Chemical Formulas
0.0(0)
Card Sorting
1/4
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
5 Terms
1
New cards
molar mass
atomic mass on periodic table
2
New cards
memorize convertions
particles (atoms/formula units) —> moles —> grams
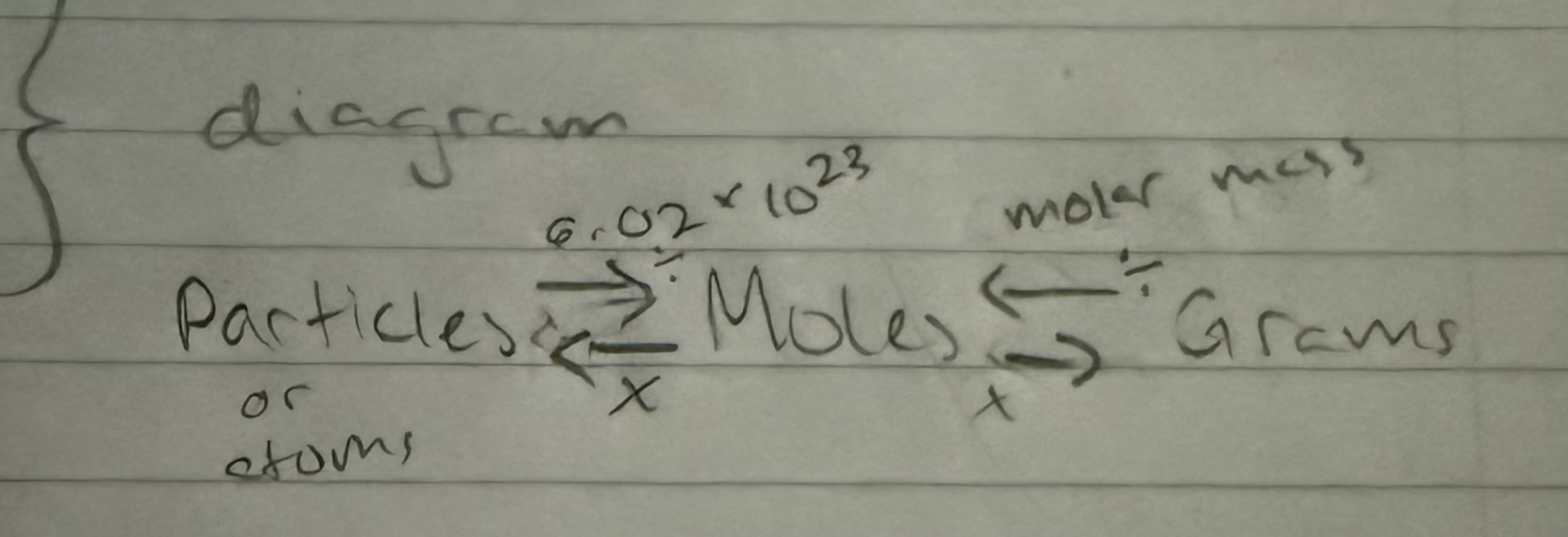
3
New cards
percent composition
(molar mass of element / total molar mass of compound) * 100
4
New cards
empirical formula
given grams/molar mass = number
number / lowest number = whole number
(round if needed)
5
New cards
molecular formula
molecular mass (given) / empirical mass (molar mass of all elements in compound) = whole number
multiply that whole number to the subscripts of the compound