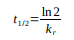CHE114 - Fundamentals of Physical Chemistry
1/150
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
151 Terms
In terms of the system–surroundings, what determines whether a system is open, closed, or isolated?
Isolated: No exchange of matter or energy
Closed: Energy can pass, matter cannot
Open: Both energy and matter can pass
In thermodynamics, what determines the state of a system?
Its macroscopic properties (e.g., pressure, volume, temperature, composition).
Do thermodynamic properties tell us how a system reached its current state?
No. Properties describe only the present state, not the path or history.
Quantities whose changes depend only on initial and final states (not the path) are called what?
State functions.
What is the difference between extensive and intensive properties?
Extensive: Depend on amount (mass, volume)
Intensive: Independent of amount (temperature, pressure, density
In the molecule and liquid energy diagrams, what types of motion contribute to internal energy U?
Translational
Rotational
Vibrational
Electronic
(In liquids) Intermolecular interactions
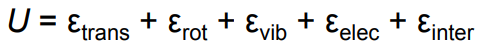
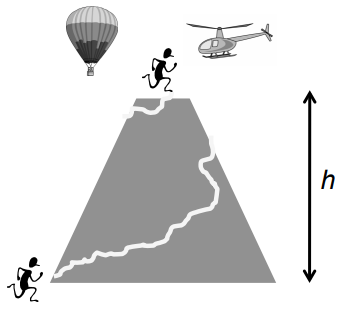
The hill-climbing diagram compares height gained to path taken. What thermodynamic quantity is this analogy used for?
Internal energy (U) — it depends only on initial and final states, not the path.
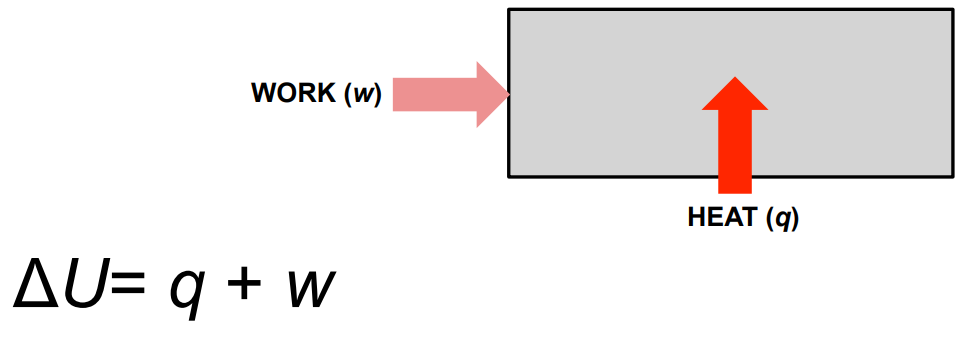
In the diagram showing HEAT (q) and WORK (w) entering a system, what equation does this represent?
First Law of Thermodynamics:
Heat and work are two ways to change internal energy.
What are diathermic and adiabatic boundaries?
Diathermic boundary: Allows heat transfer
Adiabatic boundary: Does not allow heat transfer
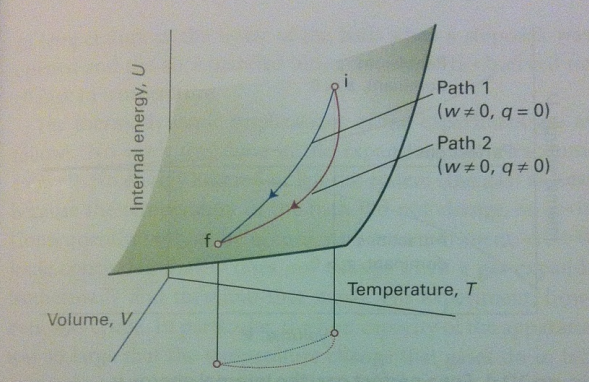
The surface diagram with two different paths leading to the same point shows what about q and w?
Heat (q) and work (w) depend on the path taken, not just the initial and final states — they are path functions.
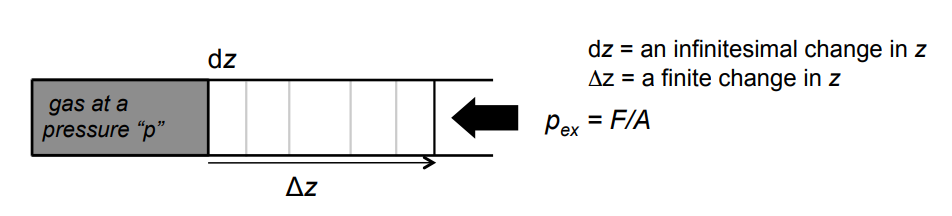
In the piston diagram, why is the infinitesimal work written as
dw=−pextdV
Because work depends on the external pressure opposing expansion and the change in volume of the gas. The negative sign shows energy leaves the system during expansion.
In the constant-pressure expansion diagram, what happens to the equation for work?
Since pext is constant:
In the reversible piston diagram, what condition must be met for expansion to be reversible?
The external pressure equals the internal gas pressure at every step:
pext=p
So dw=−p dV
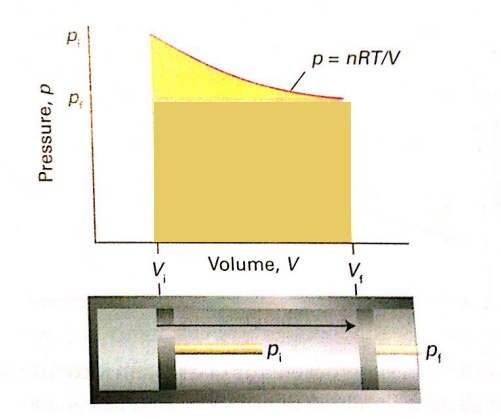
In the P–V graph, what does the area under the curve represent?
The work done during a reversible isothermal expansion. The curved area is larger than the rectangle for an irreversible expansion.
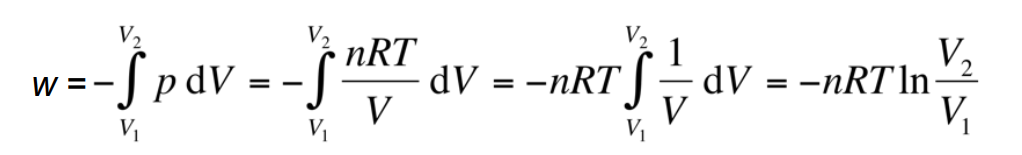
For an ideal gas, why is ΔU = 0 in the isothermal diagram?
Because internal energy depends only on temperature for an ideal gas, and temperature does not change.
If heat is supplied to the system, it must do work on the
surroundings for the temperature to remain constant.
Alternatively, if work is done on the system, it must lose heat to
the surroundings.
From the heat capacity diagram, what does CV=dU/dT mean physically?
It tells us how much the internal energy increases when temperature rises at constant volume.

What happens to internal energy at constant volume?
All heat goes into changing internal energy (no expansion work).
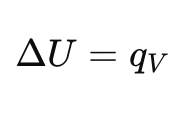
Define heat capacity at constant volume (Cv).
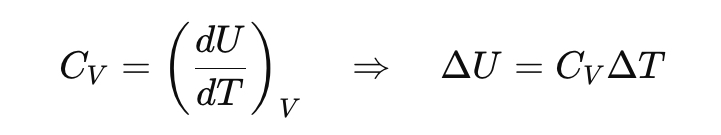
How is Cv measured experimentally?
Using constant-volume calorimetry (e.g., bomb calorimeter):
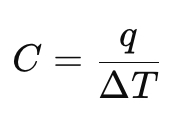
Why is internal energy (U) inconvenient in many experiments?
Because most processes occur at constant pressure, not constant volume.
Define enthalpy (H).
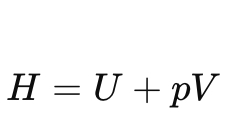
Show the differential form of enthalpy.
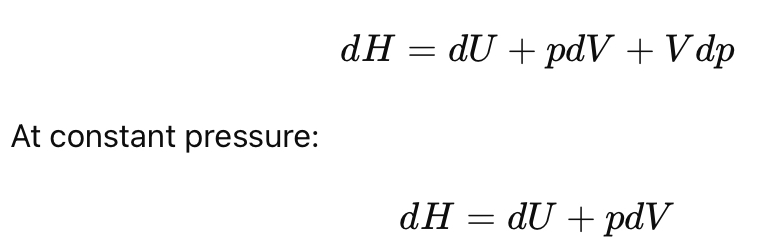
What is the key result for enthalpy at constant pressure?
Heat at constant pressure equals change in enthalpy.
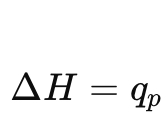
For an ideal gas reaction, how are ΔH and ΔU related?
where Δn₍g₎ is change in moles of gas.
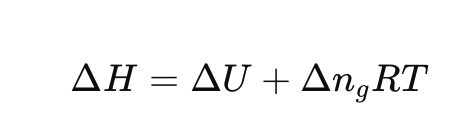
How are heats at constant pressure and volume related?
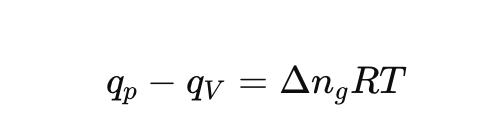
Define heat capacity at constant pressure (Cp).

Why is Cp usually larger than Cv?
At constant pressure, some heat causes expansion work, so more heat is needed for the same temperature rise.
What is the relation between Cp and Cv for an ideal gas?
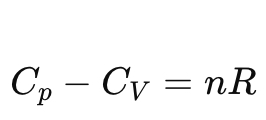
What sign of ΔH corresponds to exothermic and endothermic processes?
Exothermic: ΔH < 0
Endothermic: ΔH > 0
What is a standard state?
Pure substance at 1 bar pressure (and specified temperature).
Define standard enthalpy of fusion (ΔfusH°).
Enthalpy change when 1 mol solid → liquid at standard conditions.
Define standard enthalpy of vaporization (ΔvapH°).
Enthalpy change when 1 mol liquid → gas.
What is the relation for sublimation enthalpy?
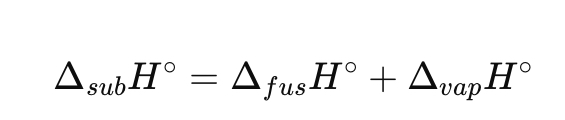
Define ionization enthalpy.
Enthalpy change when 1 mol electrons are removed from 1 mol gaseous atoms. Always positive.
Define electron affinity (conventional definition).
Enthalpy change of surroundings when 1 mol electrons attach to 1 mol gaseous atoms. Often exothermic.
Define bond dissociation enthalpy (ΔbdH°).
Enthalpy change of surroundings when 1 mol electrons attach to 1 mol gaseous atoms. Often exothermic.
Define atomization enthalpy (ΔatmH°).
Enthalpy change when all bonds in 1 mol of molecules are broken to form atoms.
What drives hydration enthalpy?
Electrostatic attraction between ions and polar water molecules. Stronger for small, highly charged ions.
Define lattice enthalpy (ΔLEH°).
Enthalpy change when 1 mol of ionic solid forms from gaseous ions. Driven by electrostatic attraction.
What is the standard enthalpy change (ΔH°)?
The enthalpy change for a process where all reactants and products are in their standard states.
What is the standard state of a substance?
Its pure form at 1 bar pressure (and usually a specified temperature).
What does ΔfusH° represent?
The standard enthalpy change when 1 mol of solid melts to form liquid.
What does ΔvapH° represent?
The standard enthalpy change when 1 mol of liquid vaporizes to gas.
How is enthalpy of sublimation related to fusion and vaporization?
ΔsubH° = ΔfusH° + ΔvapH°
What is solvation?
The process in which an ionic solid dissolves and its ions become surrounded by solvent molecules.
What two steps are involved in solvation of an ionic solid?
Lattice breakdown into gaseous ions
Hydration (or solvation) of ions
How is enthalpy of solution related to lattice and hydration enthalpies?
ΔsolH° = −ΔLEH° + ΣΔhydH°(ions)
What determines the spontaneous direction of a process?
The direction in which the entropy of the universe increases.
State the Second Law of Thermodynamics in terms of entropy.
For a spontaneous process, the total entropy of the universe increases (ΔS_total > 0).
What is Boltzmann’s entropy equation?
S = k ln W
What does W represent in S = k ln W?
The number of microstates (ways the system can be arranged).
When is entropy zero according to the Third Law?
For a perfect crystal at 0 K.
What happens to entropy when two gases mix?
Entropy increases.
Define entropy (thermodynamic definition).
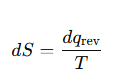
State the fundamental equation of thermodynamics.
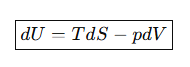
State the Clausius inequality.
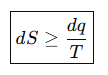
State the Second Law for an isolated system.
The entropy of an isolated system cannot decrease in a spontaneous process.
Write the entropy change of the universe.
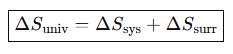
What is the criterion for spontaneity?
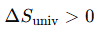
What is the entropy change of the surroundings at constant pressure?
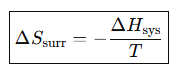
Entropy change for isothermal expansion of an ideal gas.
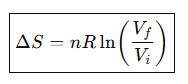
What is ΔStot for a reversible process?
0
What is ΔStot for a spontaneous irreversible process?
Positive.
Entropy change at a phase transition.
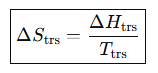
Entropy change for heating at constant pressure.
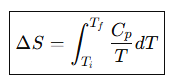
Entropy change for heating at constant pressure, Simplified form if Cp is constant.
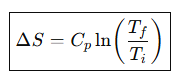
Define Helmholtz free energy.
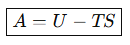
When is Helmholtz energy minimized?
Constant temperature and volume.
Why can processes with ΔS < 0 occur spontaneously?
Because ΔH < 0 increases the entropy of the surroundings.
Relationship between standard Gibbs energy and equilibrium constant.
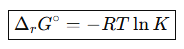
Molar Gibbs energy of an ideal gas.
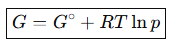
Which thermodynamic state function best determines spontaneity at constant pressure?
Gibbs free energy (ΔG)
What condition defines equilibrium in terms of Gibbs energy?
quilibrium occurs when Gibbs energy is minimized and ΔrG = 0.
What is the relationship between ΔrG° and K?
ΔrG∘=−RTlnK
What is the molar Gibbs energy of a perfect gas at pressure p?
G=G∘+RTlnp
Why does Gibbs energy depend on pressure for gases?
Because volume changes with pressure and dG=Vdp
What happens to temperature during adiabatic expansion of a perfect gas?
The temperature decreases.
Why does temperature drop during adiabatic expansion?
Work is done at the expense of internal energy, lowering molecular kinetic energy.
What is the key adiabatic relation between temperature and volume?
VTc=constant
State the Kelvin form of the Second Law of Thermodynamics.
No process can convert heat completely into work without other effects.
State the entropy form of the Second Law.
The entropy of an isolated system increases in a spontaneous process (ΔSₜₒₜ > 0).
How is entropy change defined?
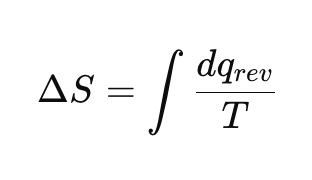
Why is entropy a state function?
Because the integral of dqrev/Tdq_{rev}/Tdqrev/T around a closed cycle is zero.
What are the four steps of the Carnot cycle?
Isothermal expansion
Adiabatic expansion
Isothermal compression
Adiabatic compression
Why is the Carnot engine the most efficient possible engine?
Because it operates reversibly between two heat reservoirs.
Why is refrigeration not spontaneous?
Because heat flows from cold to hot only with external work input.
What is the coefficient of performance (COP) of a refrigerator?
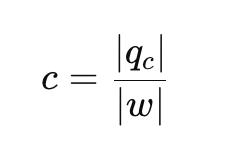
What is the maximum COP for an ideal (Carnot) refrigerator?
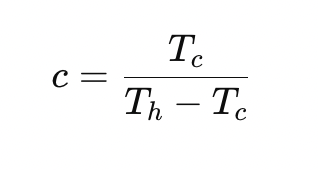
Why are real refrigerators less efficient than ideal ones?
Due to irreversibility, friction, and heat losses.

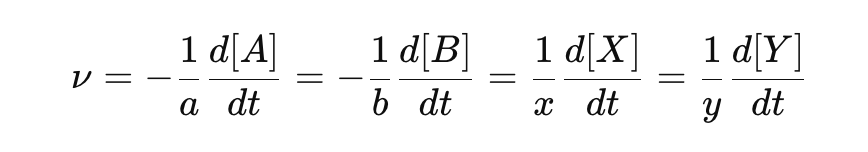
What is the rate of a chemical reaction?
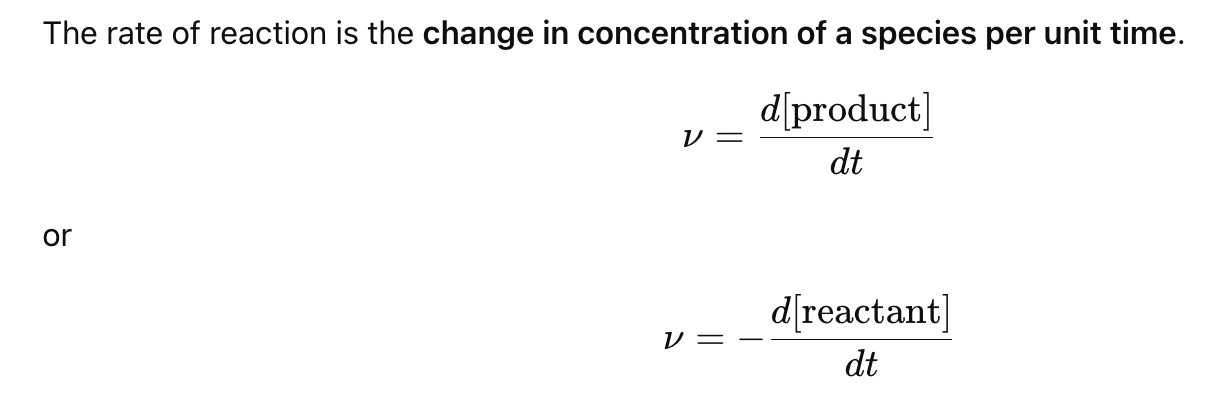
What is a rate law?
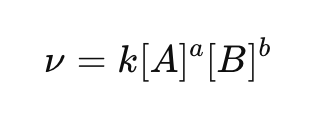
Can reaction order be predicted from stoichiometry?
❌ No
Reaction orders must be determined experimentally, except for elementary reactions.
What is molecularity?

What does the rate constant represent?
A proportionality constant relating rate to concentration.
Important facts:
depends on temperature
depends on reaction
units depend on reaction order
What is the definition of half-life t1/2?
The time required for the concentration of a reactant to decrease to half of its initial concentration.
What does a short half-life indicate?
A fast reaction.
What does a long half-life indicate?
A slow reaction.
What is the integrated rate law for a first-order reaction?
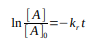
What is the half-life equation for a first-order reaction?