Biology Chapter 1 Pt 1 Basics
5.0(1)
Card Sorting
1/161
Earn XP
Description and Tags
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
162 Terms
1
New cards
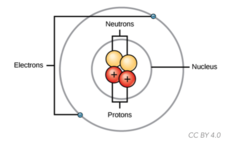
What is the fundamental building block of matter composed of protons, neutrons, and electrons?
atom
2
New cards
What is a group of 2 or more atoms held together by chemical bonds?
molecule
3
New cards
What term describes the ability of an atom to attract electrons?
electronegativity

4
New cards
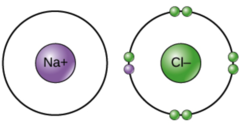
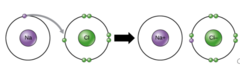
Which chemical bond involves the transfer of electrons from atom to atom where both atoms have different electronegativities?
ionic
5
New cards
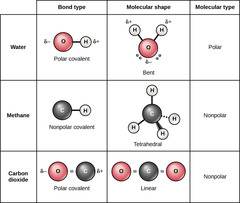
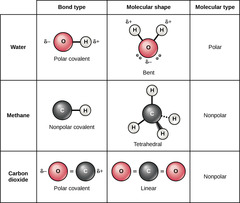
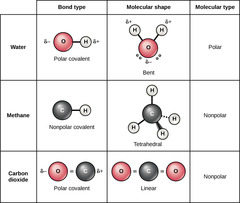
Which chemical bond involves electrons shared between atoms of similar electronegativities?
covalent
6
New cards
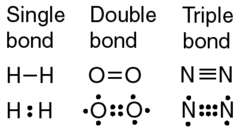
What number of covalent bonds can form between two atoms?
1(single), 2(double), or 3(triple)
7
New cards
Which covalent bond involves equal sharing of
electrons between two atoms of identical electronegativity?
electrons between two atoms of identical electronegativity?
non-polar
8
New cards
Which covalent bond involves unequal sharing of electrons
between two atoms of different electronegativities?
between two atoms of different electronegativities?
polar
(Note: leads to the
formation of a dipole)
(Note: leads to the
formation of a dipole)
9
New cards
Which chemical bond involves a weak bond between molecules with a hydrogen attached to a highly electronegative atom and is attracted to a negative charge on another molecule (F, O, or N)?
hydrogen bond
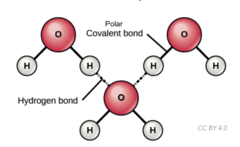
10
New cards
Which property of water describes its ability to dissolve substances with its dipole?
excellent solvent
11
New cards
Which property of water describes its ability to absorb a large amount of energy before changing temperature?
high heat capacity
(Note: also explains water's high heat of vaporization)
(Note: also explains water's high heat of vaporization)
12
New cards
Which property of water describes its expansion upon freezing to become less dense than its liquid form?
ice floats
(Note: H-bonds are maximum distance apart)
(Note: H-bonds are maximum distance apart)
13
New cards
Which property of water describes its attraction to like substances and itself?
cohesion/surface
tension
(Note: attracted to
other substances
with H-bonds,
including itself!)
tension
(Note: attracted to
other substances
with H-bonds,
including itself!)

14
New cards
Which property of water describes its attraction to unlike substances?
adhesion
(Note: capillary
action is the flow
of water without
external force -
ex: against gravity)
(Note: capillary
action is the flow
of water without
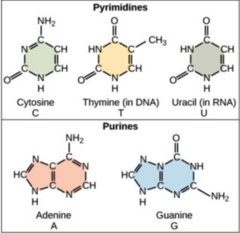
external force -
ex: against gravity)
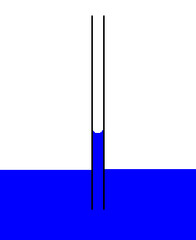
15
New cards
What are molecules composed of carbon atoms?
organic molecules
16
New cards
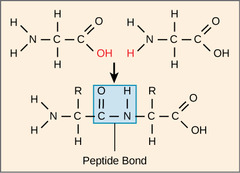
What is the simplest unit of a macromolecule?
monomer (1 unit)
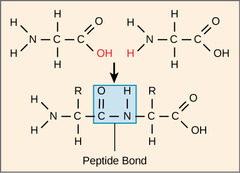
17
New cards
What is the term for the linking of monomers?
polymer
18
New cards
What are are particular clusters of atoms that give organic molecules their key properties?
functional groups
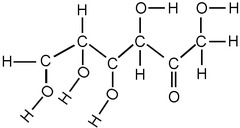
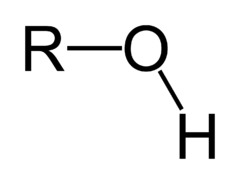
19
New cards
What is the chemical formula for the hydroxyl functional group?
OH
(Note: polar and hydrophilic)
(Note: polar and hydrophilic)
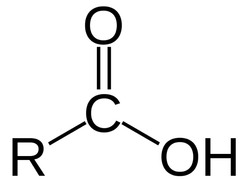
20
New cards
What is the chemical formula for the carboxyl functional group?
COOH
(Note: polar, hydrophilic, and a weak acid)
(Note: polar, hydrophilic, and a weak acid)
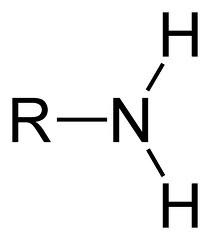
21
New cards
What is the chemical formula for the amino functional group?
NH2
(Note: polar, hydrophilic, and a weak base)
(Note: polar, hydrophilic, and a weak base)
22
New cards
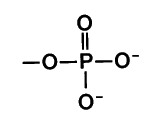
What is the chemical formula for the phosphate functional group?
(PO4)3-
(Note: polar, hydrophilic, acid)
(Note: polar, hydrophilic, acid)
23
New cards
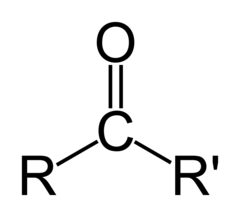
What is the chemical formula for the carbonyl functional group?
C=O
(Note: polar and hydrophilic)
(Note: polar and hydrophilic)
24
New cards
What is the chemical formula for the aldehyde functional group?
H-C=O
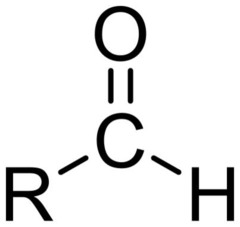
25
New cards
What is the chemical formula for the ketone functional group?
R-C=O
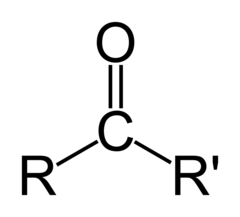
26
New cards
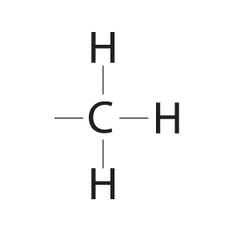
What is the chemical formula for the methyl functional group?
CH3
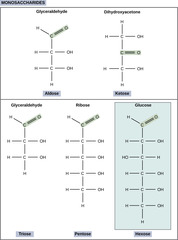
27
New cards
What is the term for a single sugar molecule with structure (CH2O)n.
monosaccharide
(ex: glucose or fructose)
(ex: glucose or fructose)
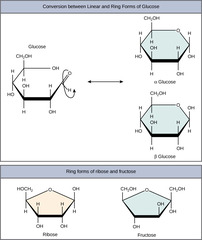
28
New cards
What structural component differentiates a monosaccharide as alpha or beta?
1. anomeric carbon -OH down = alpha
2. anomeric carbon -OH up = beta
anomeric carbon is carbon that used to be carbonyl (C=O)
2. anomeric carbon -OH up = beta
anomeric carbon is carbon that used to be carbonyl (C=O)
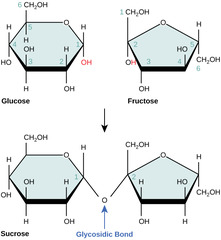
29
New cards
What is a two-sugar molecule joined by a glycosidic linkage?
disaccharide
(Note: sucrose, lactose, and maltose)
(Note: sucrose, lactose, and maltose)
30
New cards
What is a series of connected monosaccharides?
polysaccharide
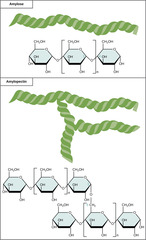
31
New cards
By what mechanism do polymers bonds form?
dehydration synthesis
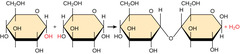
32
New cards
By what mechanism do polymers bonds break?
hydrolysis
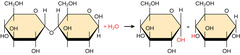
33
New cards
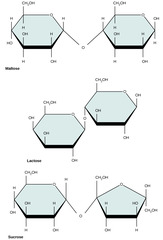
Which monomers compose sucrose?
glucose + fructose
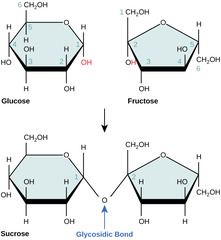
34
New cards
Which monomers compose lactose?
glucose + galactose
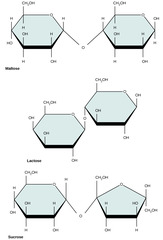
35
New cards
Which monomers compose maltose?
glucose + glucose
36
New cards
What is a polymer of alpha-glucose molecules; store energy in plant cells?
starch
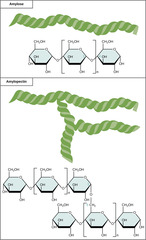
37
New cards
What is a polymer of alpha-glucose molecules; store energy in animal cells?
glycogen
(Note: differ in polymer branching from starch)
(Note: differ in polymer branching from starch)
38
New cards
What is a polymer of beta-glucose; structural molecules for walls of plant cells and wood
cellulose
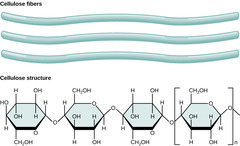
39
New cards
What is a polymer similar to cellulose, except each beta-glucose group has a nitrogen-containing group (n-acetylglucosamine) attached to the ring?
chitin
(Note: structural molecule in insect exoskeletons and fungal cell walls)
(Note: structural molecule in insect exoskeletons and fungal cell walls)
40
New cards
What are hydrophobic molecules that function in insulation, energy storage?
lipids

41
New cards
What are lipids consisting of three fatty acid chains attached to a glycerol backbone?
triglycerides
(AKA: triacylglycerols)
(AKA: triacylglycerols)
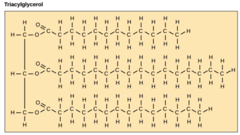
42
New cards
Which triglyceride contains no double bonds and has straight chains?
saturated
(Note: are bad for
health since the straight
chains stack densely and
form fat plaques)
(Note: are bad for
health since the straight
chains stack densely and
form fat plaques)

43
New cards
Which triglyceride contains double bonds that cause kinks in chains?
unsaturated
(Note: are better for
health since chains
stack less densely;
can be cis or trans)
(Note: are better for
health since chains
stack less densely;
can be cis or trans)

44
New cards
What are lipids comprised of two fatty acids and a phosphate group (+R) attached to a glycerol backbone?
phospholipids
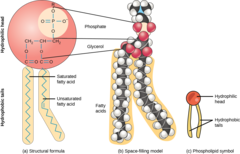
45
New cards
What is the term for a phospholipid exhibiting both hydrophobic and hydrophilic properties?
amphipathic
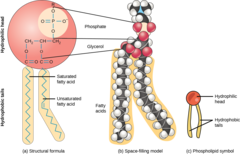
46
New cards
Which lipid derivates contain three 6 membered rings and one 5 membered ring?
steroids
(Note: sex hormones,
cholesterol,
corticosteroids)
(Note: sex hormones,
cholesterol,
corticosteroids)
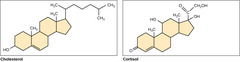
47
New cards
Which lipid derivatives are esters of fatty acids and monohydroxylic alcohols, used as protective coating or exoskeletons (lanolin)?
waxes

48
New cards
Which lipid derivatives are fatty acid carbon chains with conjugated double bonds and six-membered C-rings at each end?
carotenoids
(Note: includes
pigments which
produce colors in
plants and animals.
Subgroups are
carotenes and xanthophylls)
(Note: includes
pigments which
produce colors in
plants and animals.
Subgroups are
carotenes and xanthophylls)
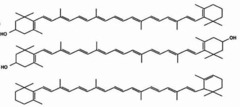
49
New cards
Which lipid derivatives are a 4 joined pyrrole ring that often complexes with a metal?
*Essential for function of hemoglobin.
*Essential for function of hemoglobin.
porphyrins
(AKA: tetrapyrroles)
(AKA: tetrapyrroles)
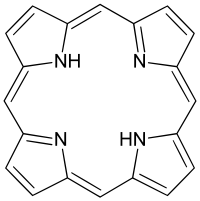
50
New cards
Which lipid derivatives are specialized in storage?
*White and brown
*White and brown
adipocytes
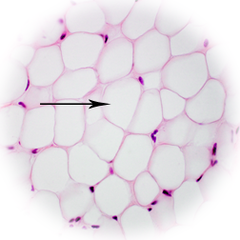
51
New cards
Which adipocyte is composed primarily of triglycerides with a small layer of cytoplasm around it?
white fat cell
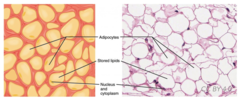
52
New cards
Which adipocyte is composed mostly of mitochondria and cytoplasm with lipid droplets scattered throughout.
brown fat cell
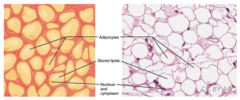
53
New cards
Which lipid derivatives are similar to phospholipids but have a carbohydrate group instead of a phosphate group?
glycolipids
54
New cards
Which lipid derivatives contain lipid cores surrounded by phospholipids and apolipoproteins to transport fats in the blood?
lipoproteins
55
New cards
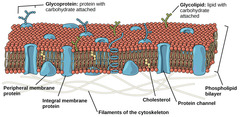
What membrane components might cells modify to maintain their cell membrane's fluidity?
fatty acids

56
New cards
In cold weather, what naturally happens to cell membranes?
become rigid
57
New cards
In warm weather, what naturally happens to cell membranes?
become more fluid
58
New cards
incorporate cholesterol
and polyunsaturated
fatty acids into the membrane.
Cholesterol acts as buffer and polyunsaturated increases membrane fluidity.
and polyunsaturated
fatty acids into the membrane.
Cholesterol acts as buffer and polyunsaturated increases membrane fluidity.
In cold weather, how does a cell compensate to prevent cell membrane rigidity?
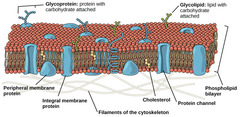
59
New cards
incorporate cholesterol
into the membrane as buffer. fatty acid tails are saturated and straight so they can pack and decrease fluidity.
into the membrane as buffer. fatty acid tails are saturated and straight so they can pack and decrease fluidity.
In warm weather, how does a cell compensate to prevent cell membrane collapse?
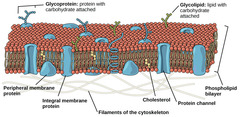
60
New cards
What are polymers of amino acids joined by peptide bonds?
proteins
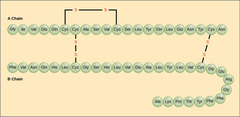
61
New cards
Casein in milk, ovalbumin in egg whites, and zein in corn seeds are examples of which type of proteins?
storage proteins
62
New cards
hemoglobin and cytochromes are examples of which type of proteins?
transport proteins
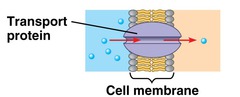
63
New cards
Which proteins catalyze reactions in both forward and reverse directions based on the substrate concentration?
enzymes
(Note: almost all
are proteins, but
RNA can also act
as an enzyme)
(Note: almost all
are proteins, but
RNA can also act
as an enzyme)
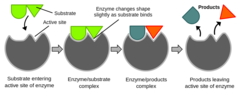
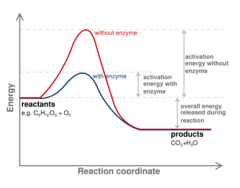
64
New cards
They do not change the spontaneity only the rate at which it occurs. It is not an equilibrium point.
Enzymes and reaction spontaneity.
65
New cards
By what factors is enzyme efficiency determined?
temperature and pH
66
New cards
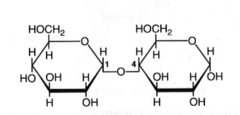
Amylase catalyzes the breaking of which bonds in starch?
alpha-glycosidic
67
New cards
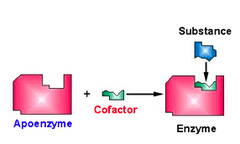
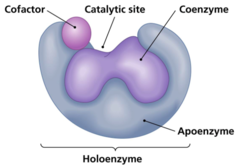
What are non-protein molecules that assist enzymes?
cofactors
68
New cards
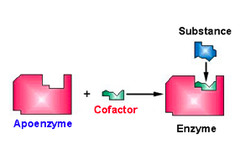
What is an enzyme called that is not combined with its cofactor?
apoenzyme/apoprotein
69
New cards
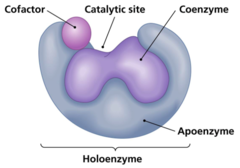
What is an enzyme called that is combined with its cofactor?
holoenzyme
70
New cards
What is a cofactor that is organic?
coenzyme
(ex: vitamins)
(ex: vitamins)
71
New cards
What is a cofactor that is covalently bound to its enzyme?
prosthetic group
72
New cards
What is the classification of proteins that are formed entirely of amino acids?
ex: albumin, globulin, histones, albuminoid/sleroprotein
ex: albumin, globulin, histones, albuminoid/sleroprotein
simple proteins
73
New cards
What is the classification of functional proteins that act as carriers or enzymes?
albumins and globulins
74
New cards
What is the classification of fibrous proteins that have
structural function (ex: collagen)?
structural function (ex: collagen)?
scleroproteins/albuminoid
75
New cards
Complex proteins composed of simple proteins and cofactors.
ex: lipoprotein, glycoprotein, chromoproteins, metalloprotein, nucleoprotein
ex: lipoprotein, glycoprotein, chromoproteins, metalloprotein, nucleoprotein
conjugated proteins
76
New cards
What is the classification of a protein bound to a lipid?
lipoprotein
77
New cards
What is the classification of a protein bound to a
carbohydrate?
carbohydrate?
glycoprotein
78
New cards
What is the classification of a protein bound to a pigmented molecule?
chromoprotein
79
New cards
What is the classification of a protein complexed
around a metal ion?
around a metal ion?
metalloprotein
80
New cards
What is the classification of a protein that contains histone or
protamine, bound to nucleic acid?
protamine, bound to nucleic acid?
nucleoprotein
81
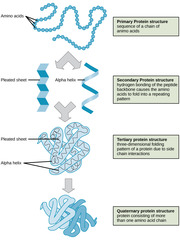
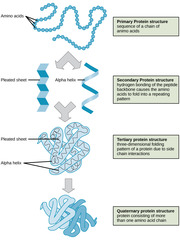
New cards
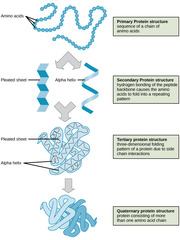
Which protein structure involves the sequence of amino acids connected by peptide bonds?
primary structure
82
New cards
Which protein structure involves the 3D shape resulting from hydrogen bonding between amino and carboxyl groups of adjacent amino acids?
secondary structure
(Note: alpha helices and beta sheets)
(Note: alpha helices and beta sheets)
83
New cards
Which protein structure involves the 3D structure that forms due to non-covalent interactions between amino acid R groups (subunit interaction)?
tertiary
84
New cards
1. H-bonds
2. ionic bonds
3. hydrophobic effect
4. disulfide bonds
5. Van Der Waals forces
2. ionic bonds
3. hydrophobic effect
4. disulfide bonds
5. Van Der Waals forces
What are the non-covalent interactions found in tertiary structure?
85
New cards
Which protein structure involves the 3D shape of a protein that is a grouping of two or more separate peptide chains?
quaternary structure
86
New cards
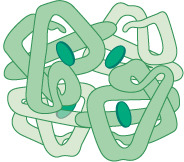
Which proteins are somewhat water-soluble, dominated by tertiary structure, and have a diverse range of functions?
ex: hemoglobin, insulin, immunoglobin.
ex: hemoglobin, insulin, immunoglobin.
globular proteins
87
New cards
1. enzymatic
2. hormonal
3. inter/intracellular storage and transport
4. osmotic regulation
5. immune response
2. hormonal
3. inter/intracellular storage and transport
4. osmotic regulation
5. immune response
What are the functions of globular proteins?
88
New cards
Which proteins are not water soluble, dominated by secondary structure, are long polymers, and add strength to cells?
fibrous/structural proteins
(Note: collagen and keratin)
(Note: collagen and keratin)

89
New cards
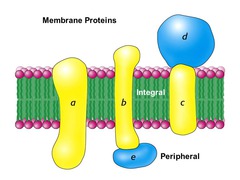
Which proteins function as membrane pumps, channels, or receptors?
membrane proteins
90
New cards
What process can occur when proteins are taken out of their ideal temperature, pH range, or solvent?
denaturation
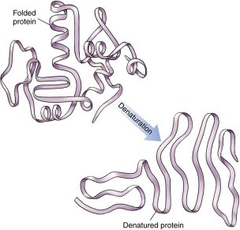
91
New cards
reversed back to primary structure
What happens to the structure of the protein following denaturation?
92
New cards
usually irreversible, but in some cases, it can be reversed with the removal of the denaturing agent
Is protein denaturation permanent?
93
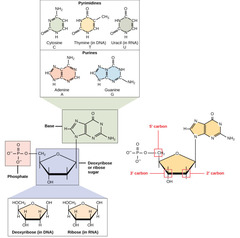
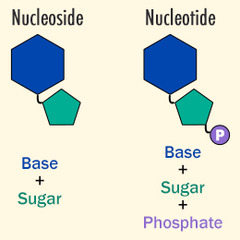
New cards
What are monomers that make up nucleic acids?
nucleotides
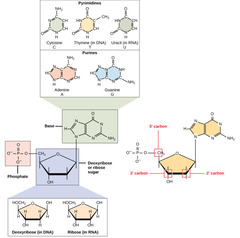
94
New cards
1. nitrogenous base
2. five carbon deoxyribose sugar
3. phosphate group
2. five carbon deoxyribose sugar
3. phosphate group
What are the components of nucleotides?
95
New cards
What unit consists of a sugar and nitrogenous base?
nucleoside
96
New cards
What is a nitrogen-containing compound that makes up a nucleotide?
nitrogenous base
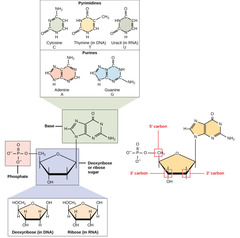
97
New cards
1. adenine (A)
2. thymine (T)
3. cytosine (C)
4. guanine (G)
2. thymine (T)
3. cytosine (C)
4. guanine (G)
What are the nitrogenous bases found in DNA?
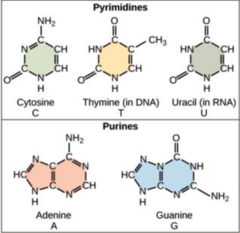
98
New cards
2
How many hydrogen bonds connect A and T?
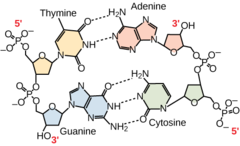
99
New cards
3
How many hydrogen bonds connect C and G?
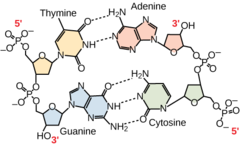
100
New cards
1. adenine (A)
2. uracil (U)
3. cytosine (C)
4. guanine (G)
2. uracil (U)
3. cytosine (C)
4. guanine (G)
What are the nitrogenous bases found in RNA?