BIOS 301 - Glycolysis
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Homo vs. heteropolysaccharides
Homo - consist of one monomer unit
Hetero - consist of multiple different monomer units
Linear vs. branched polysaccharides
Linear - contain one type of glycosidic bond
Branched - contain multiple different types of glycosidic bonds
Glycogen (structure, function)
Homopolymer of glucose
Contains mainly alpha1→4 glycosidic linkages
Branching with alpha1→6, every 8-12 residues
Acts as a storage polysaccharide in animals, water insoluble, highly branched
Contains many non-reducing ends for rapid phosphorylation (breaks down glycogen) so glucose can be mobilized
Starch
Homopolymer of glucose
Made up of two polysaccharides:
Amlyose - contains alpha1→4 glycosidic linkages, linear
Amylopectin - contains branching with alpha1→6 every 24-30 residues
Acts as storage polysaccharide in plants, water insoluble, moderately branched
Cellulose (structure, function)
Homopolymer of glucose
Contains beta1→4 glycosidic linkages
Linear
Hydrogen bonding occurs between adjacent monomers and chains - forms stable, dense sheets
Most abundant polysaccharide in nature (plant cell walls), water insoluble, cannot be digested by humans
How do linkages affect structure in polysaccharides?
Alpha glycosidic linkages between monomers introduces a slight kink in the chain → leads to helical polymers
Starch detection
Starch forms long helices than bind polyiodine
Transfer of electrons from starch to iodine allows the complex to absorb yellow-red light (appears as blue color)
Major pathways of glucose utilization
Extracellular matrix/cell wall polysaccharides (e.g. cellulose)
Storage (e.g. glycogen, starch)
Oxidation via glycolysis (convert to pyruvate)
Oxidation via pentose phosphate pathway (convert to ribose-5-phosphate)
Glycolysis (overall purpose, outcome)
Glycolysis is a series of enzyme catalyzed reactions in which glucose is converted to pyruvate
Pyruvate can be further oxidized in the citric acid cycle or serve as a precursor in biosynthesis
Glycolysis (overall reaction mechanism)
Breakage of the C3-C4 bond in one D-glucose to produce 2 net ATPs and 2 pyruvate molecules
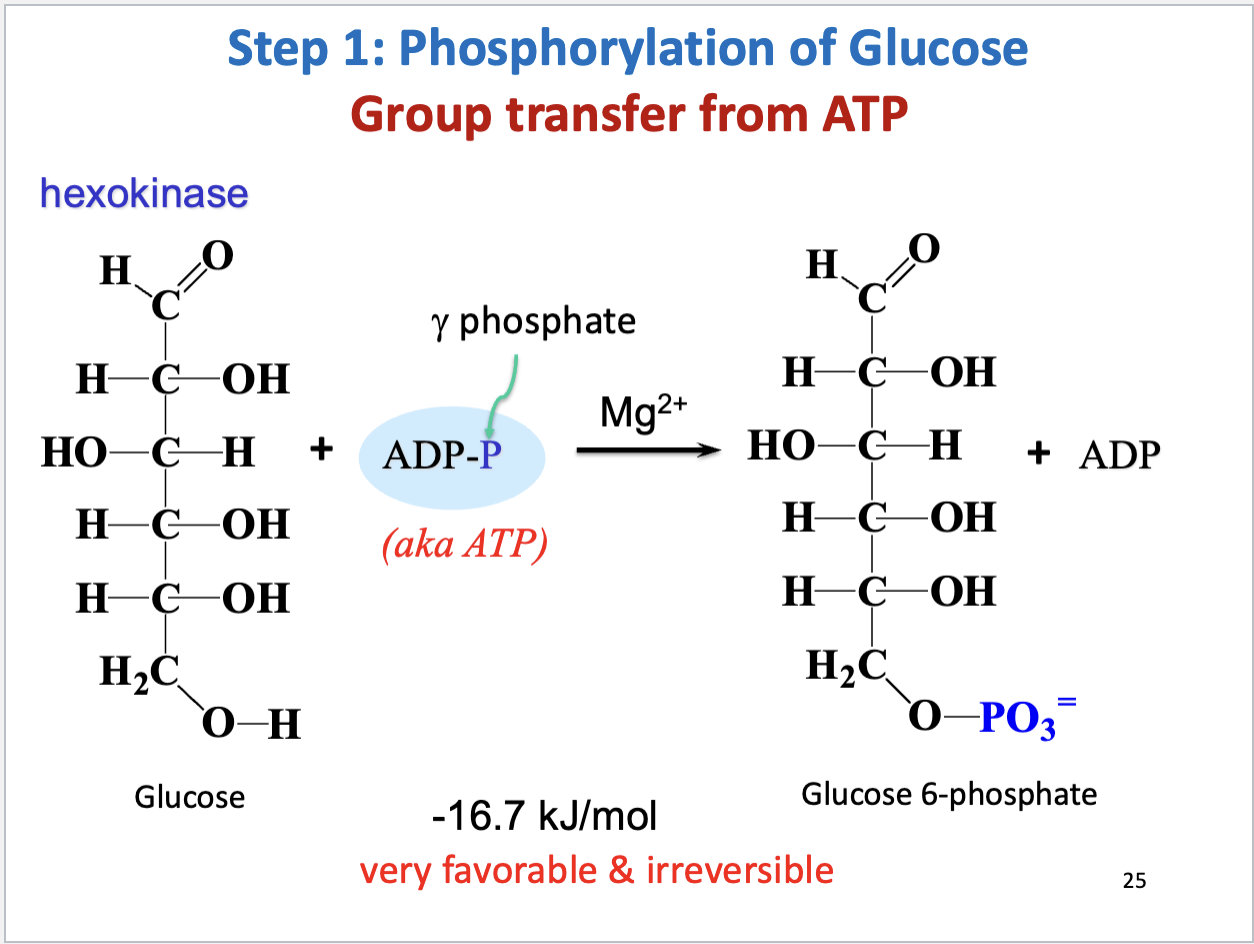
Glycolysis Step 1
Phosphorylation of glucose
Substrate: glucose, Product: glucose-6-phosphate
Enzyme: hexokinase, Cofactor: Mg2+
Mg2+ binds ATP to shield its negative charges
Negative delta G (very favorable, irreversible)
Regulated by substrate inhibition (excess substrate binds allosterically to enzyme)
Hexokinase I
Found in muscle, brain
Ensures that cells receive enough glucose for basic energy needs, regardless of fluctuations in glucose levels
Has a low Ks (high affinity for glucose)
Inhibited by G-6-P
Plot vi vs. [glucose] → sigmoidal binding curve
Hexokinase IV
Found in liver
Clears excess glucose from the blood for storage as glycogen
Very high Ks (low affinity for glucose)
High blood glucose → transported into cell → G-6-P → F-6-P → promotes release of kinase from nucleus to cytoplasm → increased production of G-6-P
If F-6-P builds up, hexokinase release is inhibited (acts as signal that liver is well-stocked with energy)
Plot vi vs. [glucose] → positive cooperativity
Glucose-6-Phosphatase
Liver enzyme
Removes phosphate group from G-6-P to release glucose into bloodstream
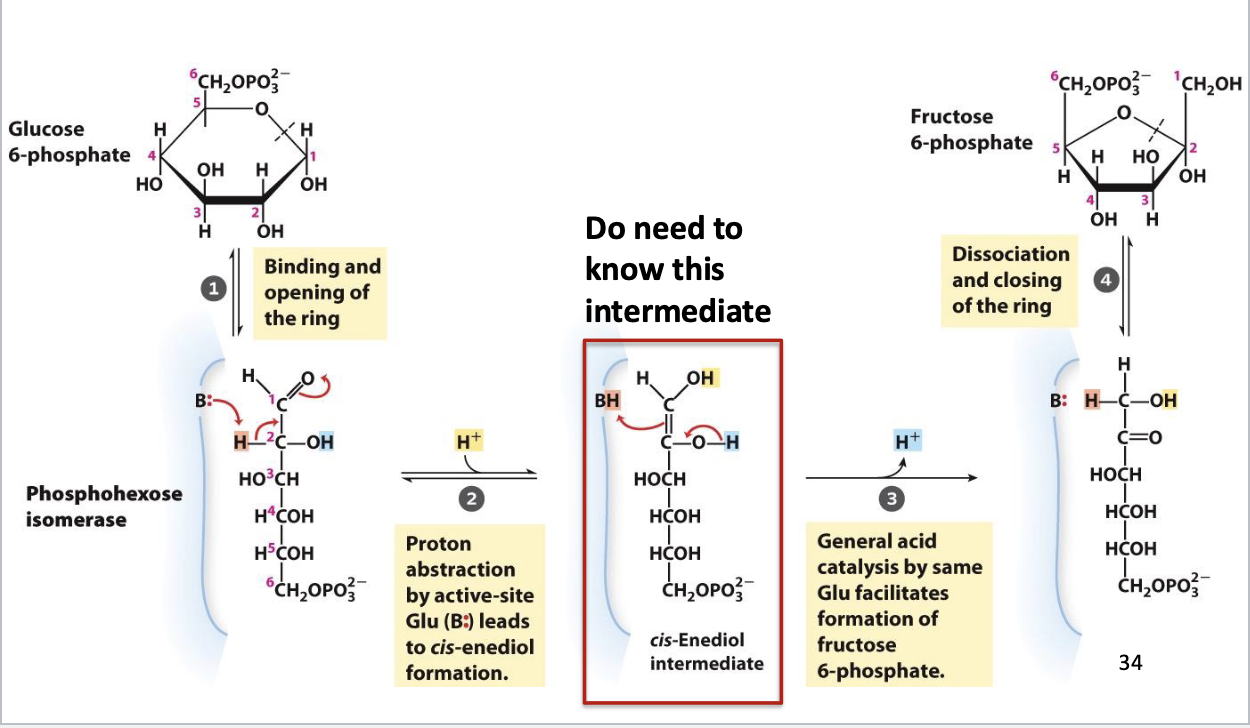
Glycolysis Step 2
Phosphohexose isomerization
Substrate: G-6-P, Product: F-6-P
Enzyme: phosphoglucoisomerase, Cofactor: Mg2+
Postive delta G (reversible)
Paired with favorable next step to drive reaction forward
Regulated by substrate inhibition (excess substrate binds allosterically to enzyme)
Glycolysis Step 2: intermediate
Enediol
Formed through acid-base catalysis
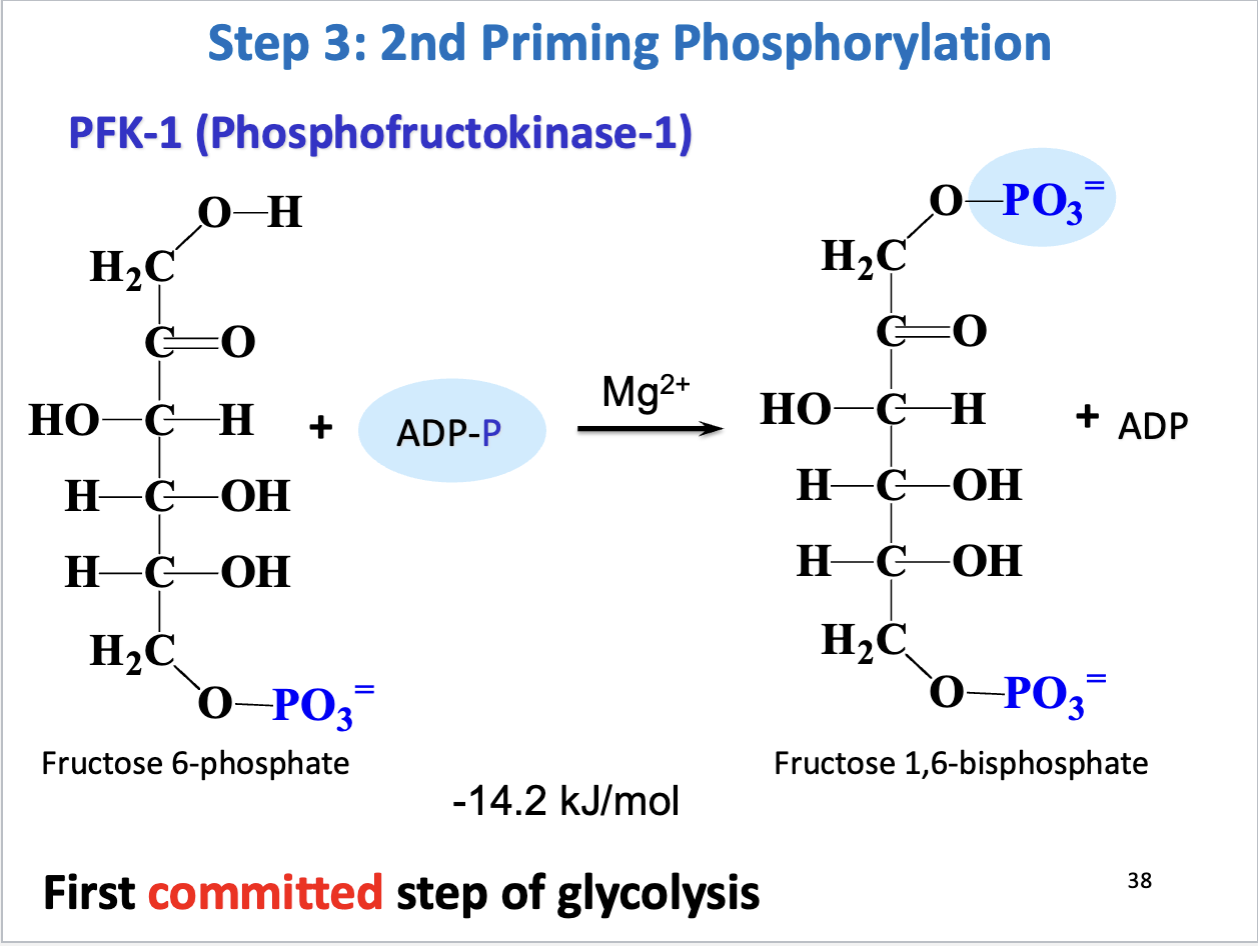
Glycolysis Step 3
2nd priming phosphorylation
First committed step of glycolysis
Substrate: F-6-P, Product: frustose 1,6-biphosphate
Enzyme: PFK-1 (phosphofructokinase-1), Cofactor: Mg2+
Negative delta G (favorable, irreversible)
Phosphofructokinase-1 regulation
Positively regulated by ADP, AMP
Low energy in the cell promotes glycolysis
Negatively regulated by ATP, citrate
High energy in the cell/bottleneck in TCA inhibits glycolysis
Plot enzyme activity vs [F-6-P]
Low [ATP] → high substrate affinity
High [ATP] → low substrate affinity
![<ul><li><p>Positively regulated by ADP, AMP</p><ul><li><p>Low energy in the cell promotes glycolysis</p></li></ul></li><li><p>Negatively regulated by ATP, citrate</p><ul><li><p>High energy in the cell/bottleneck in TCA inhibits glycolysis</p></li></ul></li><li><p>Plot enzyme activity vs [F-6-P]</p><ul><li><p>Low [ATP] → high substrate affinity</p></li><li><p>High [ATP] → low substrate affinity</p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/d62924d9-b479-4b48-a743-4e66a152a0f0.png)
Glycolysis Step 4
Aldol cleavage of F-1,6-bP
Substrate: fructose 1,6-bisphosphate, Products: glyceraldehyde 3-phosphate, dihydroxyacetone phosphate
Enzyme: aldolase
Positive delta G (standard conditions don’t exist in cell, reaction driven by product consumption)
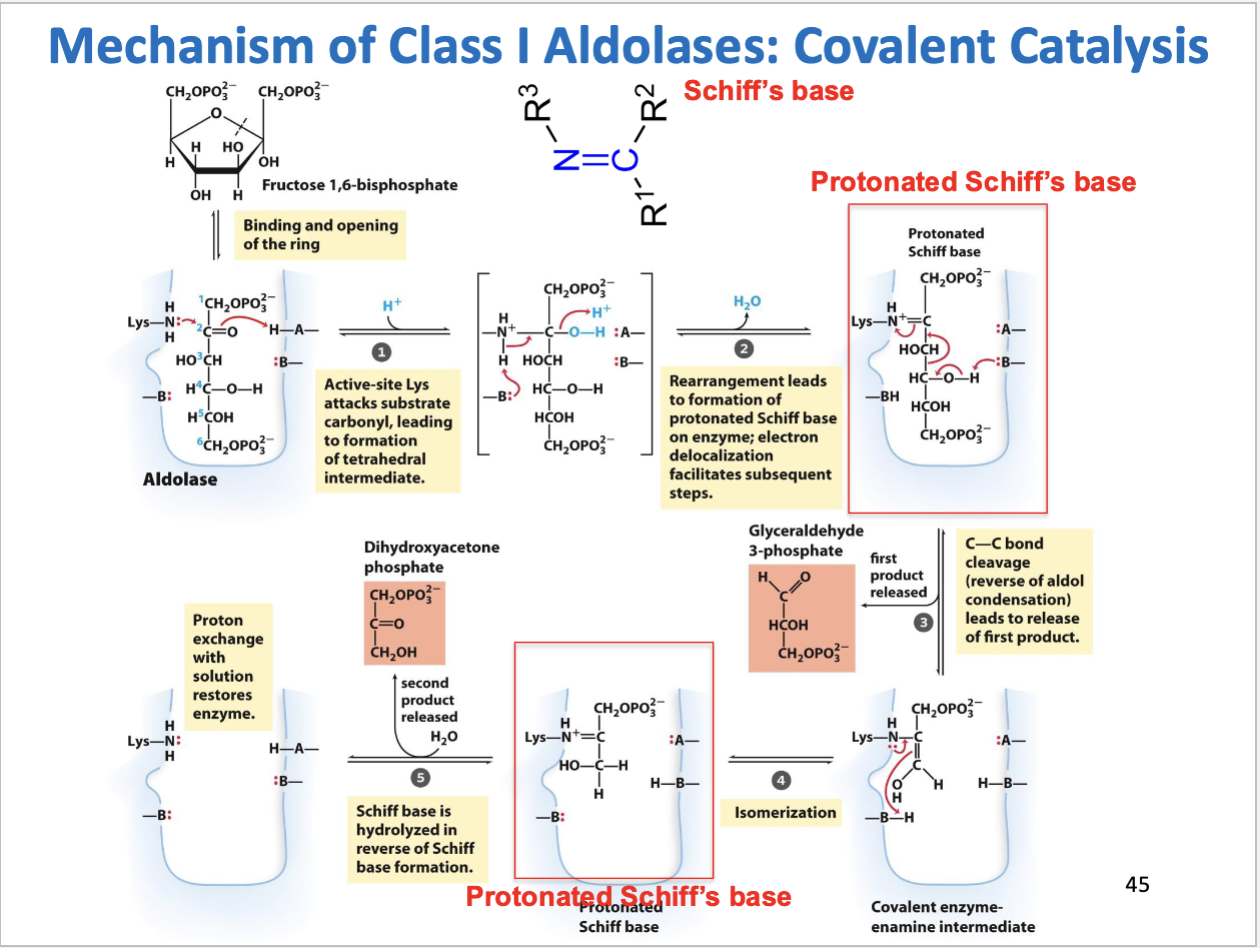
Mechanism of class I aldolases
Covalent catalysis
Intermediate = protonated Schiff’s base
Glycolysis Step 5
Triose phosphate interconversion
Substrate: dihydroxyacetone phosphate, Product: glyceraldehyde 3-phosphate
Enzyme: triose phosphate isomerase
Positive delta G (unfavorable/reversible, keep GAP concentration low to drive reaction forward)
DHAP must be converted to GAP to proceed with next phase of glycolysis
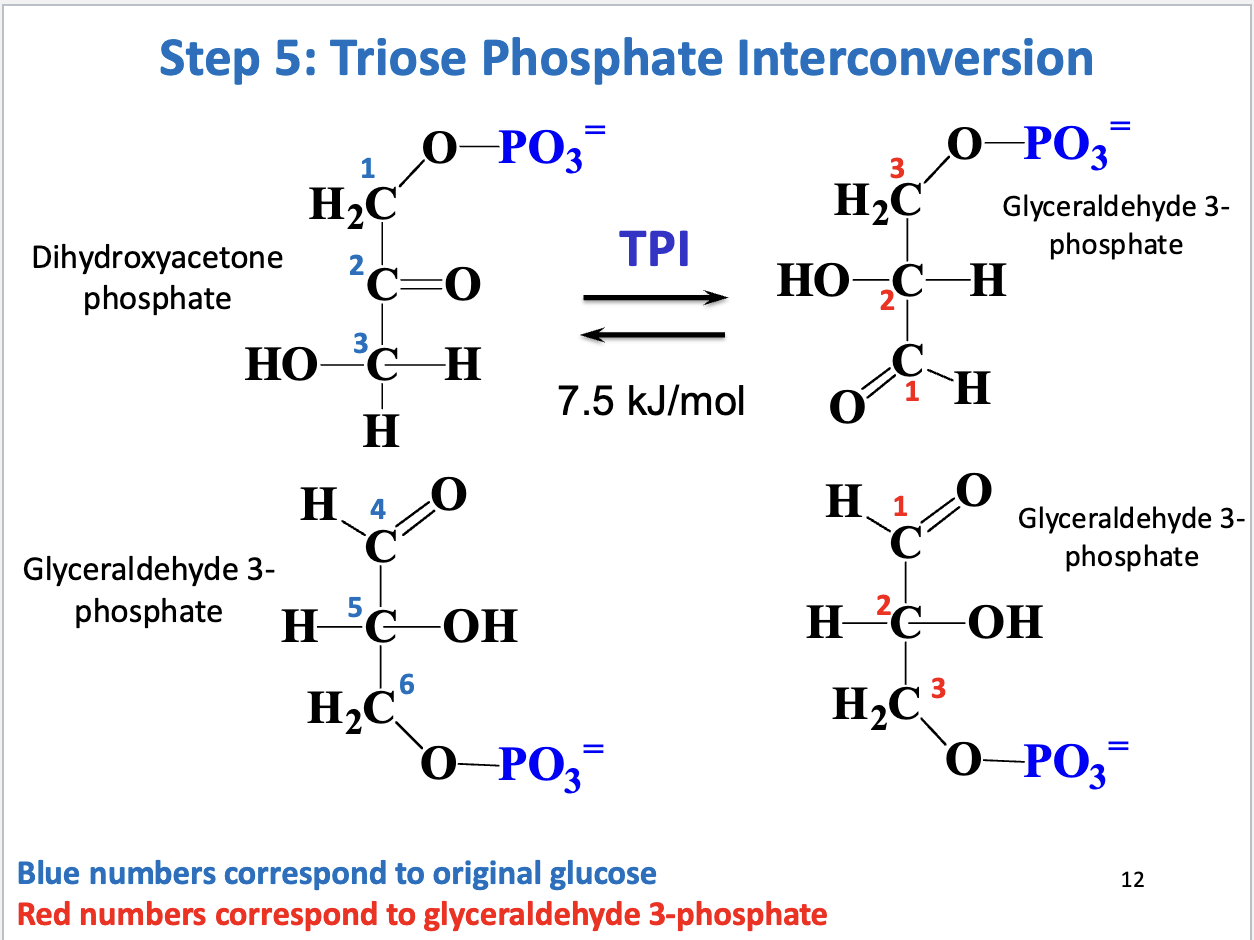
Overall product of glycolysis preparatory phase
Lysis of C3-C4 bond in D-glucose yields 2 D-glyceraldehyde-3-P
Costs 2 ATP
Glycolysis Step 6
Oxidation of GAP (oxidation-reduction reaction)
First energy yielding step in glycolysis
Substrate: glyceraldehyde 3-phosphate, Product: 1,3-biphosphoglycerate
Also, oxidation of aldehyde with NAD+ gives NADH
Enzyme: glyceraldehyde 3-phosphate dehydrogenase
Postive delta G (coupled with next reaction to drive forward)
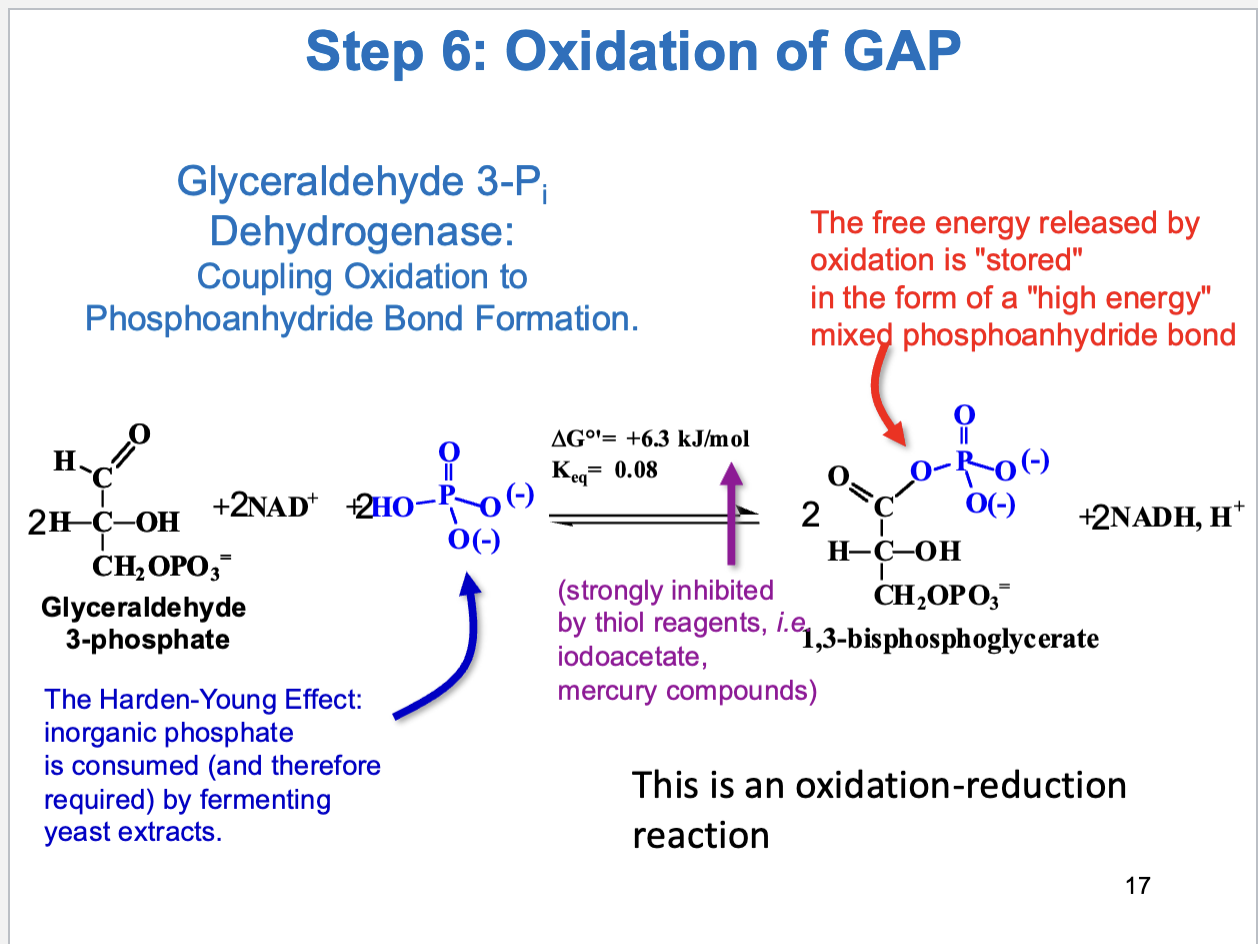
Glyceraldehyde-3-Phosphate catalysis
Performs covalent catalysis
Has cysteine residue in active site
Produces thiohemiacetal and thiohester intermediates
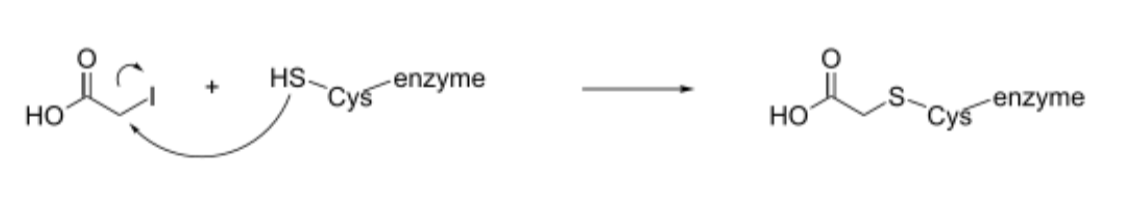
Glyceraldehyde-3-Phosphate inhibition
Iodoacetate irreversibly inhibits GAPDH by covalently modifiying its active site Cys and preventing formation of thiohemiacetal and thiohester intermediates
Arsenate reacts with the thiohester intermediate (instead of phosphate group), decoupling 1,3-bisphosphoglycerate and ATP synthesis
No ATP produced → glycolysis is unfavorable
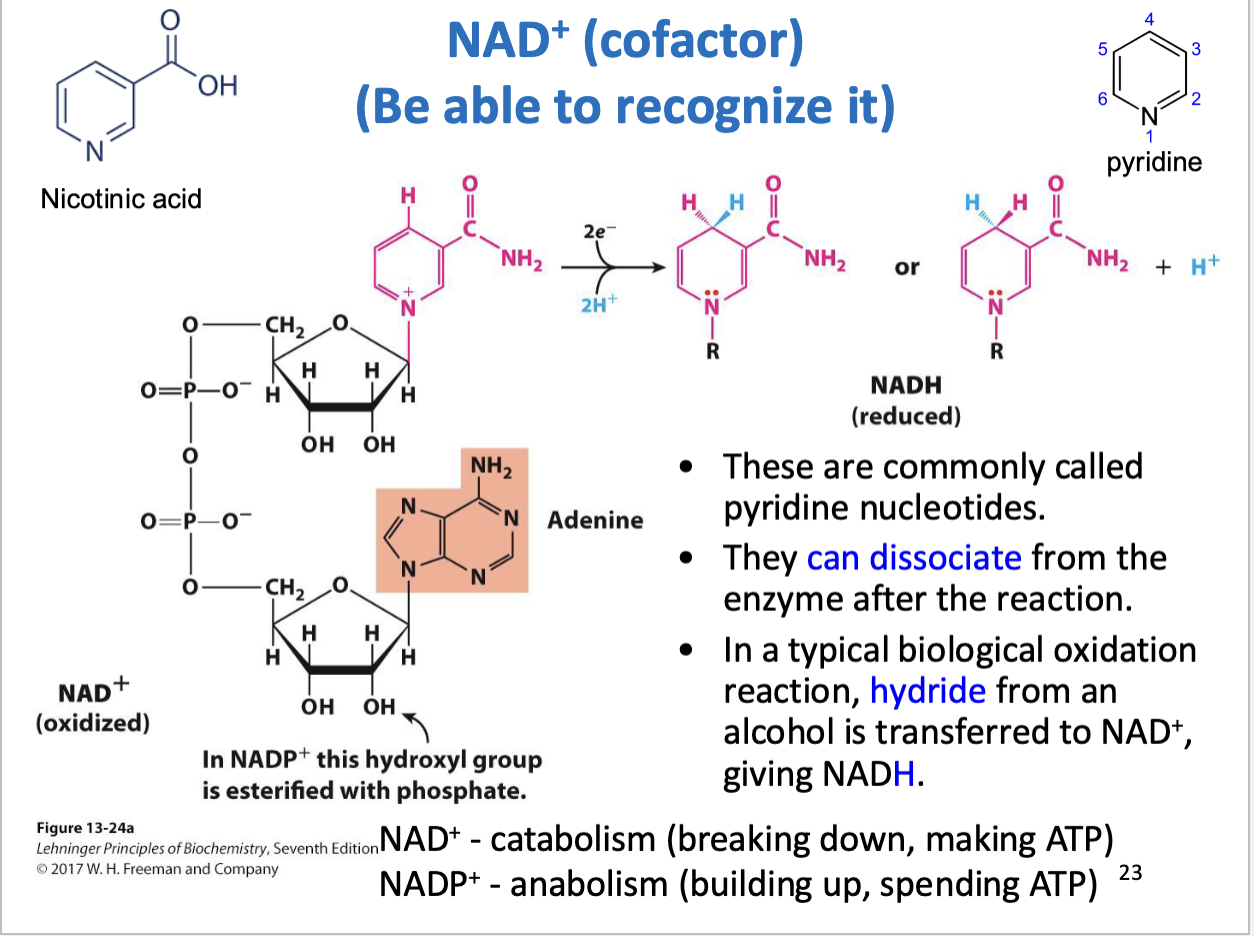
NAD+ (recognize structure)
NAD+ cofactor is used in catabolism (breaking down, making ATP)
NADP+ cofactor is used in anabolism (building up, spending ATP)
NADP+ has similar structure as NAD+, 2’ hydroxyl on ribose is esterified with phosphate
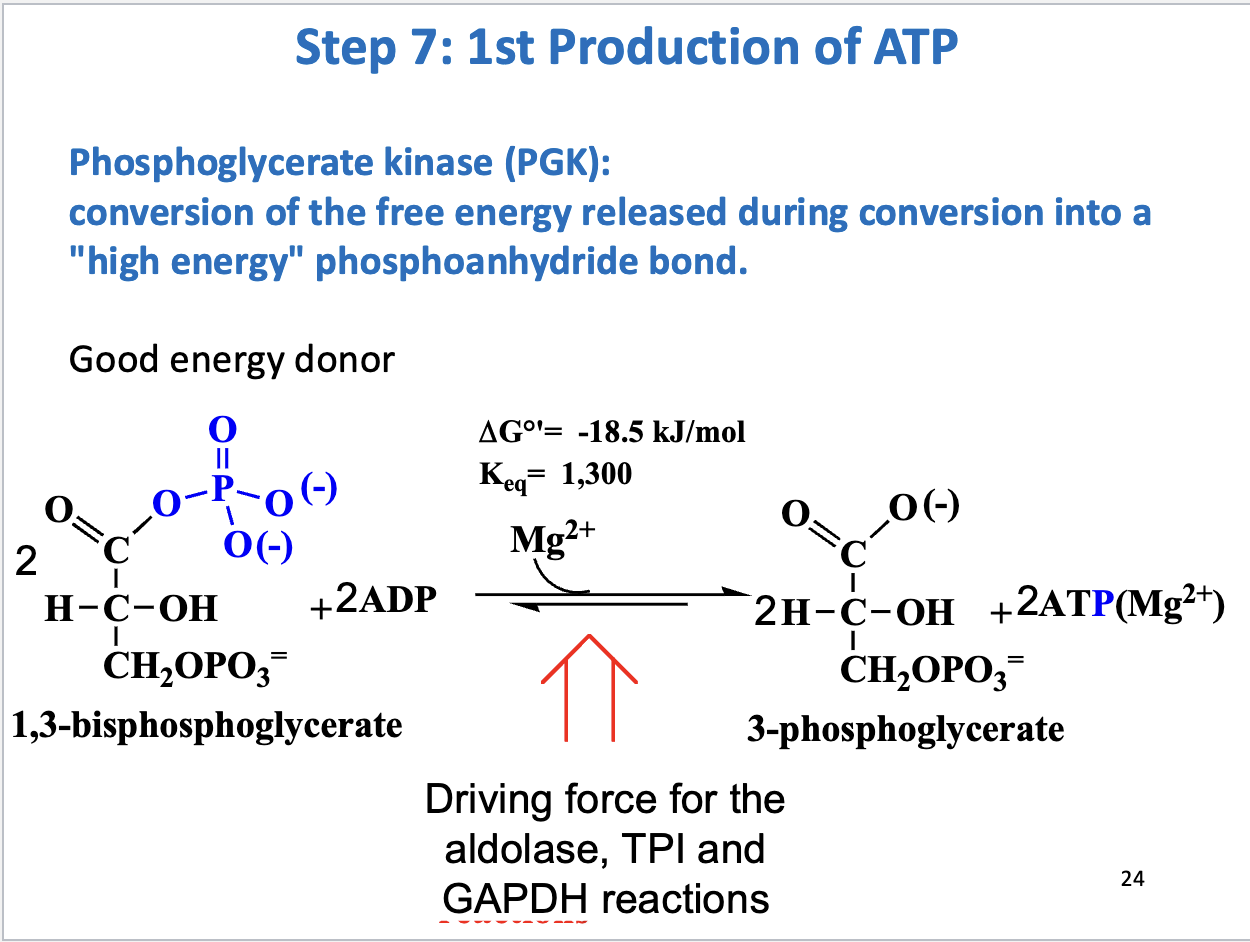
Glycolysis Step 7
1st production of ATP
Substrate: 2 1,3 bisphosphoglycerate, 2 ADP, Product: 2 3-phosphoglycerate, 2 ATP
Enzyme: phosphoglycerate kinase
Negative delta G (drives aldolase, TPI, and GAPDH reactions forward)
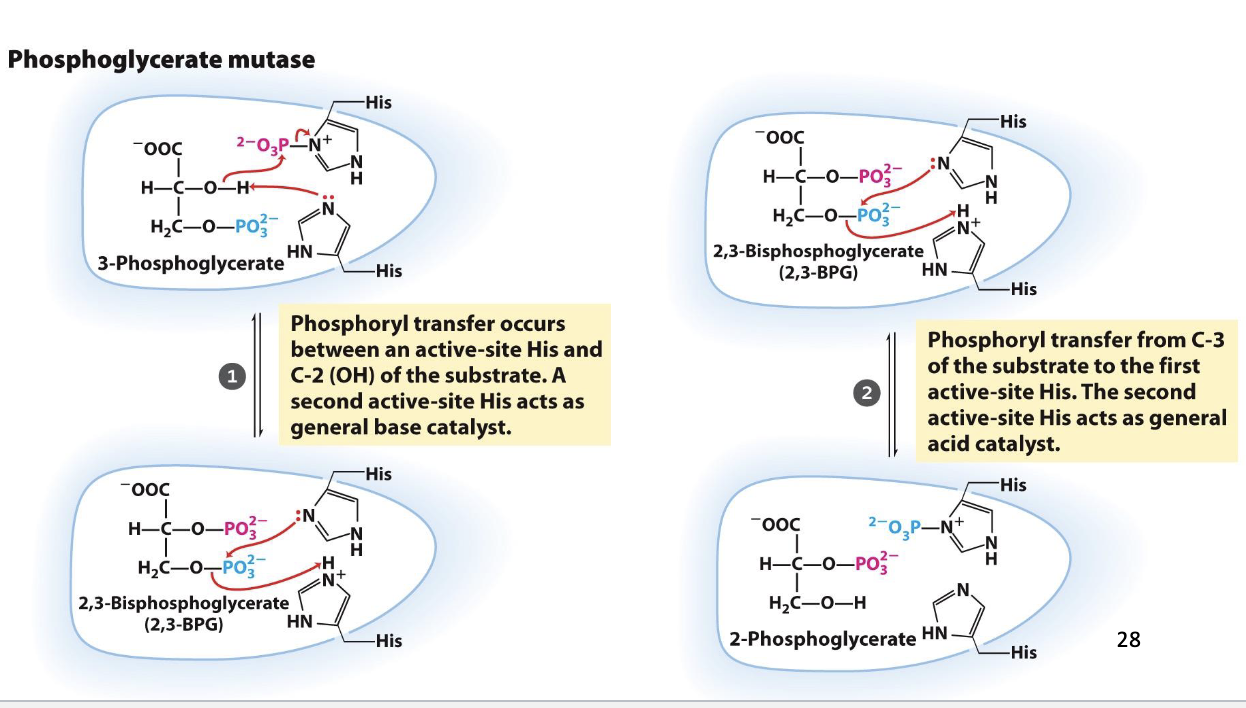
Glycolysis Step 8
Migration of the phosphate by PGMutase
Substrate: 3-phosphoglycerate, Product: 2-phosphoglycerate
Enzyme: phosphoglycerate mutase
Positive delta G (unfavorable/reversible, reactant concentration kept high to push reaction forward)
PGMutase mechanism
One active-site histidine is posttranslationally modified to phosphohistidine
Phosphohistidine donates phosphate to 3-phosphoglycerate at C2, before retrieving another phosphate from C3
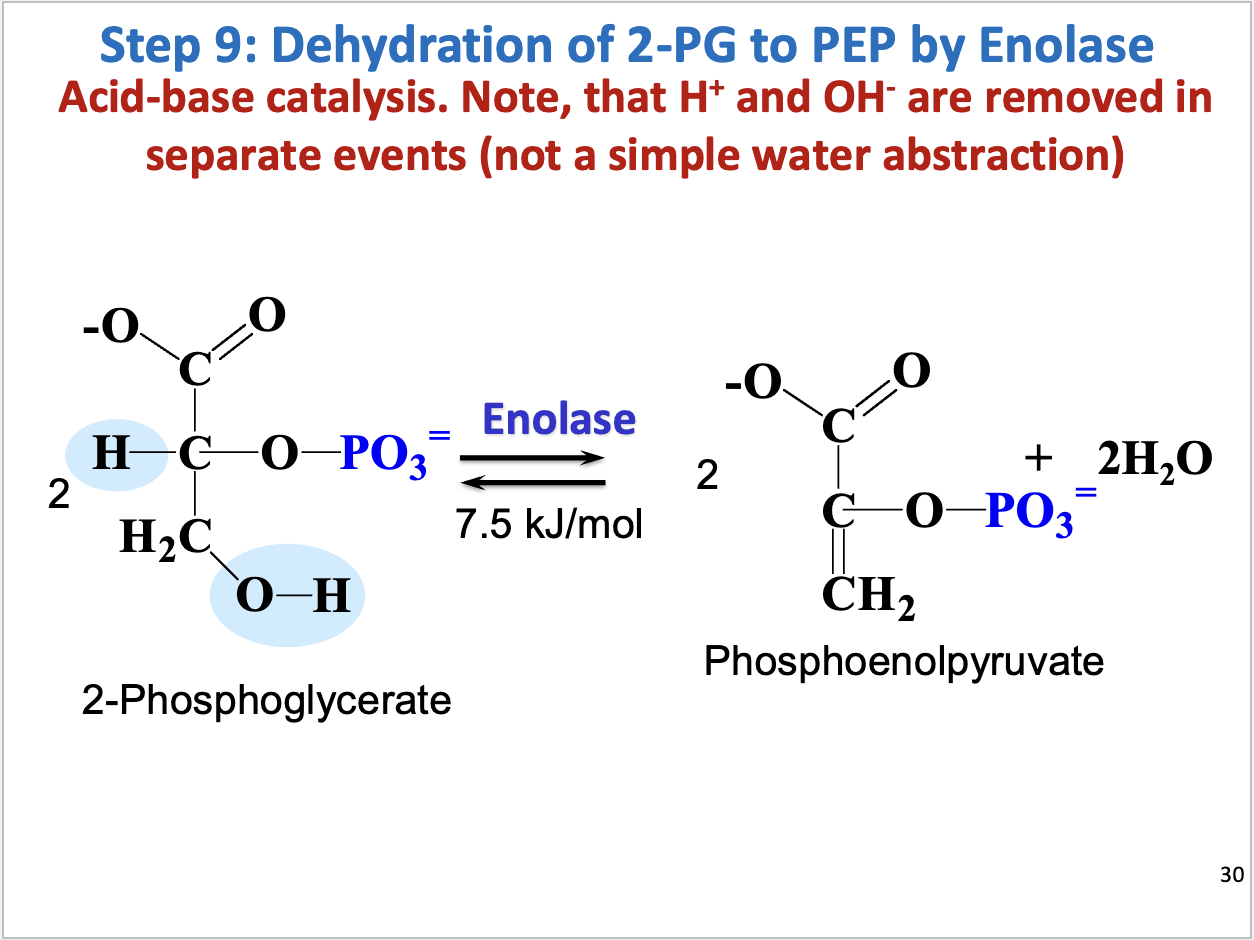
Glycolysis Step 9
Dehydration of 2-PG to PEP (acid-base catalysis)
Substrate: 2-phosphoglycerate, Product: phosphoenolpyruvate, water
Enzyme: enolase
Positive delta G (product concentration kept low to drive reaction forward)
Enolase mechanism
Acid-base catalysis
Goes through enolate intermediate (not covalent)
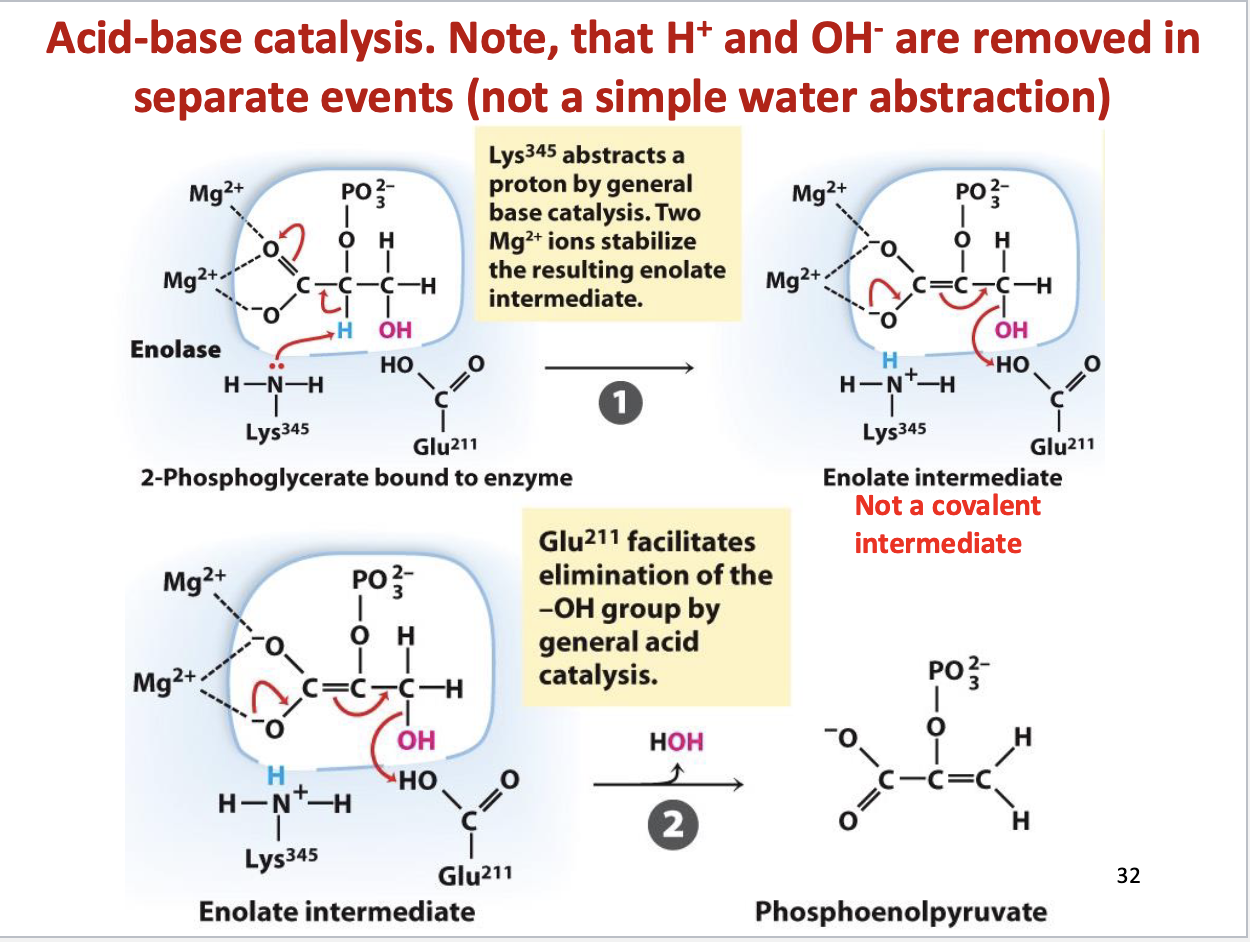
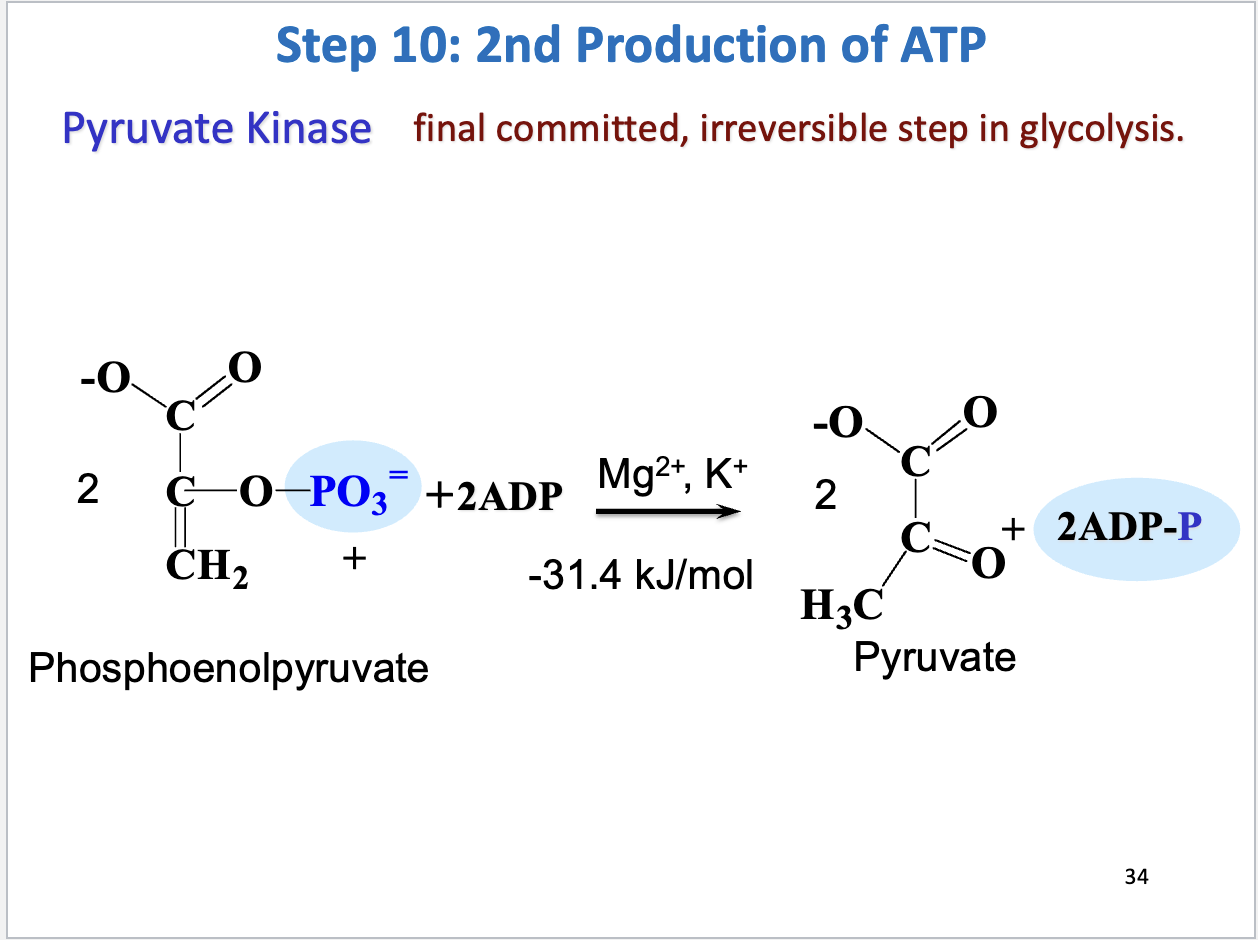
Glycolysis Step 10
2nd production of ATP
Substrate: 2 phosphoenolpyruvate + 2 ADP, Product: 2 pyruvate, 2 ATP
Enzyme: pyruvate kinase
Negative delta G