Control of Blood Glucose
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
40 Terms
Why is glucose homeostasis important?
- The CNS is almost entirely dependent on glucose as a source of energy (as
well as rbc’s)
- Blood glucose concentrations fluctuate in the fed and fasting state: too low
and too high concentrations are harmful
What is the normal circulation concentration?
3.9-6.2 mM
What is the average fasting concentration?
4.4-5 mM
What level is dangerously low?
2.5 and less induces coma/death
Note: If it rises for an extended time can induce come and death
What are the main hypoglycaemic effects?
- Dizzy/drowsy/coma
- RBC membrane rupture compromising O2 delivery
What are the main hyperglycaemic effects?
- Osmotic loss of cell water = dehydration and
hyperosmolar coma
- Protein glycosylation (e.g. glycosylated haemoglobin) = tissue malfunction
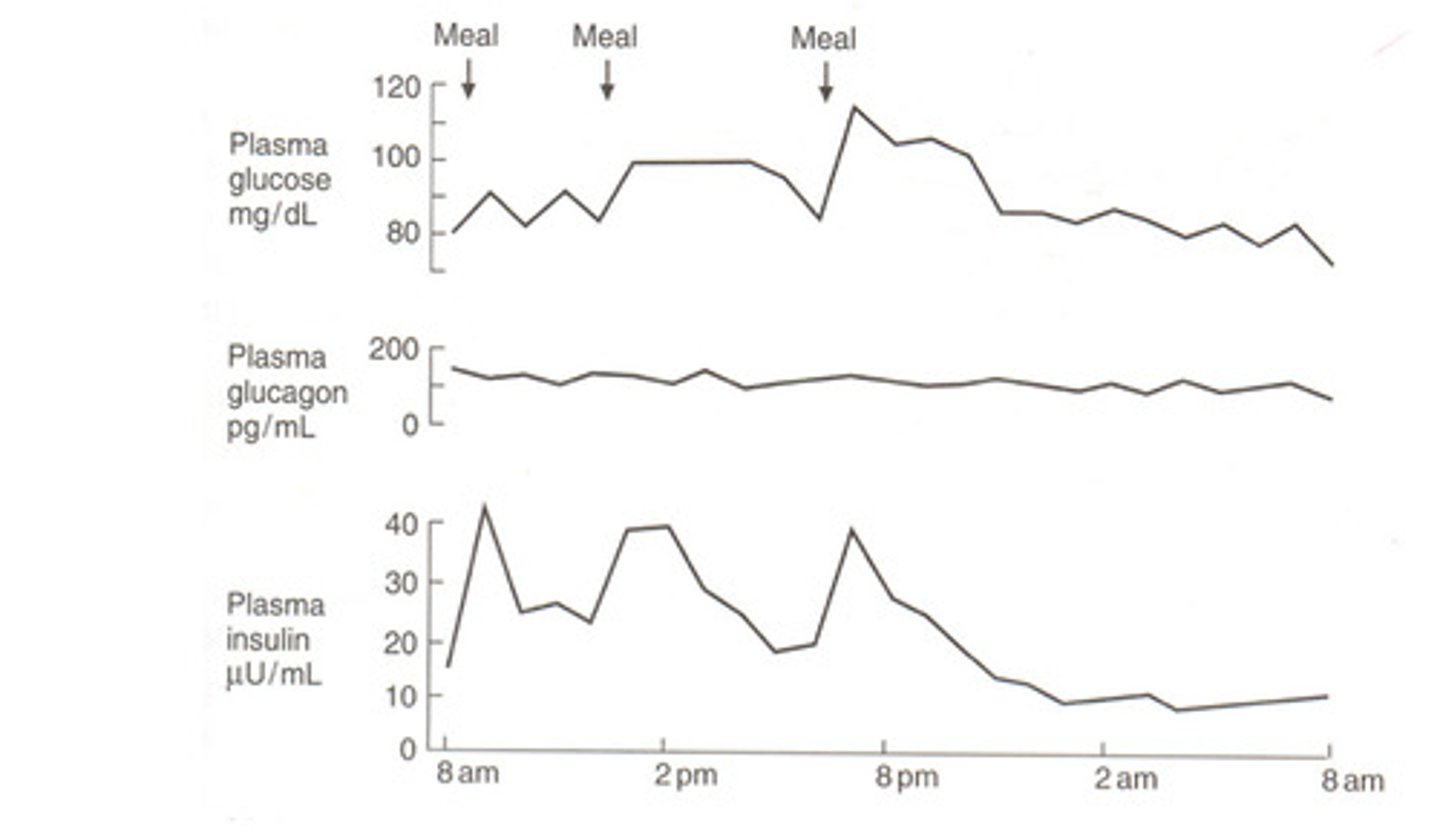
Blood glucose, insulin and glucagon in a normal person over 24 hours of normal eating behaviour (mixed meals) - Graph and trends
-Blood glucose increases when you eat and comes down over time
-Blood glucose is maintained over night
-Two main hormones that control it is insulin and glucagon
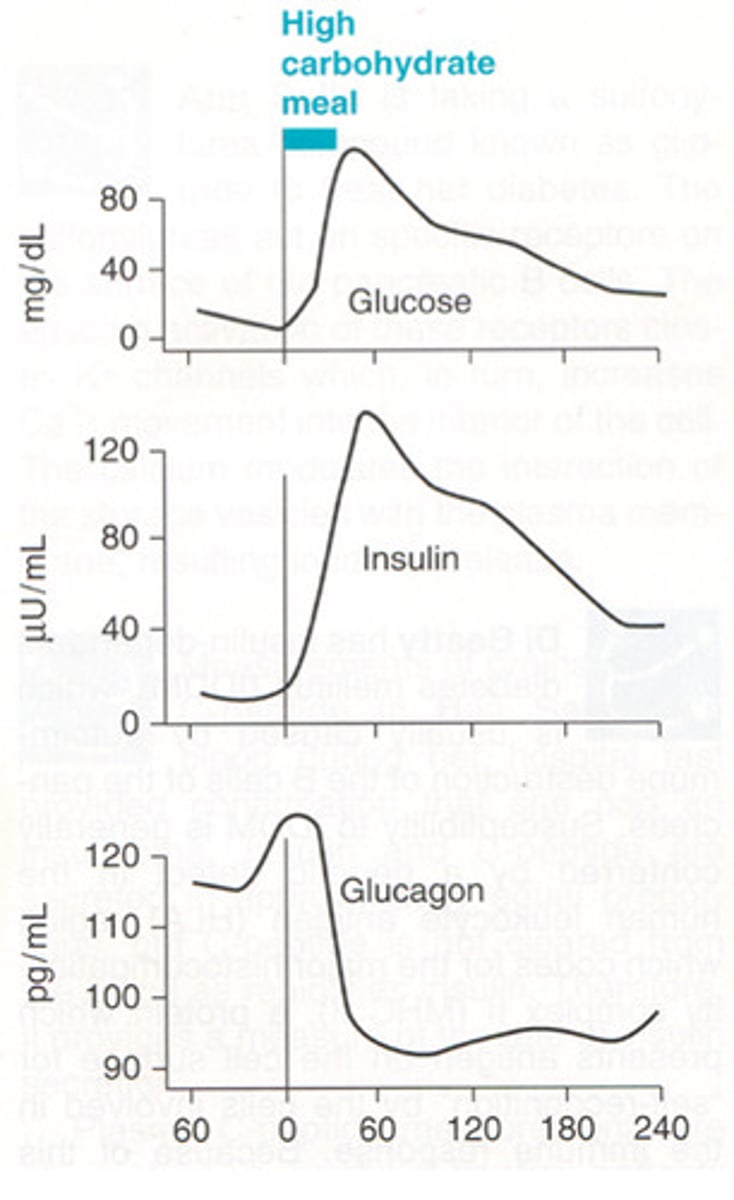
Blood glucose,
insulin
and glucagon
after a high-
carbohydrate
meal - graph
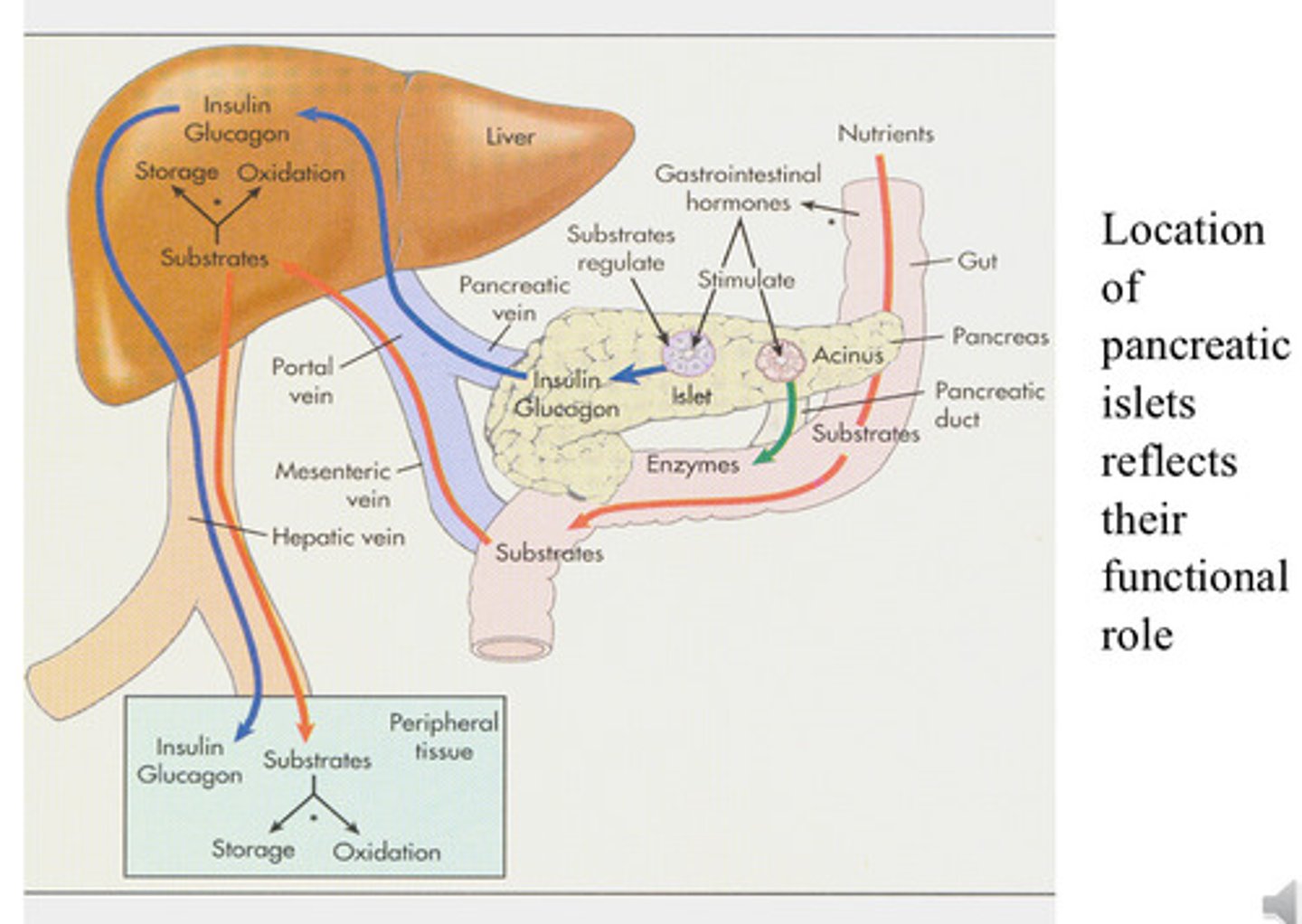
How does the location of pancreatic islets reflect their functional role?
Situated close to gut to respond to glucose changes quickly
-Secretes insulin if food is coming in or glucagon in a fasted state
-Effects the liver which controls metabolism
What are the prime glucose regulators?
Insulin (is the only hypoglycaemia hormones) and glucagon
What are the other hormones which counteract insulin?
adrenaline (adrenal medulla), cortisol (adrenal cortex),
growth hormone (anterior pituitary)
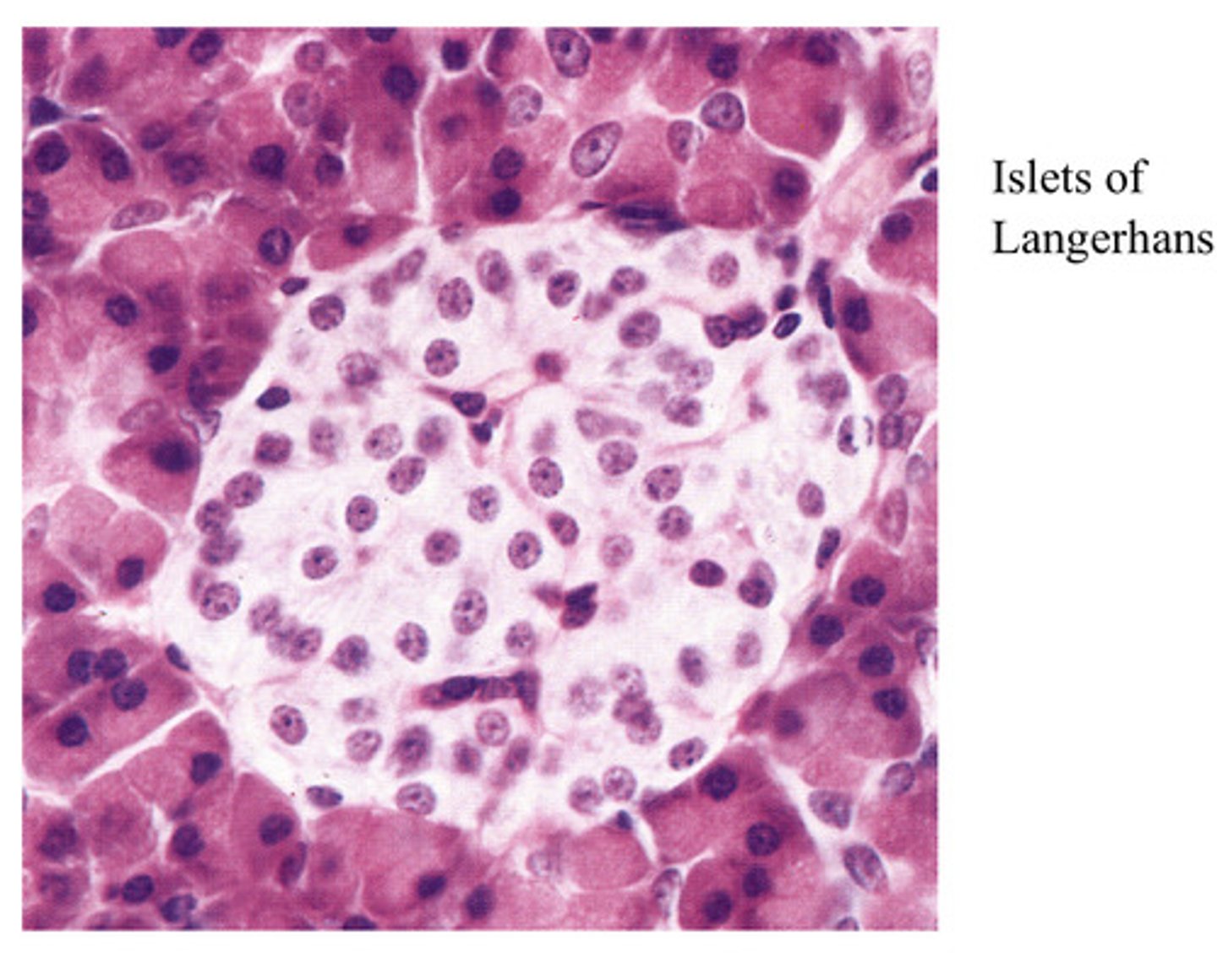
What are the Islets of Langerhans and what is its significance to to blood glucose control
-make up the endocrine (hormones secreted into circulation) part of the pancreas
Contents of the Islets of Langerhans:
- Beta cells (60-70%) secrete insulin
- Alpha cells (30-40%) secrete glucagon
- delta cells secrete somatostatin (Somatostatin inhibits the secretion of other hormones, such as growth hormone, thyroid stimulating hormone, insulin, and glucagon.)
How is insulin made in beta cells?
- Nucleus transcribed pre-pro- insulin mRNA
- rough endoplasmic reticulum (RER) translates pre-proinsulin and cleaves to pro-form
- Golgi packages pro-insulin into secretory granules and processing enzymes
- Secretory granules converts pro-insulin into insulin and C-peptide
- Insulin remains in crystals with Zn 2+ until secretion
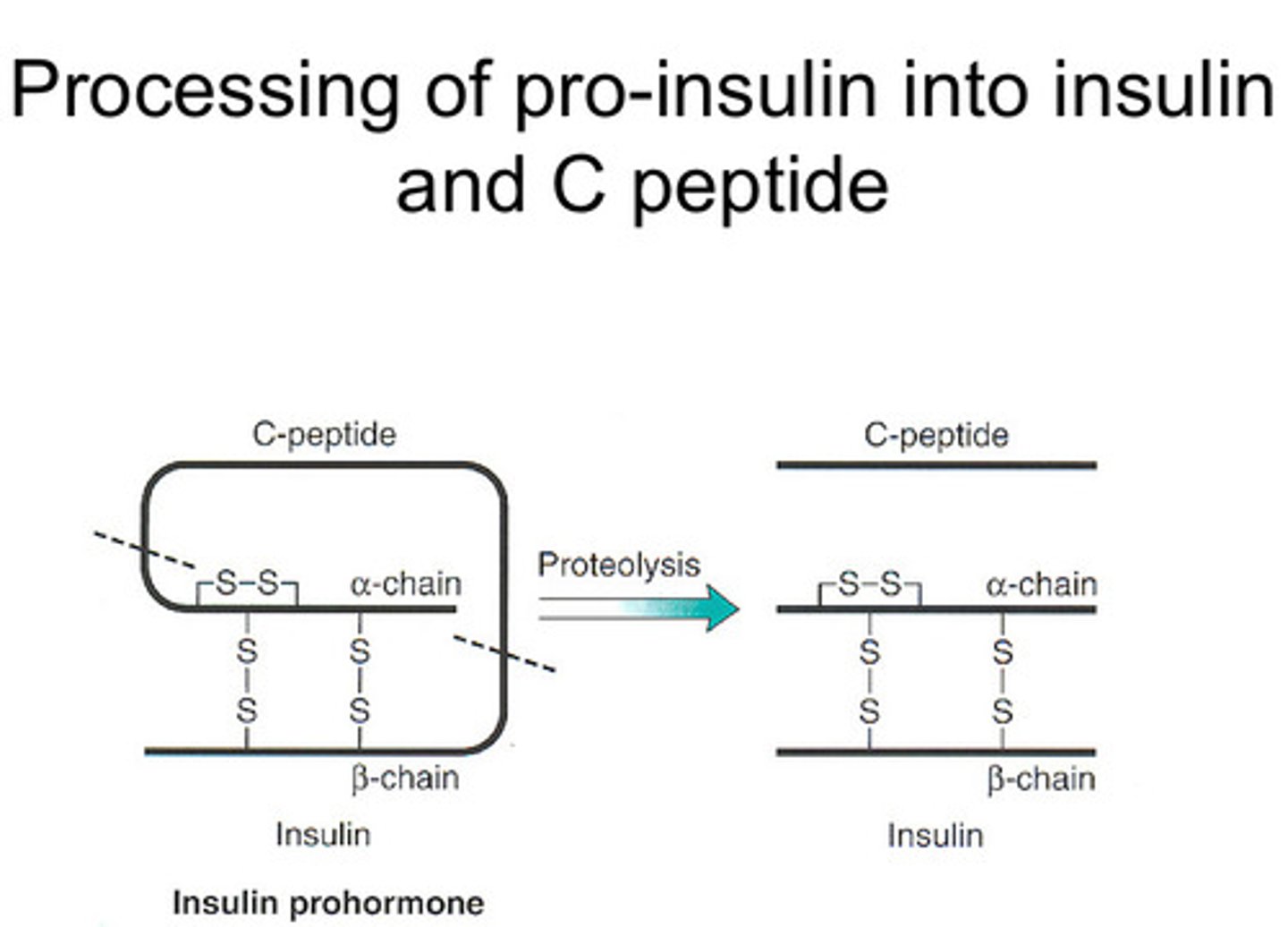
Why is C-peptide important to insulin release?
Allows transition from inactive to active and used diagnostically (If there is no C peptide in circulation = individual is not producing insulin at all )
How is insulin released from cells?
- Glucose gets into pancreatic beta cells via Glu2 transporter and is metabolised to give ATP
- ATP blocks potassium channels in membrane = polarisation changes = calcium
channels open and calcium enters (increasing calcium concentration in the cell) causing insulin to be released from cell
(action of some Type II drugs when insulin not sufficiently
released/biologically active)
Key short summary
- ATP blocks membrane channels
- Polarisation change
- Ca channels open and Ca enters
- Insulin released
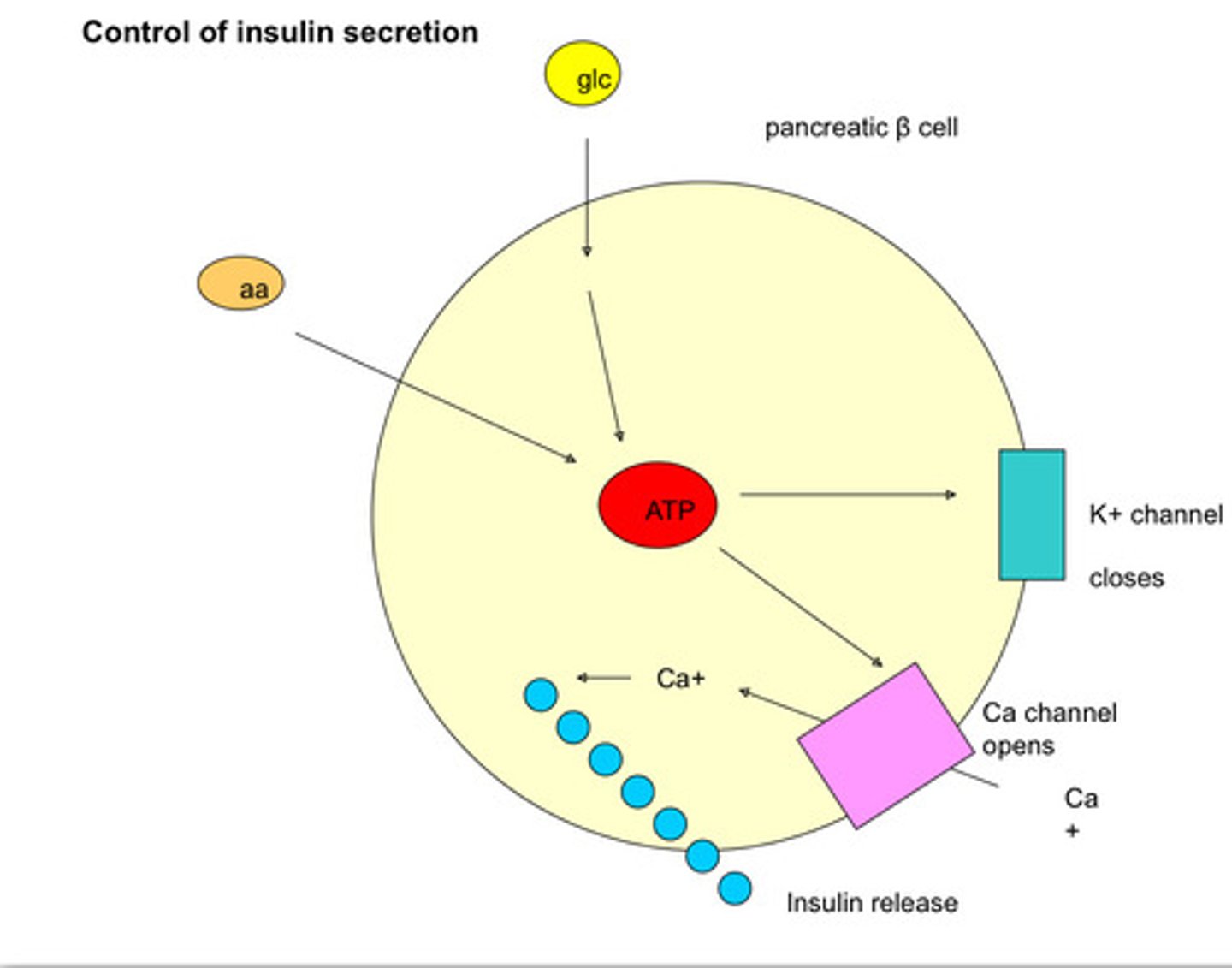
How is insulin secretion stimulated?
- Rise in glucose
- Rise in amino acid conc
- Gut hormones: Secretin and other GI hormones after food intake before glucose spike
- Glucagon risen - insulin provides fine tuning of blood glucose
homeostasis (prevents blood glucose extreme fluctuations- keeps it balanced)
What inhibits insulin secretion stimulation?
Adrenaline
How is glucagon secretion stimulated?
- Low blood glucose
- High amino acid in blood (prevents hypoglycaemia after protein-rich meal)
- Adrenaline (in stress, glucagon rises despite blood glucose because insulin is suppressed)
Sites of Insulin Action on metabolism summary diagram
What are the sites of insulin action on metabolism?
- Glucose gets into liver when high enough, in muscle high
glucose and insulin presence activates transporters so when
glucose is low you want it to go to brain not muscle
(prioritises)
-Insulin secreted (synthesis and storage function)- glucose converted to glycogen and enzymes related are activated and excess is converted to fatty acids which go into bloodstream to adipose tissue and stored as fat
-Glycogen difference in muscle and liver- in liver used to provide glucose when fasting and in muscle only used by muscle so will not increase blood glucose
Overall Insulin:
-Inhibits glycogen breakdown
-Inhibit Conversion of amino acids to glucose
-Will activate conversion of glucose to glycogen and storage
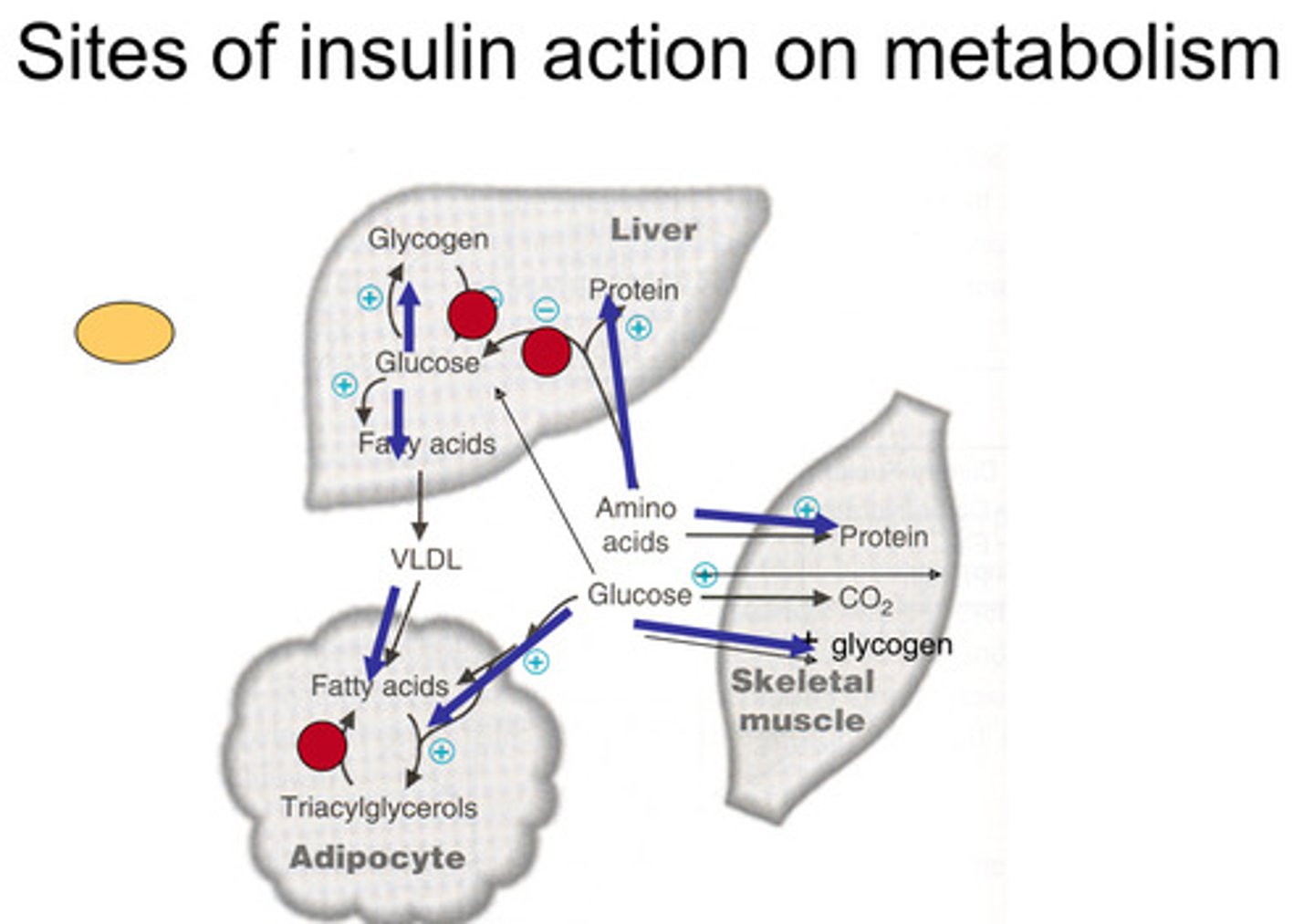
When glucose is low, how does insulin shift glucose priority?
Activates transporters to take glucose to brain not muscles
What happens to glucose once insulin is secreted?
- Glucose converted to glycogen
- excess is converted to fatty acids to be stored as fat in adipose tissue
How does insulin affect the liver?
- Inhibits gluconeogenesis so any entering amino acid doesn't turn into glucose as liver is saturated
- Activated glycogen synthesis (via activation of glycogen synthase) using glycol synthase
- Increased fatty acid and lipid synthesis
- Increases amino acid uptake and protein synthesis
What are the effects of insulin on muscle?
- Increased glucose uptake via increase in glucose transporters
- Increase amino acid uptake and protein syntheiss
- Glycogen synthesis activation (via activation of glycogen synthase)
What are the effects of insulin on adipose?
- Uptake of Fatty Acids and store as Triacylglycerol's (TAG) via activation of lipoprotein lipase
- Inhibit lipolysis
- Increased glucose uptake in adipocytes for TAG assembly and inhibit fat breakdown
Sites of glucagon Action on metabolism summary diagram
Glucagon:
-causes breakdown of glycogen turning it into glucose and sends it out of the liver into circulation
-Inhibits the synthesis of fatty acids
-Breaks down fat from the adipocyte and fatty acids in the blood stream (these can't get into the brain - only used in the muscle)
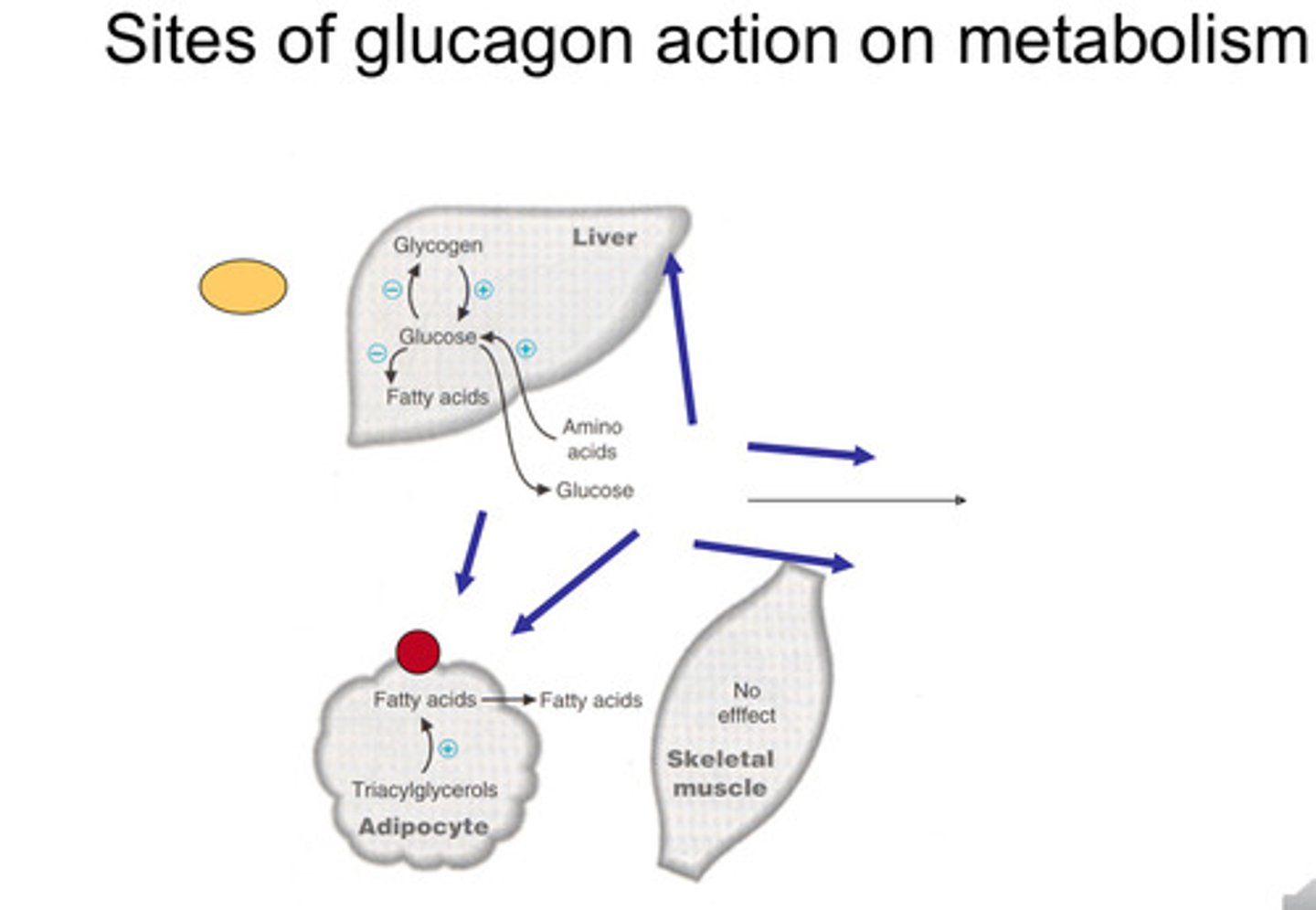
What are the effects of glucagon?
- Increase glycogenolysis
- Increase gluconeogenesis - making glucose from protein (in the liver
)
- Increase circulating fatty acids and ketone bodies
- Increase adipose lipolysis
- Decrease plasma amino acids and more uptake in liver
Graph of what happen after you stop eating
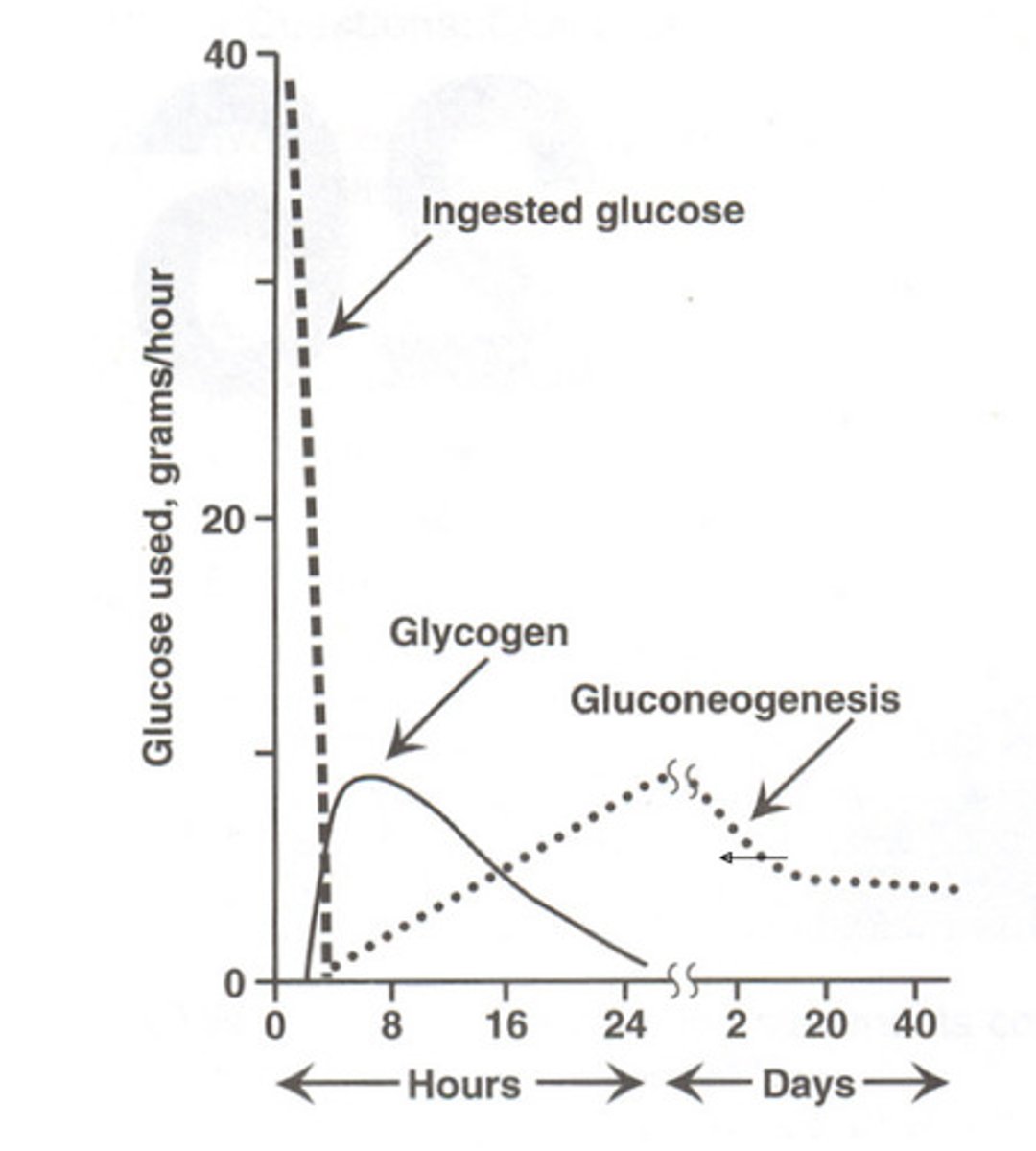
Role of Adrenaline?
mobilises fuels during stress by stimulating glycogenolysis
in muscle/liver and stimulates fatty acid release from adipose tissue
Process when the brain is fed
Fully oxidative metabolism takes place in the brain
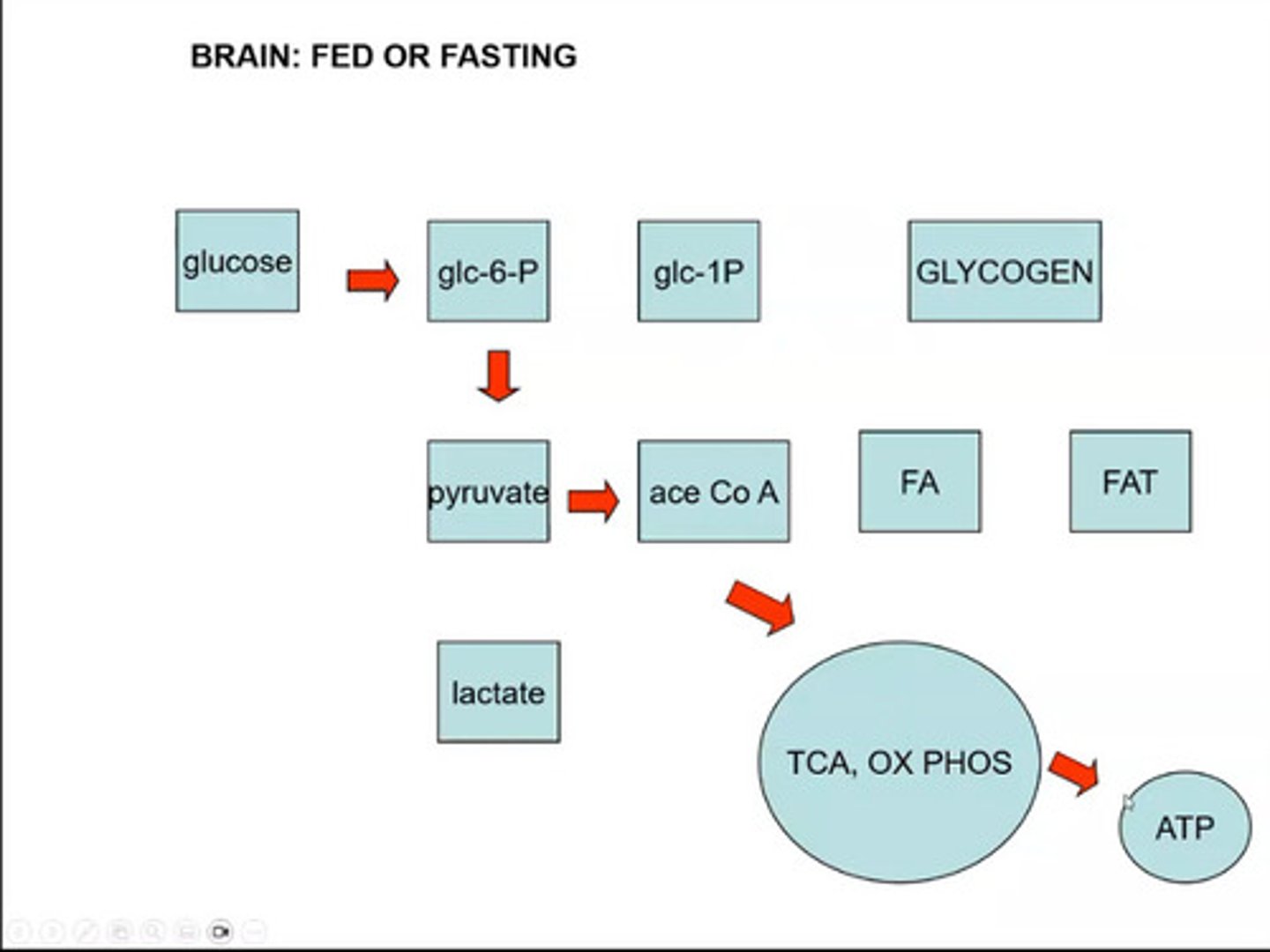
Why can erythrocytes only use glucose and not fatty acids
-Cannot use acetyl coA as it has no mitochondria so only glycolysis
-Advantage of producing lactate: regenerates NAD (reoxidise NADH) as well as lactate going back to the liver which then gets converted into pyruvate and then to glucose.
How does cortisol influence blood glucose?
- Stimulates amino acid mobilisation from muscles
- Stimulate gluconeogenesis
- Stimulate FA release from adipose
Describe Type I
- Type 1- or insulin-dependent diabetes- is the autoimmune destruction of beta
cells - No insulin production - constantly in fasted state
- Early onset
- Polyuria (passing a lot of urine), polydipsia (excessive thirst), polyphagia (extreme hunger), fatigue, weight loss, muscle wasting
weakness
- Hallmarks are hyperglycaemia and ketoacidosis - medical emergency (as you are breaking down a lot of fats)
- Requires insulin treatment
Describe Type II
- Type II- or non-insulin dependent diabetes- is resistance to insulin causing
target tissues to be unresponsive
- Later onset associated with diet and lifestyle e.g., obesity and milder
- Major incidence increase including children
- Hyperglycaemia but usually no ketoacidosis
- Often responds to diet and oral hypoglycaemic agents
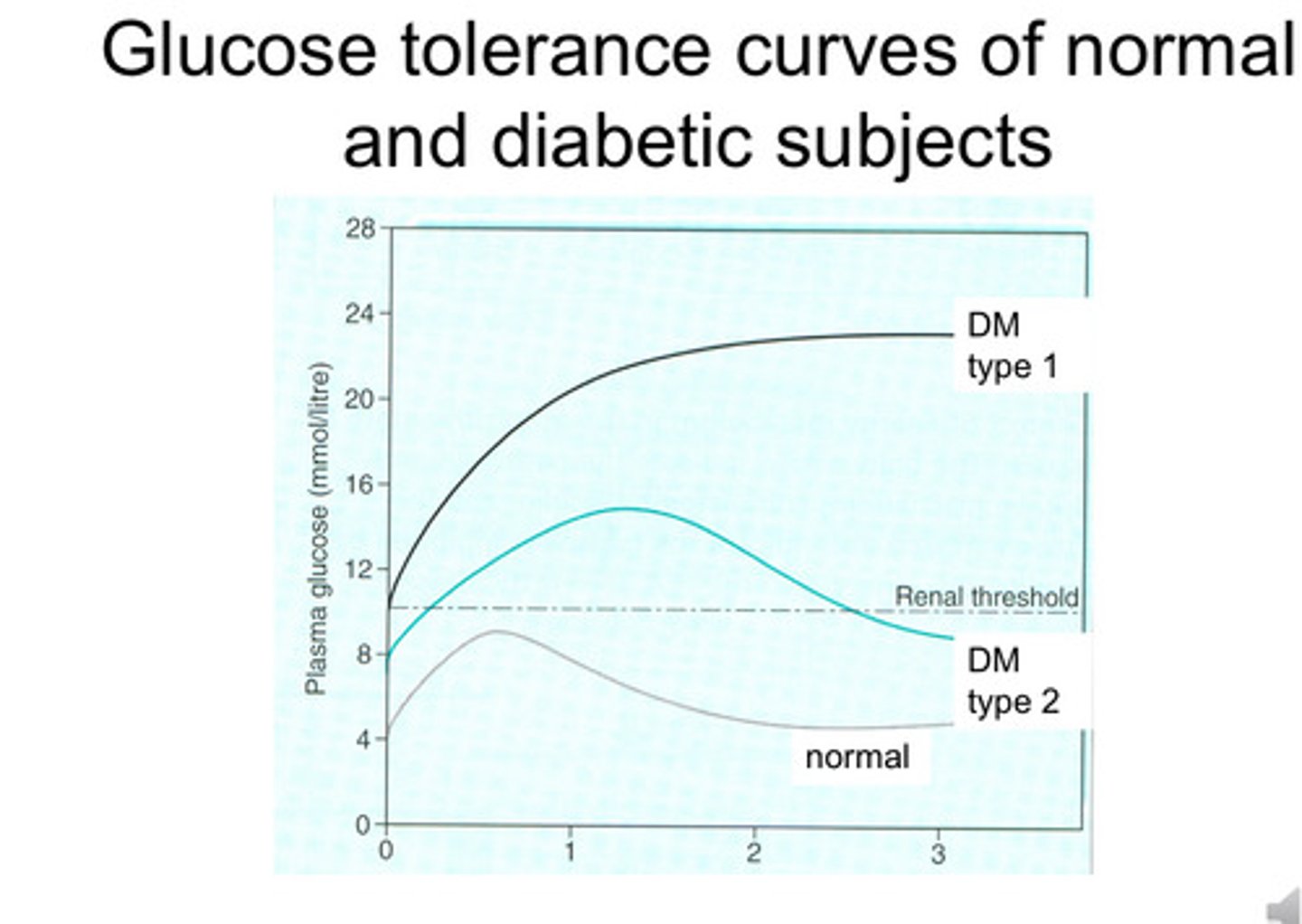
Glucose Tolerance curves of normal and diabetic subjects given glucose
Renal threshold is the conc. of glucose in the blood below which won't come out in the urine - above it will
What are the chronic complications?
- Microangiopathy: changes in walls of small blood vessels seen as thickening
of basement membrane
- Retinopathy: blindness 25x more common in patients
- Nephropathy: renal failure 17x more common
- Neuropathy: postural hypotension, impotence, foot ulcers
What are the treatments for Type I ?
- Type I: exogenous insulin via injection- it is important to balance the dosage
with amount of food to avoid hypoglycaemic incidents (most common therapy
complication)
What are the treatments for Type II?
- Type II: weight reduction, diet modification, oral hypoglycaemic agents:
biguanides (increase GLUT4 numbers) and sulphonylureas (act on Beta cell
to improve insulin secretion)
Metformin (Biguanide) - what does it do
Glucophage (brand name)
•Decreases gluconeogenesis
•Increases insulin sensitivity
•Increases GDF 15 (growth/differentiation factor 15) which reduces appetite
•May cause lactic acidosis and should not be used for patients with renal impairment
Ozempic - what does it do
•Semaglutide
•Subcutaneous or oral
•Rybelsus, Wegovy (for weight loss)
•Mimics incretin glucagon like peptide
•Increases insulin secretion
•Inhibits glucagon production and gluconeogenesis
• lowers appetite and slows down digestion
•But effects stop when administration stops
Tirzepatide
GIP/GLP-1 receptor agonist
-Slows gastric emptying
-Inhibits glucagon release
-Decreases appetite and leads to weight loss