QA/QC and Molecular Microbiology Part 1
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Regulations and governing bodies
federal laws governs implementation, usage and quality programs that surround testing of human specimens
Clinical Lab Improvement Amendments - regulations passed down by CMS (CLIA ‘88)
Joint commission (TJC), local state laws (state DOH), and other accrediting orgs (CAP, AABB)
some states have additional requirements for testing/accreditation (CLEP - clinical lab eval program, NYS DOH)
accrediting bodies will sometimes defer to whichever one is “stricter” regarding rules CLEP>CAP>CLIA
So what do they tell us how to do the tests?
must be accredited first (apply for license)
must be enrolled in PT program (blinded challenges)
must undergo periodic review by other labs (biannual inspection)
must have staff that are qualified and certified to run tests alongside ALL PAPERWORK
CAP - biyearly inspection where teams come in to review lab
Requirements for testing/reporting
whats inside?
thing you must do
what they expect you to do (why)
how you have to keep track
This covers
training/competency, safety, SOPs, IT, testing, QC/QA
IVD (in vitro diagnostic)
intended for use in diagnosis of disease or other conditions
easiest to show performance and introduce
use in collection, prep, and examination of specimens taken from human body
everything made by company therefore cannot deviate from manufacturer’s instructions
Pre-market approval (new diagnostic)
goes through initial submissions of safety and efficacy of new device in chosen field
510k Premarket notifcation
comparative analysis of new assay to “gold standard” showing substantial equivalency
Emergency use authorization
classification for emerging diseases/infectious agents - limited FDA submission data (rare) [ex. COVID testing]
Laboratory developed test (LDTs)
developed by lab (nothing on market)
need to show that it works well (validation)
vendors won’t help (no tech support)
have to state “not FDA approved” and that “characteristics were determined by X lab”
may be cheaper than IVD (can use equipment you already have)
Proficency testing/challenges
regulated analytes require annual enrollment (CLIA defines)
CMS tell us which ones are approved (PT 3× 5 samples/yr)
for non-regulated analytes, lab tests 2x/yr
if no PT avail, can work with other labs (peer/alt review) and show performance (blinded samples)
Showing your test works
IVD requires verification (unless modified)
LDT requires full validation
verfication
shows assay works like manufacturer says
FDA-cleared or approved tests only
checks accuracy, precision, reportable range, and reference range
compare new method to one already verified (test using same samples) - do this w ALL specimen types
any changes require FULL validation (don’t deviate)
Validation
checks accuracy, precision, reportable range, reference range
ALSO checks for interferences, sensitivity, specificity, LoD, LoQ
check against another method (IVD or LDT that’s approved)
define: controls, protocol, and performance chars
Qualitative tests
require +, - analytes and non-template control (NTC)
these show assay detects what it should, no false pos amplifications, and no contamination
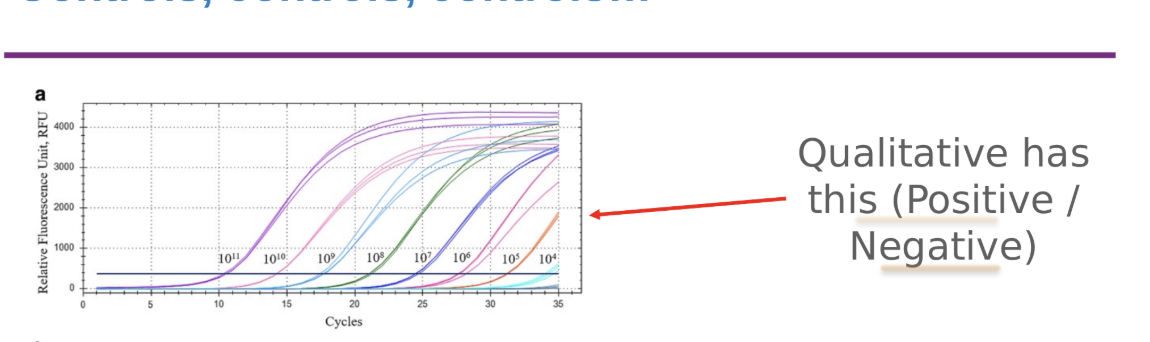
Quantitative test
require (1) high and low pos, (2) -, (3) NTC
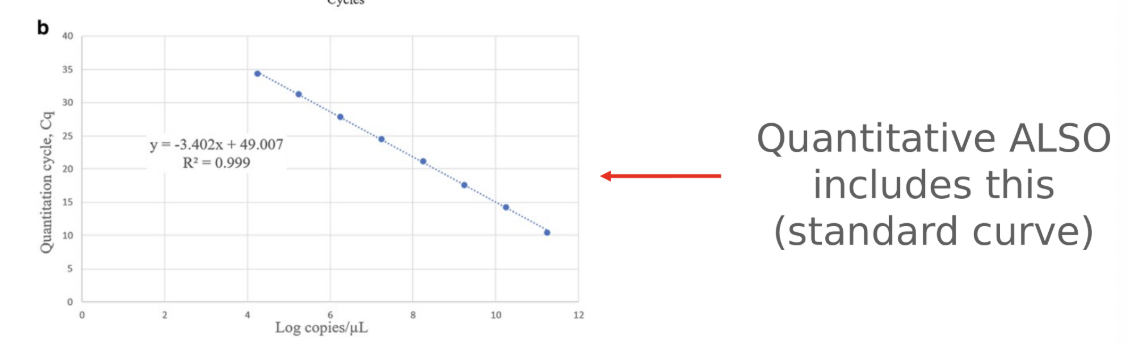
have “standard curve” of known analytes at specific concentrations
External control
typically brought by someone other than test maker
previously tested (post/neg) samples work too
shows assays detects targets/works
Internal control
typically part of the assay (checks for quality)
can be human genes or spiked in target
no amplification = can’t report = INVALID test
Extraction control
used to show that nucleic acid extraction worked
can be same control as internal/external
many companies use this as a schema to save on reagent costs (fewer tubes to run and less ctrl mats to use at once)
Why do we need so many controls?
manufacturers define conditions in IVD approval as to how many controls need to be run
typically defined within 24 hr window or per each “batch” of tests
if LDT, control frequency must be defined by lab
some tests don’t have defined frequency for QC to be run, thus lab must work on defining this (ICQP)
all control performance must be tracked (logged and tracked for compliance)
must be done prior to reporting ANY specimen results
IQCP: individualized quality control plan
QC plan that allows for decreased control mat utilization IF Assay allows for it
in NYS no less than 1 + sample/mon, 1 neg sample/week
each lab must carry risk assessment and review of performance (20-30d of runs)
monitored monthly following approval - signed by lab director
changes/updates needed if deviates from prior approval conditions
used to cut down on usage of individual cassettes/reagents (cost)
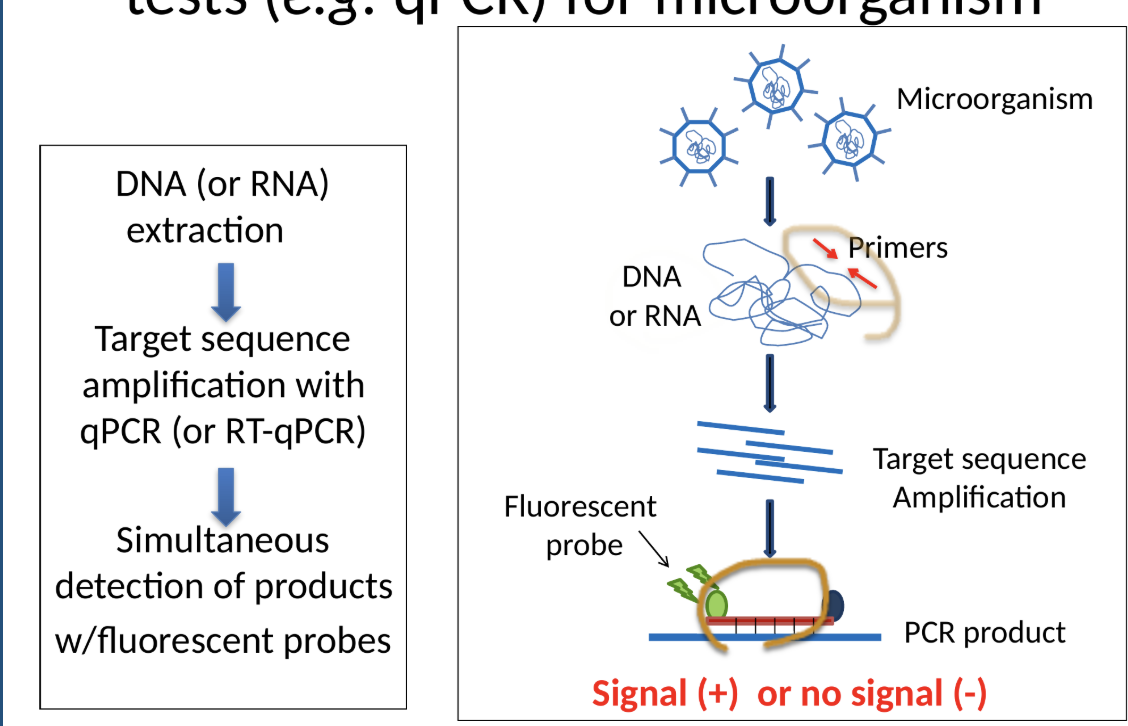
Molecular microbiology
detection, ID, and analysis of microorganism (DNA/RNA)
advantages:
rapid turn around time
better sensitivity and specificity
can be quantitative
comparison of biochemically similar organisms (epidemiological)
applications of molecular assays in microbiology
replaced viral culture in most labs
antibiotic resistance testing tailored for antimicrobial therapy
quantification of viruses
genotyping, classification, and epidemiological studies
discovery of novel pathogens
microbiome/virome/mycobiome studies
DNA detection methods
signal amplification methods
nucleic acid amplification tests (NAATs)
post-amplification analysis methods
sequencing and NGS
Signal amplification methods
typically use nucleic acid probe combined with some form of amplifying signal producer (enzyme:
branched DNA
hybrid capture
in situ hybridization
typically less sensitive than NAATs
less likely to be contaminated than NAATs (false +)
In situ hybridization (Fluorescent -ISH)
use hybridized probes of DNA (or peptide, PNA) to ID intact microorganisms
bind to rRNA molecules on microbes (higher copy # than a single gene) and aid in ID/selection of antimicrobials
Nucleic acid amplification tests (NAATs)
qPCR or RT-qPCR
predominant workhorse of most molecular diagnostic labs. End-stage PCR have mostly been replaced by qPCR
SYBR green (primers specific for ROI) and TaqMan (primers and an ROI specific probe)
region of interest - ROI
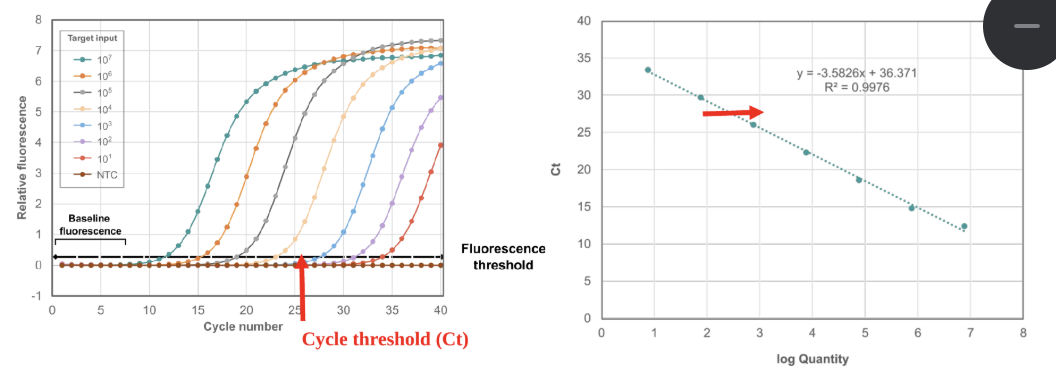
qPCR test principles
NAATs: qualitative assays
presence or absence of a gene/ROI
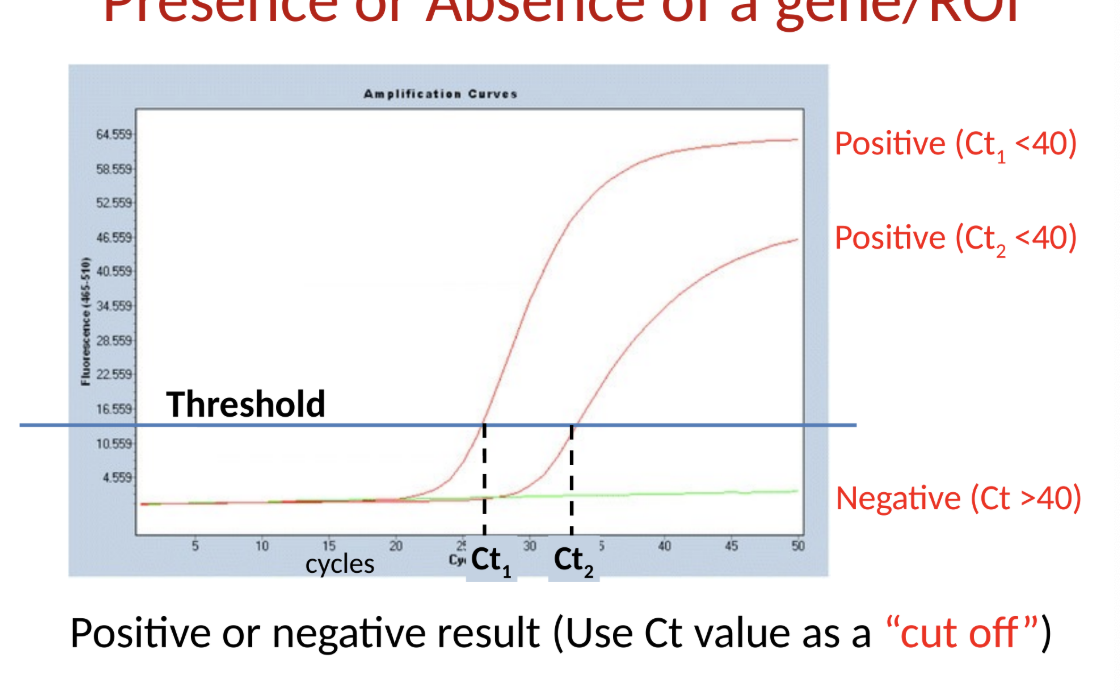
what are qualitative assays good for?
diagnosis of infection
syndromic-based diagnostic panels (test for multiple pathogens in 1 test)
examples of syndromic-based panels
many are “sample-to-answer” platforms where assay complexity is highly reduced
nucleic acids extracted onboard
PCR carried out on cartridge
self-sealed containers that can be discarded once used

NAATs: quantitative assays
uses standard curve (serial dilution of known concentrations) run in parallel to allow for estimation of titer within a sample
used for diagnosis/tracking of titers of organisms in bodily fluids
namely viral titers in serum/plasma
quantifiable range and reportable range are not always same
things can be + < or > limit of quantitation, and are still +
cannot report outside of LoQ (only that it is +)
Ct values =/= quantitation (and shouldn’t be interp that way)
How do you select target sequence for ID of microorganisms?
genomic or plasmid DNA, genomic RNA
unique gene/sequence for pathogen of interest (virulence factors)
antibiotic resistance gene
for viruses with various subtypes, use:
sequence shared by all types for initial detection
use specific sequence for further typing
Major considerations of using molecular tests
CANT differentiate between alive/dead organisms
remnant nucleic acid can persist for weeks or even months
many PCRs shouldn’t be used as “test of cure'“
not all tests and targets are equal - know test limitations
not detected=/= not present
pathogen under detection limit, wrong location
genotype =/= phenotype
presence of antibiotic resistance gene doesn’t be bacteria is resistance
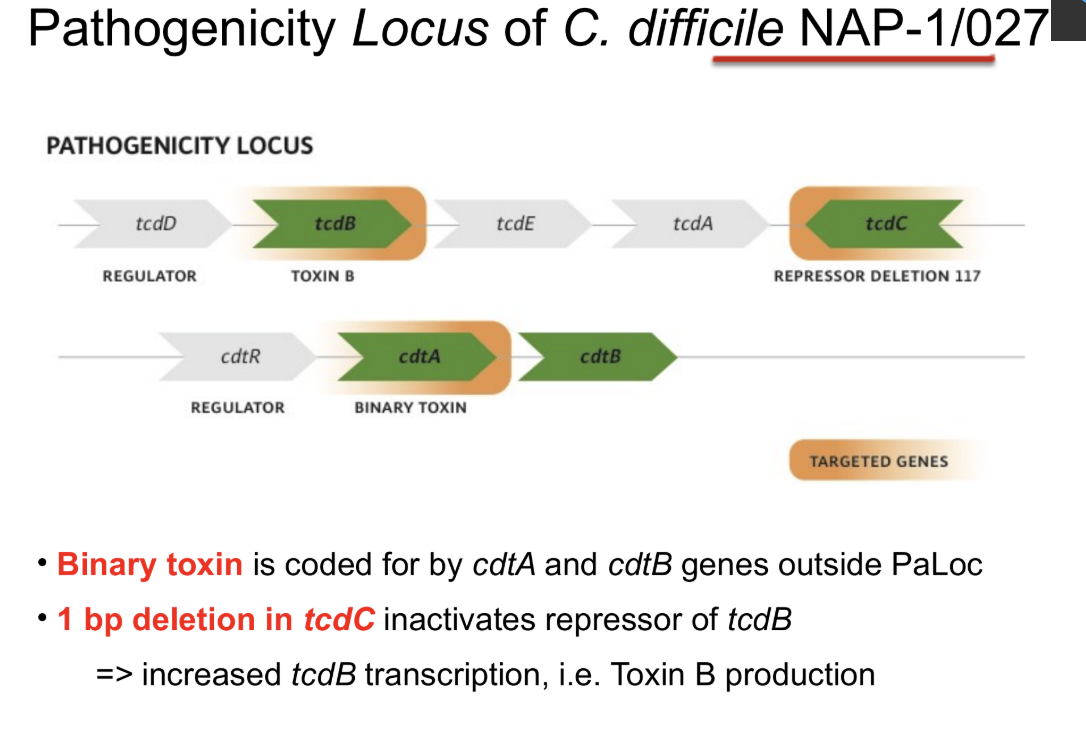
C. diff
pathogenicity locus: PaLoc
produced by tcdB and tcdA [produce toxins B and A]
most assays try to target tcdB
![<ul><li><p>pathogenicity locus: PaLoc</p><ul><li><p>produced by tcdB and tcdA [produce toxins B and A]</p><ul><li><p>most assays try to target tcdB</p></li></ul></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/8be1378c-74e7-4cee-8d36-8d7bd7fc7b32.png)
Internal control for C. diff assay
IC: spores of Bacillus globigii
tests for validity of each sample
+ IC
nucleic acid extraction worked
also checks absence of PCR inhibitors
valid test
- IC
invalid test
sample needs re-extraction
qPCR detection of C.diff using GeneXpert (Cepheid)
quick TAT: 50 min
closed system
detects ToxB gene and internal control
high sensitivity and specificity
severe epidemic strain of C. Diff
BI/NAP1/027, toxinotype III
carries extra toxin known as binary toxin (cdtB, cdtA)
increased toxin A/B production due to polymorphism in regulatory gene (tcdC)
Glutamate dehydrogenase (GDH) EIA
fast and sensitive, but not specific (false +s)
all C. diff strains (toxic or not), other clostridial species and other bacteria produce GDH