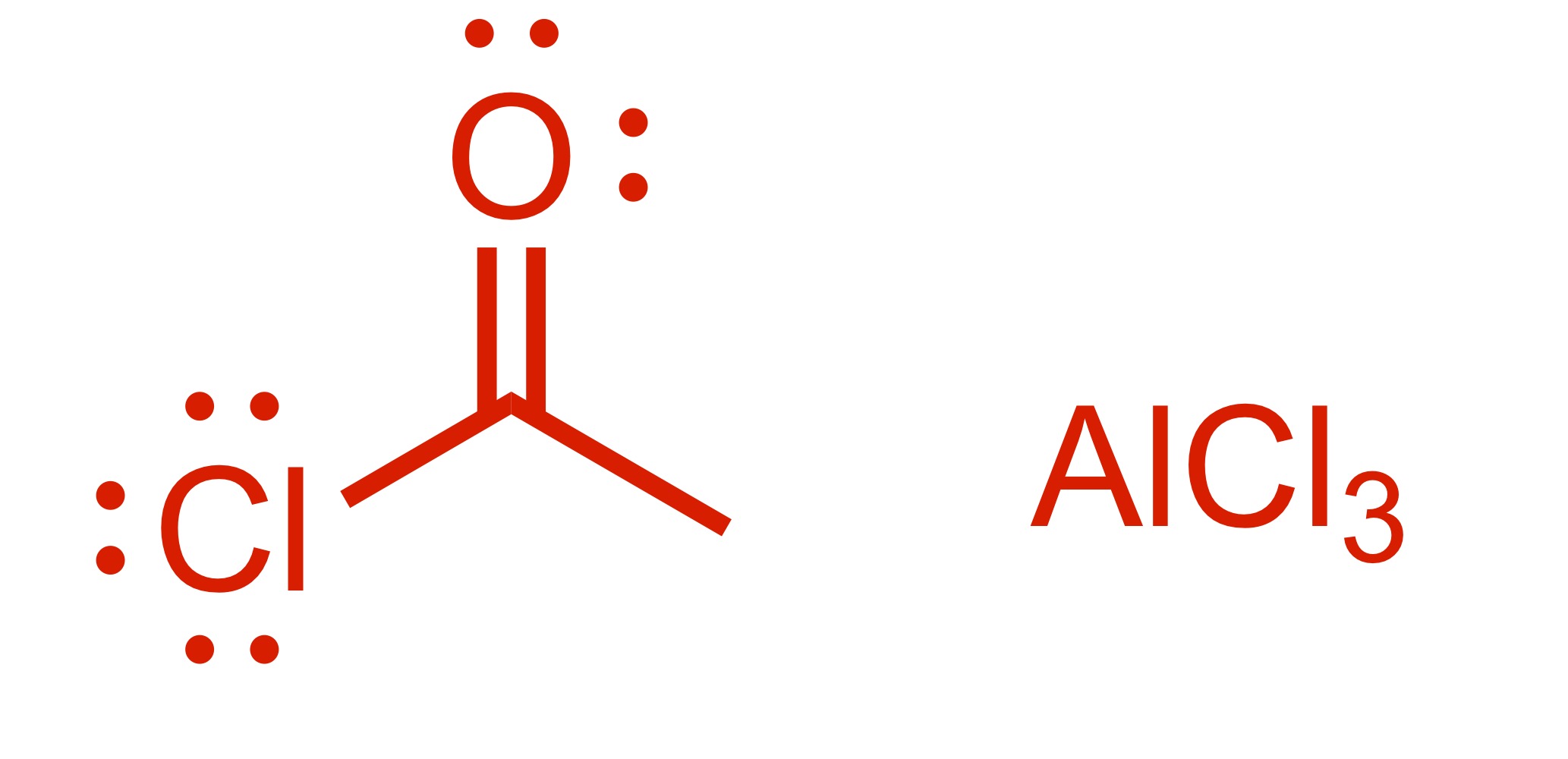
Fill in the missing reagent
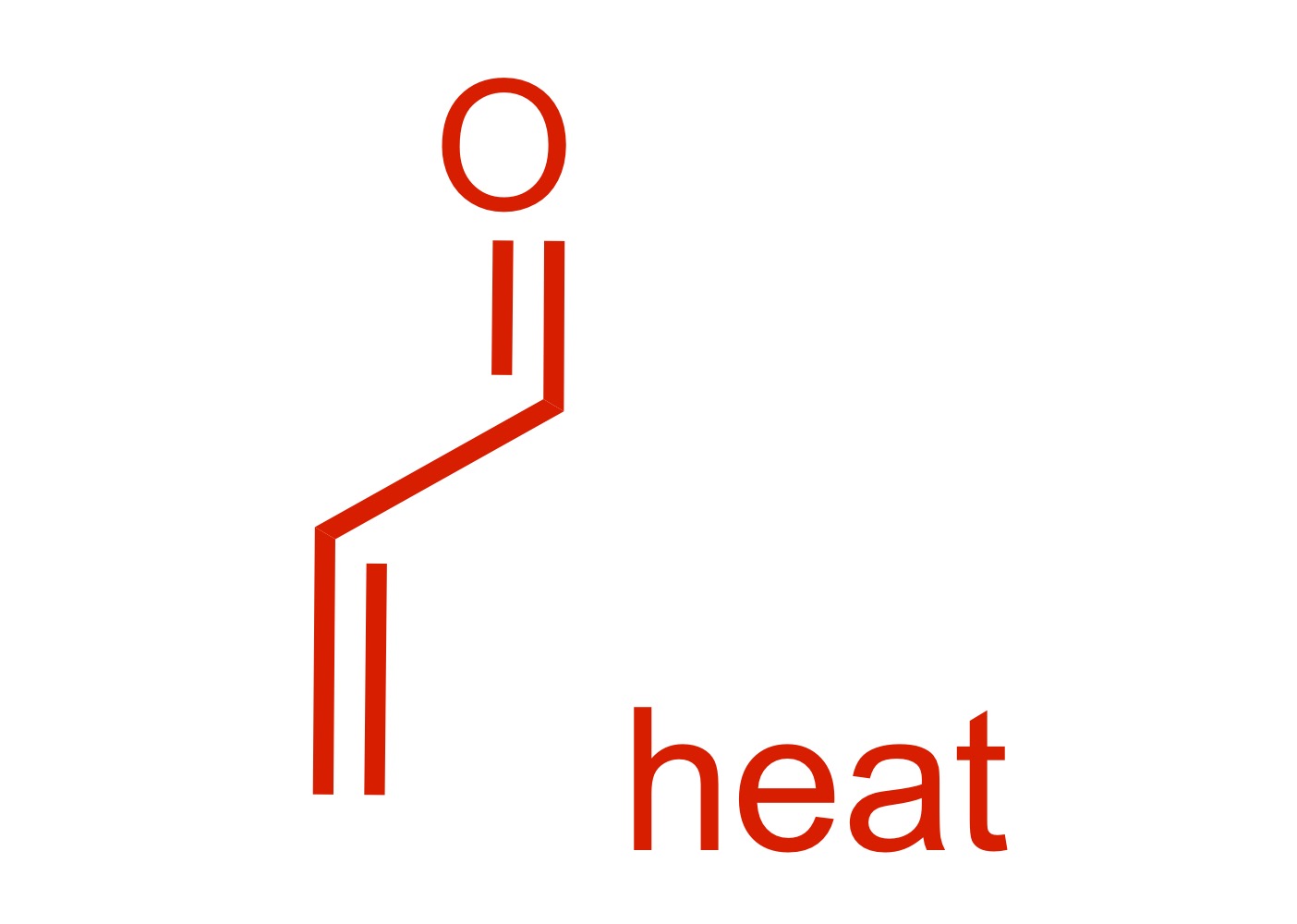
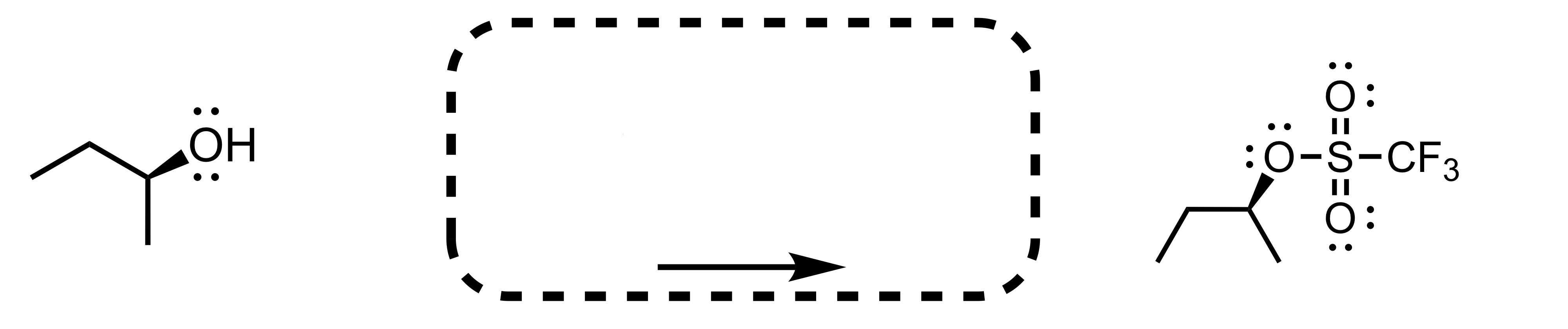
Fill in the missing reagent
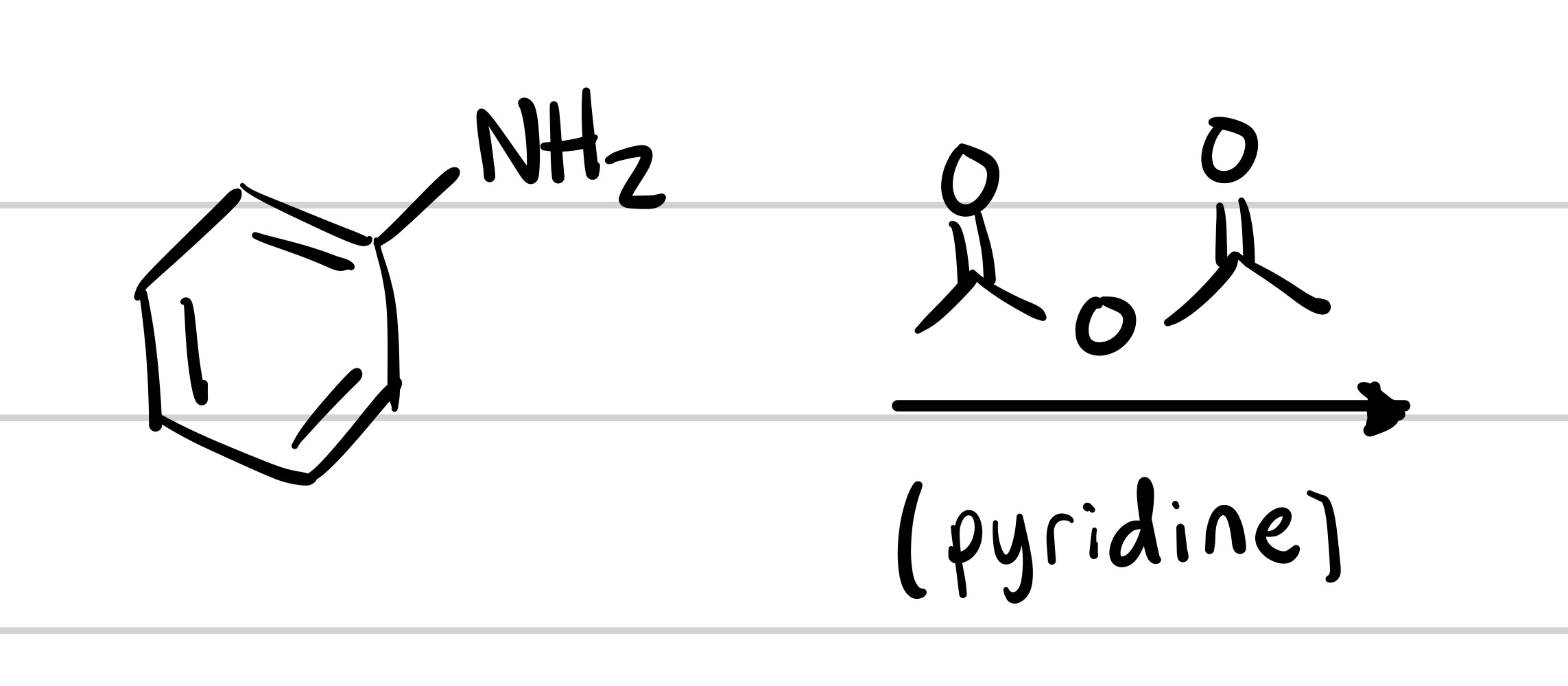
(Substitution) The H leaves the OH, and the pyridine adds. The structure and pyridine is the reagent
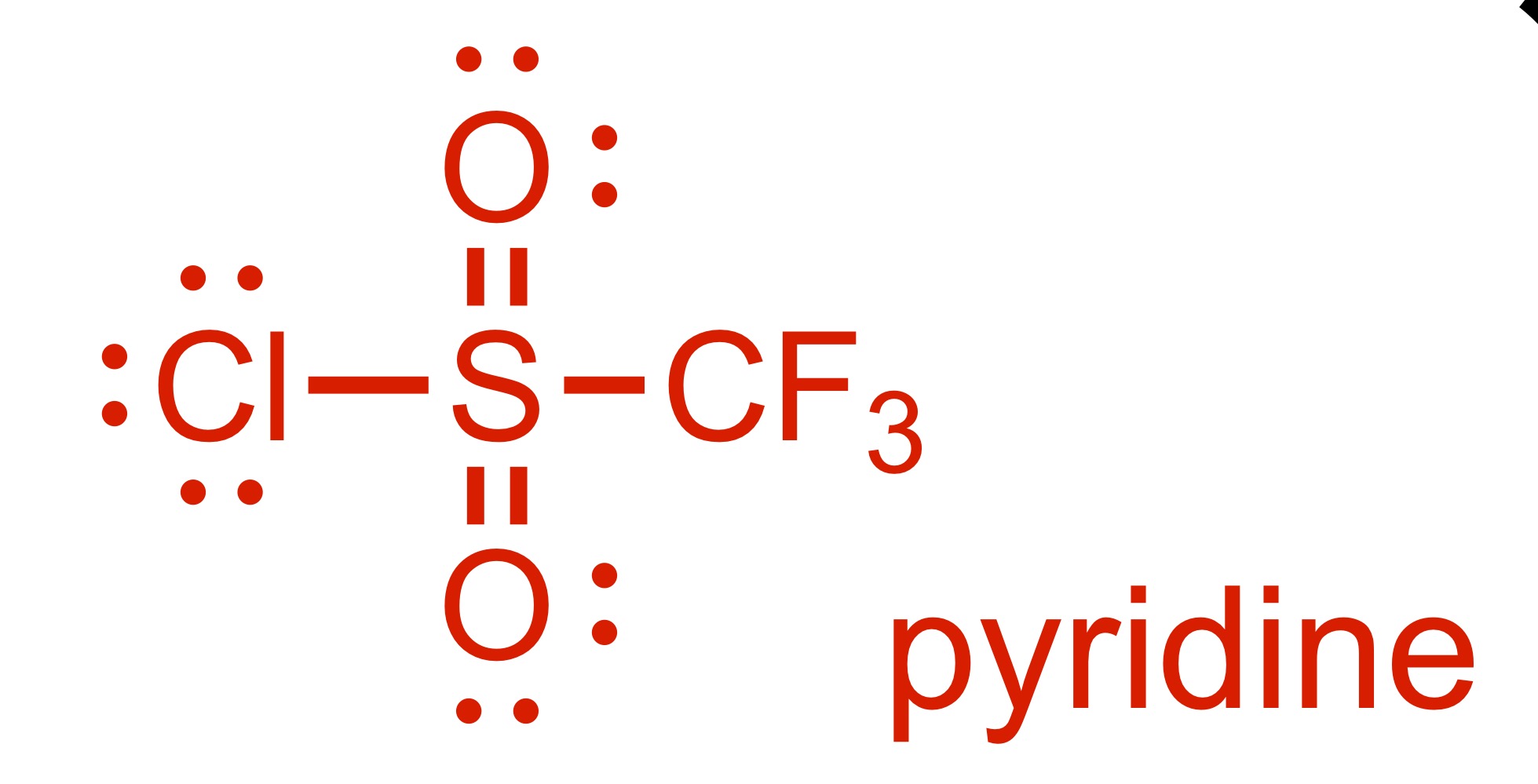
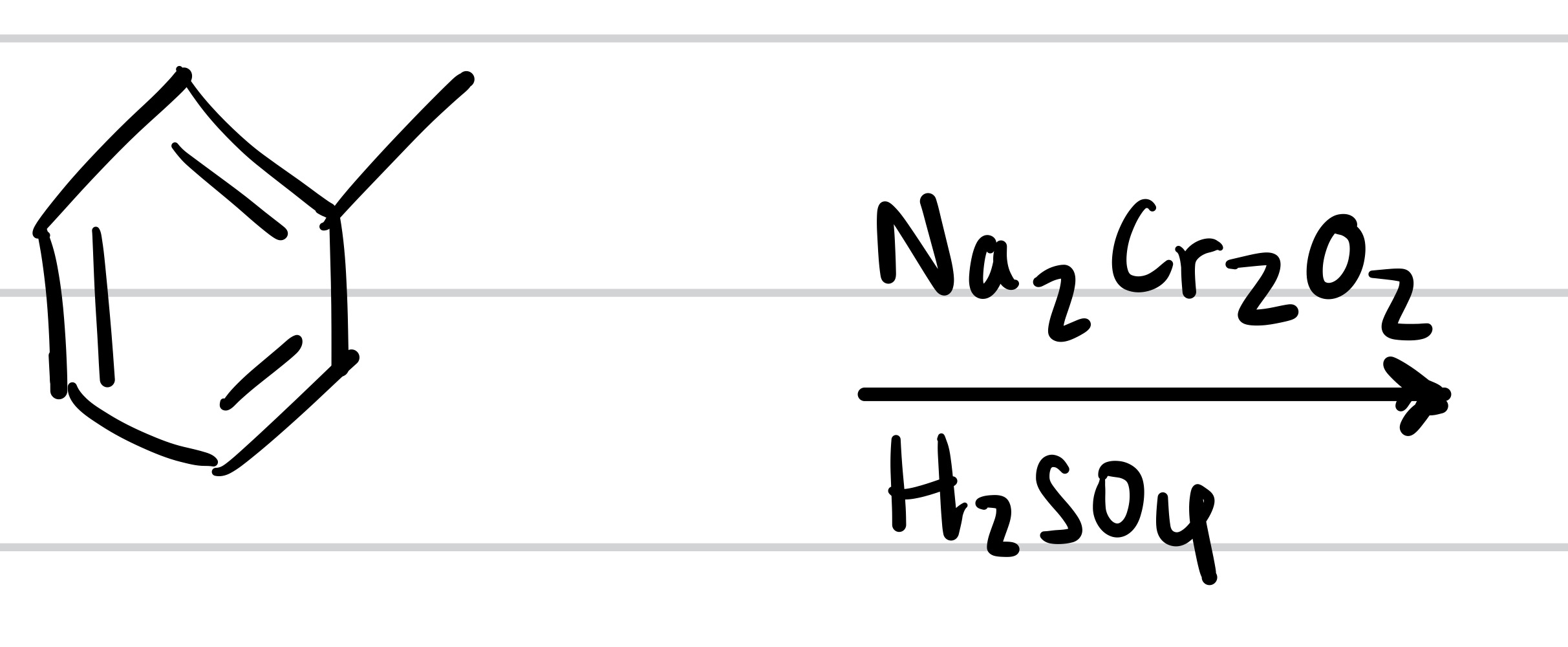
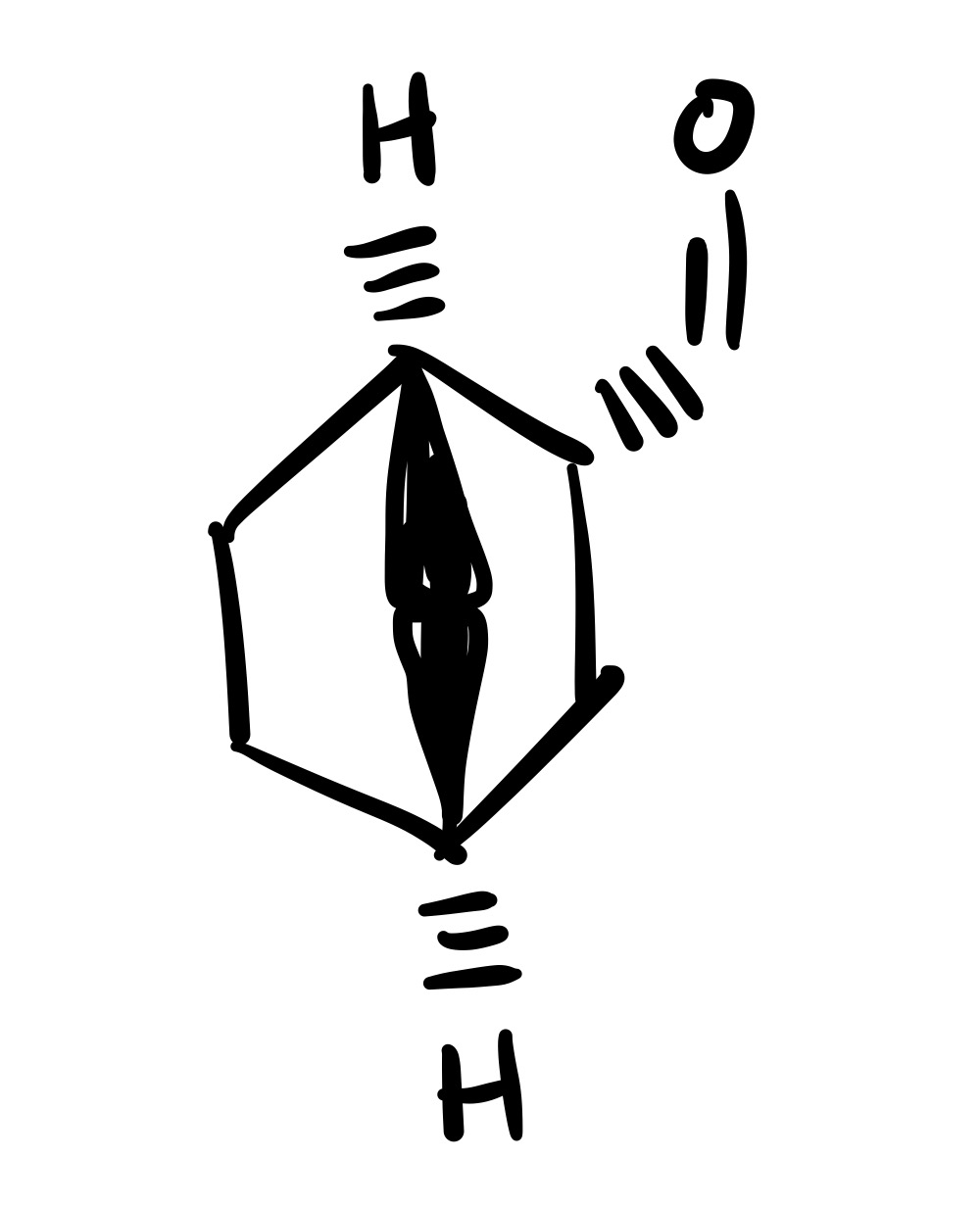
The double O bond adds to the methyl, and so does the OH
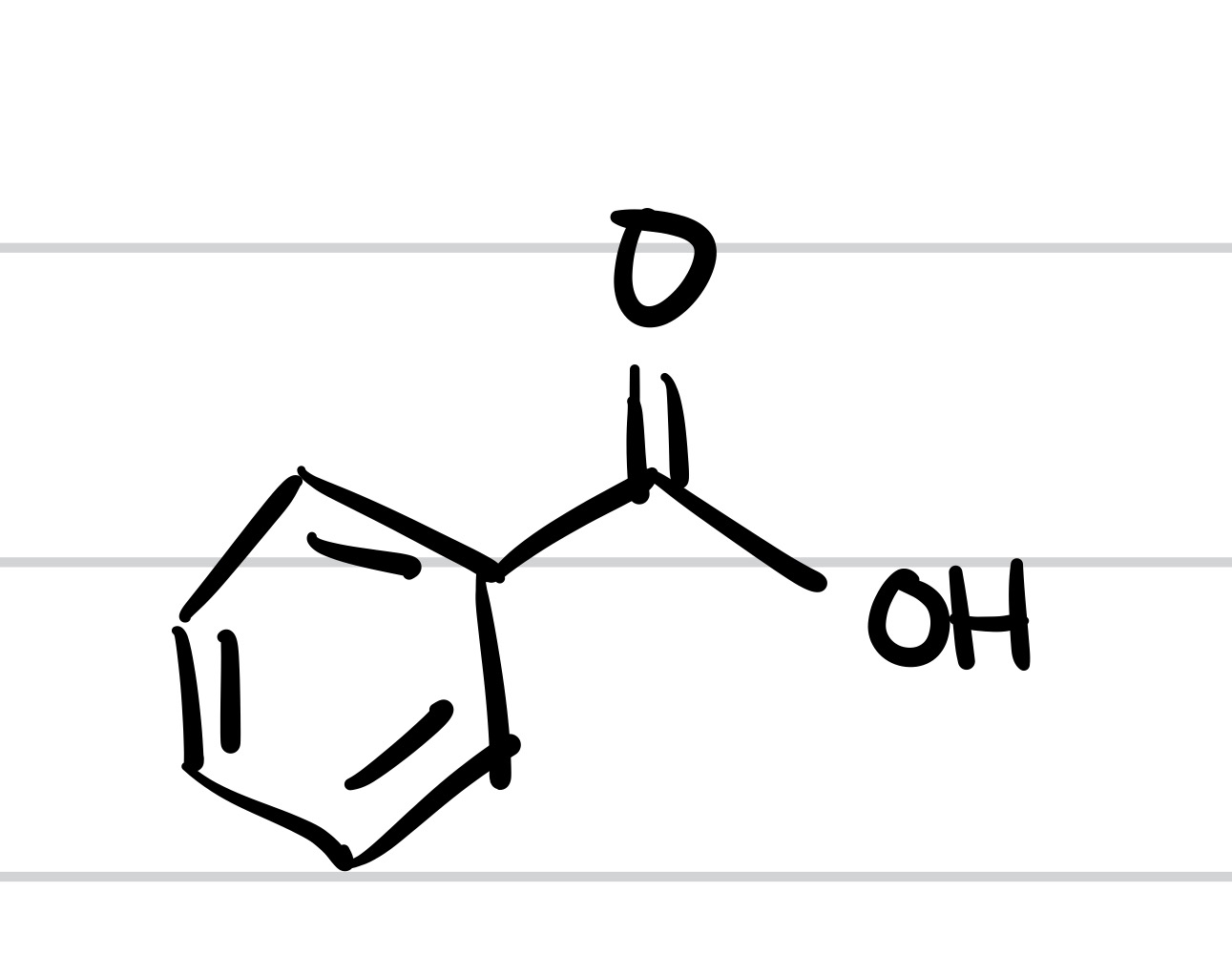
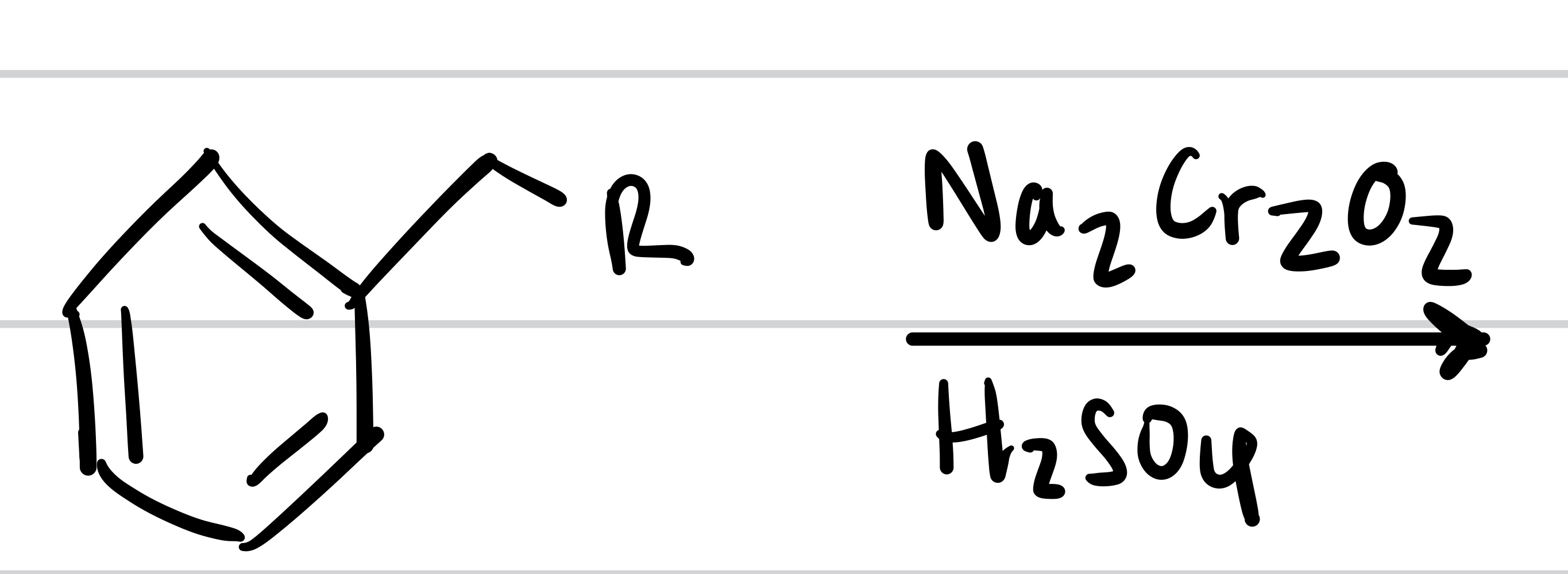
The double O bond adds to the methyl, and so does the OH
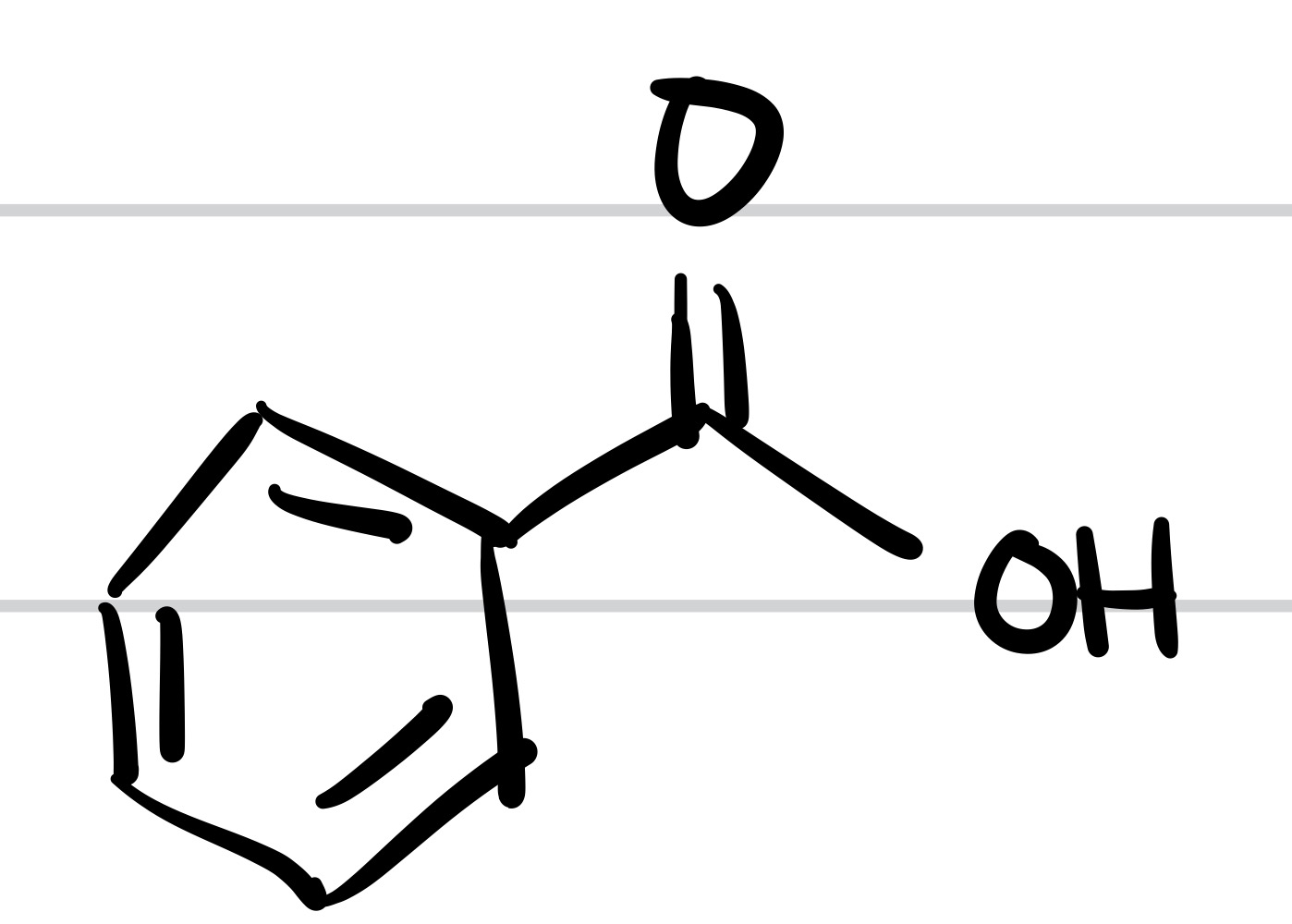
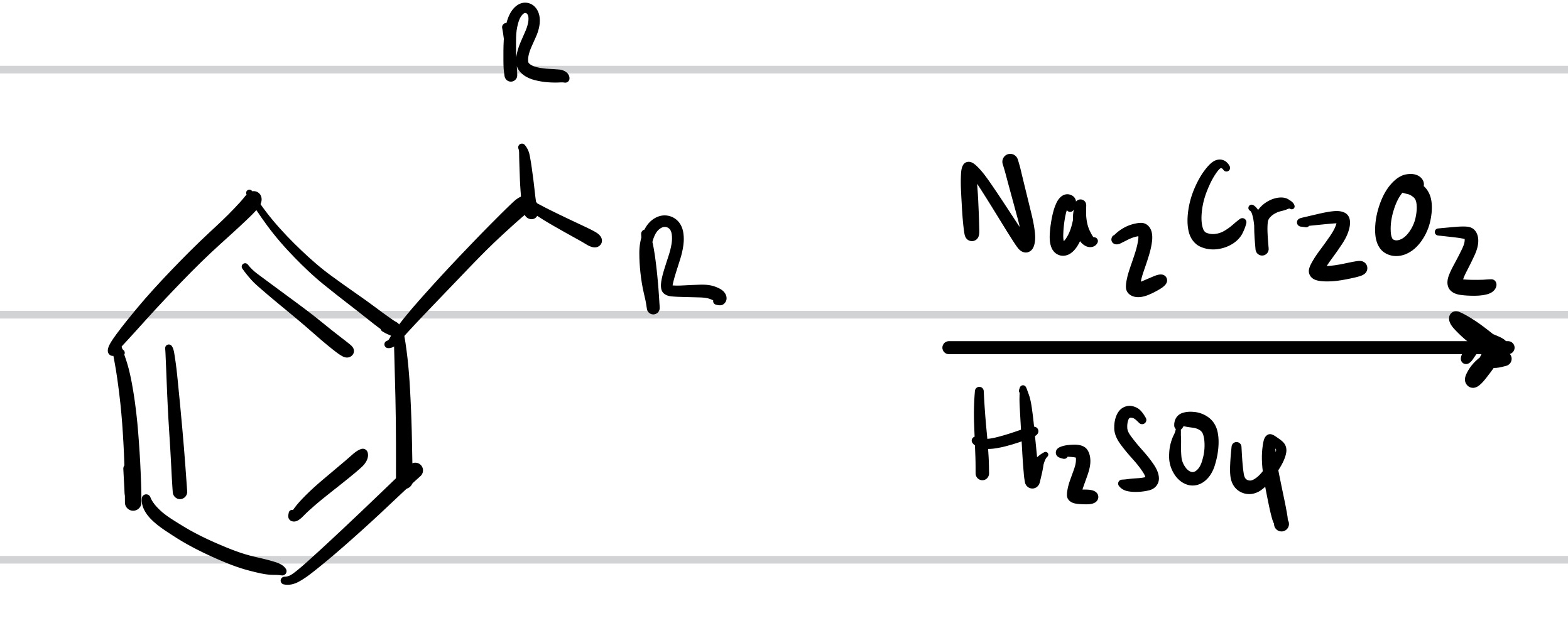
The double O bond adds to the methyl, and so does the OH
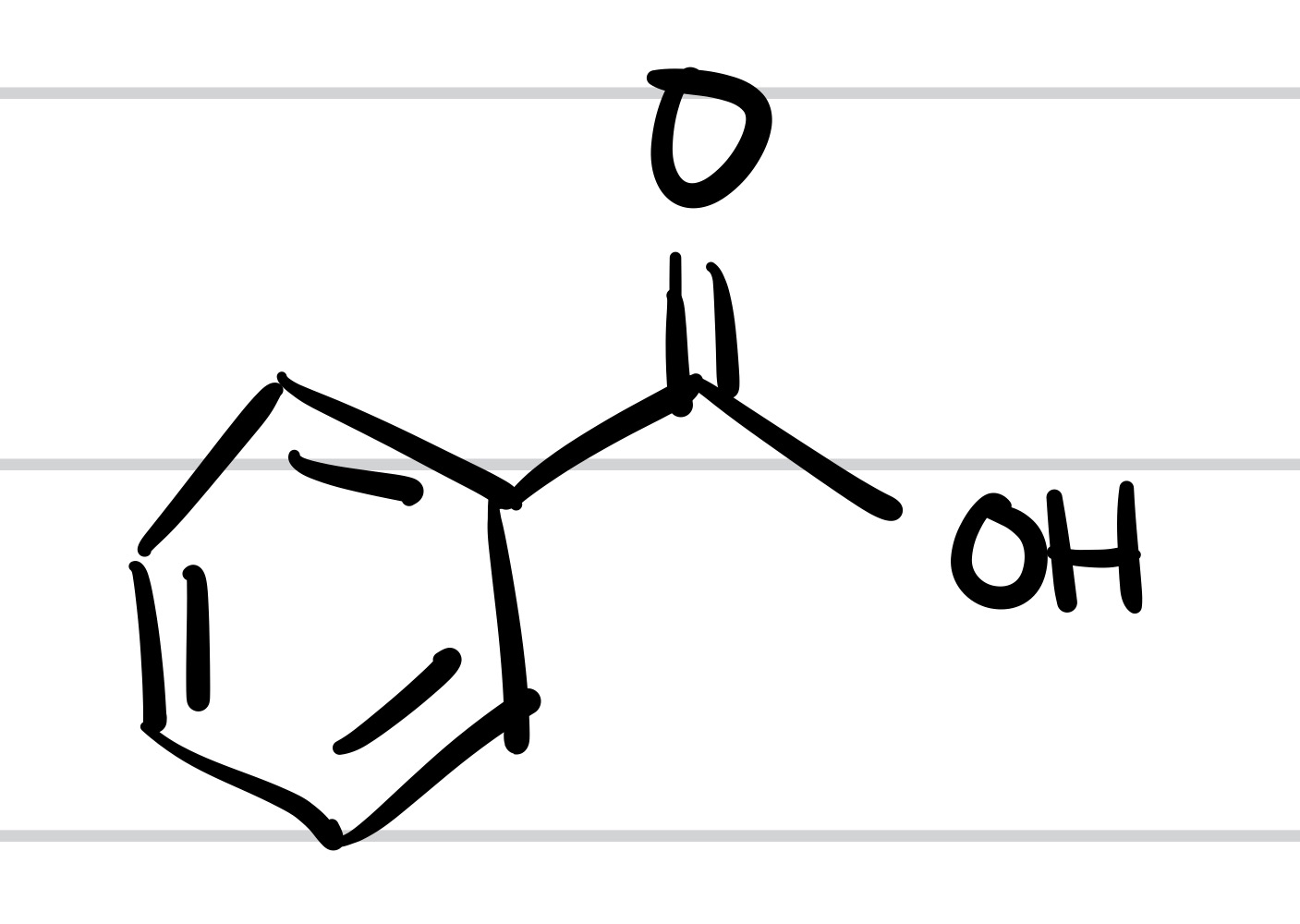
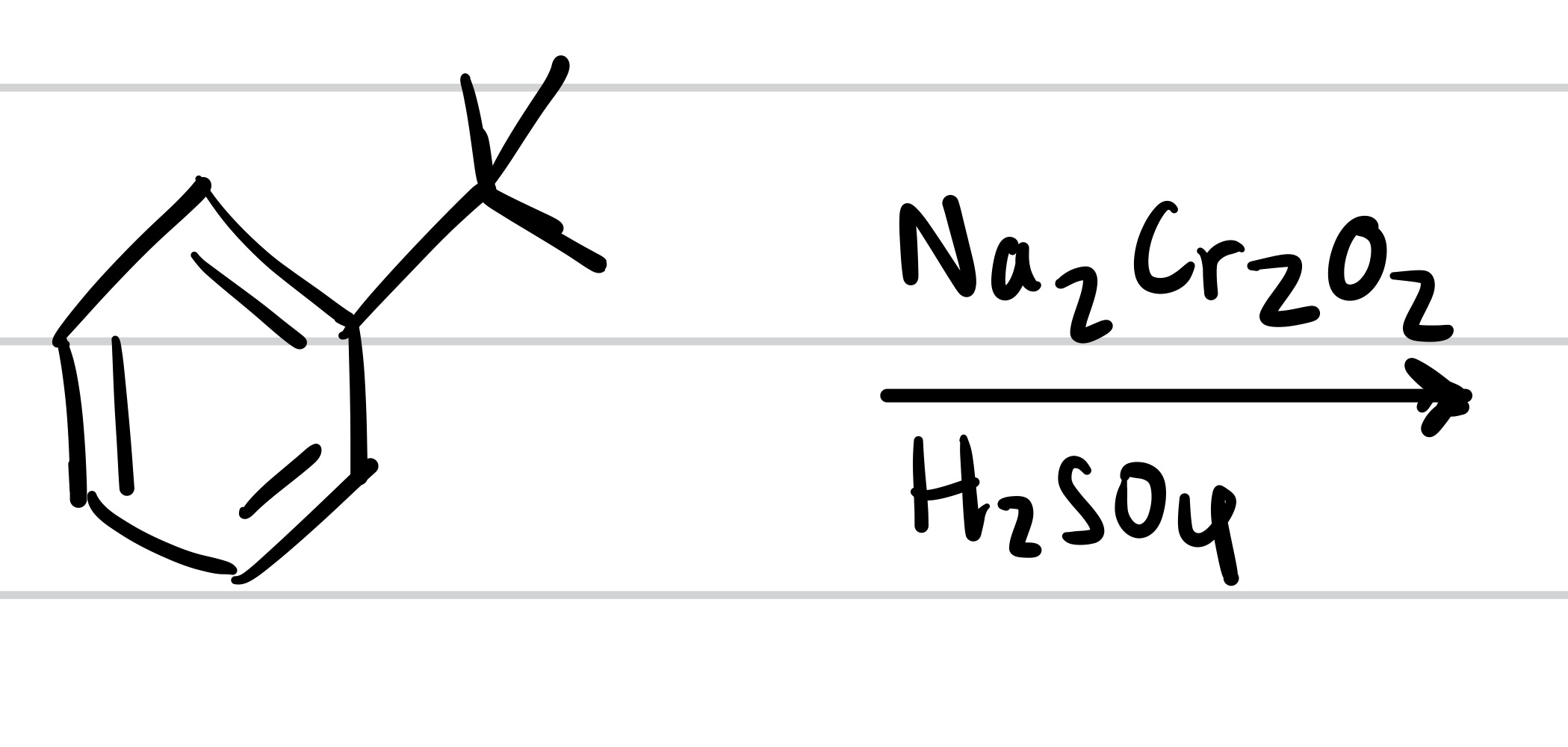
(Three methyl groups = no reaction)

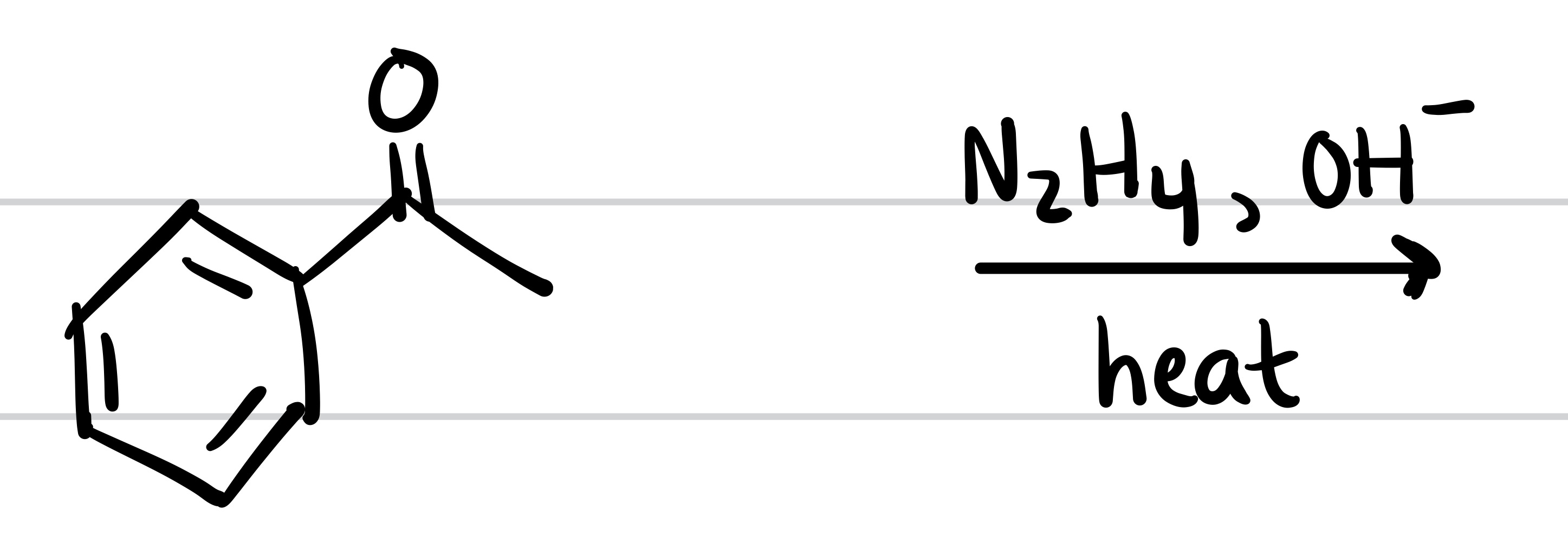
The double O bond leaves
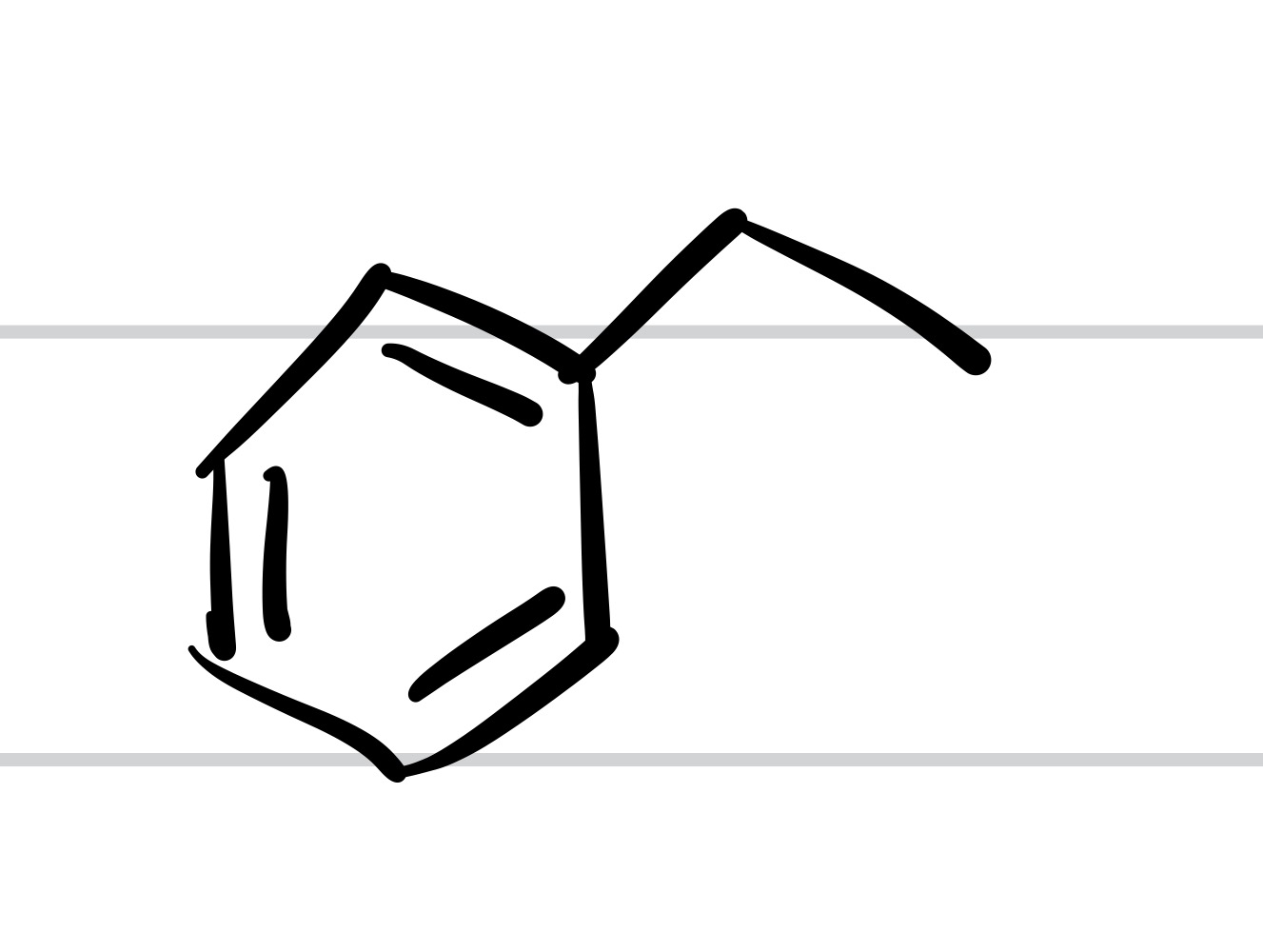

The double O bond leaves
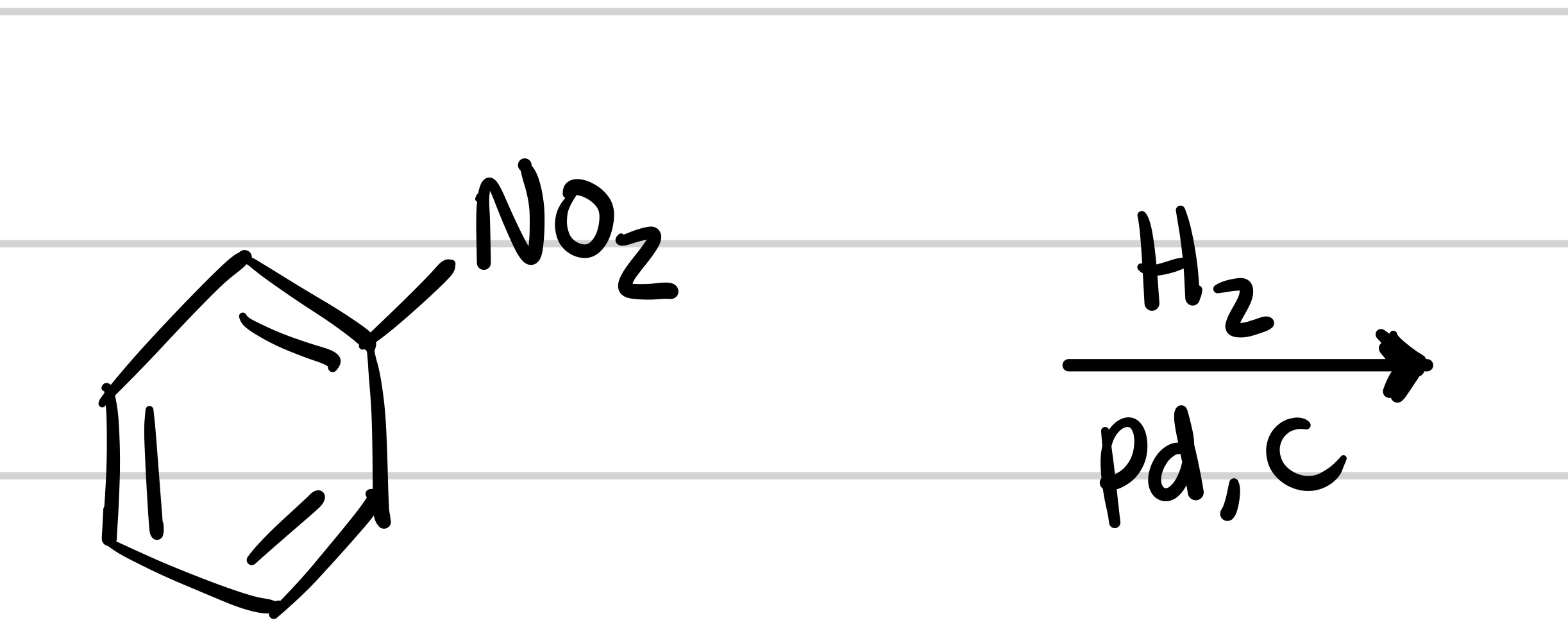
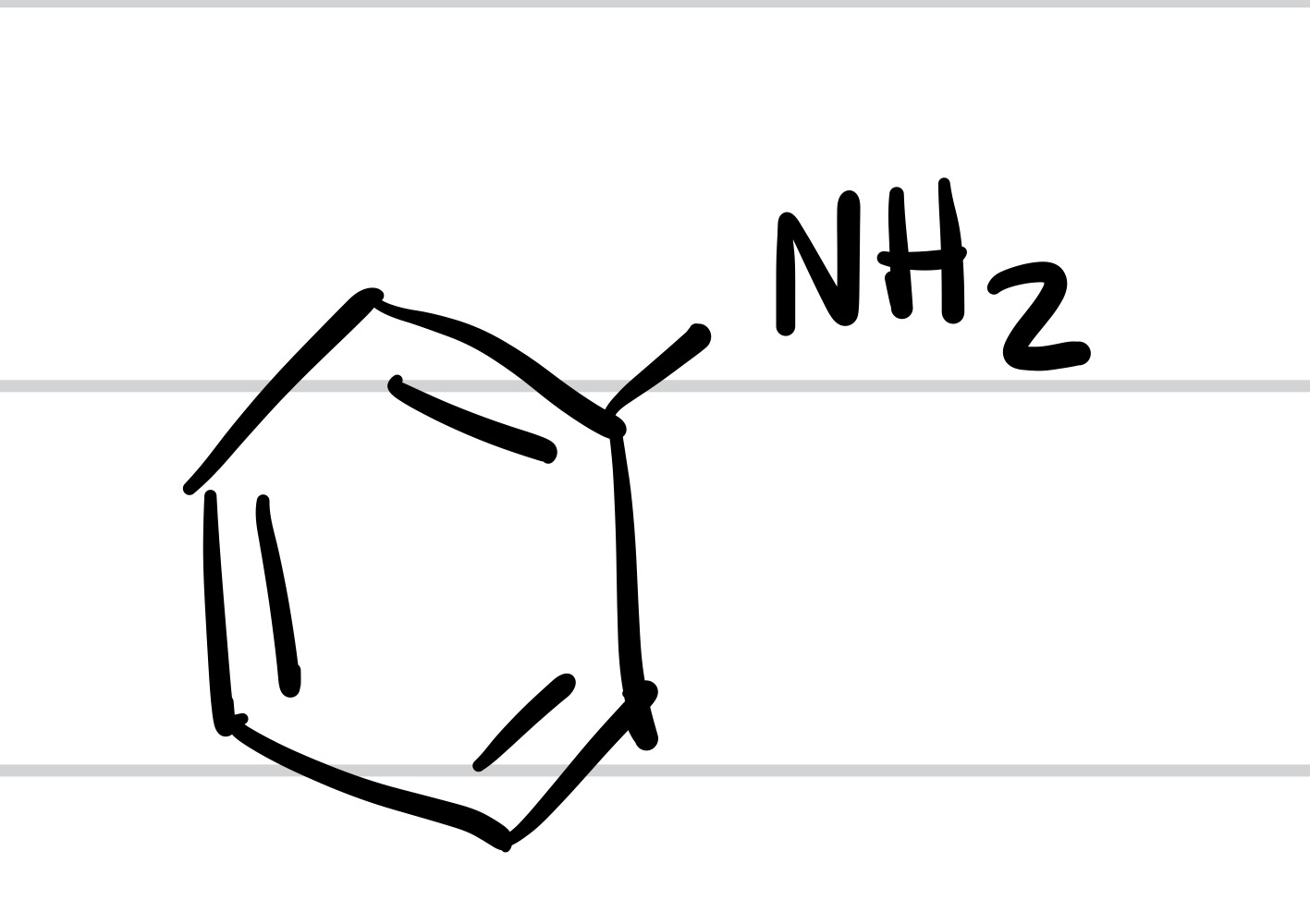
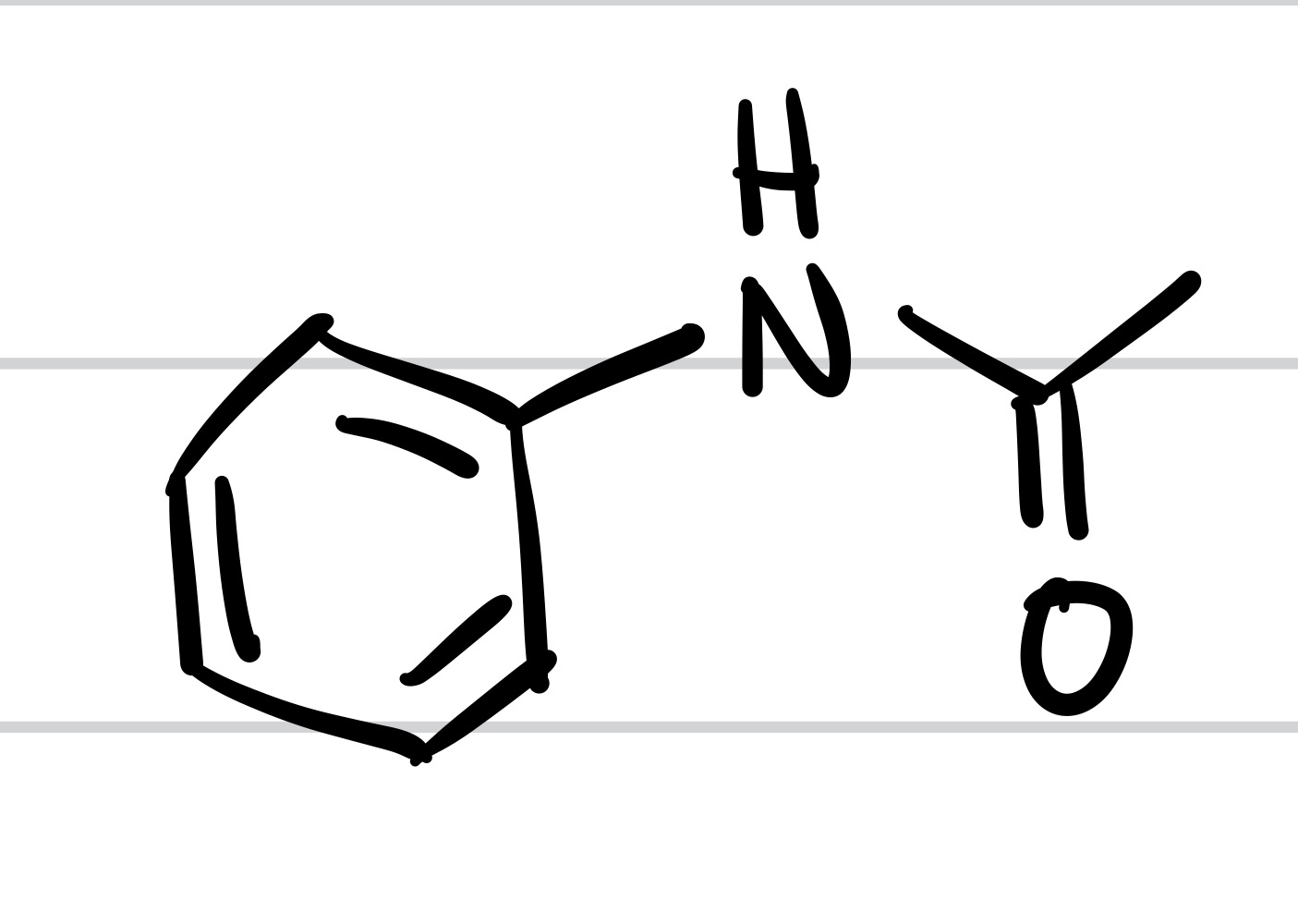
The NO2 is replaced with NH2
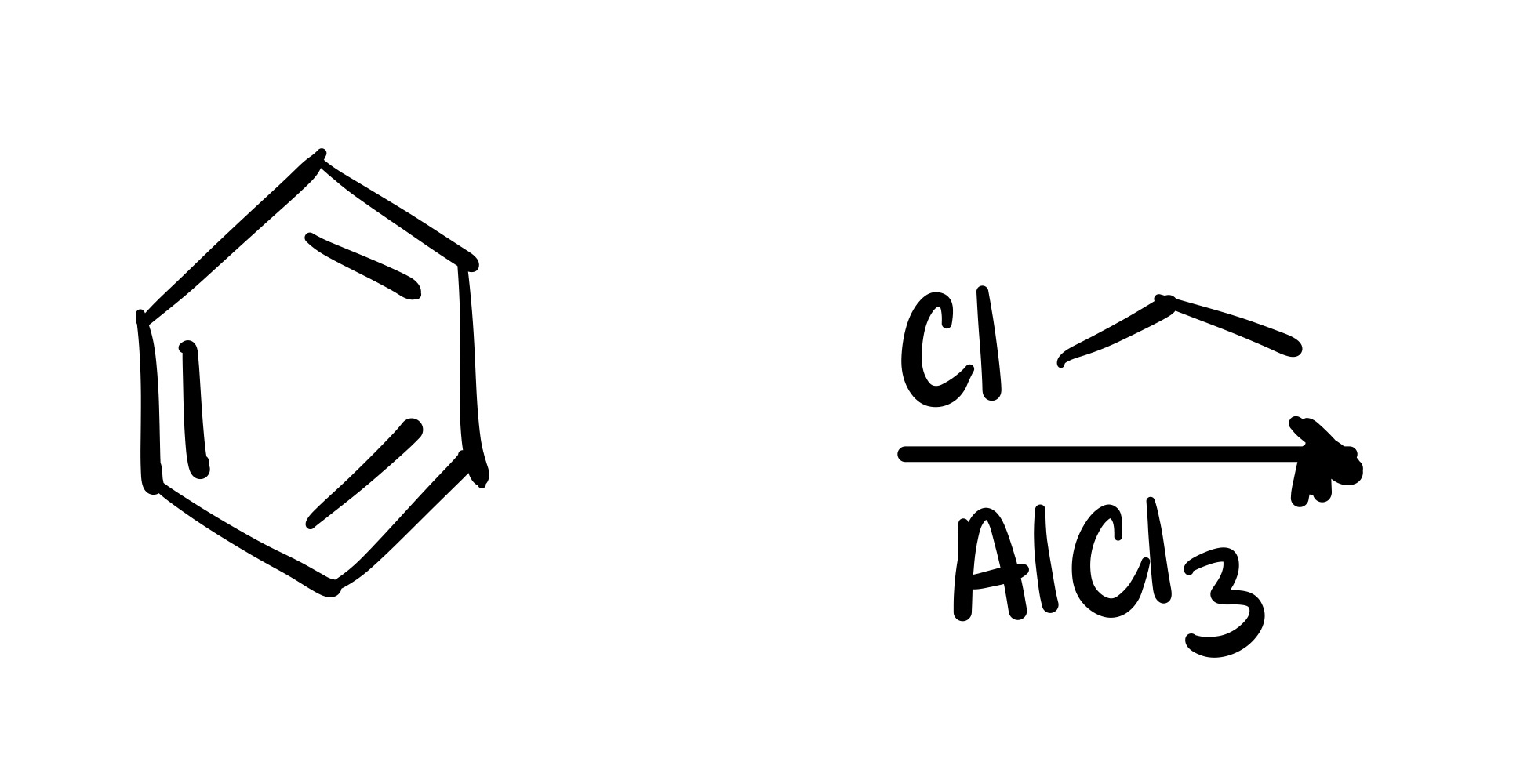
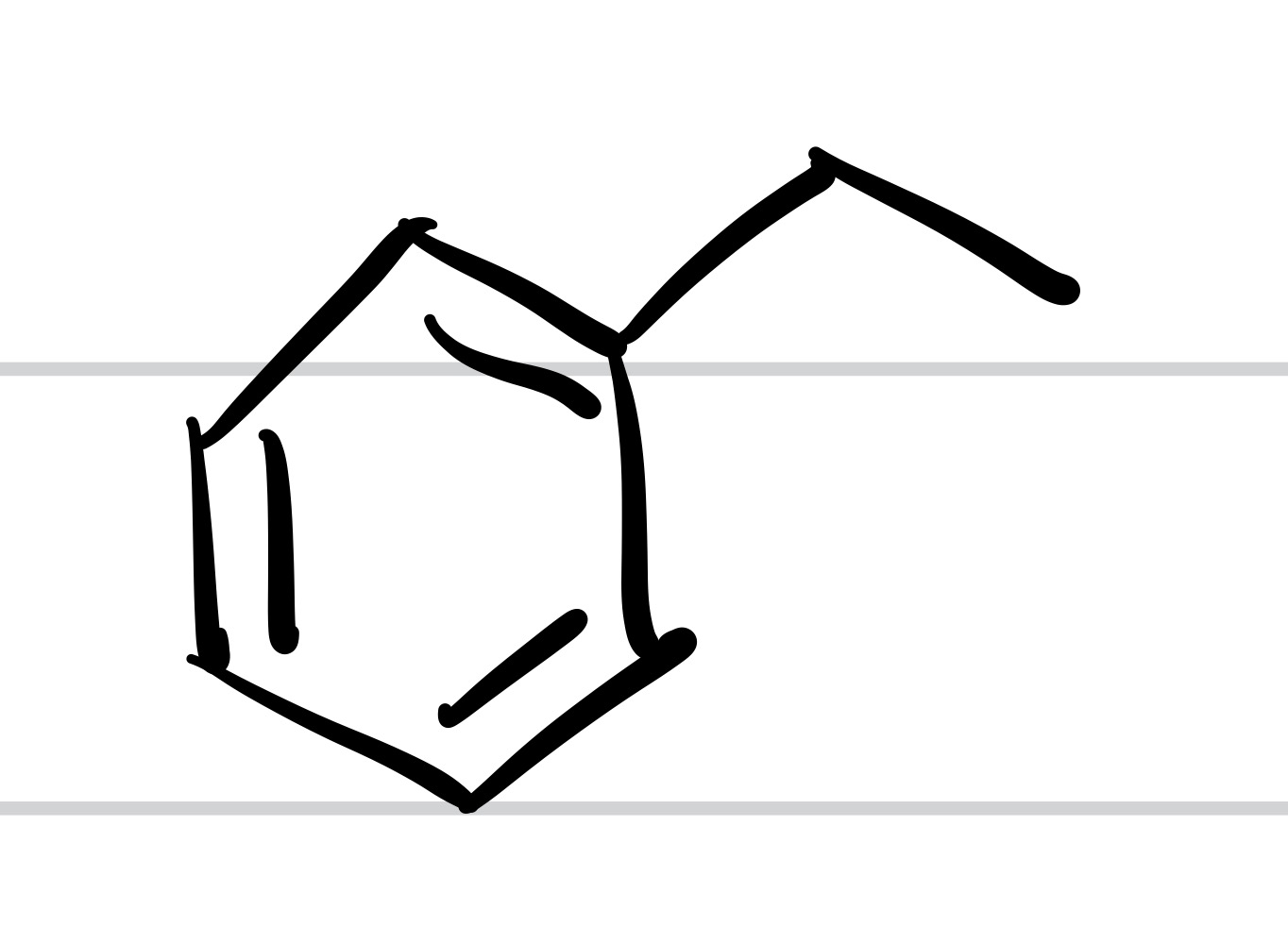
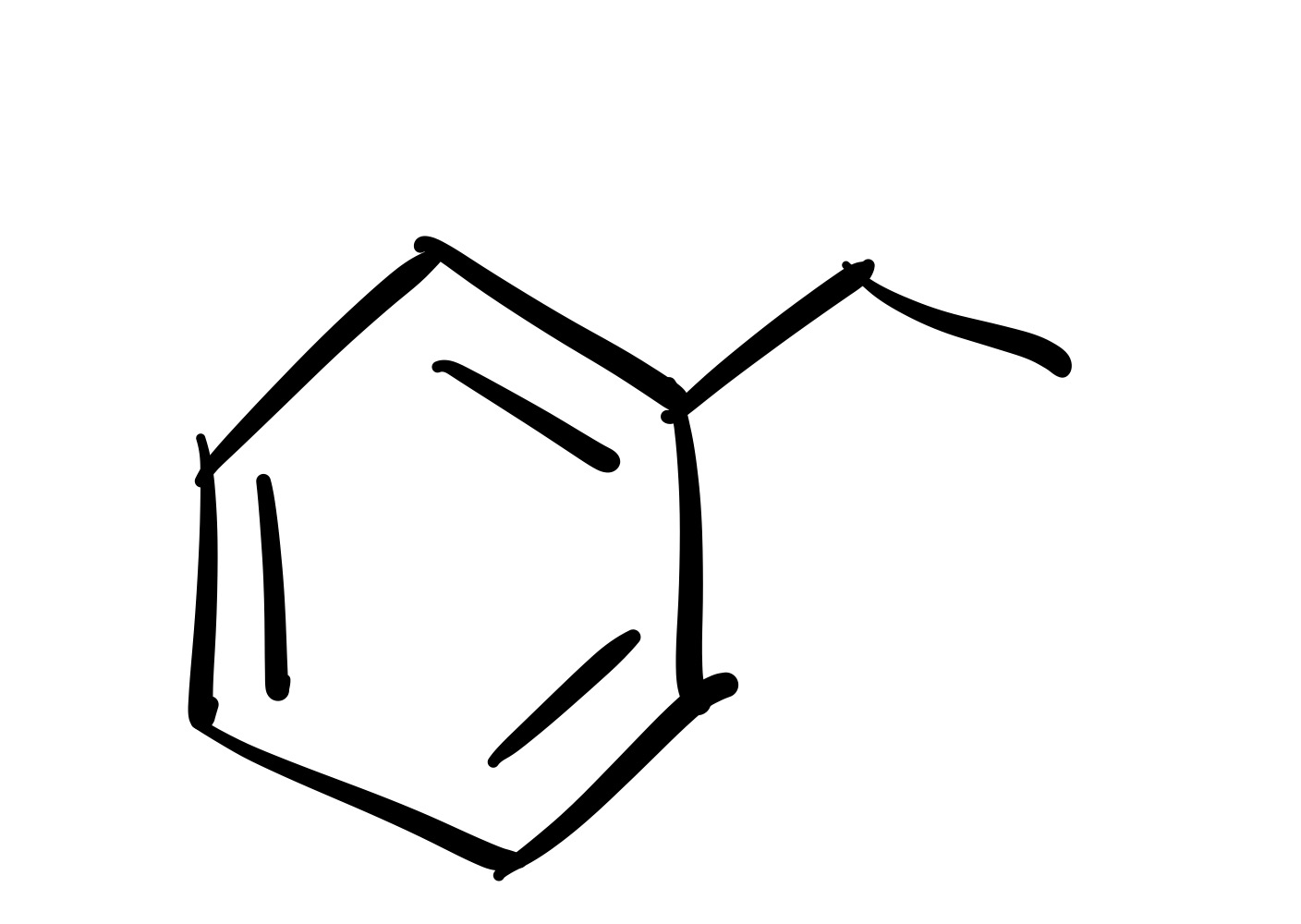
The Cl leaves the carbon chain, and the carbon chain attaches to the cyclohexane
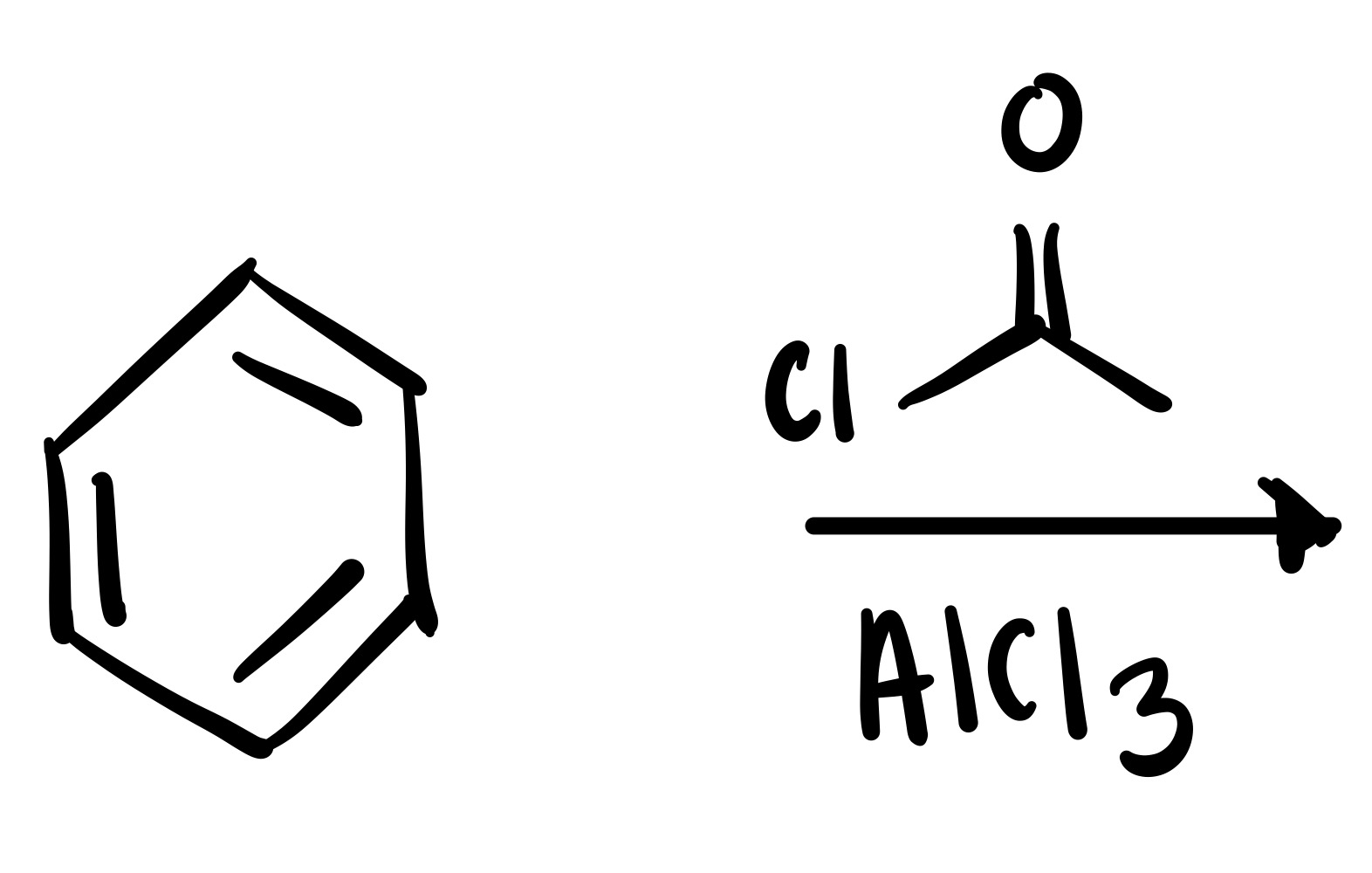
The Cl leaves the carbon chain, and the carbon chain attaches to the cyclohexane
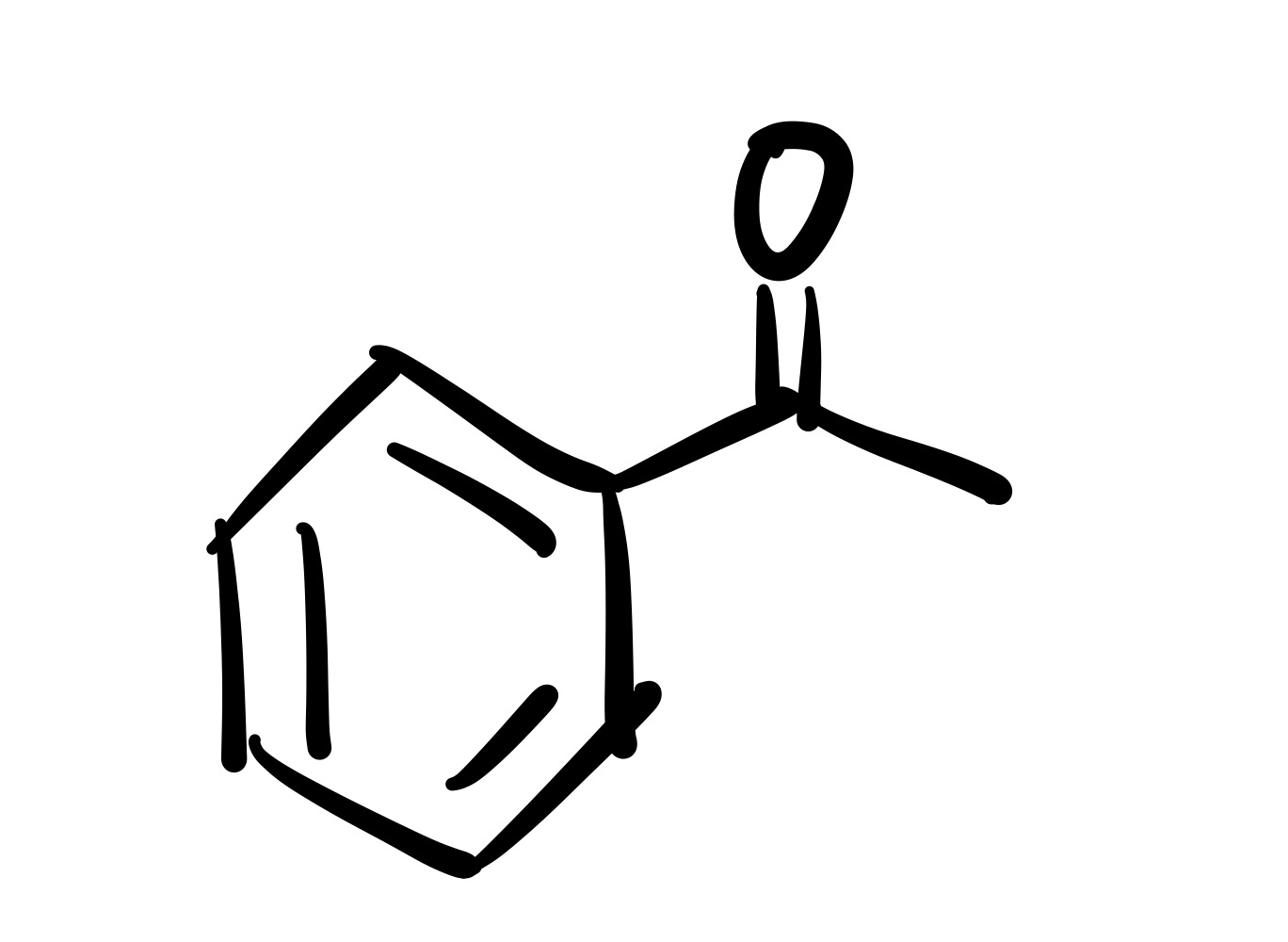
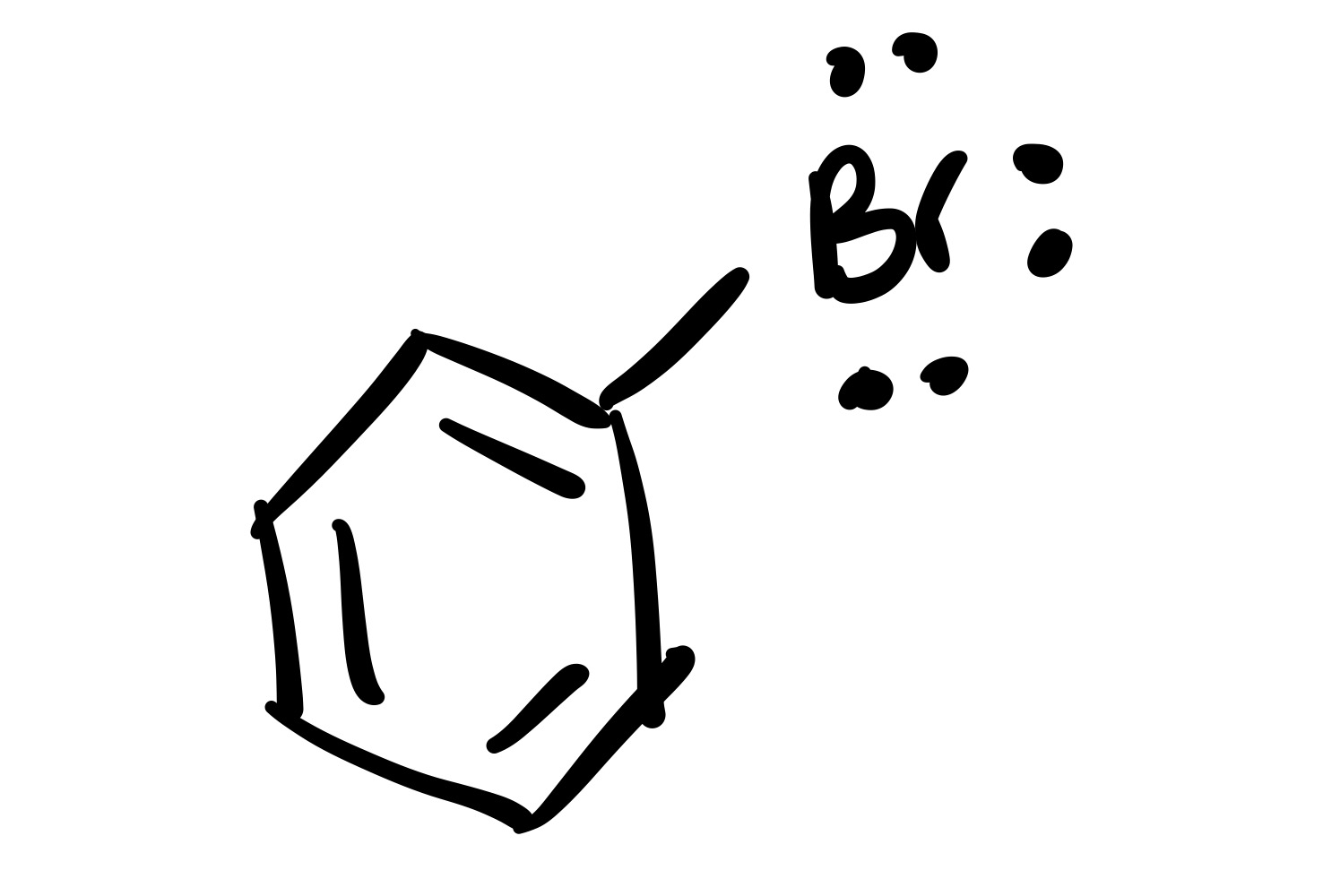
(1) Br adds to the cyclohexane (lone pairs attached)
(1) Cl adds to the cyclohexane (lone pairs attached)
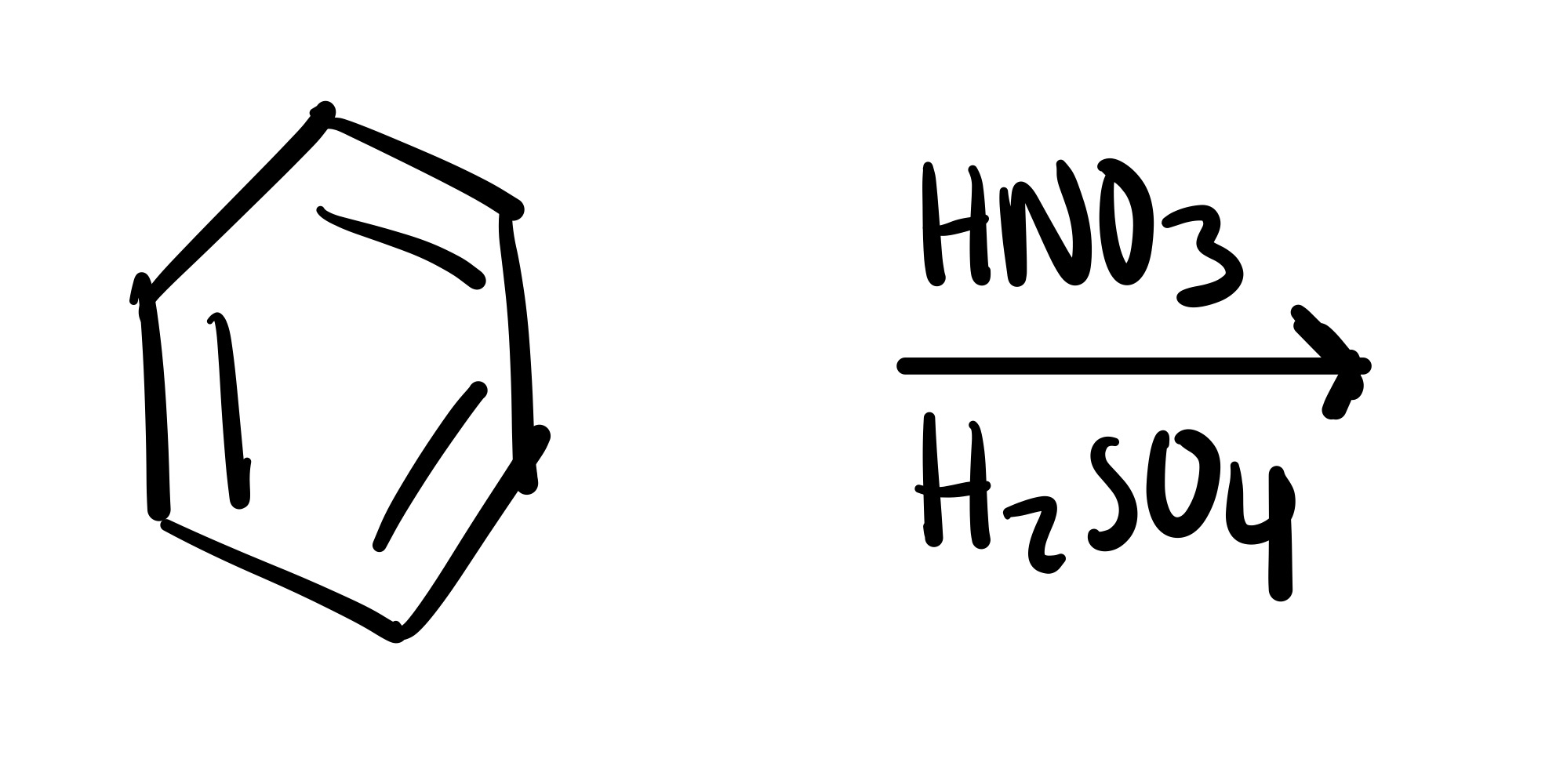
NO2 adds
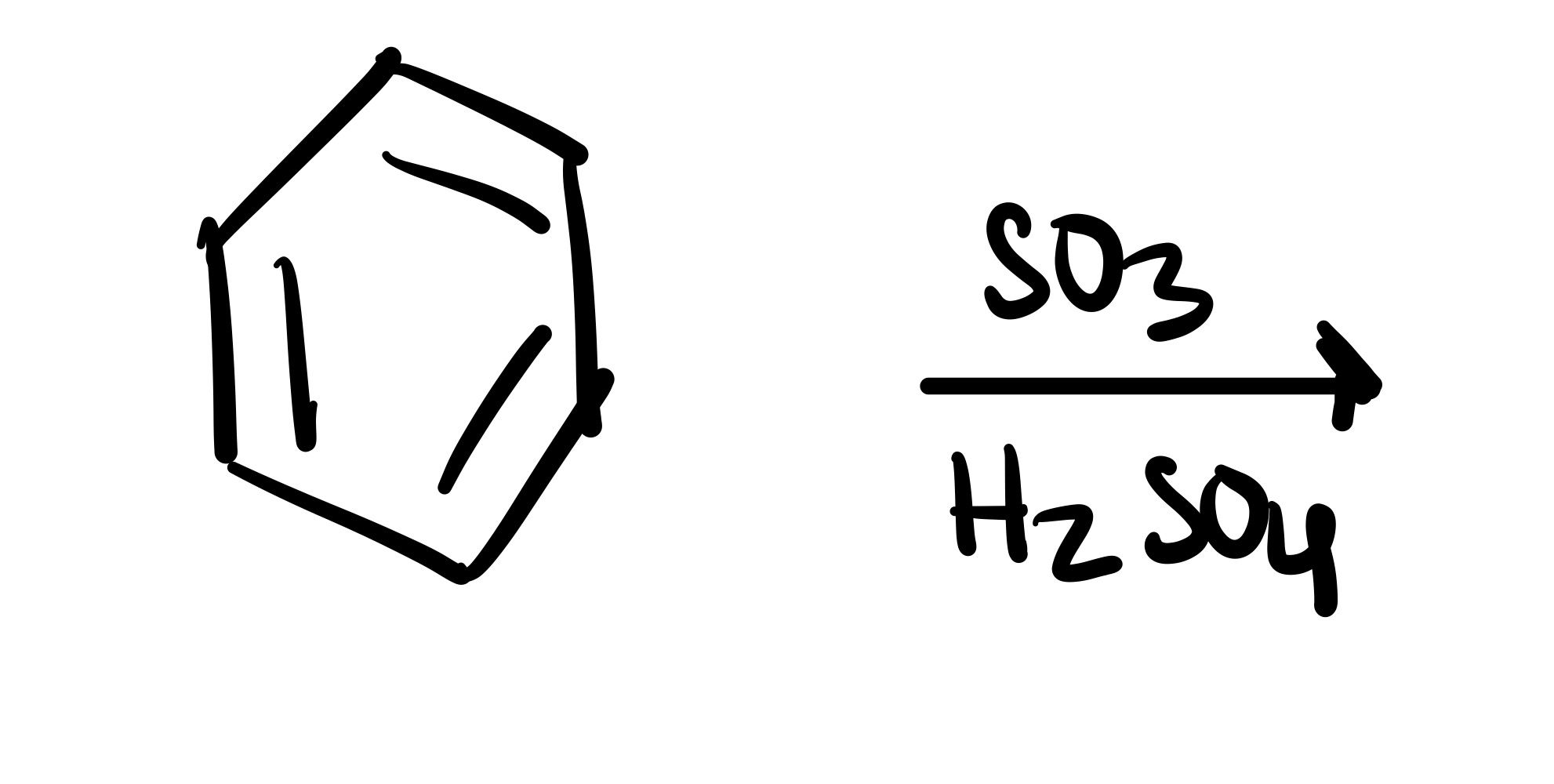
SO3H adds
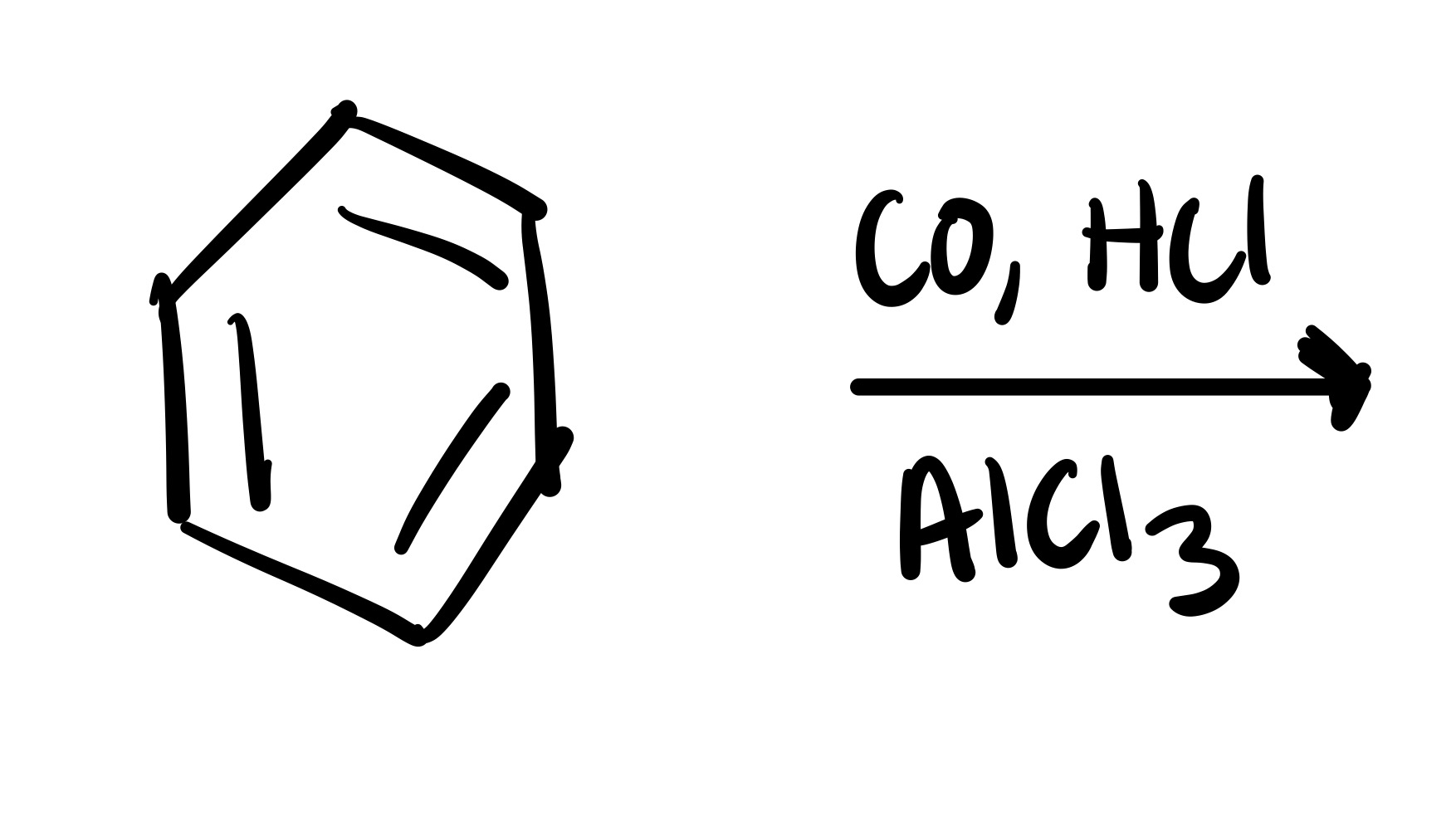
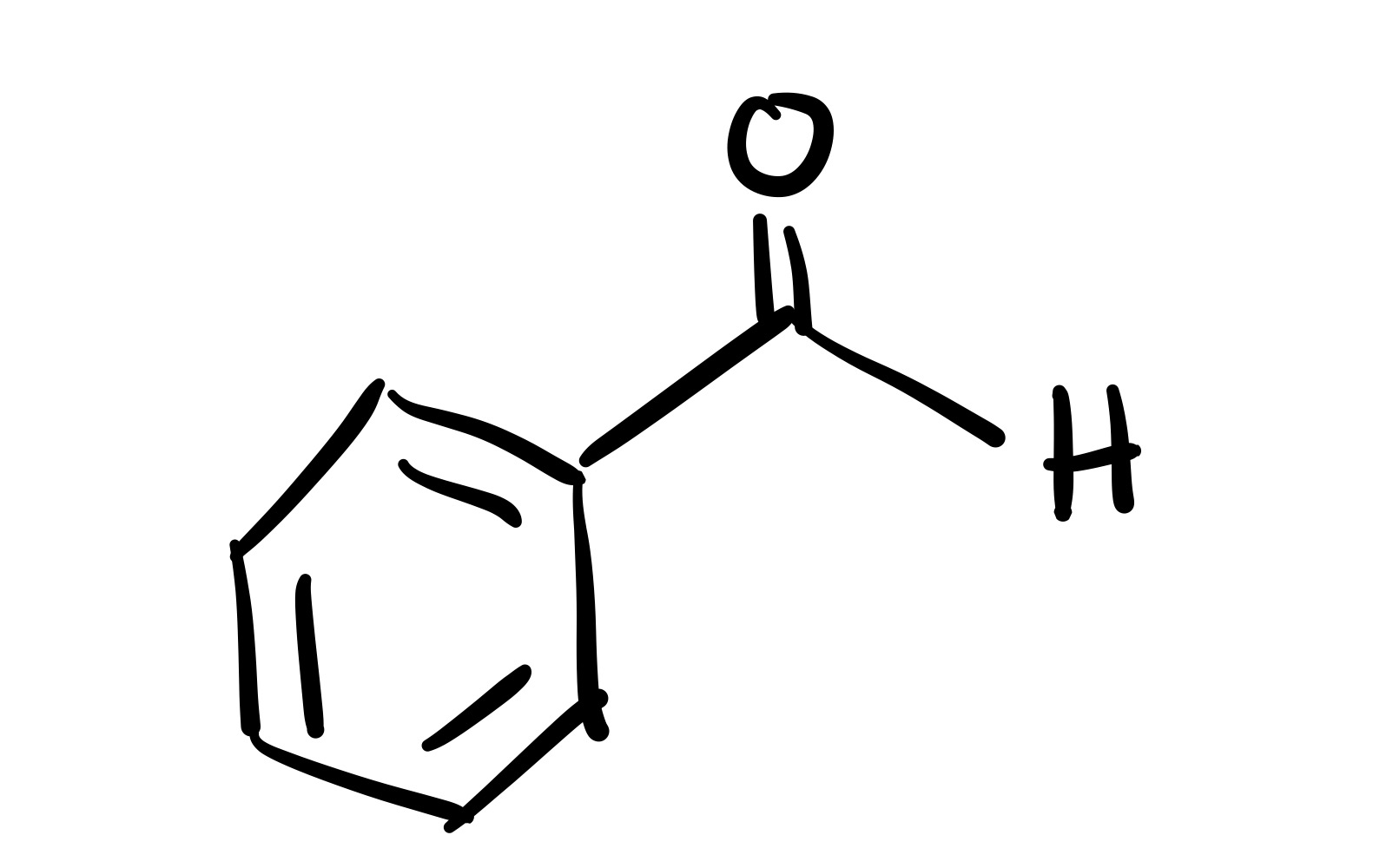
Carbon chain w/ O double bond and H attaches
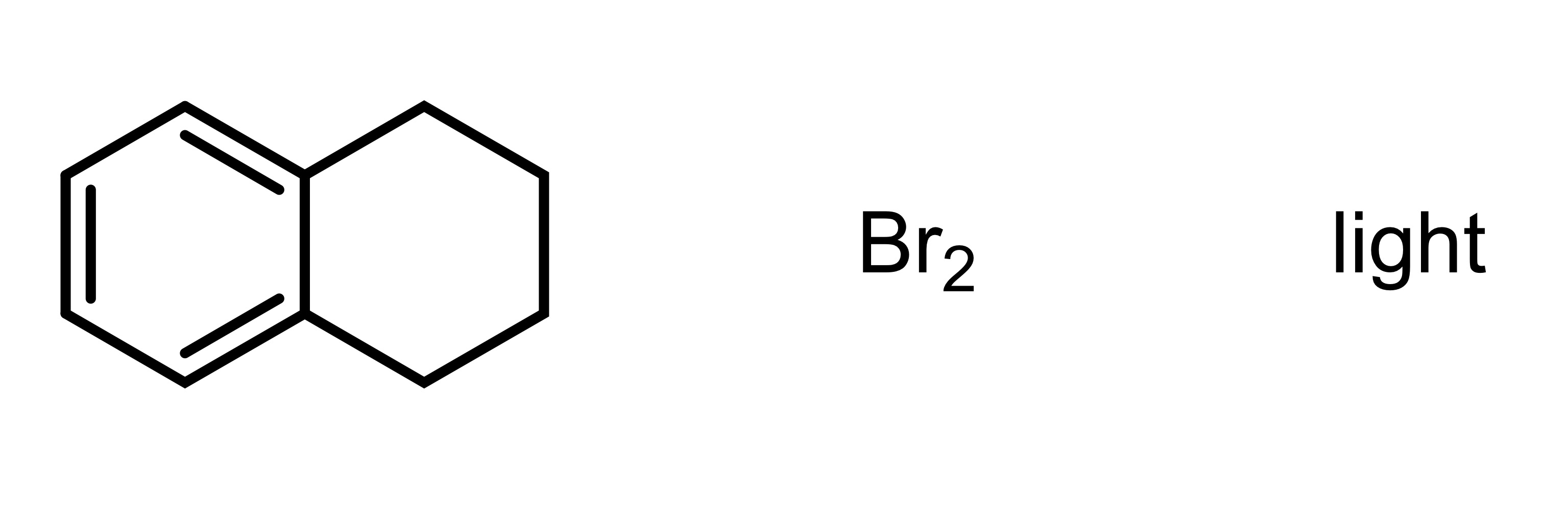
(1) Br is added to the cyclohexane (can be wedge or dash). Add plus minus sign