ALL REAGENTS Chapters 9-15 and Mechanism Pathways
1/65
Earn XP
Description and Tags
A set of vocabulary flashcards covering key terms and concepts from Chapter 9 of the lecture notes, focusing on organic reactions, reagents, and mechanisms.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
HOW TO STUDY
1) What is the starting material:
In this case, OH is the starting material (ALL OF CHAPTER 9)
2) What is the reagent?
What causes that reaction to happen to convert start to end product
3) What is the end product?
What causes the reaction to occur
-Alkoxide Reaction
-Double Bond formed
-Substitution Formed
-What is the leaving group? Good or Bad?
-How many carbons are attached to the OH?
-Any carbocation rearrangements?
4) Look at the end product first and then the starting product for retrosynthesis
CHAPTER 9 Reagents
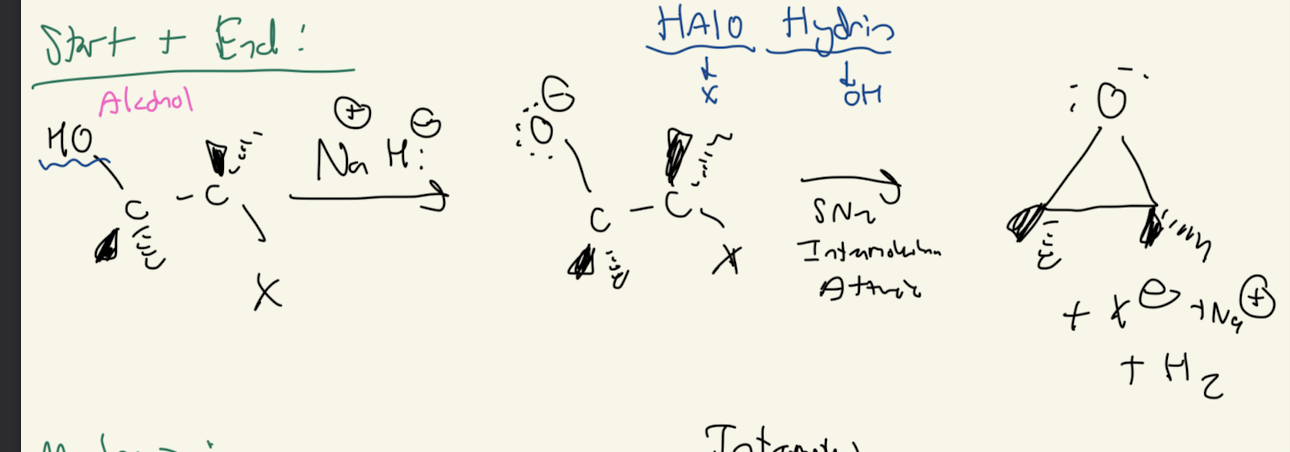
1) Reagent: Na+OH-

2) Na+H-
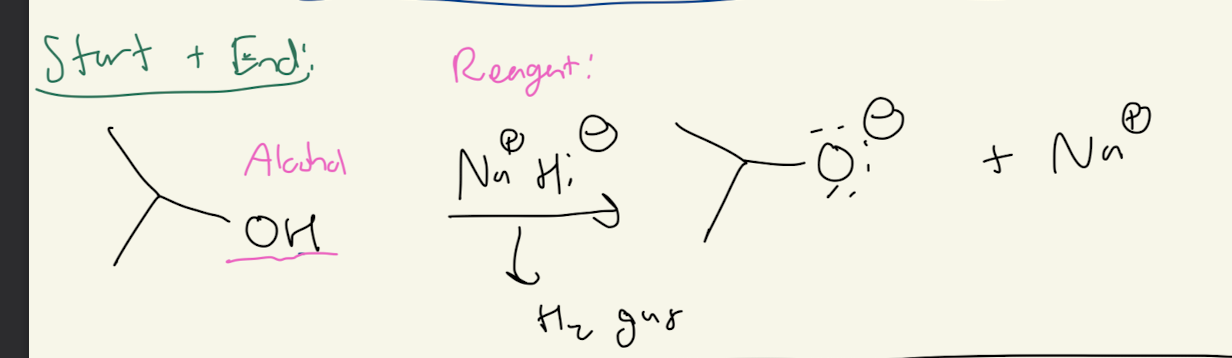
2a) Na+H-
Path #1: Alkoxide —> Ether
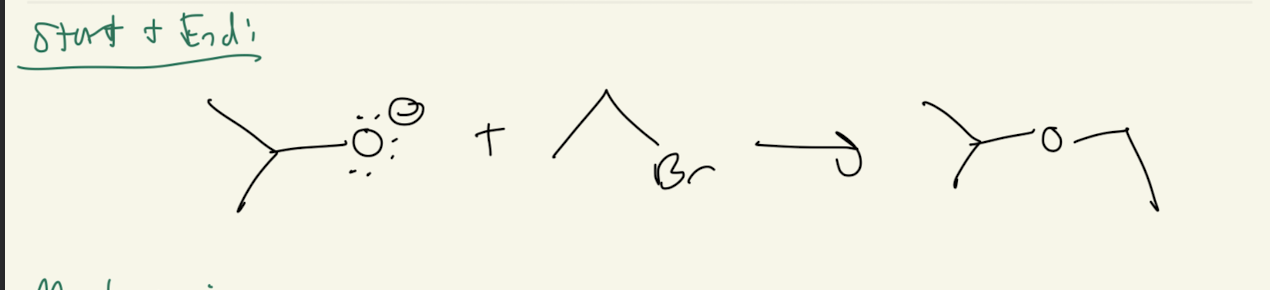
2b) Na+H-
Path#2: Alkoxide → Epoxide
A reagent used for converting alcohols into chlorides, often used in organic synthesis.
3) H2SO4
Acid Catalyzed Dehydration
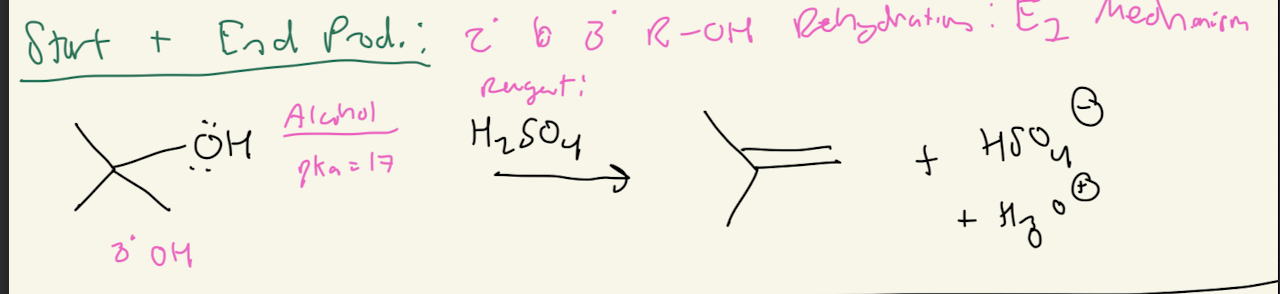
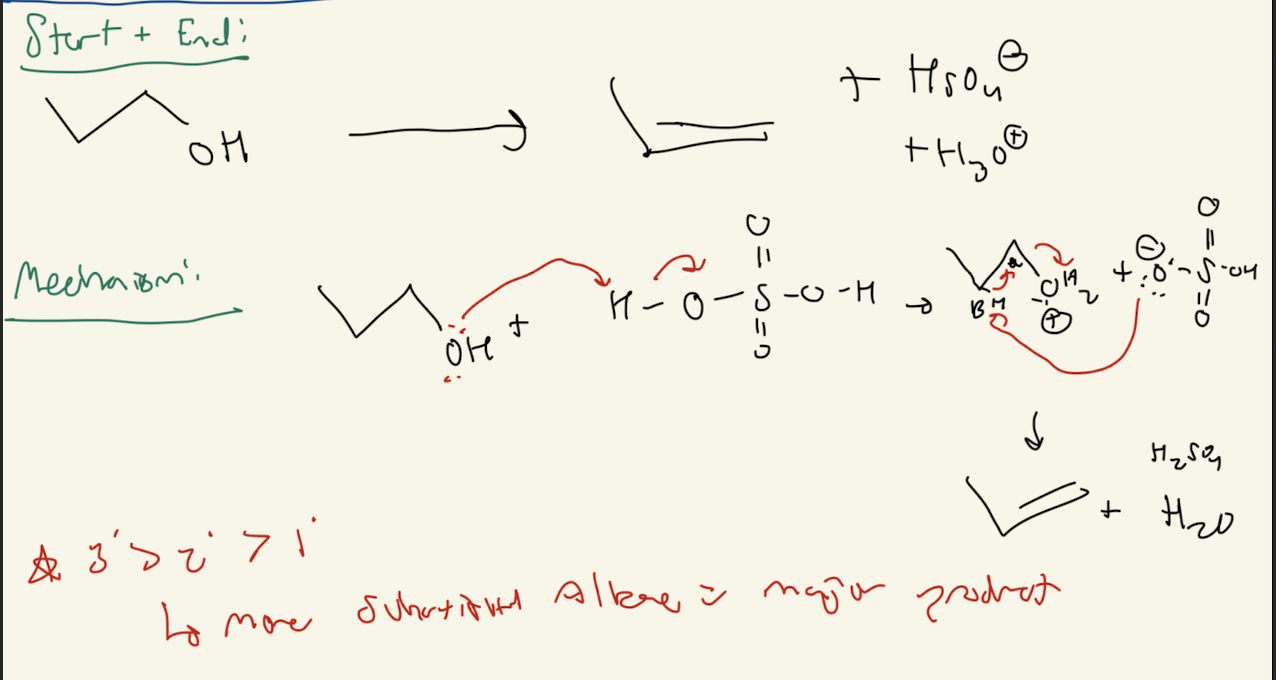
3a) H2SO4
Path #1: E1 Mechanism with 2° and 3° R-OH
Acid Catalyzed Dehydration
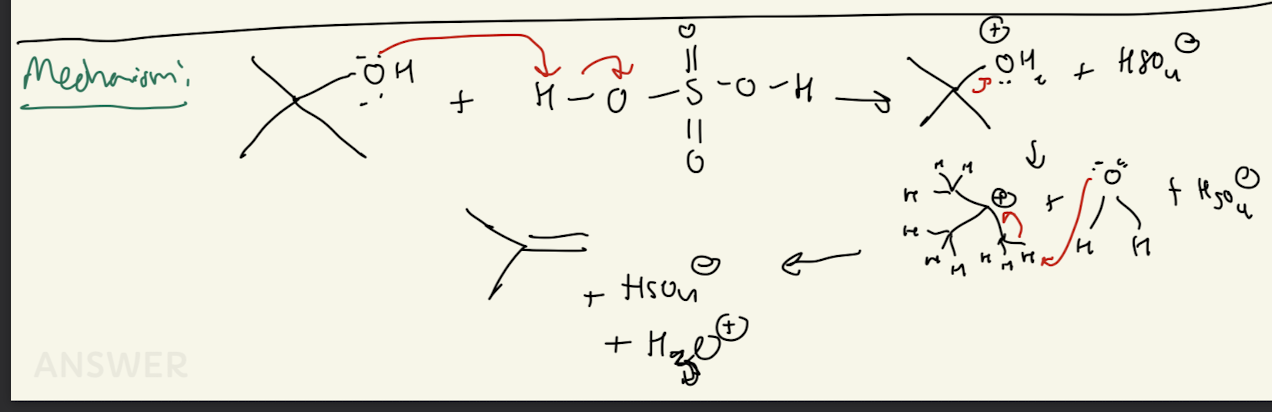
3b) H2SO4
Path #2: E2 Mechanism with 1° R-OH
Acid Catalyzed Dehydration
4) H-x (X= I, Br, Cl)
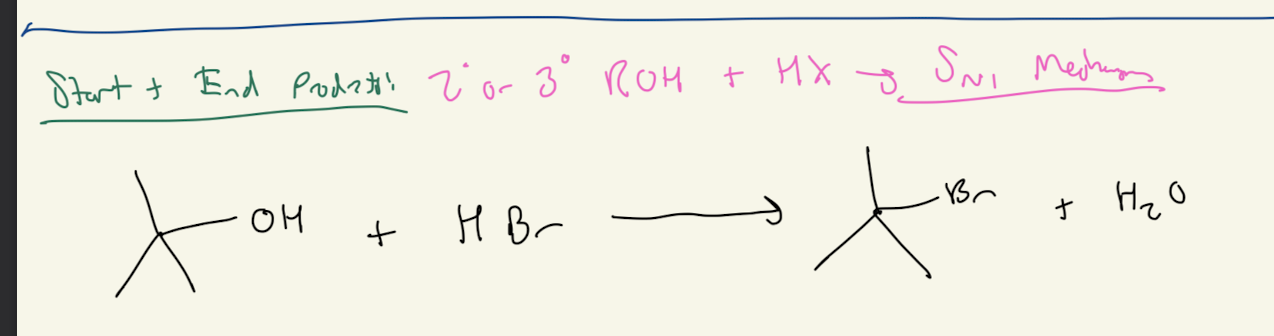
4a) H-X
Path #1: 1° ROH + HX —> SN2 Mechanism
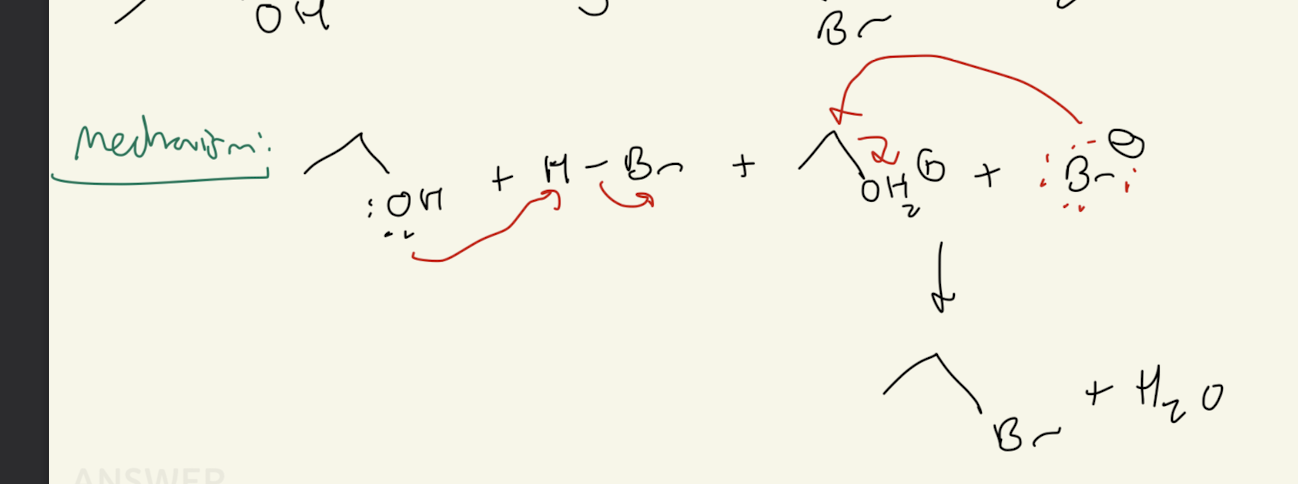
4b) H-X
Path #2: 2° or 3° ROH + HX —> SN1 Mechanism
SN1 Mechanism
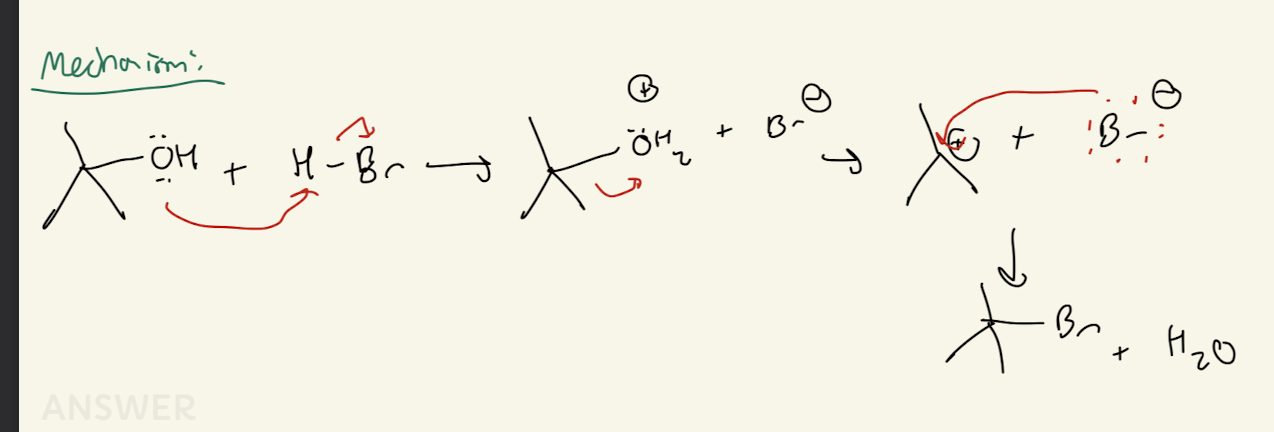
#5) SOCl2 (thionyl chlroide) + pyridine
1° and 2° only
SN2 Mechanism
So chemists prefer SOCl₂ or PBr₃ when they want clean substitution without rearranging the carbon skeleton.
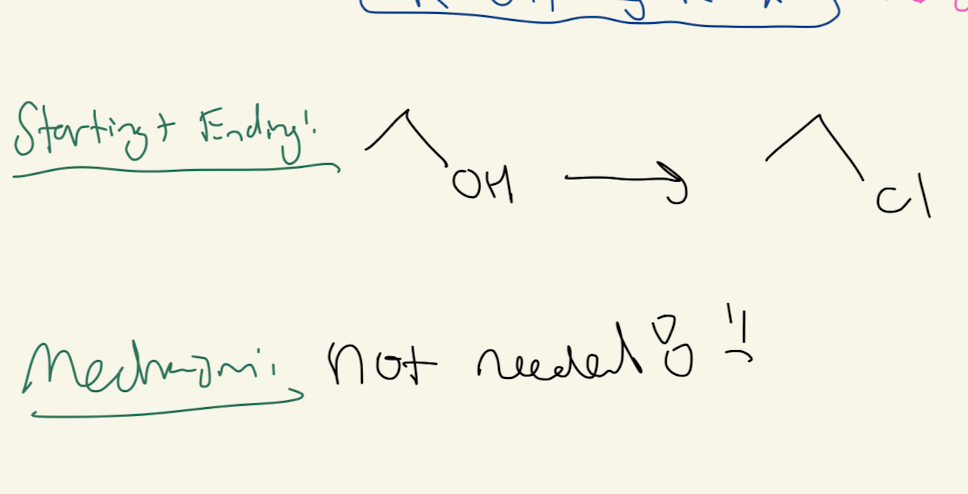
#6) PbBr3
So chemists prefer SOCl₂ or PBr₃ when they want clean substitution without rearranging the carbon skeleton.
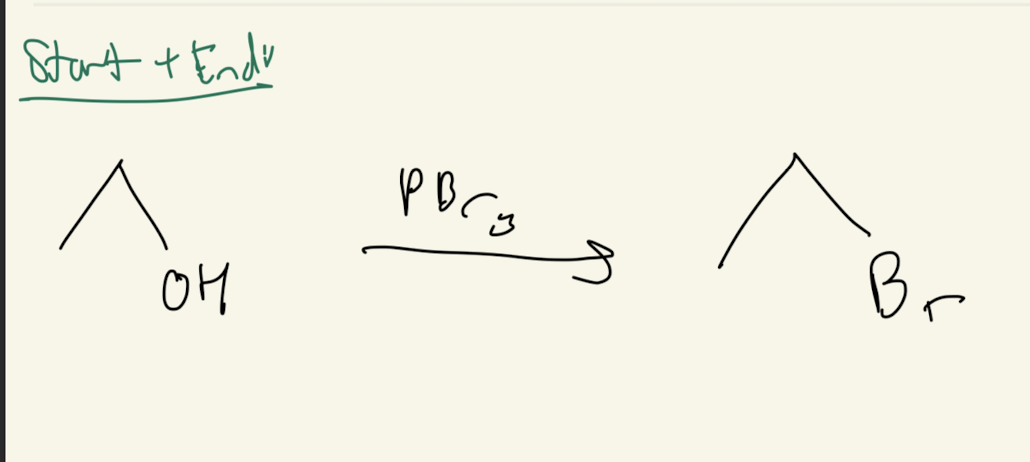
What’s the difference between using an H-X (X= Cl, Br) for substitution instead of using SOCl2 and PBr3 as reagents. What about Vice Versa? Which reagent should I use in which situation? Do some scenarios have the same product using both types of reagents?
Quick exam shortcut
Alcohol type | Best reagent |
|---|---|
1° alcohol | SOCl₂ or PBr₃ |
2° alcohol | SOCl₂ or PBr₃ (to avoid rearrangement) |
3° alcohol | HX (HCl or HBr) |
HX → SN1 (good for 3° alcohols, rearrangements possible)
SOCl₂ / PBr₃ → SN2 (best for 1° and 2°, no rearrangements
1. Using HX (HCl, HBr)
Examples: Hydrochloric acid and Hydrobromic acid
Mechanism depends on the alcohol
3° alcohol
Reaction proceeds via SN1
Forms a carbocation
Rearrangements can occur
2° alcohol
Often SN1, sometimes mixed mechanisms
Rearrangements possible
1° alcohol
Usually SN2
Rearrangements usually don’t occur
Pros
✔ Works well for tertiary alcohols
✔ Simple reagent
Cons
❌ Carbocation rearrangements possible
❌ Strong acidic conditions can cause elimination (alkenes)
2. Using SOCl₂ or PBr₃
Thionyl chloride converts alcohol → alkyl chloride
Phosphorus tribromide converts alcohol → alkyl bromide
Mechanism
Usually SN2
No carbocation intermediate
Pros
✔ No rearrangements
✔ Very clean reactions
✔ Excellent for 1° and 2° alcohols
Cons
❌ 3° alcohols don’t work well (SN2 is blocked by sterics)
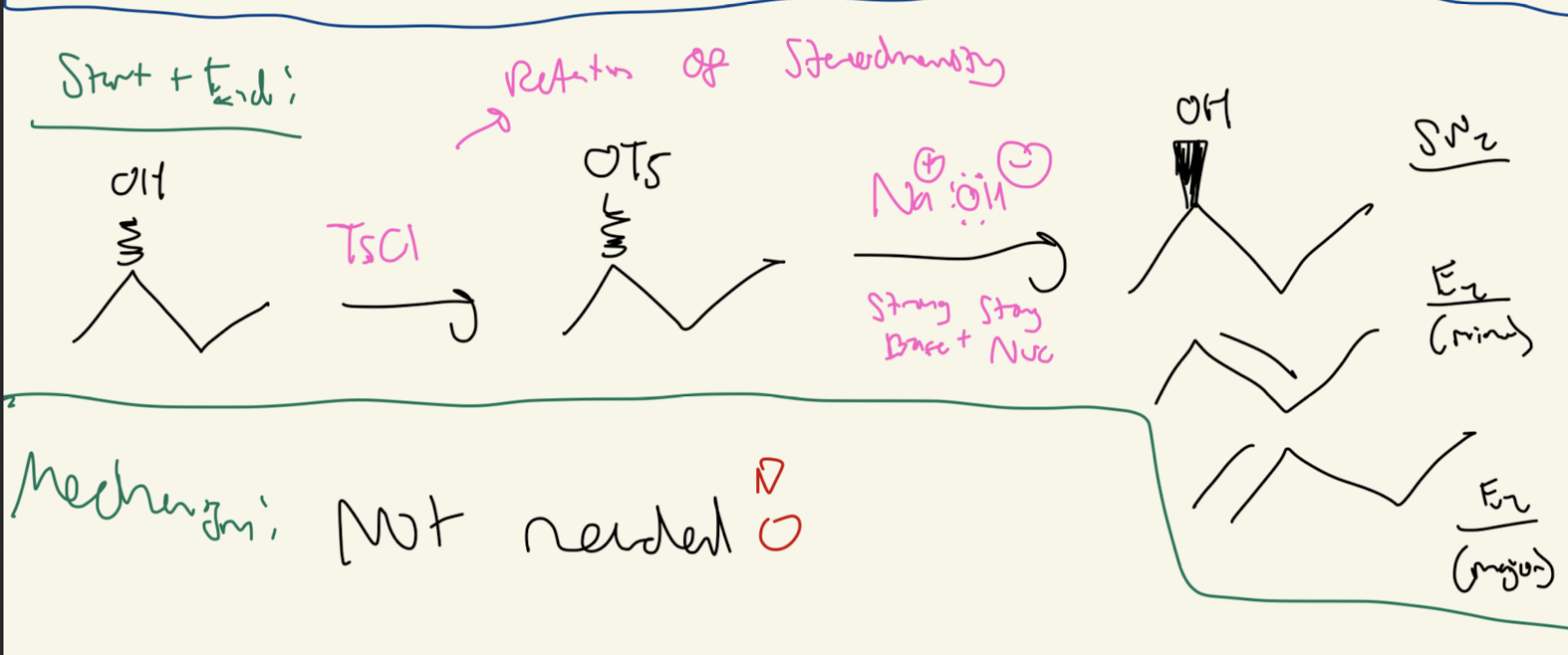
#7) TsCl pyridine
OH → OTS → back to OH using Na+OH- substitution
OTS keeps its retention of stereochemistry!!!!!
#8) Ether → Reagent: 2 HX equivalents
KEEP OH on the least substituted R and (X = Br, Cl, I) on most substituted R for 3° carbocation forms
If none, then KEEP OH on most substituted R and (X = Br, Cl, I) on least substitued R for methyl or 1° least hindered
Form carbocation 1st , if possible.
• If only 1° and/or methyl, then least hindered is attacked 1st
The first H-X attacks and protonates the MOST STABLE substituent which is 3°!
If there is no 3°, then protonate the LEAST susbtituent R group of the ether….EVEN IF you have a scenario with a 2° and a 1° R group,…. choose the 1° R group first to protoante using the H-X
NOTE: if there is a Phenoyl benzene ring with double bond resonance, KEEP IN THE ETHER!! H-X does not protonate sp2 hybridized carbons only sp3 hybridized carbons yoooo.
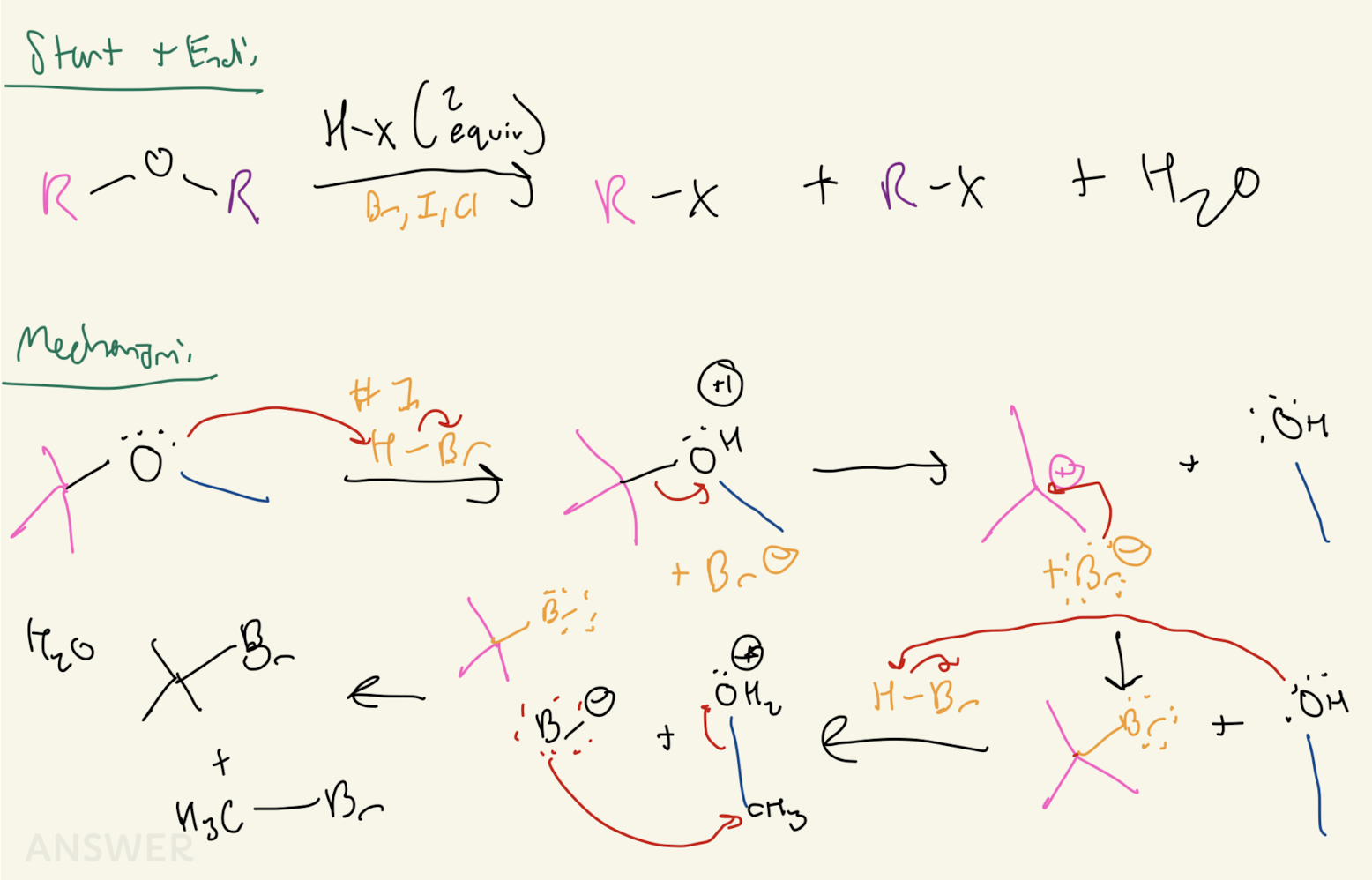
#9 Epoxide Opening:
Reagent: Strong Nuc: OH-, OR-, CN-, SR-
SN2 Mechanism
Nuc Attacks Less Substituted Carbon
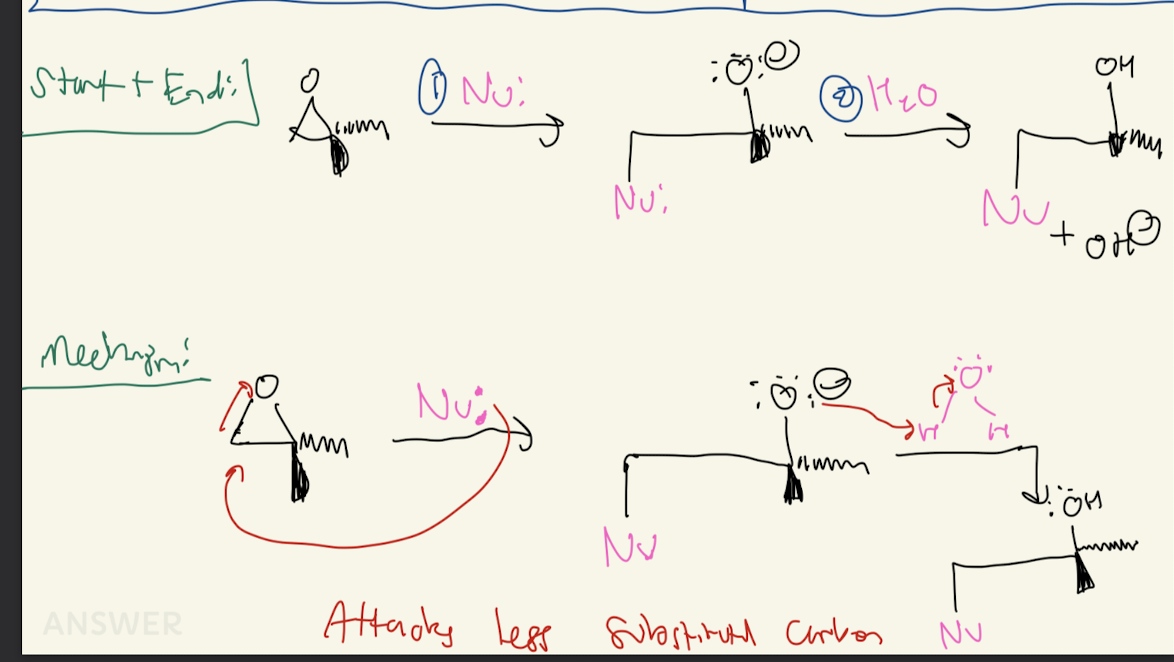
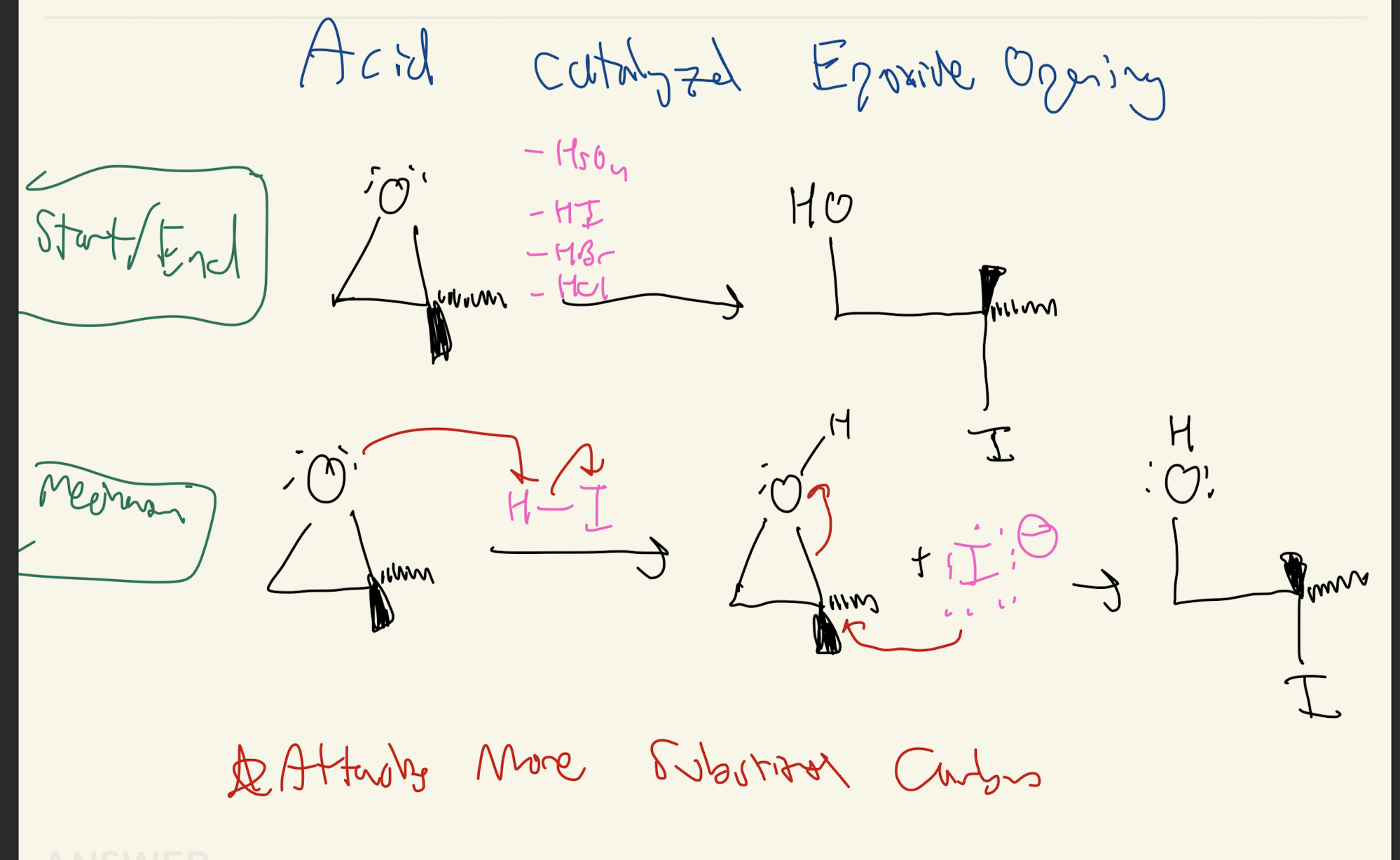
#10) Acid Catalyzed Opening
H2SO4, HI, HBr, HCl
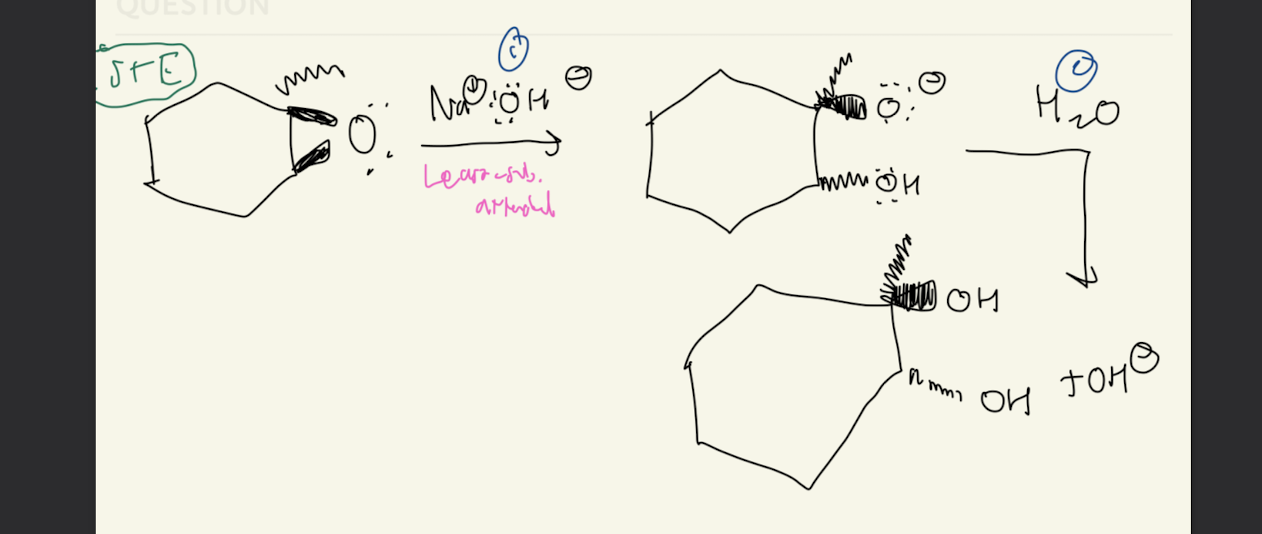
#10b) Cyclohexane Epoxide Opening
1) Na+, OH-, CN-, OR- SR-,
2) H2O
Chapter 10 Alkene Reagents
STARTING MATERIAL IS ALKENES! REMEMBER THAT! Some reagents are the same as OH reagents but the starting material is different in this case.
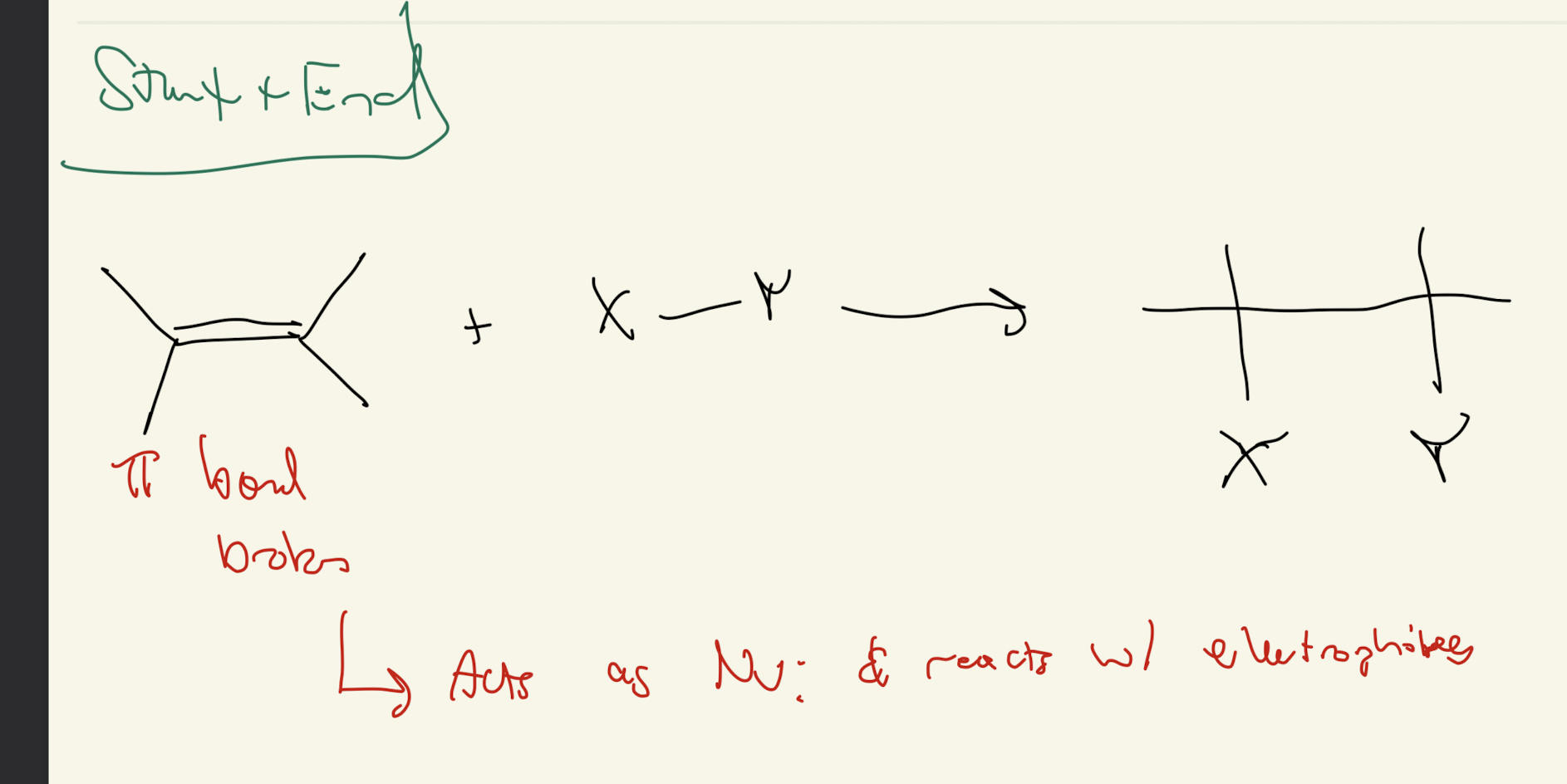
Overview
Dehydrohalogenation: E2 Reaction
Dehydration: E1 Reaction
#1) Reagent: Alkene + H-X
Hydrohalogenation
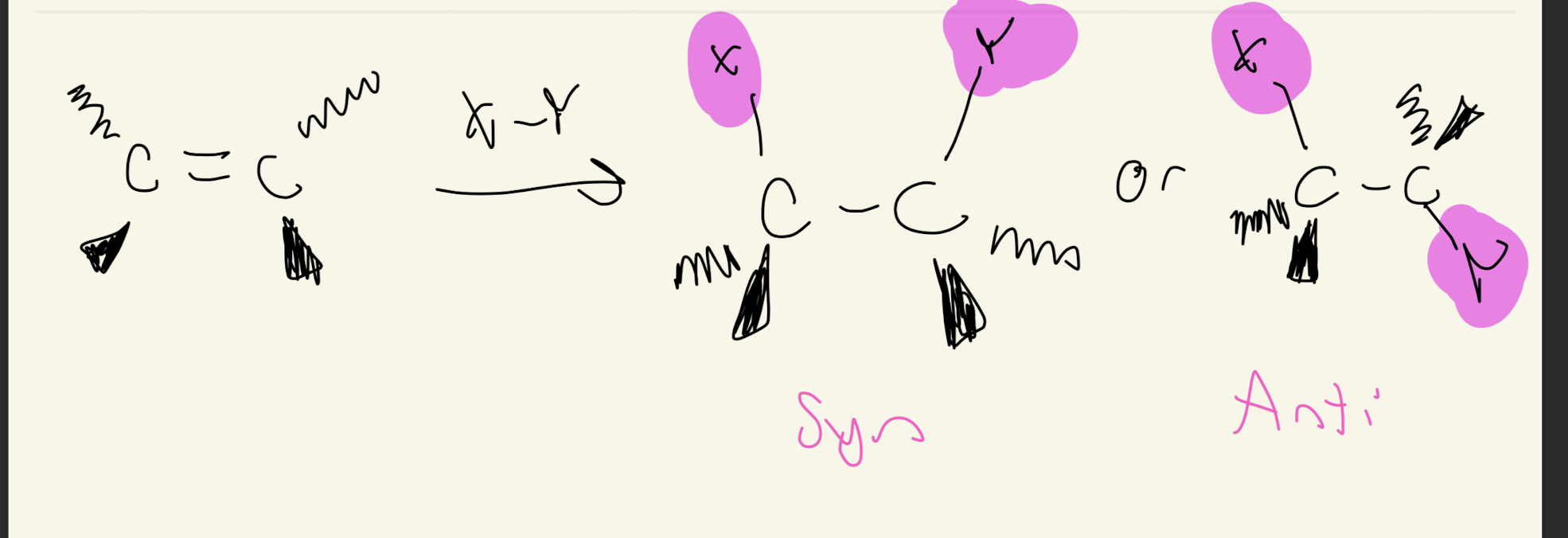
Markovnikov’s Rule
X (Cl, Br, I) rotates robot arm to more substituted carbon and H rotates robot arm to less substitutd carbon
#2) Alkene + H20 + H2SO4
Hydration

#3) Alkene + X-X
Halogenation
ANTI ADDITION
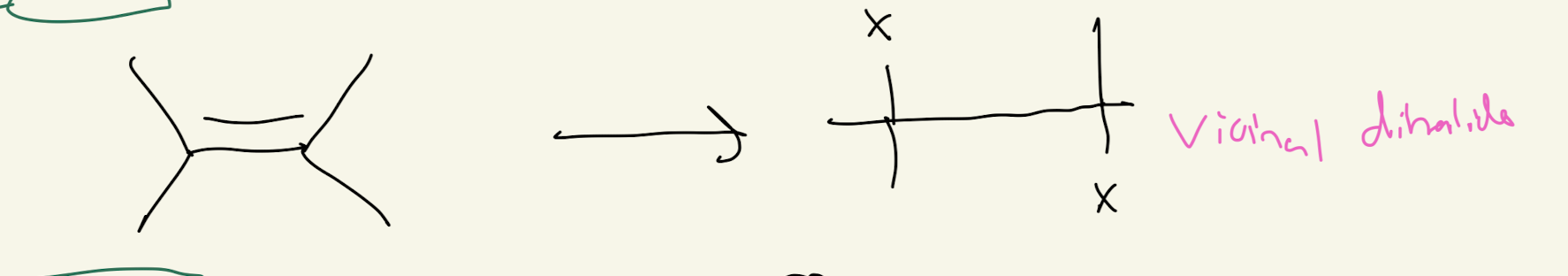
#4) X-X + H2O
HaloHydrin Formation
ANTI ADDITION
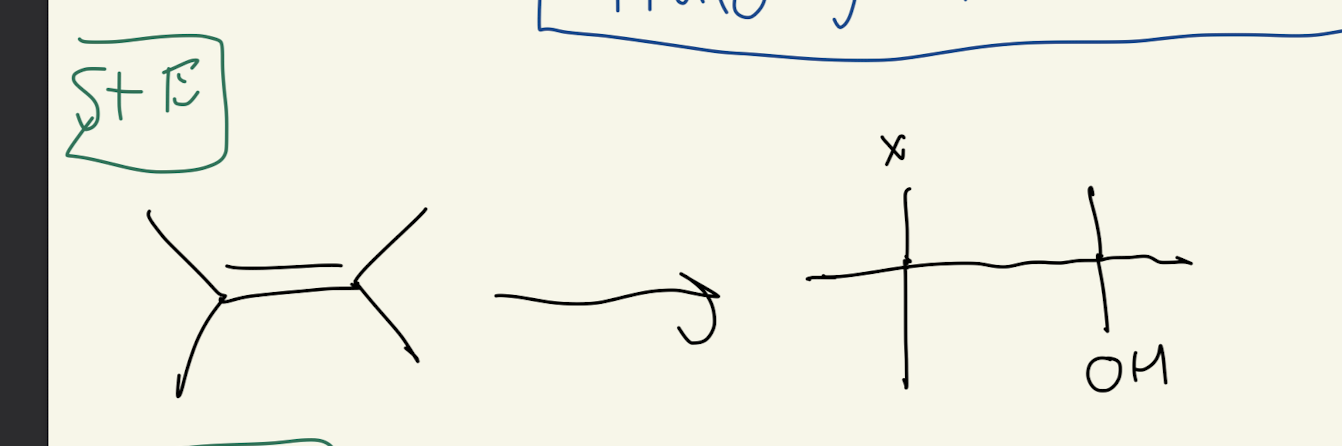
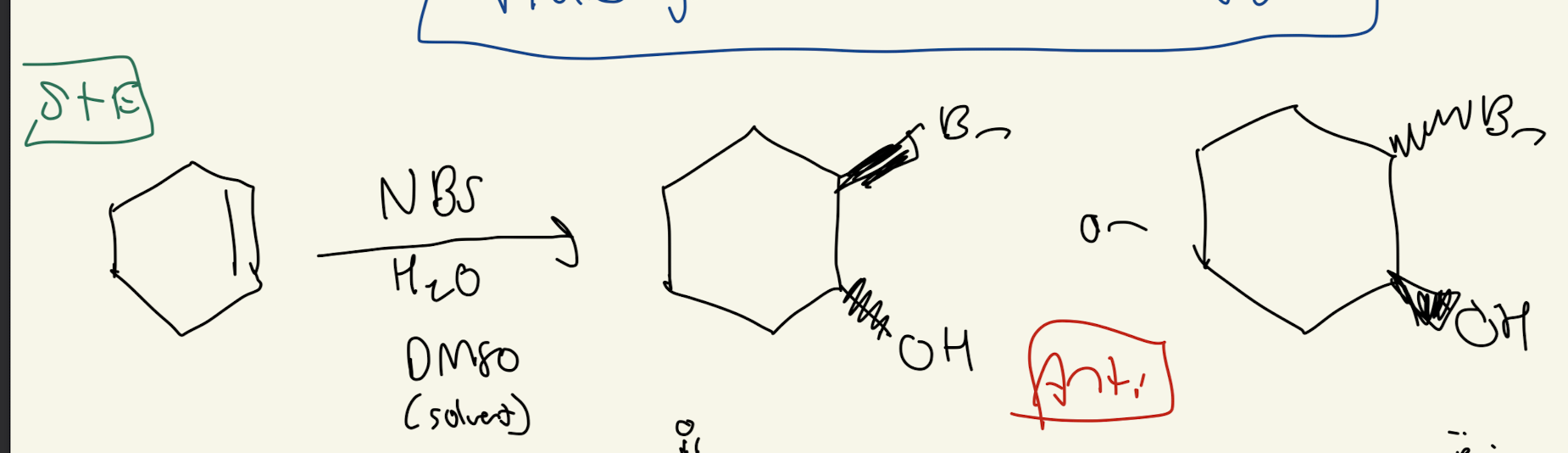
#4b) NBS (N-bromosuccinide)
ANTI addition
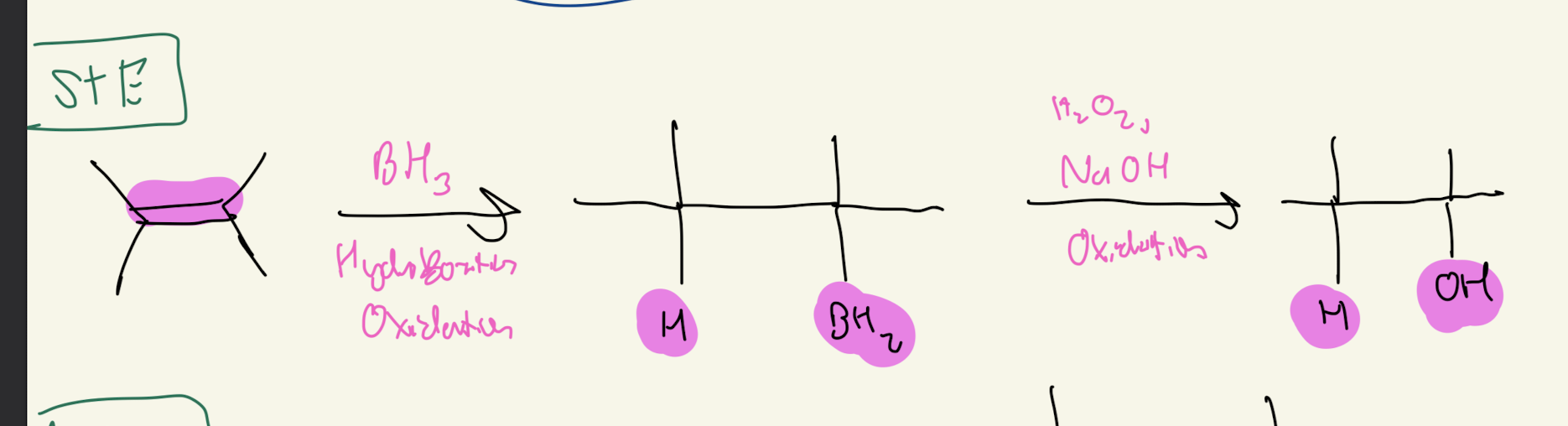
#5) 1: BH3. 2: H2O2 OH-
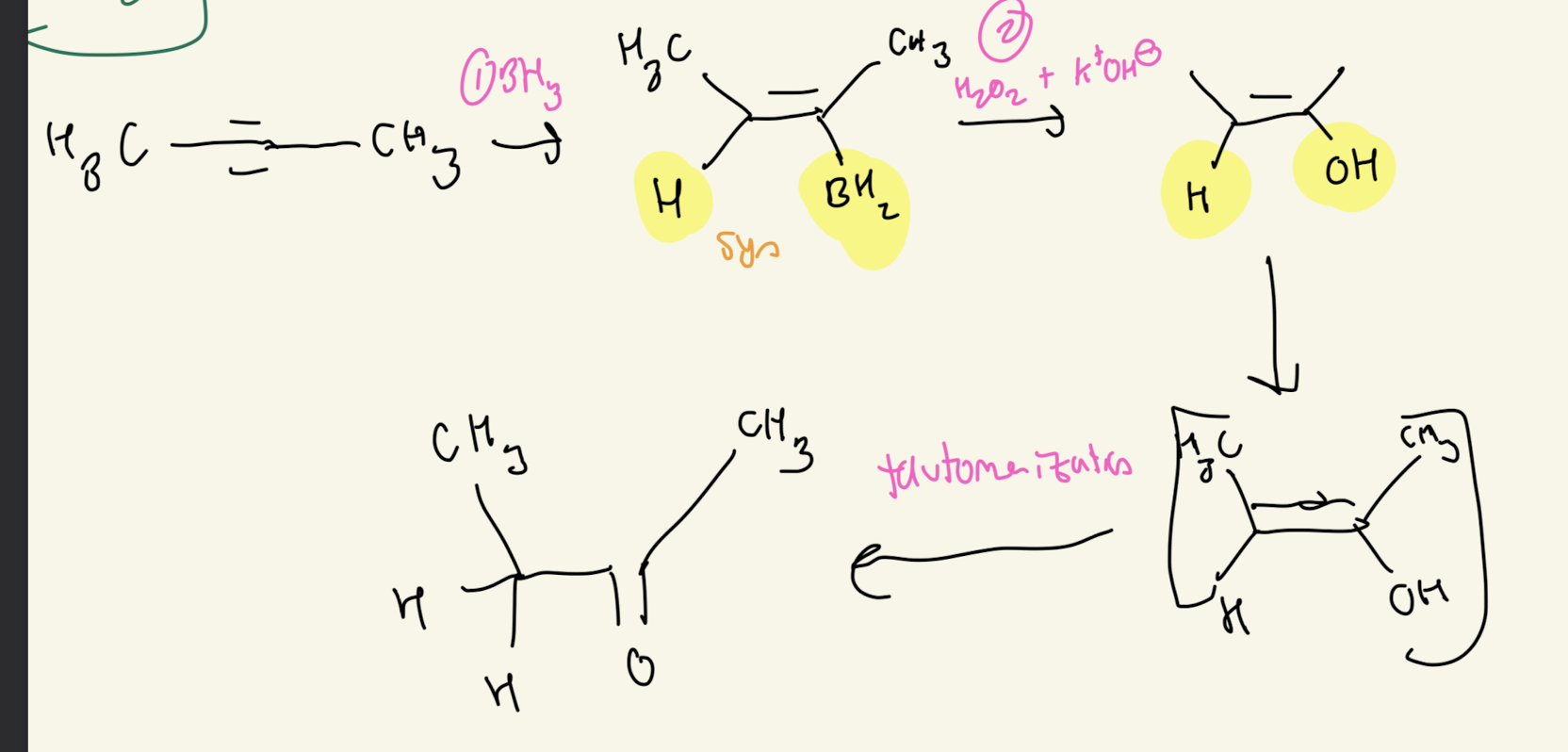
Hydroboration Oxidation
-DONT Need to Know Mechanism
-Transition State
-Syn Addition
-Retention of Configuration
ANTI Markovnikov
-H goes on MOST SUBSTITUED CARBON
Chapter 11 Alkynes
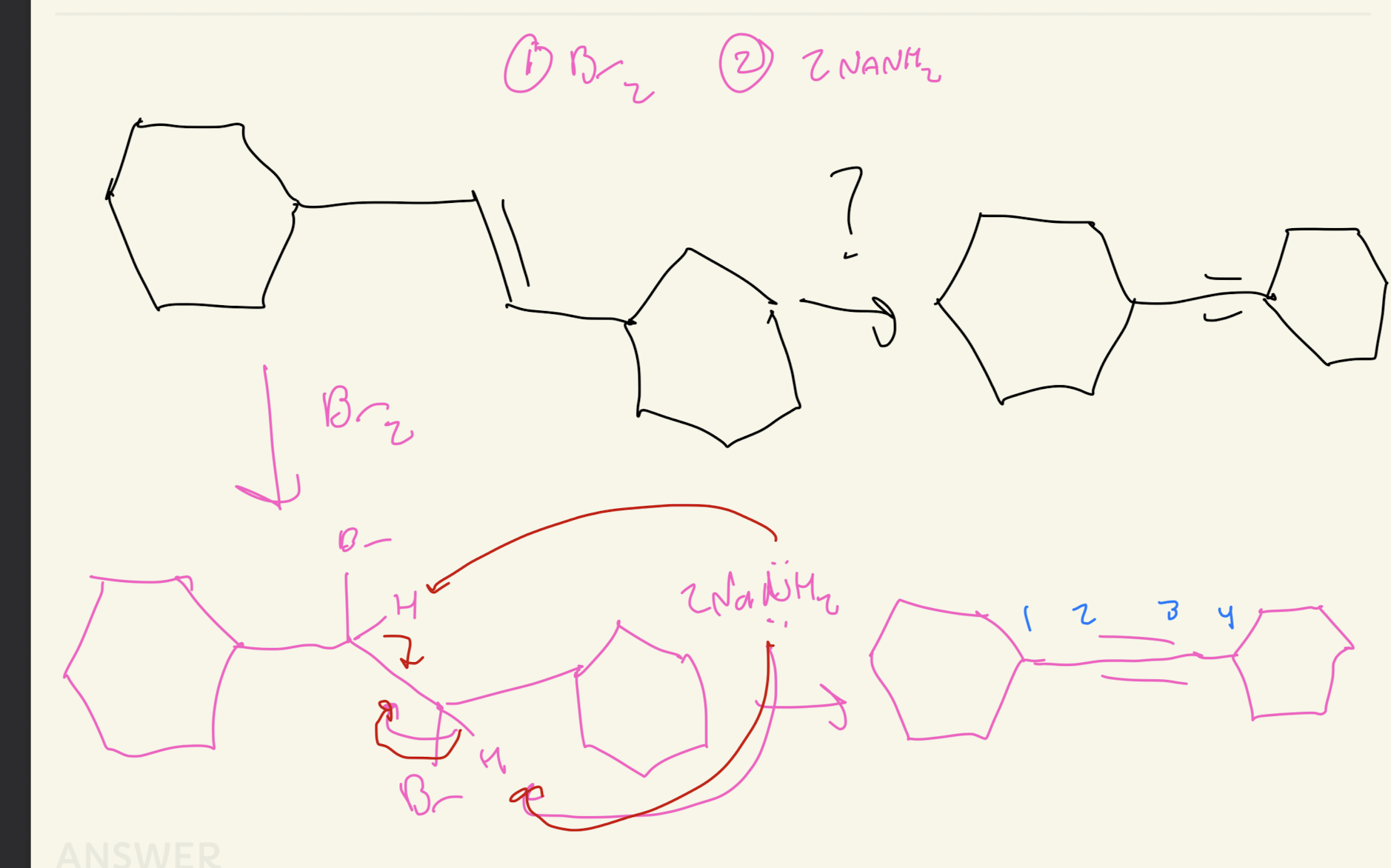
Alyne Preparation and Formation
2NaNH2
Alkene → Br2 → 2NaNH2 → Alkyne
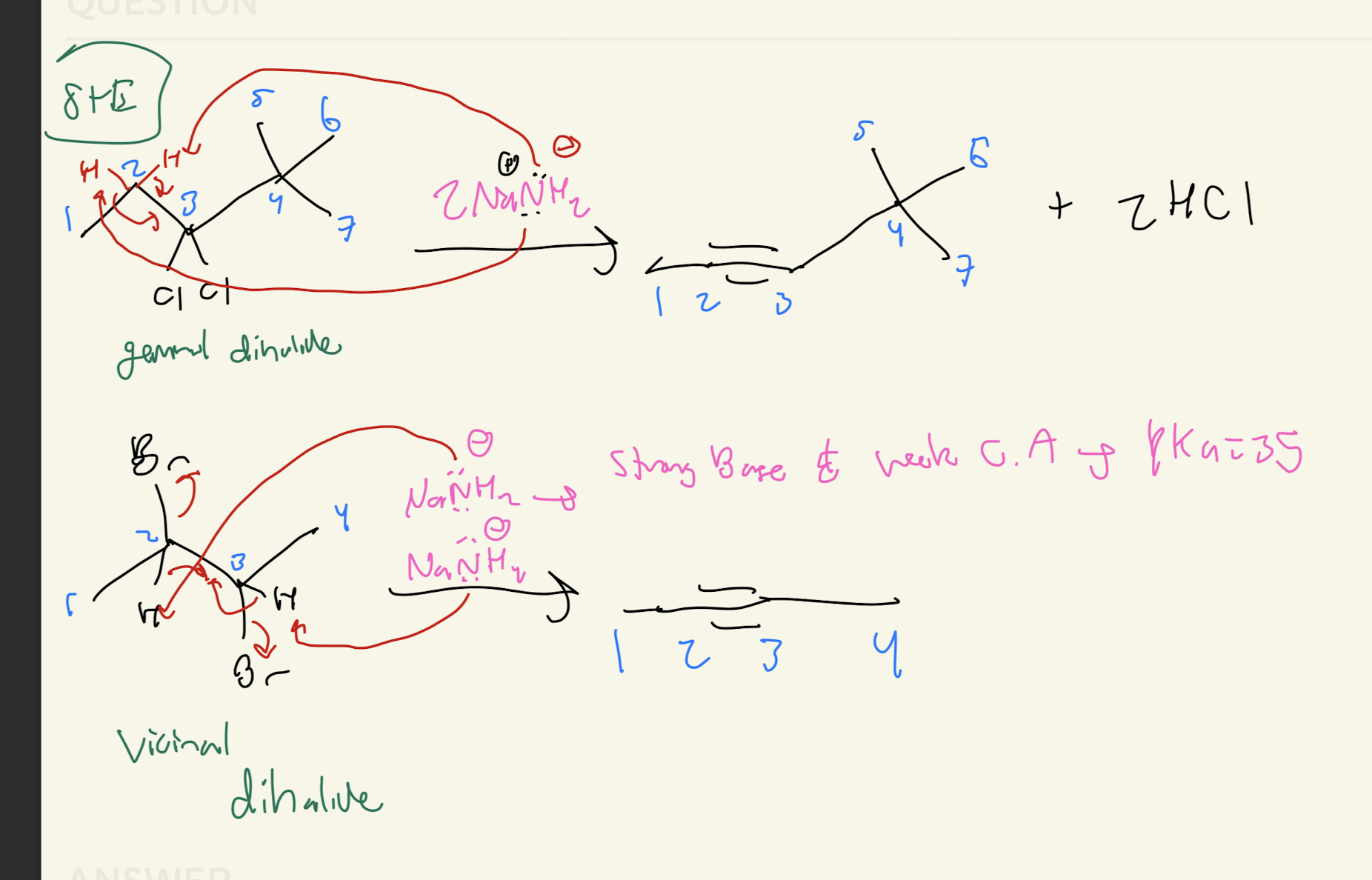
#1) Alkyne + HBr (2 times)
Hydrohalogenation
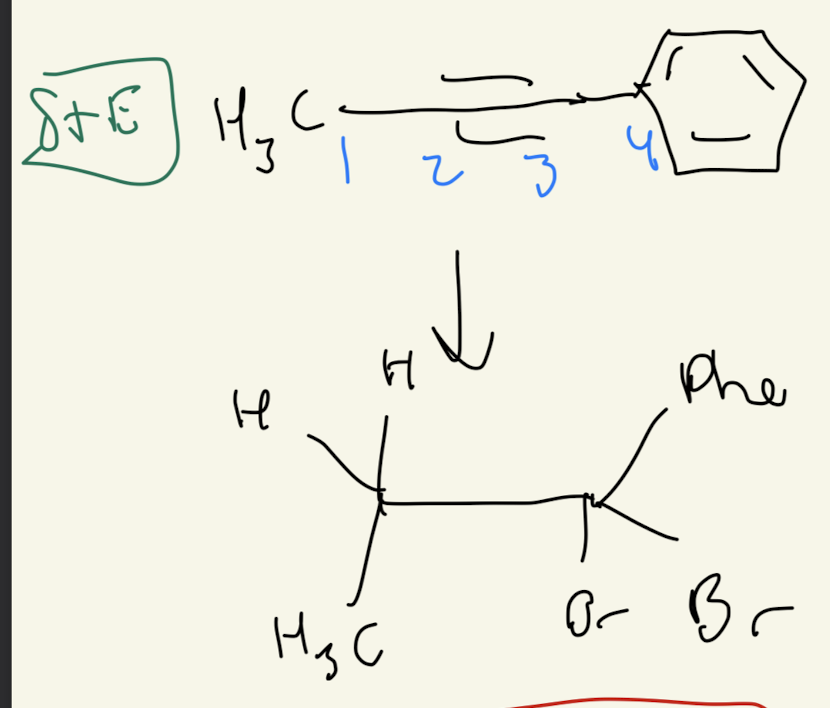
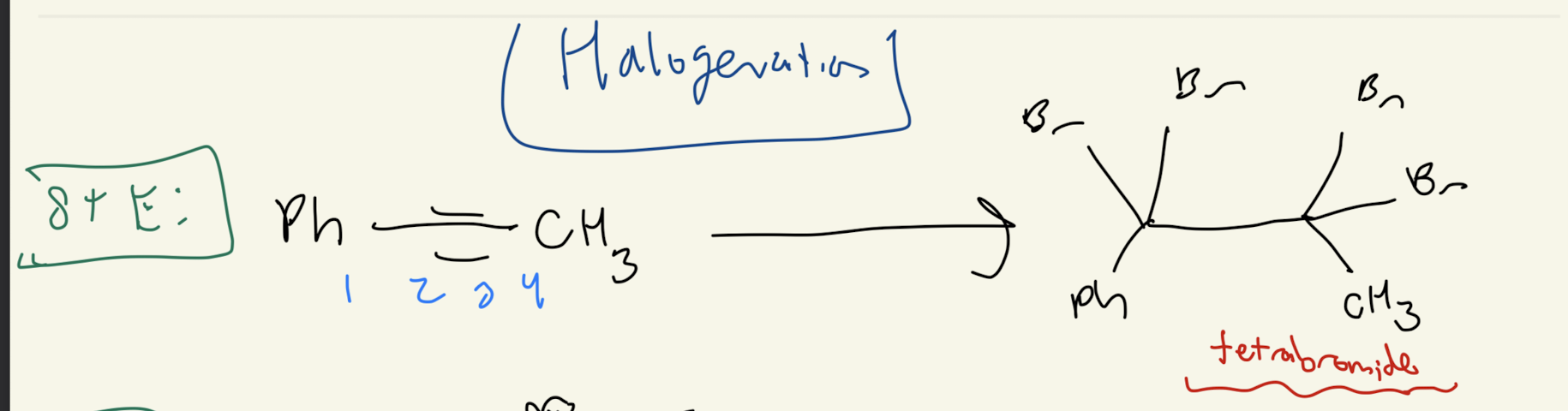
#2) Alkyne + X-X (2 times)
#3) Hydration (H20H2SO4)
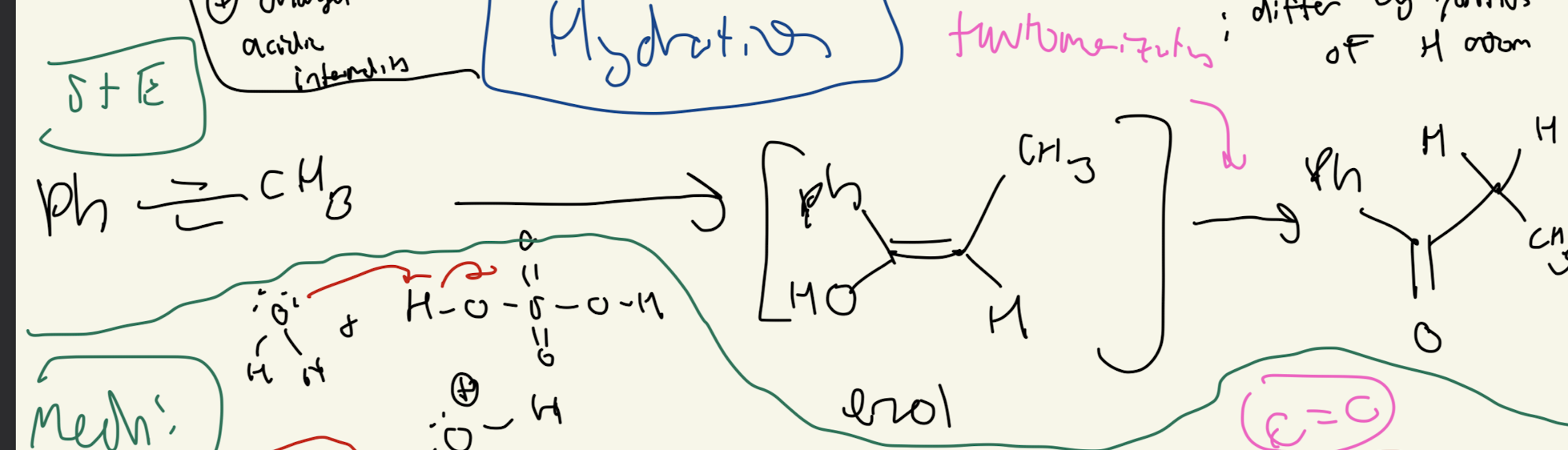
#4) Hydroboration Oxidation (BH3 and H2O2 and OH-)
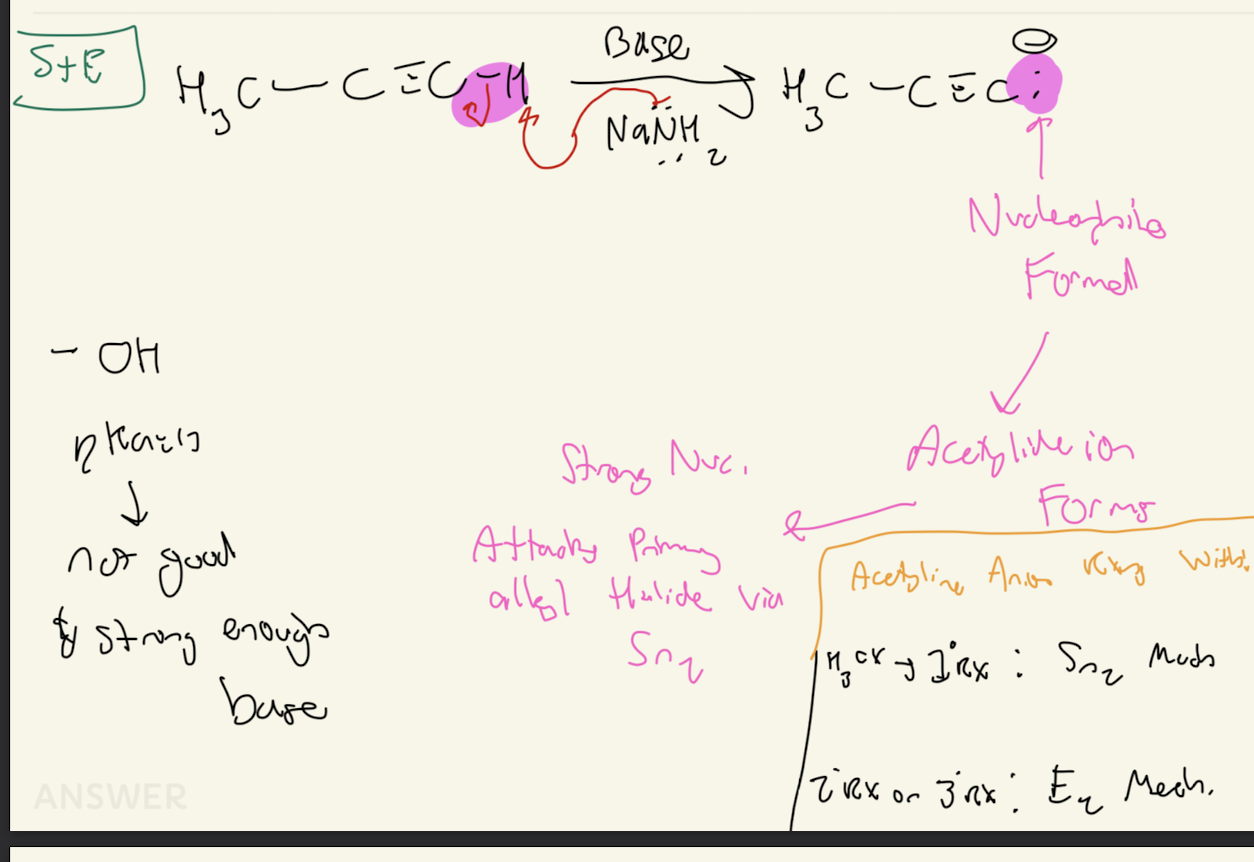
#5) Terminal Alkyne Reactions
-NaNH2- (strong base)
-NaH- (strong bases)
Forms Acetylide Ion
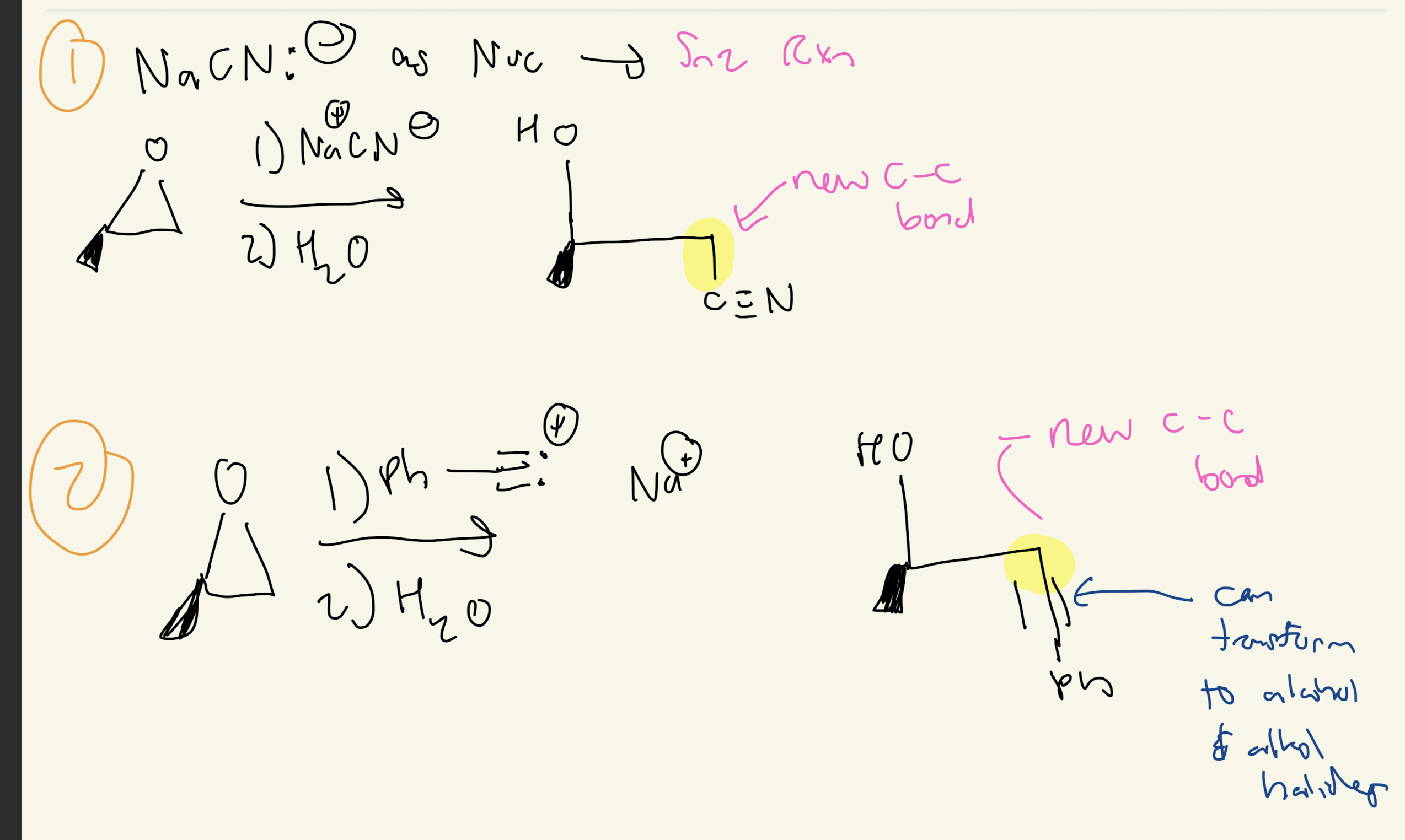
#6) 2 Ways to Make C-C bond
1) Na+ Cn-, H2O
2) New Acetylide Anion
#12 Oxidation and Reduction
OIL RIG
Oxidation is Losing electrons/C-H bonds
Reduction is Gaining electrons/C-H bonds
Reduction
High → Low oxidation state
Decrease # of C double bond C, C-O bonds, C-N, and C-X bonds (X = Cl, Br, I)
Increase # C-H bonds
Oxidation
Low → High Oxidation State
Decrease # C-H bonds
Incease # of C double bond C, C-O bonds, C-N, and C-X bonds (X = Cl, Br, I)
Oxidation State
THE AMOUNT OF OXIDATION OR A LOT OF C double bond C, C-O bonds, C-N, and C-X bonds (X = Cl, Br, I)
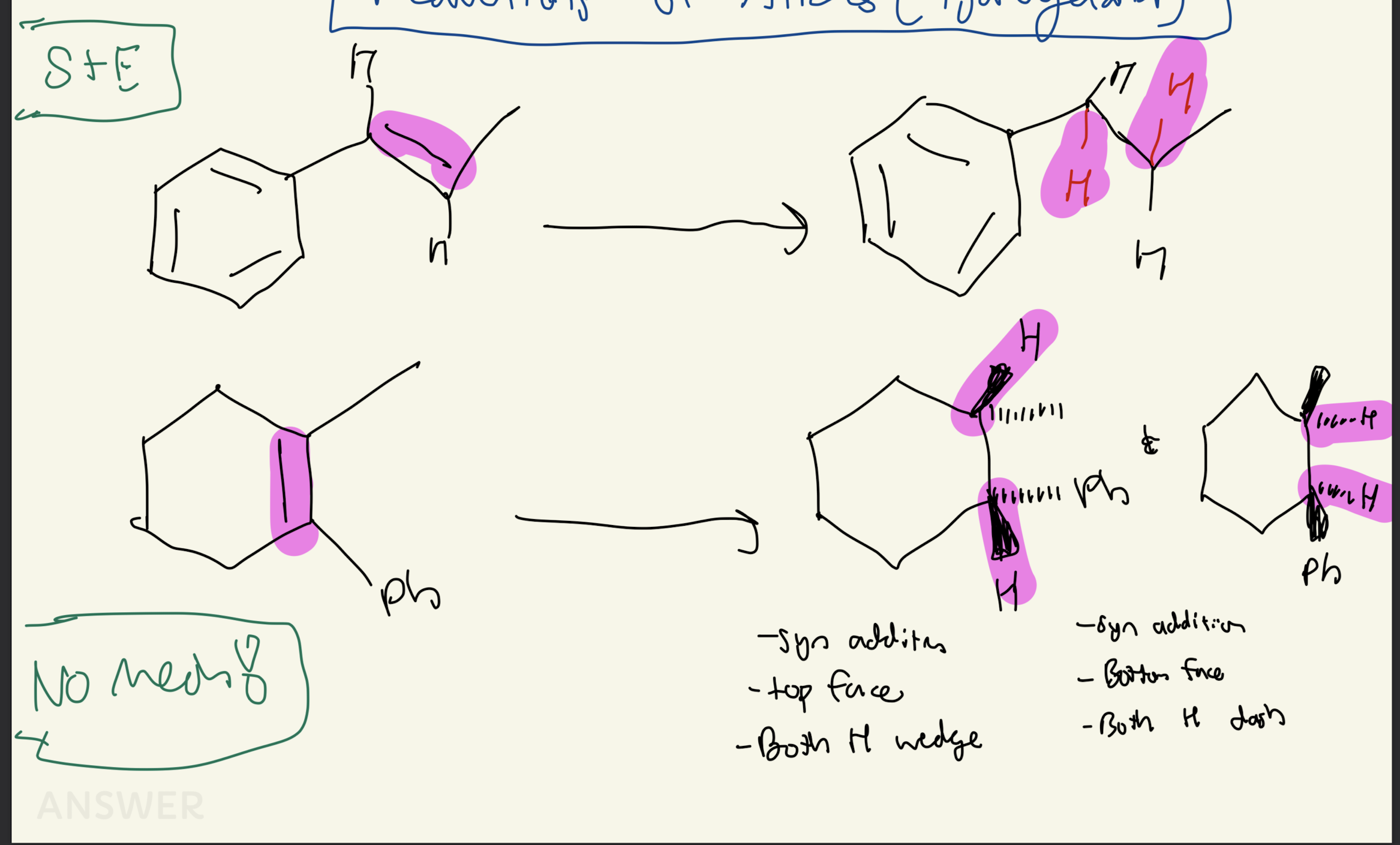
1) Reagent: Pd/C, H2
Reduction of Alkenes
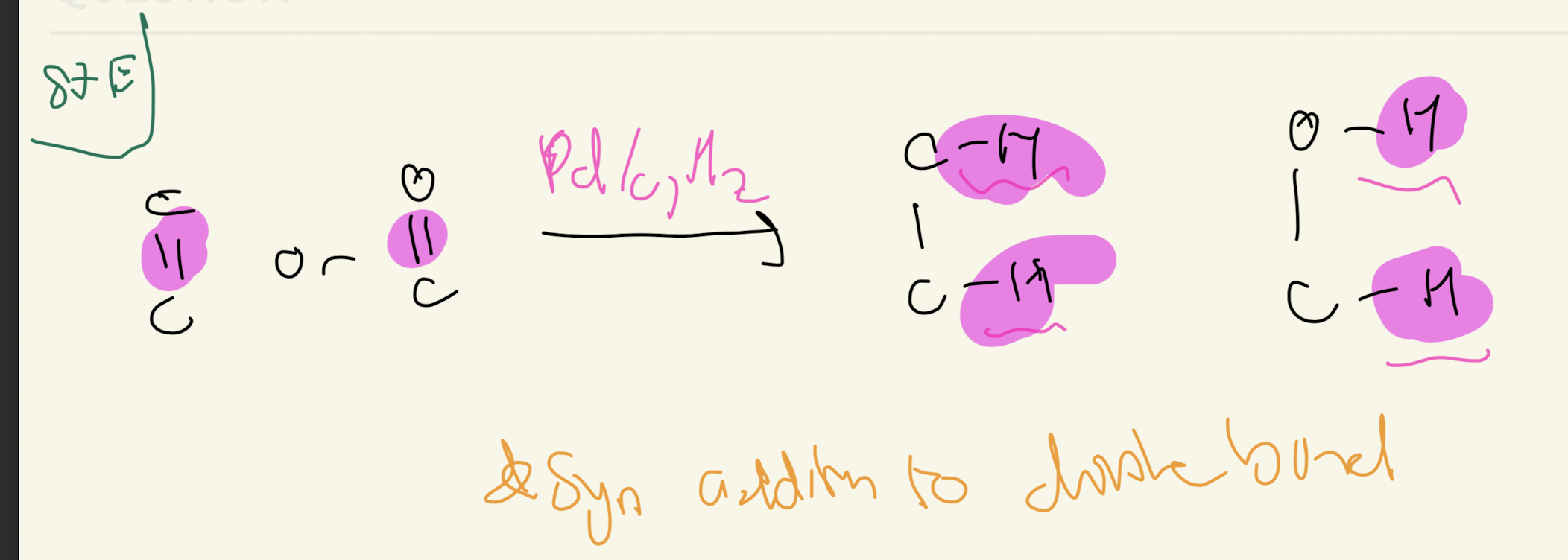
1b) Reagent: Pd/C, H2
Reduction of Carbonyls
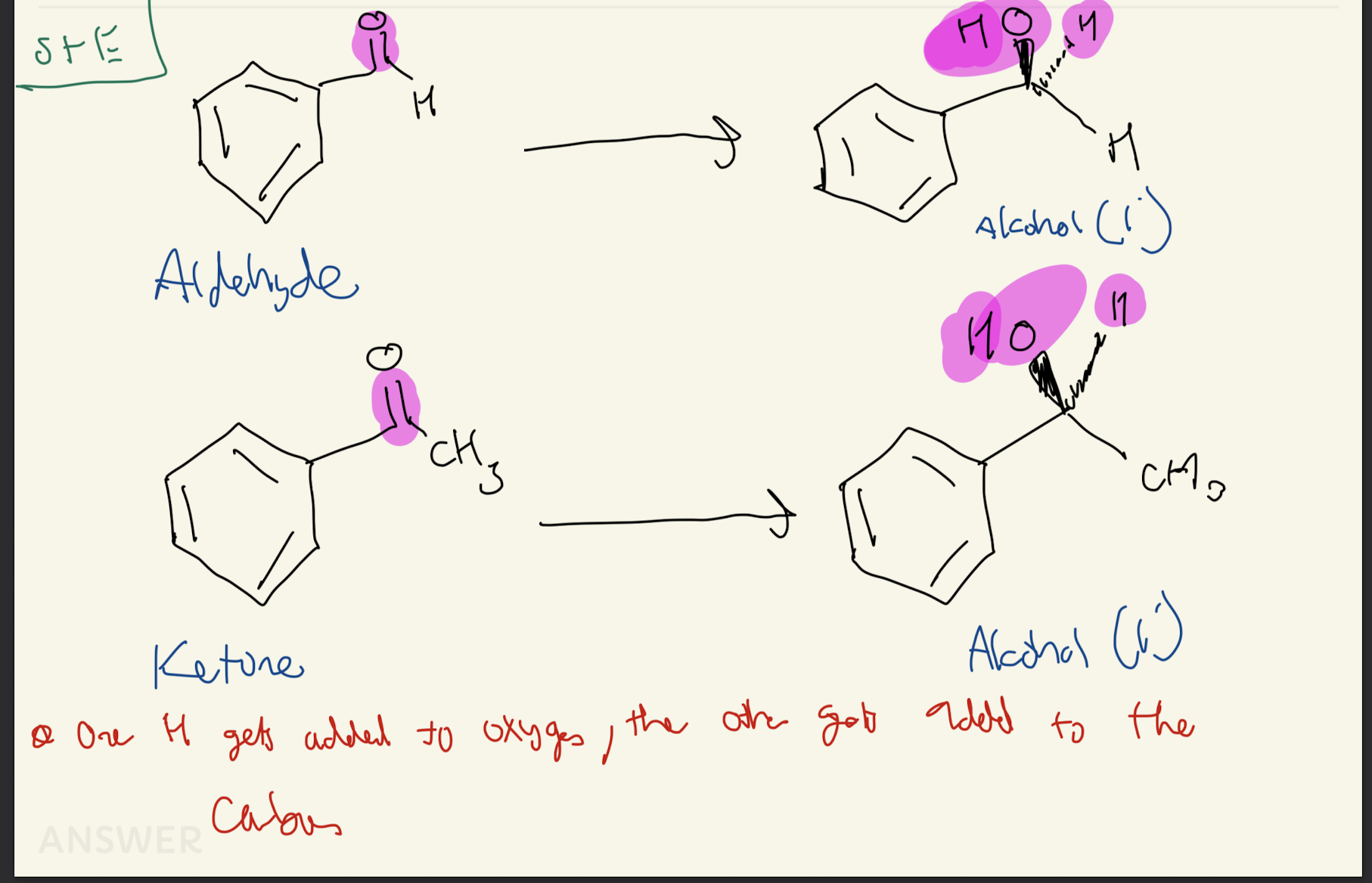
1c) Reagent: Pd/C, H2
Reduction of Aldehydes and Ketones form the C double bond O. The H attaches to the Oxygen first and then to the nearby carbon.
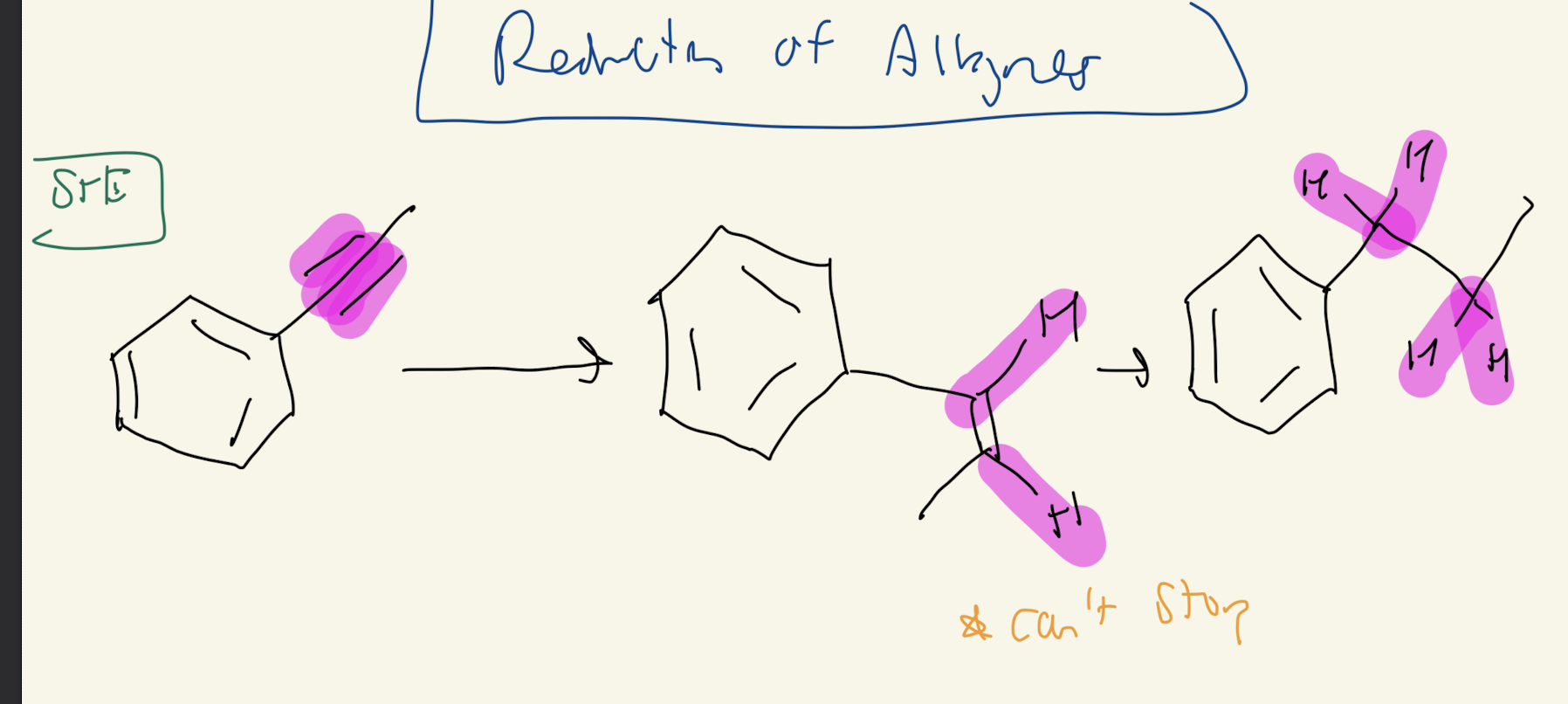
1d) Reagent Pd/C, H2
Reduction of alkynes
2) Lindlar’s Catalyst, H2
Reduction of Alkynes
Cis Alkene

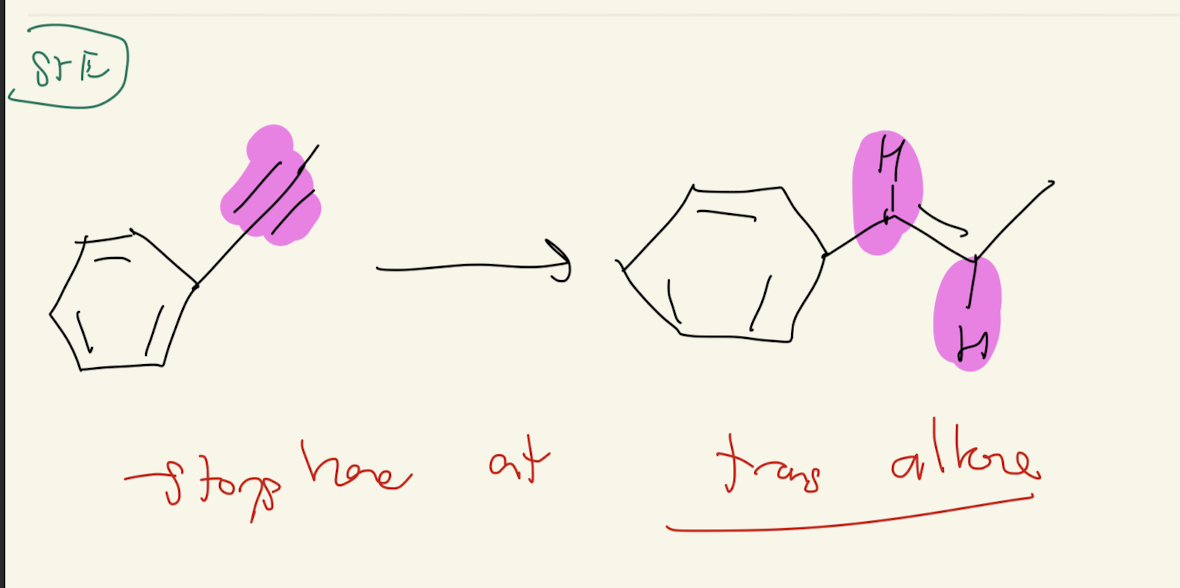
3) Na°, NH3
Reduction of Alkynes
Trans Alkene
4) LiAlH4
Reduction of Polar C-X Bonds (Alkyl Halides)
SN2 Mechanism (INVERSION of STEREOCHEMISRY) using H as nucleophile

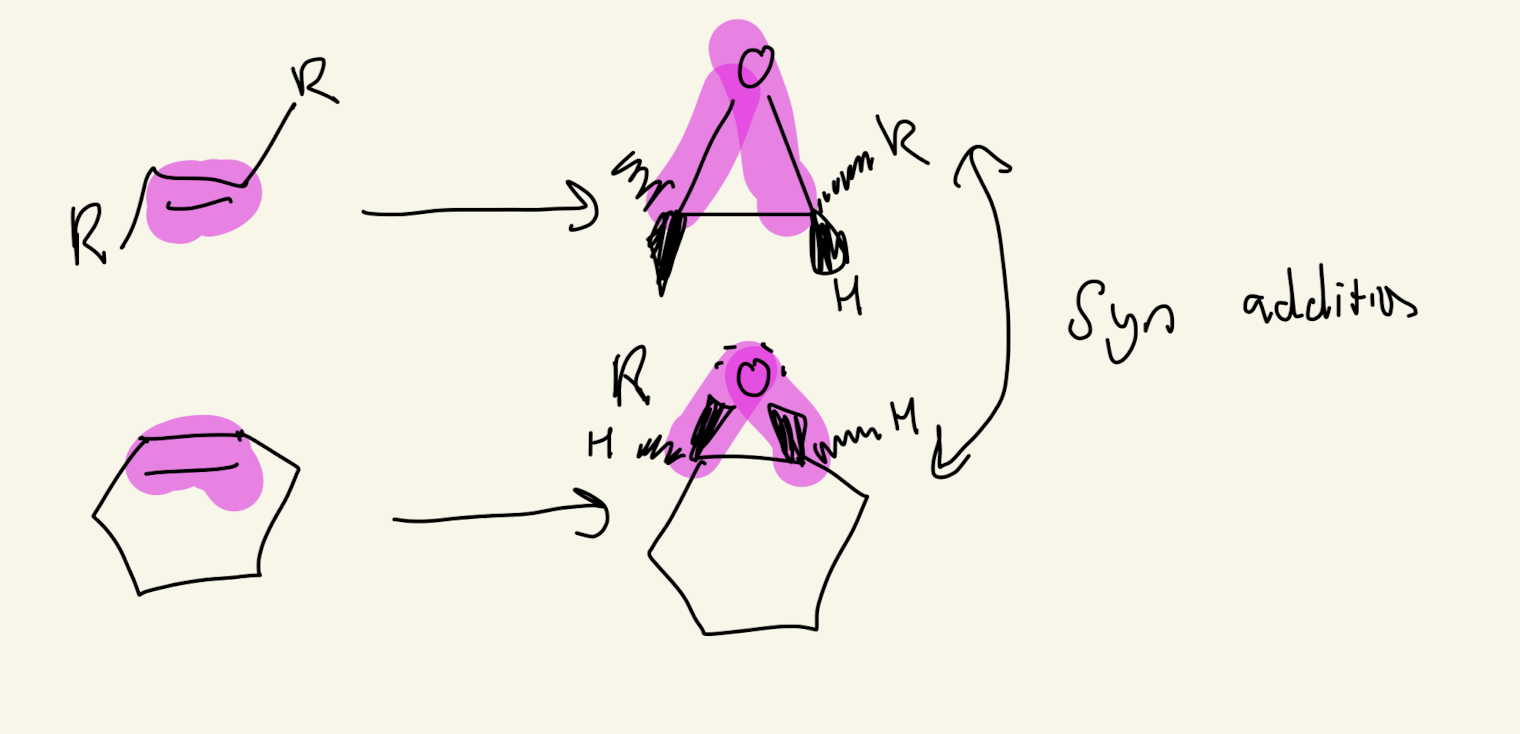
5) mPCBA
SYN ADDITION
Cis Alkene: Meso Cmpound and Identical (Superimposable mirror Images)
Trans Alkene: Enantiomers (50%/50%) (Non superimposable Mirror Images)
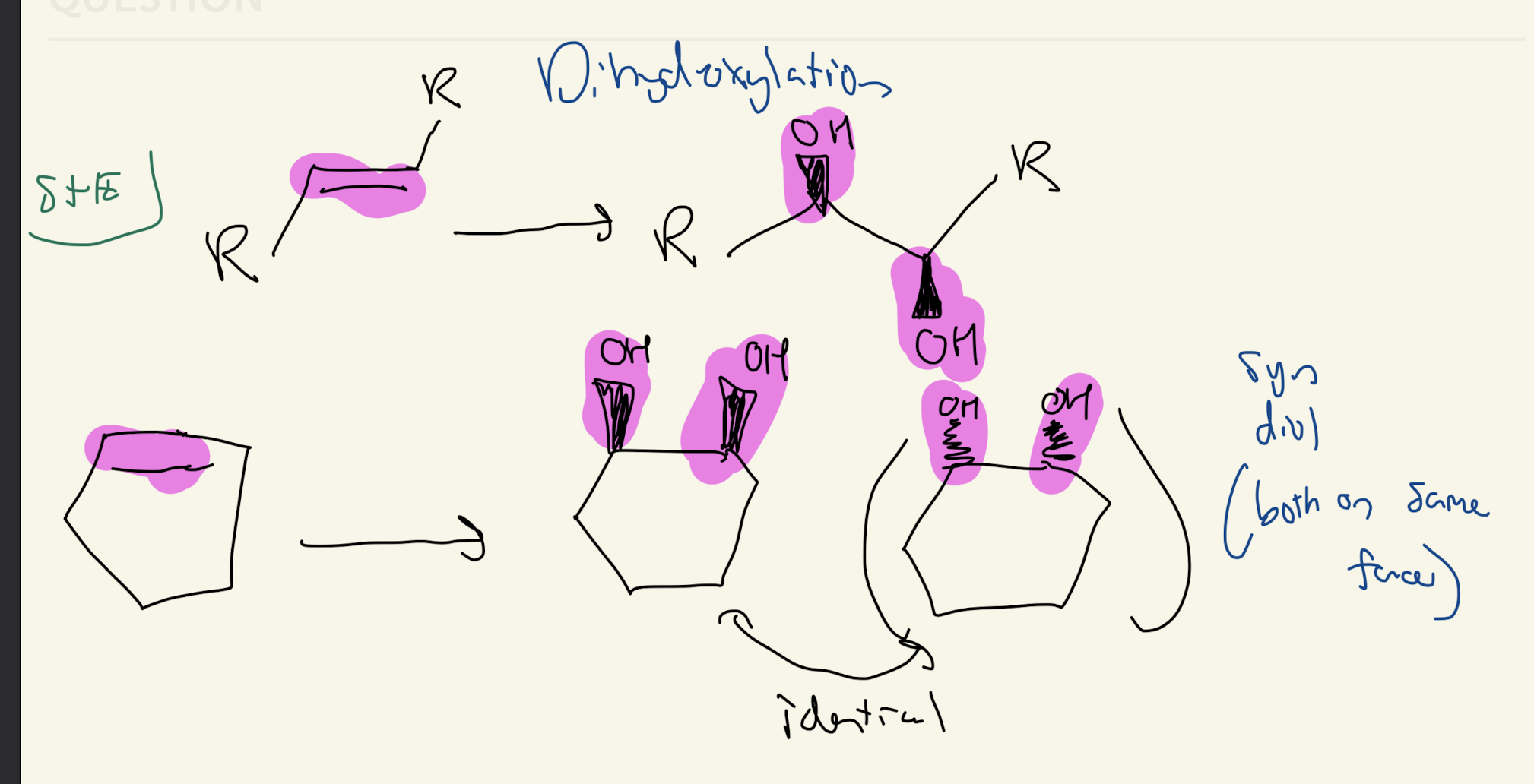
6)
1) KMnO4
2) H2O, KOH.
OR
1) OSO4.
2) NaHSO3, H2O
Dihydroxylation
SKIP MECHANISM FOR BOTH
Syn Addition for Both Cis and Trans Alkenes
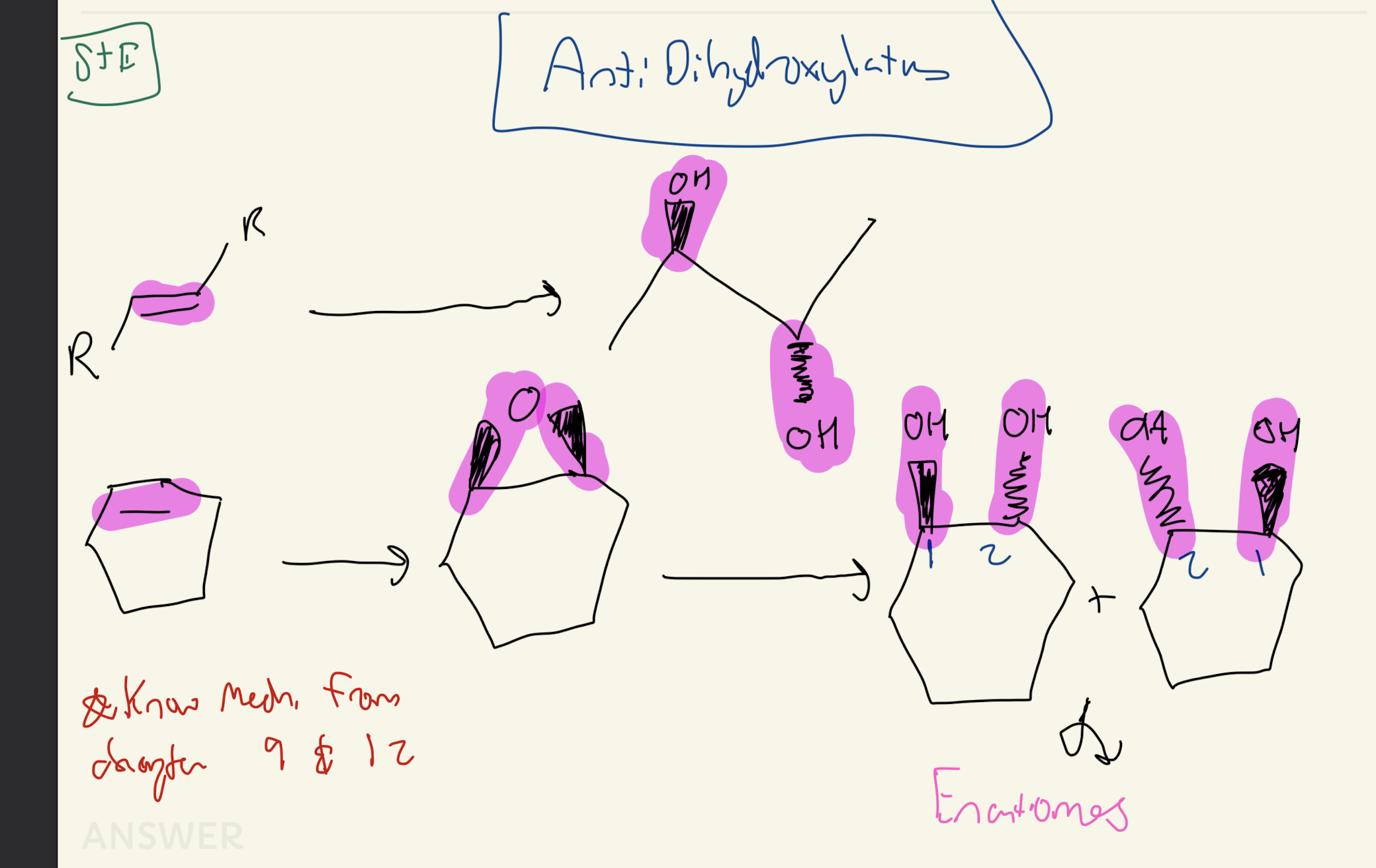
#7 1) MCPBA. 2) H2SO4, H2O
AntiDehydroxylation
Know these Mechanism from Chapter 9 and Earlier from Chapter 12
#8.
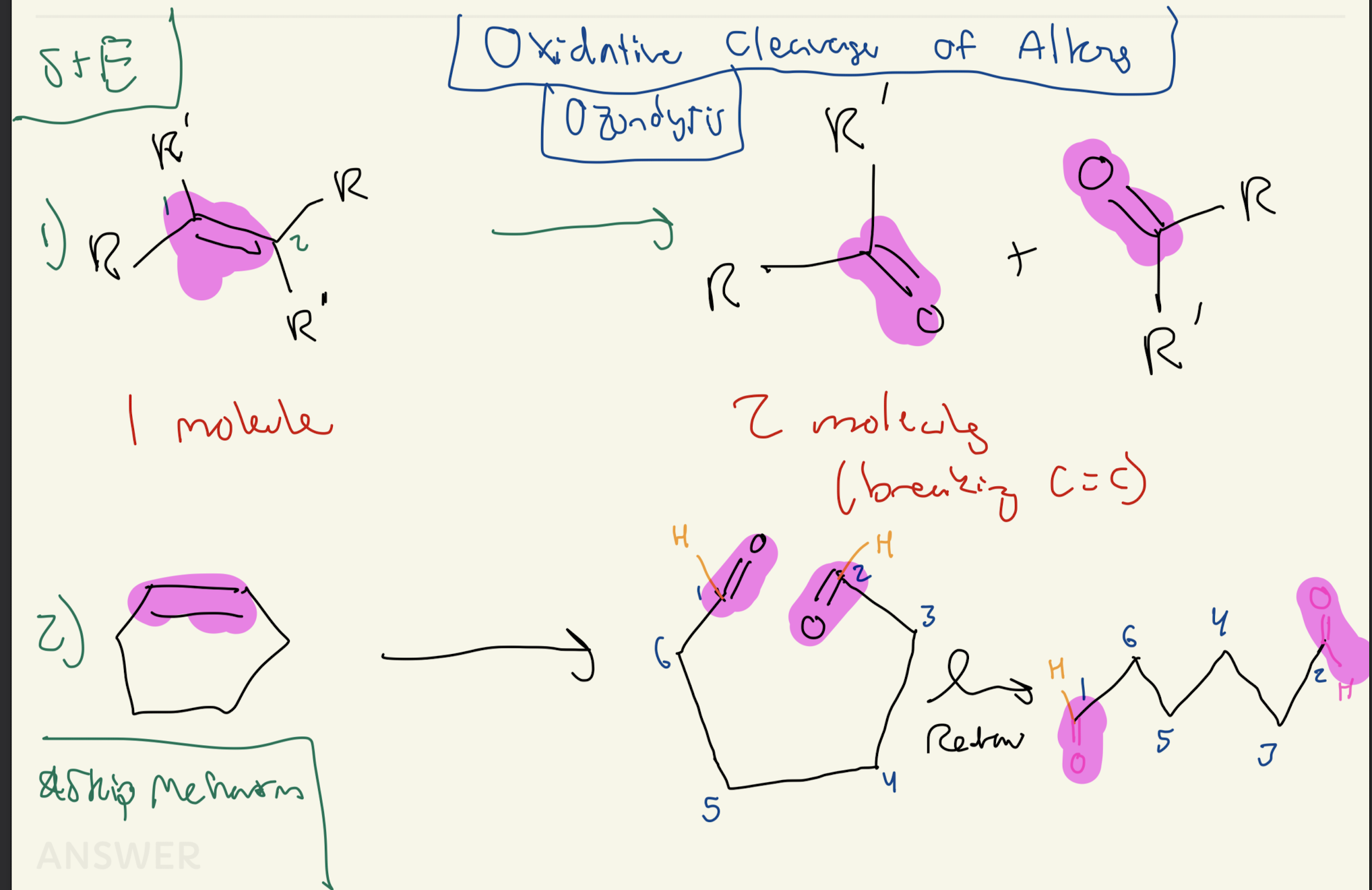
1) O3 (Ozone)
2) H3C - S - CH3. (Dimethyl Sulfide)
OZONOLYSIS
Oxidative Cleavage of ALKENES
Skip Mechanism
1 to 2 molecules
If Cyclohexane → Unwind it
#8b)
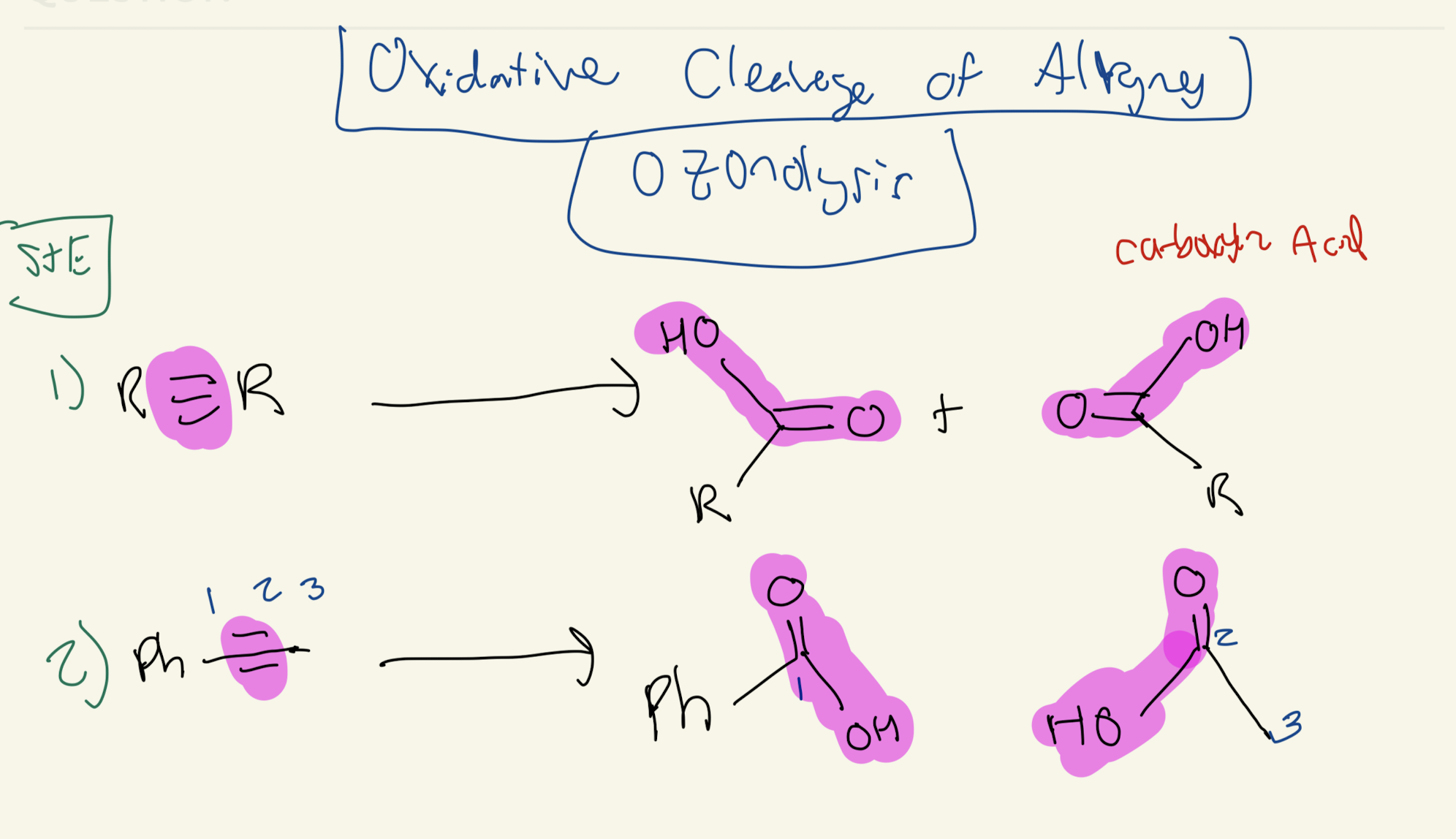
1) O3 (Ozone)
2) H3C - S - CH3. (Dimethyl Sulfide)
OZONOLYSIS
Oxidative Cleavage of ALKYNES
#9:
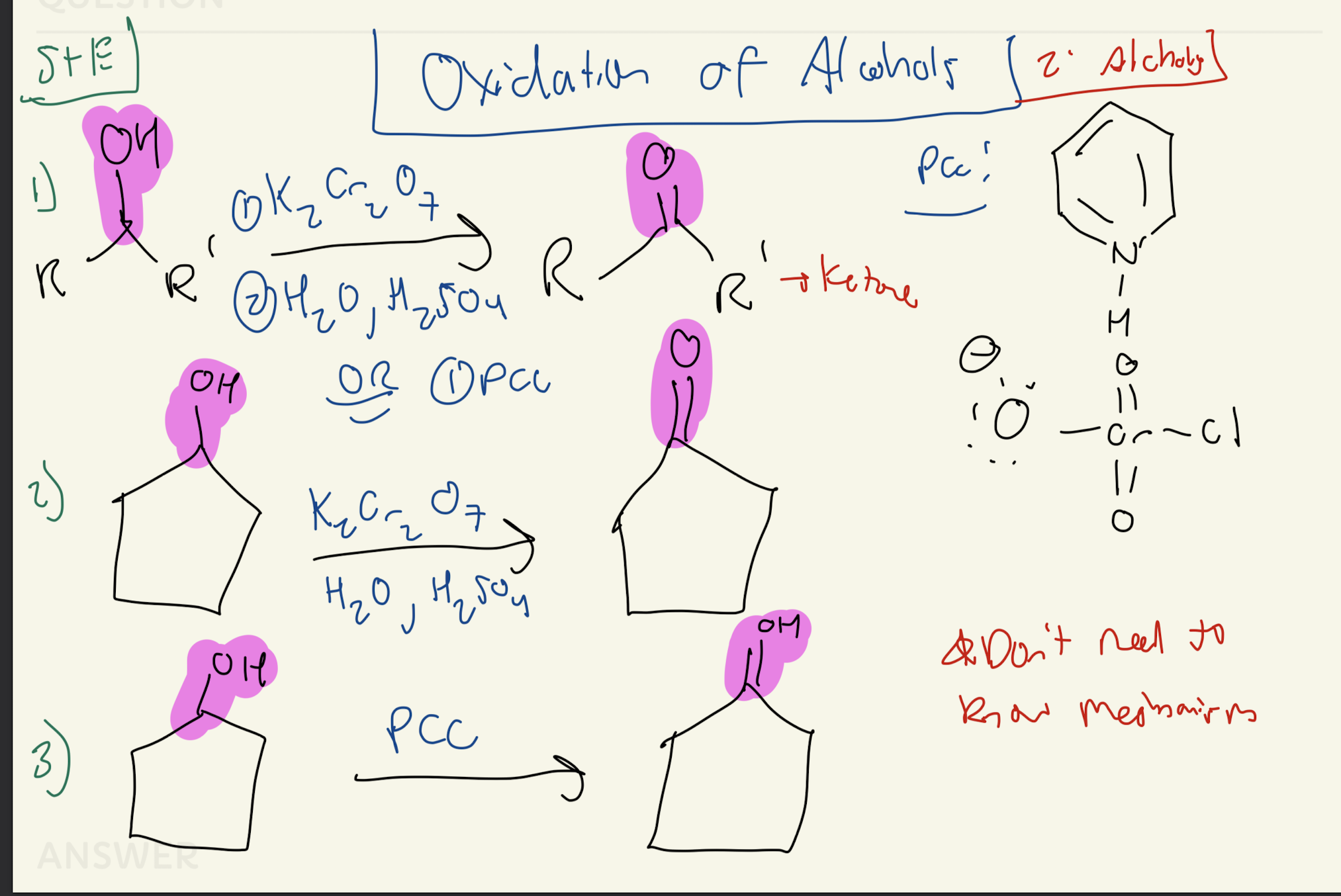
1) K2Cr2O7
2) H20, H2SO4
OR
1) PCC
2° Alcohols
#9b)
1) K2Cr2O7
2)H20, H2SO4
OR
PCC
1° Alcohols
Chapter 13 Radicals
-single unpaired electron
-homolysis
-parallels carbocation stability
-more attached carbons = more stable
-3° > 2° > 1°
-NO REARRANGEMENTS like carbocations
General Features of Radical Reactions
Halogenation of Alkanes
Hydrohalogenation of Alkenes
#1) hv or ∆
Initiation
Propogation
Chlorination vs. Bromination
Chlorination:
-smaller
-less polarizable
-less stable
-Reacts quickly WITHOUT selecting just right bond
-UNSELECTIVE mixture of products
-FASTER
Bromination:
-larger
-more polarizable
-more stable
-takes its time to make JUST the RIGHT PRODUCT
-SELECTIVE, one major product
-Slower
-More Substituted C-H bond is weakest and easiest to break
BR2:
1:99
1 = Anti-Markovnikov
99 = Markovnikov
Cl2:
1 = Anti-Markovnikov
1 = Markovnikov
#2: H-Br hv or ∆ or ROOH
Radical Addition Reactions to Alkenes
Initiation:
Propogation:
Termination:
Chapter 14: Conjugation
Overlapping p orbitals on at least 3 adjacent atoms
Electrons in pi bond spread over a great area = stabilizatioin!
Allylic carbocation more stable than non allylic carbocation
Carbocation Stability:
1°< 2 = allylic carbocatioin < 3°
Benzylic carbocation more stable than non-allylic carbocation.
Delocalization = Stability
Common Examples of Resonance
1) Allyl System
-lone pair one sigma bond away from a double bond
2) Conjugated Double Bond
3) Cations adjacent to Lone pair
4) Double Bond with Adjacent Atoms differing in electronegativity
The Resonance Hybrid: Most closely resembles best hybrid
#1: Resonance structure with more bonds and fewer charges is better
#2: Resonance structure with every atom having an octet is better
#3: Resonance structure with negative charge on the more electronegative
atom is better.
Honorable Mention: Electrophilic Addition: 1,2- vs 1,4-Addition. When there is conjugation of 2 double bonds adjacent to each other in trans position
H-Br
-1,2 Addition: Major Product is Markovnikov
-1,4 Addition: Resonance Minor Product is Anti Markovnikov
#1: ∆
The Diels Alder Reaction
DIENE + DIENOPHILE
-exothermic
-2 sigma bonds formed and 1 pi bond formed from 3 pi bonds
1. Rotate diene to s-cis conformation.
-Diene must be in s-cis conformation:
-Dienes constrained to s-cis conformation are most reactive.
2. Draw dienophile adjacent to s-cis diene.
-Dienophile reactivity:
-Electron-withdrawing substituents on dienophile increase the reaction rate.
-More oxygen groups C double bond O
3. Break 3 π bonds. Show where new bonds form with arrow.
-Stereochemistry of dienophile is retained in the product:
-CIS-Dienophile → SAME COMPOUND and CIS SUbstituents
-TRANS-Dienoophile → Enantiomers and TRANS Substituents
Retrosynthetic Analysis of the Diels-Alder Reaction
1. Find the 6-membered ring containing C=C.
2. Draw 3 arrows, beginning at the π bonds.
3. Draw the diene and dienophile.
Chapter 15: Aromaticity
• All C-C bond lengths are equal.
• Planar
• 6 Cs and 6 Hs
Molecule must be:
1. Cyclic
2. Planar
3. Completely conjugated
4. Have a particular # of π electrons (4n+2)
Aromatic compound: 4N + 2 number of electrons (STABLE)
Antiaromatic compound: 4N number of electrons (NOT STABLE)