Exam 3 Chem 2
1/56
Earn XP
Description and Tags
chapters 21 and 23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
oxidation, reducing agent
-loss in electrons
-reducing agent gives electrons to the substance being reduced
reduction, oxidizing agent
-gain of electrons
-takes electrons from the substance being oxidized
balancing redox reactions in acidic and basic solutions
1) divide into half reactions
2) balance the atoms and charges in each half reaction, (H20 and H+)
3) multiply each half reactuion, if necessary, by an integer so that the number of e- is the same in both equations
4) Add the half reactions together and cancel substances that appear on both sides
5) In a base, add OH- to the side with H+. this creates water. excess H20 molecules are cancelled in the final step
voltaic cell
-spontaneous, ΔG<0 to generate electrical energy
system does work on the surroundings
electrolytic cell
-uses electrical energy to drive a nonspontaneous reaction (ΔG > 0)
the surroudings do work on the system
construction of a voltaic cell
-each half reaction takes place in its own half cell
-each half cell has an electrode in an electrolyte solution
-connected by the external circuit
-salt bride
allows ions to flow through both cellls, maintains electrical neutraility
contains nonreacting cations and anions
-oxidation occurs in anode, source of e-
causes anode to decrease in mass and the concentration of electrolyte to increase
cathode increase in mass
-cathode= positve
-anode= negative
active and inactive electrodes
-active= active componenet in its half cell, and is a reactant or product in the overall reaction
-inactive= does not participate actively in the overall reaction, provides a surface for the reaction
necessary when none of the reaction componenets can be used as an electrode (gas or aqueous)
ex. graphite, platinum
notation for a voltaic cell
-anode on left, cathode on right
-single line = phase boundary
-double line= physically seperated half cells
-inert electrode is on the end
-comma= used to show componenents are in the same phase
cell potential
E cell
-depends on the difference in electrical potential between the two electrodes
-also called the voltage of the cell or the elctromotive force (emf)
E cell > 0 (spontaneous, voltaic/galvanic)
E cell < 0 (nonspontaneous, electrolytic)
E cell = 0 at equilibrium
E cell = E cathode- E anode
standard hydrogen electrode (SHE)
= 0.00V
consists of a Pt electrode with H2 gas at 1atm bubblinh though it
Pt is in strong acid
SHE can act as a cathode or anode
the more positive the E value
-the better it is at gaining electrons, better at reduction
the more negative the E value
-the better it is at losing electrons, better at oxidizing
activity series of the metals
-metals that can displace H2 from acid are metals that are better at losing electrons than H2
-the more negative its E cell, the better is is at oxidizing, more active
concentration cell
-explotis the effect of concentration changes on cell potential
-has the same half reaction in both cell compartments but with different concentrations of electrolyte
-cell potential should be greater than 0 and the cell can do work
electrolytic cells
-uses electrical energy from an external source to drive a nonspontaneous redox reaction, electrolysis
-cathode= negative
-anode= positive
-ex. electroplasting of jewelry and autoparts, the electrolysis (breaking down) of chemical compounds
electrolysis of a molten salt
-no H20 involved
-cations: highest IE is the species that is reduced
-anions: lowest EA is the species that is oxidized
electrolysis of aqueous salt soltuons
-overvoltage- the additonal voltage needed to produce gases at metal electrodes (for H20)
0.4-0.6 V for H2 or O2
-highest E in the cathode gets electrolyzed
-lowest E in the anode gets electrolyzed
-anions not oxidized include F- and common oxo-anions (NO3-, SO42-, CO32-, etc), bc in highest oxidation state, can’t lose anymore
-cations of less active metals including gold, silver, etc are reduced to the metal
-cations of more active metals including those in Group 1A 2A and Al from 3A are not reduced, water is reduced instead
stoichiometry of electrolysis
1) mass (g) of substance oxidized or reduced
2) amount (mol) of substance
3) amount (mol) of electrons transferred
4) faradays constant to get coloumbs, charge
5) time to get to currect (A)
faraday’s law of electrolysis
-the amount of substance produced at each electrode is directly proportional to the quantity of charge flowing through the cell
-current= amount of charge/ time
measured in amperes A
d block elements condensed ground state configuration
[Noble Gas] ns²(n-1)dx
f sublevel general condesed ground state configuration
[noble gas] ns² f14 (n-1)dx
atomic size across a period for transition metals
-decreases at first then remains relatively constant
-the d electrons fill inner orbitals, so they shield outer electrons very efficiently and the 4s electrons are not pulled closer by the incresing nuclear charge
-period 4 and 5 vs period 5 and 6
increases from period 4 to 5 but not from period 5 to 6
lanthanide contraction
electronegativity across a period for transition metals
-tend to increase slightly, the decrease
—> ←
-period 4 to 5 vs period 5 to 6
increases within a group from period 4 to 5
remains unchanged from period 5 to 6
heavier elements often have higher EN values
ionization energy values tend to increase down a transition group
density increases dramatically down a group since atomic volumes change little while atomic masses increase significantly
first ionization energy across a period for transition metals
-increase relatively little
-higher at d10 ex. Zn bc d orbital filled so harder to take an electron from
lanthanide contraction
-the extra shrinkage from the increase in nuclear charge is roughly equal to the normal size increase due to adding an extra energy level
metallic behavior of transition metals
-lower the oxidation state, the more metallic, more basic
ionic bonding more prevalent
-higher the oxidation state, more covalent, more acidic
Eo for period 4
-ability to gain electrons better across a period
-period 4 transition metals active enough to recude H+ to H2 except for Cu
-most transition metals have an oxide coating that allows rapid reaction only with hot water or steam
color and magnetic behavior
-colorless= diamagnetic
-color= paramagnetic
lanthanides and actinides
-atomic properties of lanthanides vary little across the period, similar chemical properties
-actinides are radioactive, have very similar physical and chemical properties
+3 oxidation state is common
coordination compound
-contains at least one complex ion, central metal cation bonded to ligands
-associated with counter ions of opposite charge
-covalently bonded
coordination number
-number of ligand atoms bonded directly to the central metal ion
-most common is 6, but 2 and 4 are often seen
d8 metal ions = square planar (C.N. = 4) dsp2
d10 metal ions= tetrahedral (C.N. = 4) sp3
octahedral (C.N. = 6) d2sp3
linear (C.N. = 2)
monodentate ligands
-bond through a single donor atom
-water, ammonia, fluroide ion, chloride ion, cyanide ion, thiocyanate ion (SCN), hydroxide ion, nitrite ion
bidentate ligands
-have two donor atoms, each of which bonds to the metal ion
-ethylenediamine (en), oxolate ion (C2O42-)
polydentate ligands
-have more than two donor atoms
-triphosphate ion, diethenetriamine, EDTA
ambidentate
-bind to a different atom
-ex. nitrite
chelates
-bidentate and polydentate ligands give rise to rings in the complex ion, a chelate
-the ligand grabs the metal ion like claws
formula for coordination compounds
-cation is written first before the anion
-charge of the cation(s) are balanced by the chage of the anion(s)
-neutral ligands are written before anionic ligands
-formula of the while complex ion is placed in square brackets
naming coordination compounds
-cation is named before the anion
-ligands are named in alphabetical order before the metal ion
anionic ligands drop ide and add o
ex. chloride —> chloro
-a numerical prefix is used to indicate the number of ligands of a particular type
prefixes do not affect the alphabetical order
bis, tris, tetra
-a roman numveral is used to indicate the oxidation state for a metal that can have more than one state
-if the complex ion is an anion we drop the ending of the metal name and add -ate
ligand names
-ammonia= ammine
-water= aqua
-carbon monoxide= carbonyl
-phosphine= phosphine
-nitrogen oxide= nitrosyl
-chloride= chloro
-fluoride= fluoro
-cyanide= cyano
-hydroxide= hydroxo
-nitrite= nitro
names of metal ions in complex anions
-iron= ferrate
-copper= cuprate
-lead= plumbate
-silver= argentate
-gold= aurate
-tin= stannate
constitutional isomers
-compounds with the same formula, but with the atoms connected differently
coordination isomers
-composition of the complex ion, but not the compound is different
can occur by the exchange of a ligand and a counter ion, or by the exchange of ligands
ex. [Fe(NH3)5F] Br —> [Fe(NH3)5Br]F
linkage isomers
-occur when the composition of the complex ion is the same but the ligand donor atom is different
ex. [Fe(NH3)5(NO2)]Br and [Fe(NH3)5(ONO)]Br
stereoisomers
-compunds that have the same atomic connections, but different spatial arrangments of their atoms
geometric or cis-trans isomers
-occur when atoms or groups can either be arranged on the same side or on opposite sides of the compund relative to the central metal ion
cis- close as possible, usually 90 degrees apart
trans- far away as possible, usually 180 degrees apart
optical isomers (enantiomers)
-non-superimposable mirror images of each other
-species must be tetrahedral and all four ligands must be different, not square planar
-bidentate ligands- can only go “cis” isomerism, not trans, because of their size
-optically active (no plane of symmetry)
coordinate covalent bond
-when a complex ion is formed, each ligand donates an electron pair to the metal ion
-valence bond theory- the filled orbital of the ligand overlaps with an empty orbital of the metal ion
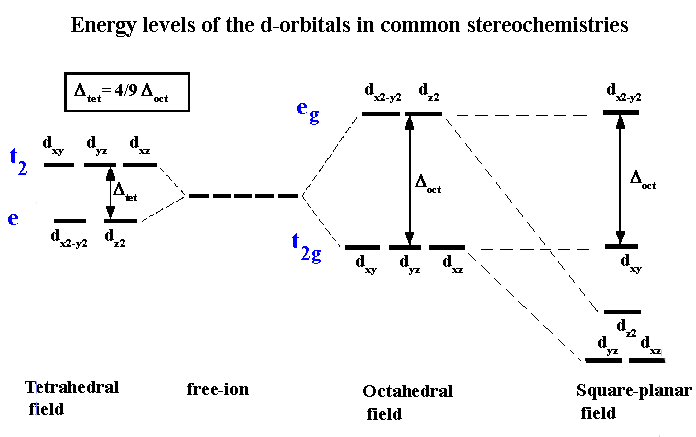
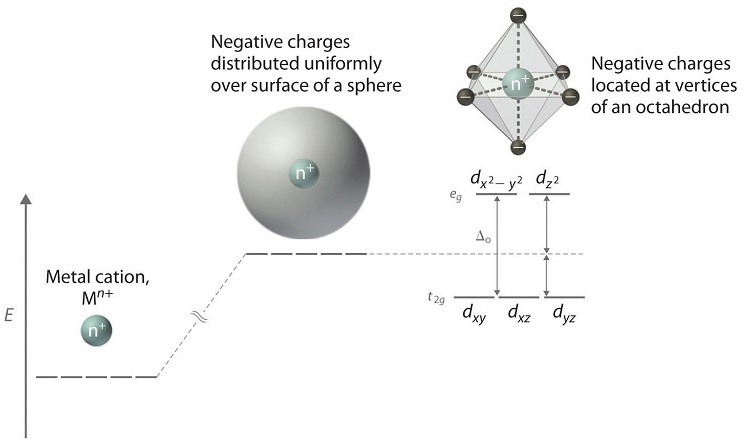
crystal field theory
-explains color and magnetism in terms of the effect of the ligands on the energies of the d-orbitals of the metal ion
-the bonding of the ligands to the metal ion cause the energies of the metal ion d-orbitals to split
-the splitting of the d-orbitals depends on the relative orientation of the ligands
splitting energy
-eg = higher level
-t2g = lower level
-the more strongly the ligands bond to the metal atom, the larger the energy gap between e and t
induces a low spin complex (number of unpaired e- is less than in the free ion)- fills up the lower level first
usually diamagnetic
-weak field ligands lead to a smaller splitting energy which leads to a high spin complex (max # of unpaired e-)
usually paramagnetic
seperation between strong field and weak field ligands
between EDTA and NH3
color due to CFT
-when the ion absorbs light, electrons can move from the lower t2g level to the higher eg level
-color is determined by the crystal field splitting energy
for a given ligand, the color depends on the oxidation state of the metal ion
for a given metal ion, the color depends on the ligand
spectrochemical series
-as 𝝙increases, shoter wavelengths (higher energies) of light must be absorbed to excite electrons
pairing energy
when all lower energy orbitals are half filled:
the next e- can enter a half filled orbital and pair up by overcoming repulsive pairing energy or
the next e- can enter an empty, higher, energy orbital by overcoming 𝝙
electron configuration of d1-d3 and d8-d10
-d1-d3: electrons only occupy the lower energy level, high spin
-d8-d10 : only show one type of spin
electron configuration of d4-d7
-can either be high spin or low spin depending on field strength
octahedral field
-2 on top, 3 on bottom
tetrahedral and square planar field of ligands
-The splitting of d orbitals energies is less in a tetrahedral than an octahedral complex, and the relative d-orbital energies are reversed
-only high-spin tetrahedral complexes are known bc 𝝙 is small
-square planar complexes are low spin and usually diamagnetic because the four pairs of d electrons fill the four lowest-energy orbitals