Enthalp Changes
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
What is meant by an enthalpy change (ΔH)?
Enthalpy change is the heat energy transferred to or from the surroundings during a chemical reaction at constant pressure. It is the difference between the enthalpy of the products and the enthalpy of the reactants:
ΔH = H(products) − H(reactants).
What is the difference between an exothermic and an endothermic reaction in terms of ΔH and energy transfer?
In an exothermic reaction, the system releases heat energy to the surroundings — the products have lower enthalpy than the reactants, so ΔH is negative and the surroundings get warmer (e.g. combustion, neutralisation).
In an endothermic reaction, the system absorbs heat from the surroundings — the products have higher enthalpy than the reactants, so ΔH is positive and the surroundings get cooler (e.g. thermal decomposition).
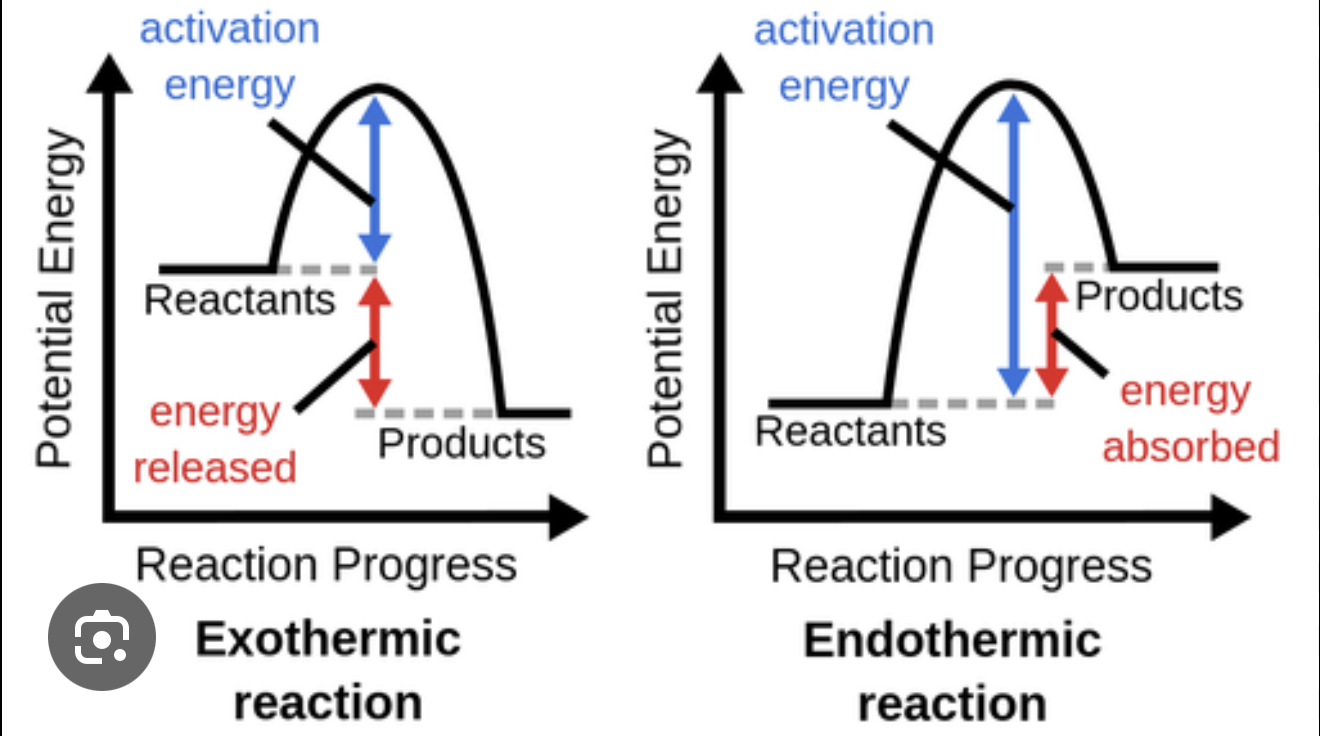
What does an enthalpy profile diagram show, and how does it differ for exothermic and endothermic reactions?
An enthalpy profile diagram plots enthalpy on the y-axis against the reaction pathway (progress of reaction) on the x-axis.
What is activation energy (Ea)?
Activation energy is the minimum energy required for a reaction to take place. It is the energy needed to break bonds in the reactants so that new bonds can form in the products. On a reaction profile diagram, it is the difference in enthalpy between the reactants and the peak of the energy curve (the transition state).
What are the standard conditions used for measuring enthalpy changes?
Standard conditions are:
pressure of 100 kPa and
specified temperature of 298 K (25°C)
all substances in their standard states (their normal physical state under these conditions)
a concentration of 1 mol dm⁻³, for solutions only.
Define the standard enthalpy change of formation (ΔfH°)
The standard enthalpy change of formation is the enthalpy change when 1 mole of a compound is formed from its elements in their standard states under standard conditions. The standard enthalpy of formation of any element in its standard state is zero by definition.
Example: C(s) + 2H₂(g) → CH₄(g), ΔfH° = −75 kJ mol⁻¹
Define the standard enthalpy change of combustion (ΔcH°)
The standard enthalpy change of combustion is the enthalpy change when 1 mole of a substance is completely burned in excess oxygen, with all reactants and products in their standard states under standard conditions. Enthalpies of combustion are always negative (exothermic). The equation must be written so there is 1 mole of the substance being burned, even if this requires fractional coefficients elsewhere.
Example: C₃H₈(g) + 5O₂(g) → 3CO₂(g) + 4H₂O(l), ΔcH° = −2220 kJ mol⁻¹
Define the standard enthalpy change of neutralisation (ΔneutH°).
The standard enthalpy change of neutralisation is the enthalpy change when 1 mole of water is formed in a reaction between an acid and a base under standard conditions. It is always negative.
Example: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l), ΔneutH° = −57 kJ mol⁻¹
Why is the equation for enthalpy of formation sometimes balanced with fractions?
Because the definition requires exactly 1 mole of the product compound to be formed. If using whole number coefficients would give more than 1 mole of product, fractions must be used for the reactants instead.
For example, to form 1 mole of water: H₂(g) + ½O₂(g) → H₂O(l).
what is the entalhpy change of a reaction
Δr Hθ
The enthalpy change when the reactants in the stoichiometric equation react to form the products, with all species in their standard states
Can be both exothermic and endothermic