Sed Strat Week 3
1/36
Earn XP
Description and Tags
Carbonates
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Carbonate formation
Form chemically and biochemically. Dissolved ions are carried to the site of deposition where they precipitate to form solid minerals. Hard to tell the difference between chemical and biochemical
Stromatolites
Mounds of cyanobacteria that are bulbous masses of trapped CaCO3 material. Occur at different scales
left is modern stromatolite while right is ancient

Carbonate chemistry
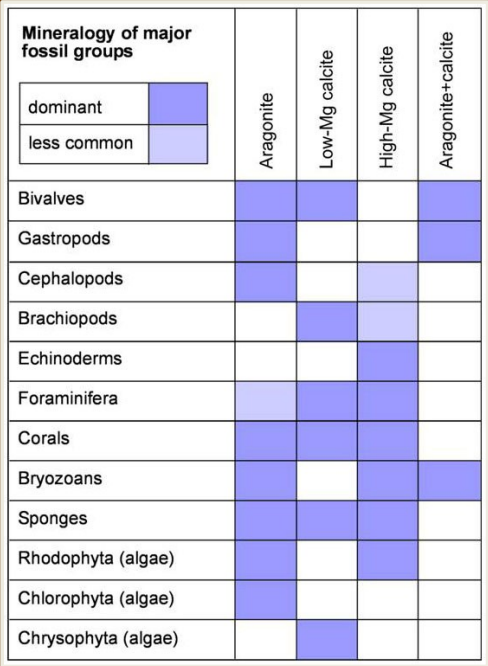
Composed of a divalent cation with a carbonate anion. Calcite, dolomite, and aragonite are the only carbonates available at the surface
Calcite and aragonite (polymorphs)
Calcite has a rhombohedral structure while aragonite’s is orthorhombic. Aragonite’s open lattice allows for more substitution of different cations where calcite’s is more limited. As a result, arag often reverts to calcite. Calcite prefers to substitute calcium for magnesium, where you can get high or low-mag calcite
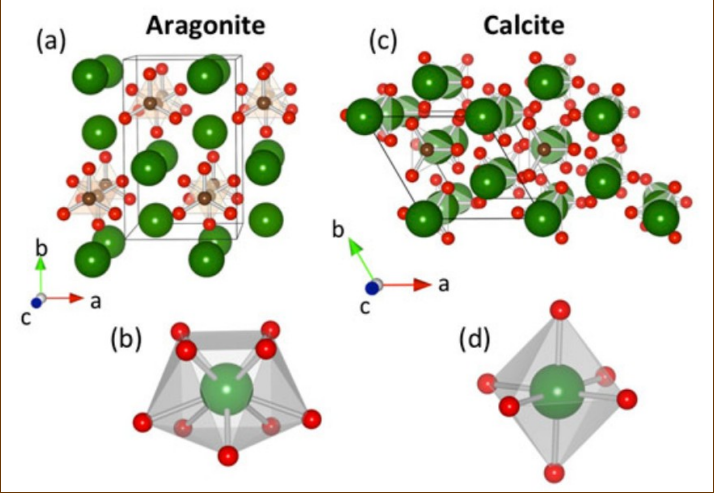
Aragonite cannot incorporate
magnesium in its structure, unlike calcite
High mag calcite (>5% Mg)
Secreted by many different calcareous organisms, less stable than low mag calcite (susceptible to dissolution)
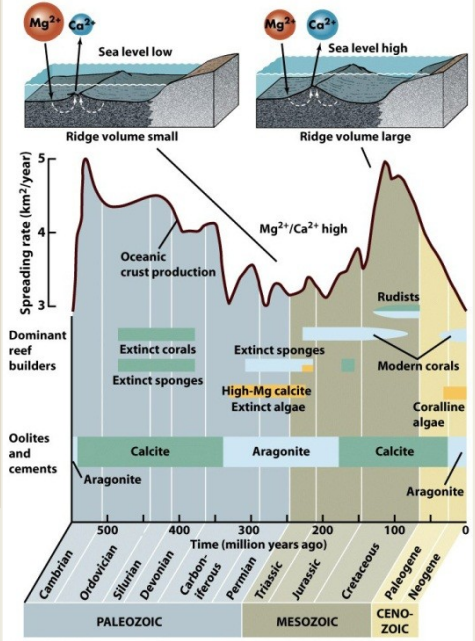
Low rates of seafloor spreading (fusing of continents)
Decreased ratio of Ca/Mg - aragonite and high-mag calcite precipitation
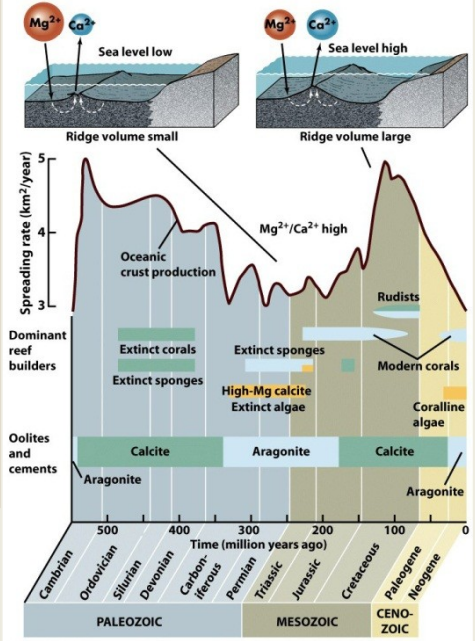
High rates of seafloor spreading (breakup of continents)
Increased ratio of Ca/Mg - low-mag calcite precipitation
Dolomite (CaMg(CO3)2)
Trigonal-rhombohedral. Contains about 50/50 Ca and Mg. Fe²+ may sub for Mg. If there is enough iron, ankerite will form
Carbonates don’t generally form in dolomite-dominated environments today
Siderite (FeCO3)
Most common iron carbonate with same structure as calcite. Rarely pure, often contains traces of Mg or Mn in lattice
Major factors controlling dissolved CO2
Temperature, pressure (water depth), degree of agitation (wave action)
Lesser factors contributing to carbonate formation
Salinity (low salinity is preferred, due to lower carbonate concentration)
Organic activity (plants and animals precipitate CaCO3)
Light (required for photosynthetic organisms and calcareous algae)
Favorable carbonate conditions
Warm temperature, shallow depth, and high-energy waves that drive carbonate ions away, raising pH
these conditions concentrate CO2 at the surface
CCD (calcite compensation depth)
Depth at which calcium carbonate is no longer stable and will dissolve. Not constant; ranges from 4500-5000 m in warm water and 3000 m in polar waters
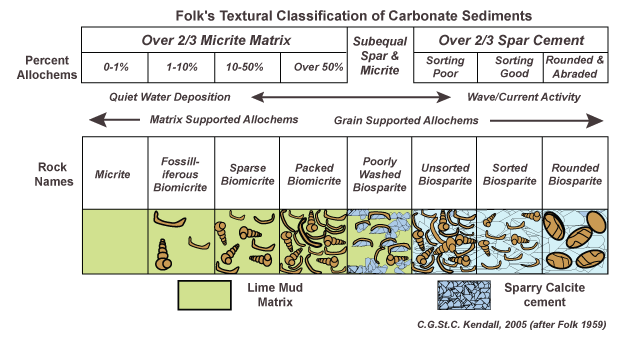
Bob Folk scheme
Separates allochems (in-situ organic grains, skeletal or non-skeletal) and orthochems (in-situ micrite or spar). Focused on the types of both of these
Common skeletals in thin section
image
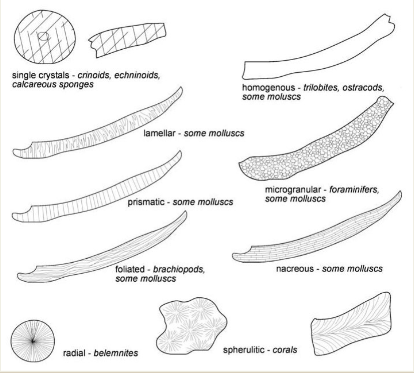
Non-skeletal allochems
Coated grains (ooids, oncoids, pisoids, sand sized)
Peloids (silt - sand size)
Clumped/aggregated grains (grapestones)
Intraclasts (pre-existing, eroded limestone/mud chunks with internal structure)
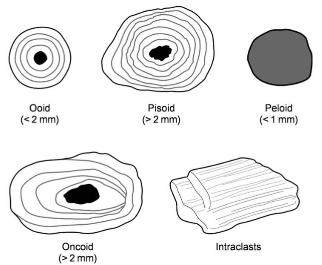
Ooid vs peloid formation
Ooids are theorized to form in high energy waters, giving them their more stable form and internal structure while peloids form in low energy, calm waters (lagoons). They are less compact and often softer than ooids, with no internal structure
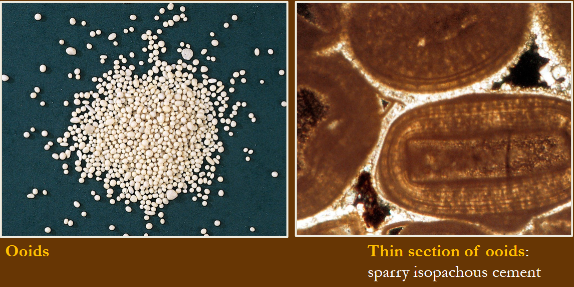
Grapestones
Clusters of cemented ooids, peloids, and skeletal pieces bound together by encrusting organisms/micrite and then reworked. Form in low energy settings

Polygenetic (micrite)
More than one source of formation. Micrite can be formed through the breakdown of green algae (halemeda, penicillus), whitings, and micritic envelopes (endolithic boring algae)

Spar
Carbonate cement. Coarser than micrite, optically clear. May form as pore-filling or recrystallizing micrite (neomorphism)
Dunham scheme
Ignores allochem type. Based on presence or absence of micrite. All except boundstone accumulate as clastic carbonates (components not bound at deposition)
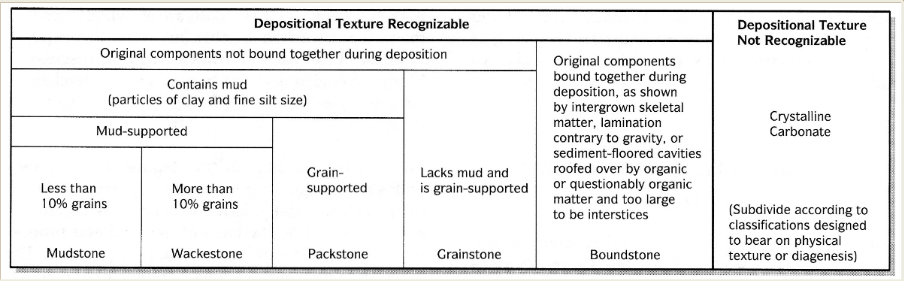
Modified Dunham scheme
Modified to provide niches for limestones that contain allochems coarser than 2 mm.
Matrix supported rocks like these are floatstones, grain supported are rudstones
Evaporites
Form from evaporation of saline water. Minerals precipitated out of solution become more concentrated
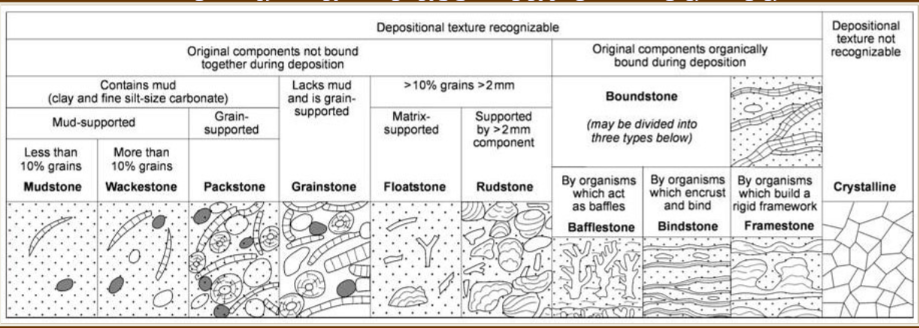
Seawater composition
Contains 35g/l (35,000 ppm) of dissolved ions.
Saline lakes have same ions, but in different proportions. May include trona, mirabilite, epsomite
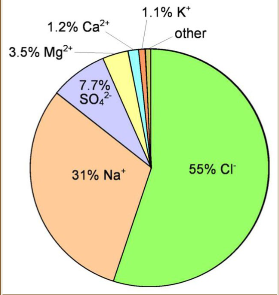
Marine evaporite precipitation
The most soluble compounds precipitate first when seawater is evaporated, so CaCO3 is first, followed by CaSO4, NaCl, and KCl once the seawater is fully concentrated
Deep-water basin evaporites
In a deep ocean basin, past the reef, evaporite salts and minerals will settle on the ocean floor, precipitating crystals until they bind together and lithify in ascending layers. Dark layers may represent organic matter while white layers may represent higher salt concentration
Shallow-water basin evaporites
A shallow basin is flooded, and evaporites form as that water dries up. This often results in evaporites with only modest thickness, but layers can deepen as the basin subsides
Deep-basin, shallow basin evaporites
A deep basin is separated from the ocean by a barrier. Water floods over the barrier into the basin, where it evaporates to form evaporites. Repetition of this event allows evaporites to stack and increase in volume
Gypsum and anhydrite
Most common evaporite minerals. Gypsum is hydrous and precipitates at the surface under all but the most arid conditions, and may dehydrate to anhydrite when buried. Anhydrite has no water in its structure and forms by precipitation on arid shorelines or the alteration of gypsum
Halite
Precipitates from seawater at 9.5% of its original volume. Occurs in thick beds of cubic crystals; only preserved in rocks with absence of dilute groundwater due to high solubility. Rock salt
Sylvite (KCl) may form with halite


Chert
fine grained siliceous rock made up of silt-sized interlocking quartz crystals. Chert beds are formed as primary sediment or from diagenesis; Diatom/radiolaria/sponge skeletons form siliceous ooze on the seafloor that consolidates to form chert (thin bedded), or opaline silica recrystallizes to chalcedony and quartz

Diagenetic chert (chalcedony from replacement) occurs as
Nodules and irregular layers within mudstones/limestones. Precipitates under acidic conditions

Sedimentary phosphates (PO4)3
Occurs in igneous rocks as apatite, an accessory mineral in granite. Generally, phosphates occur in solution and are absorbed by plants or animals. Often occurs in bones, teeth, or scales in biogenic and clastic rock. High concentrations found in shallow marine continental shelf deposits.
Most occurrences happen in conditions with low oxygen and high organic activity. Not fully anoxic. Composed of francolite (Ca((PO4)3)
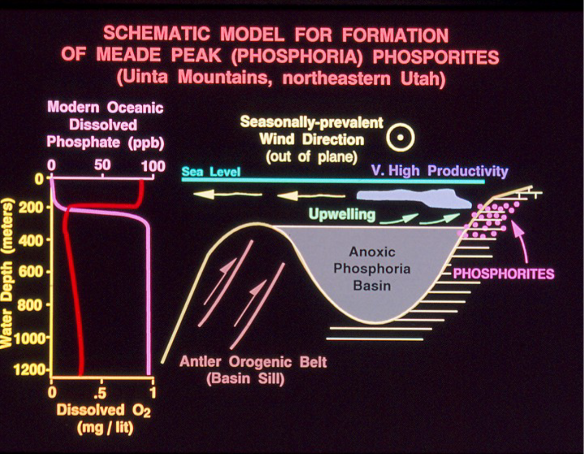
Oxygen minimum at 200m (phosphorite formation)
Aerobic decomposition creates anoxic conditions that ripen the area for phosphorite formation. When biogenic particles reach the seafloor, there is little oxygen, so they are decomposed via sulfate reducers, which create phosphate crystals in pore spaces. Nutrients are brought to the surface from upwelling, effectively fixating phosphate minerals there

Sedimentary ironstone
Contain at least 15% iron in the form of oxides, hydroxides, carbonates, sulfides, silicates. Occur in all environments; often include magnetite, hematite (most common), goethite, and limonite. Opaque in thin section w/ various colors

Banded iron formation
All banded iron formations are Precambrian in age. Thought to have originated in shallow or marginal marine environments, probably on continental shelves or shallow basins. Early microbes could have oxygenated waters, releasing Fe2+ ions that oxidized on the seafloor while chert layered on top in alternating bands
