Chemistry: Topic 2 & 1.3
1/18
Earn XP
Description and Tags
volumetric analysis and managing chemical processes
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
limiting reagents
whichever reactant has less mols.
collision theory
reactants must collide for chemical reactions to occur.
successful collisions: particles must have sufficient energy and orientation to react and form products.
unsuccessful collisions: particles collide but bounce off each other without a reaction occurring.
activation energy
the minimum energy required for a chemical reaction to occur
when does the rate of reaction speed up
the rate of reaction will speed up if:
the frequency of collisions increases
the proportion of successful collisions increases
average rate of change formula
y2-y1/x2-x1
measures the change in quantity being measured over a defined period of time.
instantaneous rate
measure of the rate at a particular instance
represented as the slope of the tangent
tangents
steep tangent: fast rate of reaction
shallow tangent: slow rate of reaction
zero gradient: reactants have stopped converting to products.
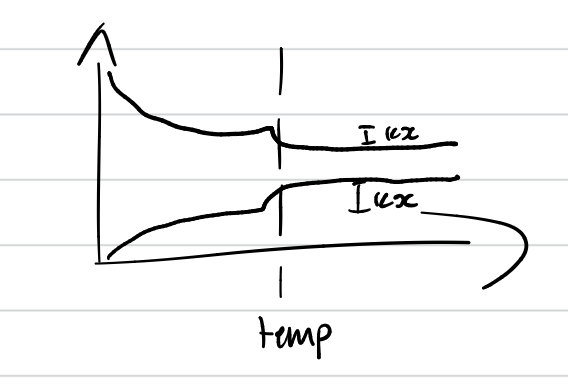
Explain the effect of higher temperatures on the rate of the chemical reaction
at a higher temperature, the reacting particles have increased average kinetic energy. this increases the number of collisions per unit time. A greater proportion of reacting particles reach or exceed the activation energy and collide in the correct orientation. This increases the number of successful collisions.
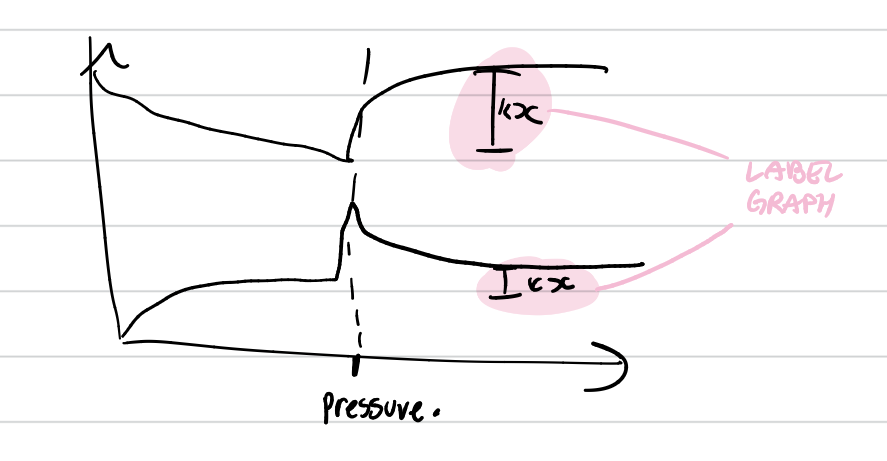
Explain the effect of higher pressure on the rate of the chemical reaction
at a higher pressure, there are more mols of gaseous particles per unit space . this increases the number of collisions per unit time. A greater proportion of reacting particles reach or exceed the activation energy and collide in the correct orientation. This increases the number of successful collisions.
Explain the effect of increased surface area on the rate of the chemical reaction
increased surface are increases the number of sittes for collision to occur. . this increases the number of collisions per unit time. A greater proportion of reacting particles reach or exceed the activation energy and collide in the correct orientation. This increases the number of successful collisions.
Equilibrium
occurs when the rate that reactants are being converted to products is the same as the rate where products are being converted to reactants.
characteristics of equilibrium
closed system: no matter is lost to surroundings
dynamic process: neither forwards or backwards reactions ever stop
rate of forwards and backwards reactions are equal
amount of reactants/products at equilibrium remains constant: no observable change.
when does the Kc value change
temperature
Le Châtliers principle
if an external change is made to the system, the change will be counteracted.
Le Châtliers (wording)
according to le châtliers principle, the system will attempt to counteract the increase in XYZ by decreasing XYZ. this favours the forwards/backwards reaction, shifting eq position to the right/left
identify change
state le chatliers
outcome
pressure graph
temperature graph
gradual increase/decrease
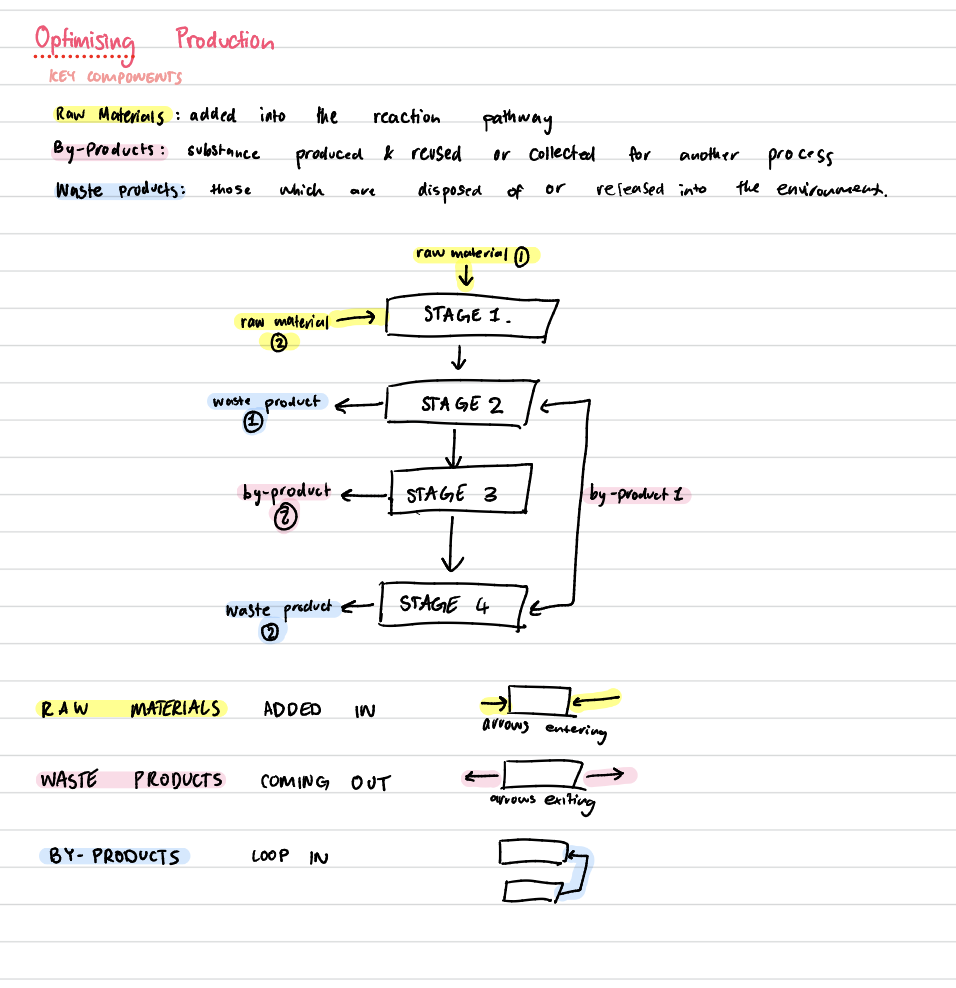
optimising production
Yield
quantity of product formed during a process, referring to the efficiency of the system.
theoretical: quantity expected from stoichiometric calculations
actual: quantity obtained through chemical process
percent: % ratio comparing actual to theoretical
%yield= actual/theoretical x100