Energy changes
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
What is an exothermic reaction?
A reaction that transfers energy to the surroundings, so the temperature of the surroundings increases
What is an endothermic reaction?
A reaction that takes in energy from the surroundings, so the temperature of the surroundings decreases
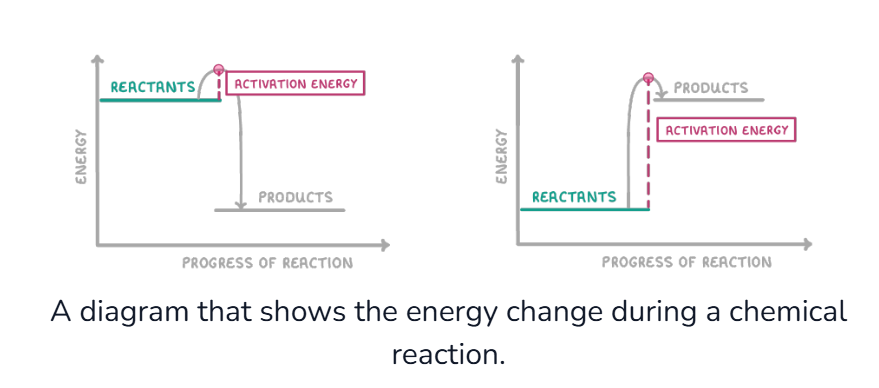
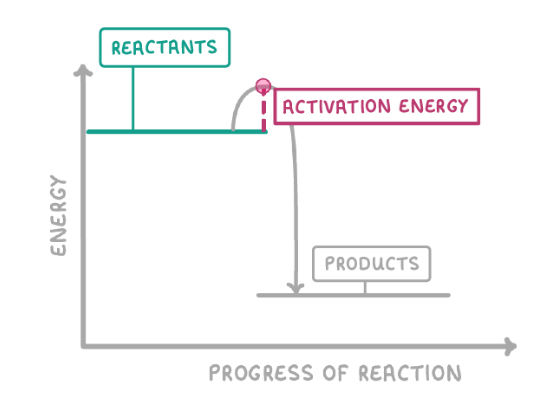
What is a reaction profile diagram?
examples of exothermic reactions
combustion
neutralisation
oxidation
Examples of endothermic reactions
citric acid + sodium hydrogen carbonate
thermal decomposition
what does an exothermic reaction look like on a graph?
The energy of the products is lower than the reactants, so energy has been lost to the surroundings
what does an endothermic reaction look like on a graph?
The energy of the products is higher than the reactants, so energy has been gained from the surroundings
Is bond breaking endothermic or exothermic?
endothermic because energy is required to break bonds
is bond forming endothermic or exothermic?
exothermic because energy is released when new bonds are formed
what is the formula for calculating the energy change of a reaction using bind energies?
energy required to break bonds - energy released by forming bonds
What is activation energy?
the energy needed to break the bonds of the reactants