W6, Acid-Base Imbalances
1/95
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
96 Terms
What are acids composed of?
Acids are end products of protein, carbohydrates, and fat metabolism
What must happen to acids to maintain homeostasis?
Acids (H+) must be neutralized or excreted to maintain homeostasis
What are major organs involved in acid regulation?
Bones, lungs, and kidneys
What are bases?
A negatively charged particle looking to associate with an H+
What are bases also known as?
Alkali
What ion do bases accept?
Hydrogen (H+)
True/False: Bases are end products of protein, carbohydrate, and fat metabolism
False; acids are end products of protein, carbohydrate, and fat metabolism whereas bases are negatively charged particles looking to associate with an H+
What is the carbonic acid-bicarbonate pair?
Primary buffer system in human blood that maintains pH stability at approximately 7.4
How does the carbonic acid-bicarbonate pair function?
When blood becomes too acidic (excess H+), bicarbonate (HCO3-) binds with H+ to form carbonic acid. When the blood is too basic, carbonic acid (H2CO3) dissociates into H+ and HCO3- to increase acidity
True/False: The carbonic acid-bicarbonate pair is primary buffer system in human blood that maintains pH stability at 7.4. When blood becomes too acidic (excess H+), bicarbonate (HCO3-) binds with H+ to form carbonic acid. When the blood is too basic, carbonic acid (H2CO3) dissociates into H+ and HCO3- to increase acidity
True
How is the carbonic acid-bicarbonate pair linked to respiratory system?
Carbonic acid is formed from carbon dioxide (CO2) and water (H2O) via carbonic anhydrase. Lungs regulate pH by breathing out excess CO2, which shifts equilibrium to reduce acid
What is ideal ratio of bicarbonate to carbonic acid (think carbonic acid-bicarbonate pair)?
20:1 ratio of bicarbonate to carbonic acid is vital to maintain 7.4 pH
True/False: 20:15 is the ideal ratio of bicarbonate to carbonic acid, which is vital to maintain 7.4 pH (think carbonic acid-bicarbonate pair)
False; 20:1 is ideal ratio of bicarbonate to carbonic acid, which is vital to maintain 7.4 pH
What is ideal pH in body to maintain homeostasis?
7.35 to 7.45, with an average of 7.40
What does carbonic acid-bicarbonate pair buffer system do when blood becomes too acidic (excess H+)?
Bicarbonate (HCO3-) binds with H+ to form carbonic acid, which reduces acidity in blood as free H+ ions are removed (which define acidity)
What does carbonic acid-bicarbonate pair buffer system do when blood becomes too basic?
Carbonic acid (H2CO3) dissociates into H+ and HCO3- to increase acidity
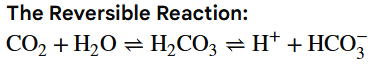
What is this?
Carbonic acid-bicarbonate pair buffer system in human blood that maintains pH stability at approximately 7.4
Does respiratory compensation (to maintain homeostasis of acids/bases) work quickly or slowly?
Quickly as it usually begins within minutes to hours of an acid-base disturbance
Does renal compensation (to maintain homeostasis of acids/bases) work quickly or slowly?
Slowly as kidneys require hours to days to fully correct acid-base imbalances
What are 2 types of compensation in maintenance acid/base balance?
Respiratory compensation (rapid, via lungs), renal compensation (slow, via kidneys)
What is major stimulus for inhalation in healthy people?
CO2 concentration in blood
True/False: Major stimulus for inhalation in healthy people is O2 concentration in blood
False; major stimulus for inhalation in healthy people is CO2 concentration in blood
What is a VERY STRONG ACID?
H+ ion
What is most significant weak base in human physiology?
HCO3-
How do lungs increase CO2 in blood?
Primarily through hypoventilation (slow/shallow breathing) or inefficient gas exchange (hypercapnia)
What is respiratory compensation?
Body’s rapid, automatic adjustment of breathing rate and depth to stabilize blood pH during metabolic acid-base imbalances. It acts within minutes to hours to regulate CO2 levels, with hyperventilation treating metabolic acidosis (by exhaling CO2) and hypoventilation treating metabolic alkalosis (by retaining CO2)
How do lungs decrease CO2 in blood?
Through a process of gas exchange in alveoli, where moves from blood capillaries into the lungs and is exhaled, and hyperventilation works too
What is renal compensation?
Slow-acting (24–72 hours) but powerful mechanism where kidneys regulate blood pH during acid-base imbalances by altering hydrogen ion (H+) excretion and bicarbonate (HCO3-) reabsorption. It specifically corrects respiratory-driven pH changes by retaining or excreting bicarbonate to restore normal blood chemistry
What happens to urine in renal compensation?
Kidneys alter urine composition to balance blood pH by producing more acidic or alkaline urine
What is relationship between serum bicarbonate levels and renal compensation?
Correlation is direct and vital for maintaining pH balance, particularly in response to respiratory acid-base disorders as kidneys compensate for respiratory issues by regulating bicarbonate retention or excretion, typically taking hours to days to reach maximal effectiveness
True/False: Increased PaCO2 (serum bicarbonate) causes kidneys to retain more bicarbonate, raising serum levels to buffer excess acid whereas decreased PaCO2 causes kidneys to increase bicarbonate excretion, lowering serum levels to reduce pH
True (this is relationship between renal compensation of acids/bases and serum bicarbonate levels)
True/False: Renal compensation is quicker to respond to pH changes than lungs
False; renal compensation is slower to respond to pH changes than lungs
What does ABGs stand for?
Arterial blood gases
What are ABGs (arterial blood gases)?
Concentrations of oxygen and carbon dioxide in bloodstream
Which arterial blood gas is involved in calculation of blood pH and HCO3- concentration?
Carbon dioxide
True/False: Oxygen is the arterial blood gas involved in calculation of blood pH and HCO3- concentration
False; carbon dioxide is the arterial blood gas involved in calculation of blood pH and HCO3- concentration
What is normal arterial blood pH?
7.35 to 7.45
Which test is used to obtain normal arterial blood pH?
Arterial blood gas (ABG) sampling
What is acidosis?
Increase in H+ concentration or decrease in bicarbonate (base)
What is alkalosis?
Decrease in H+ concentration or increase in bicarbonate

What are values in #1?
7.35 to 7.45

What are values in #2?
80 to 100

What are values in #3?
35 to 45

What are values in #4?
22 to 26

What are values in #5?
95% to 100%
What are 2 types of acidosis?
Metabolic acidosis, respiratory acidosis
What causes metabolic acidosis?
Decrease in bicarbonate
What causes respiratory acidosis?
Increase in carbonic acid
What are 2 types of alkalosis?
Metabolic alkalosis, respiratory alkalosis
What causes respiratory alkalosis?
Decrease in carbonic acid
What causes metabolic alkalosis?
Increase in bicarbonate
What is etiology?
Cause or set of causes of a disease or condition
What is etiology of respiratory acidosis?
Drugs, edema (pulmonary), pneumonia, respiratory damage, emboli (pulmonary), spasm like a bronchospasm causing airway obstruction
What type of drugs could cause respiratory acidosis?
Opioids, recreational drugs
What is example of respiratory damage that could cause respiratory acidosis?
COPD
What is mnemonic for etiology of respiratory acidosis?
DEPRESS (for drugs, pulmonary edema, pneumonia, respiratory damage, pulmonary emboli, spasm like a bronchospasm causing airway obstruction)
What are arterial blood gas (ABG) values in respiratory acidosis (used to aid in diagnosis too)?
Decrease pH, increased PaCO2
What are clinical manifestations of respiratory acidosis?
Headache, confusion, dyspnea, shallow respirations, tachycardia, flushed skin
What do COPD patient have high amounts of in their lungs?
Have high CO2 in their lungs as their lungs cannot get rid of CO2 adequately
Why are COPD patients hypercapnic and thus susceptible to acidosis?
COPD patients have high CO2 in their lungs as their lungs cannot get rid of CO2 adequately, and CO2 lowers pH levels in body (making them acidic)
True/False: Patients with COPD have high CO2 in their lungs as their lungs cannot get rid of CO2 adequately, so COPD patients are hypercapnic and are susceptible to acidosis. H+ accumulates and respiratory acidosis can easily develop
True
What are signs and symptoms of respiratory acidosis?
Hypoventilation leading to hypoxia, rapid shallow respirations, decreased BP, skin/mucosa pale to cyanotic, headache is big sign, hyperkalemia, dysrhythmias (due to increased K+), drowsiness, dizziness, disorientation, muscle weakness, hyperreflexia
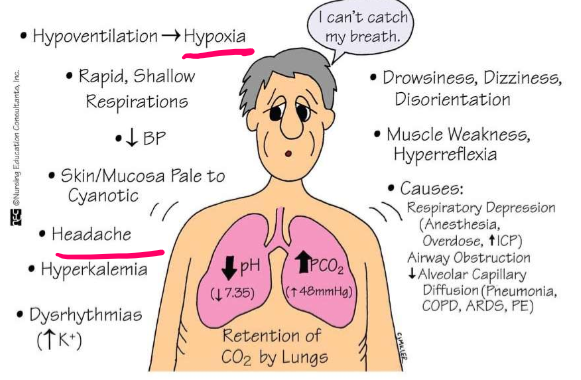
What condition is displayed by signs and symptoms shown here?
Respiratory acidosis
What could cause respiratory acidosis?
Respiratory depression (due to anesthesia, overdose, increased ICP), or airway obstruction which leads to decreased alveolar diffusion (pneumonia, COPD, ARDS, PE)
What are arterial blood gas (ABG) values in respiratory alkalosis (used to aid in diagnosis too)?
pH greater than 7.45 and PaCO2 below 35 mmHg
What is etiology of respiratory alkalosis?
Excessive ventilation (breathing fast) due to anxiety or pain, fever, hypoxemia, mechanical ventilation
True/False: Respiratory alkalosis is pretty common
False; respiratory alkalosis is not very common (unless you’re working with lots of ventilators)
What are clinical manifestations of respiratory alkalosis?
Lightheadedness, tingling, muscle cramps, tachycardia, anxiety
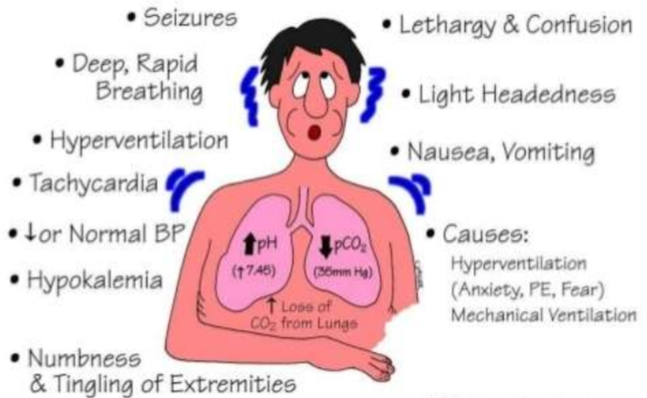
What condition is displayed by signs and symptoms shown here?
Respiratory alkalosis
What are signs and symptoms of respiratory alkalosis?
Seizures, deep rapid breathing, hyperventilation, tachycardia, low or normal BP, hypokalemia, numbness and tingling of extremities, lethargy and confusion, lightheadedness, nausea and vomiting, red and flushed in color (not cyanotic)
What are causes of respiratory alkalosis?
Hyperventilation due to anxiety, PE or fear as well as mechanical ventilation
What is main difference between metabolic and respiratory acidosis/alkalosis?
In metabolic acidosis/alkalosis, stem of problem is somewhere other than lungs, whereas in respiratory acidosis/alkalosis, problem is due to lungs and breathing
What are arterial blood gas (ABG) values in metabolic acidosis (used to aid in diagnosis too)?
pH below 7.35 and HCO3- less than 22 mEq/L
What is etiology of metabolic acidosis?
Diabetic ketoacidosis (DKA) so pancreas is not working, renal failure, lactic acidosis or sepsis, diarrhea, toxin ingestion
How could diarrhea cause metabolic acidosis?
Excessive diarrhea drains base (HCO3-) from intestines, breaking body's buffering system (carbonic acid-bicarbonate pair)
What are clinical manifestations of metabolic acidosis?
Kussmaul respirations, fatigue, headache, hypotension, confusion
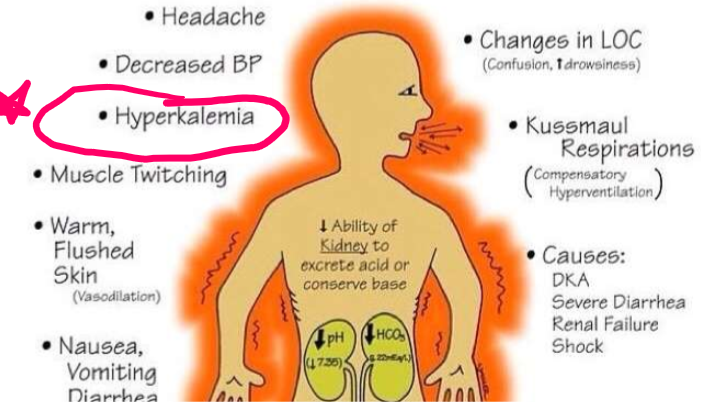
What condition is displayed by signs and symptoms shown here?
Metabolic acidosis
What are signs and symptoms of metabolic acidosis?
Headache, decreased BP, hyperkalemia especially, muscle twitching, warm flushed skin (vasodilation), nausea, vomiting, diarrhea, changes in LOC (confusion, increase drowsiness), Kussmaul respirations (compensatory hyperventilation)
What are causes of metabolic acidosis?
DKA, severe diarrhea, renal failure, shock
Why is hyperkalemia a big sign of metabolic acidosis?
When increased H+ in blood goes into cells, this H+ pushes K+ out of cells, which leads to hyperkalemia (increased K+) in bloodstream
What are arterial blood gas (ABG) values in metabolic alkalosis (used to aid in diagnosis too)?
pH above 7.45 and HCO3- greater than 26 mEq/L
What is etiology of metabolic alkalosis?
Excess bicarbonate from too many antacids or diuretics, acid loss from excessive vomiting, suctioning
True/False: Metabolic alkalosis is extremely common
False; metabolic alkalosis is not very common finding and is very rare
What are clinical manifestations of metabolic alkalosis?
Muscle weakness, cramps, tremors, confusion
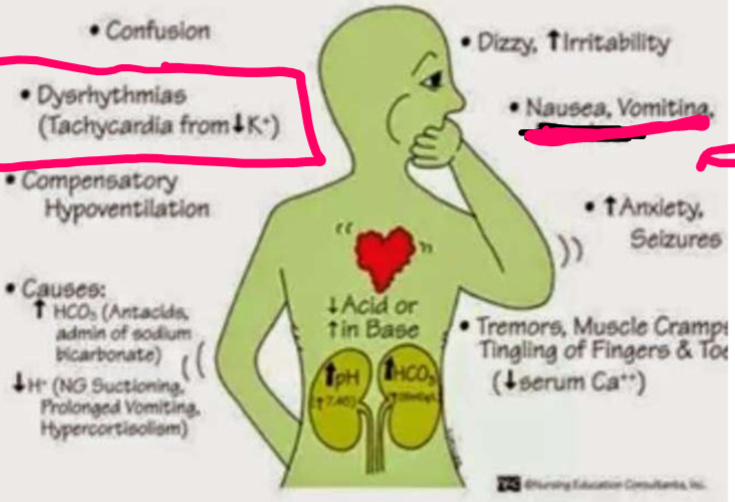
What condition is displayed by signs and symptoms shown here?
Metabolic alkalosis
What are signs and symptoms of metabolic alkalosis?
Confusion, dysrhythmias (tachycardia from decreased K+), compensatory hypoventilation, dizzy, increased irritability, nausea, vomiting, increased anxiety, seizures, tremors, muscle cramps, tingling of fingers and toes (due to decreased serum Ca2+)
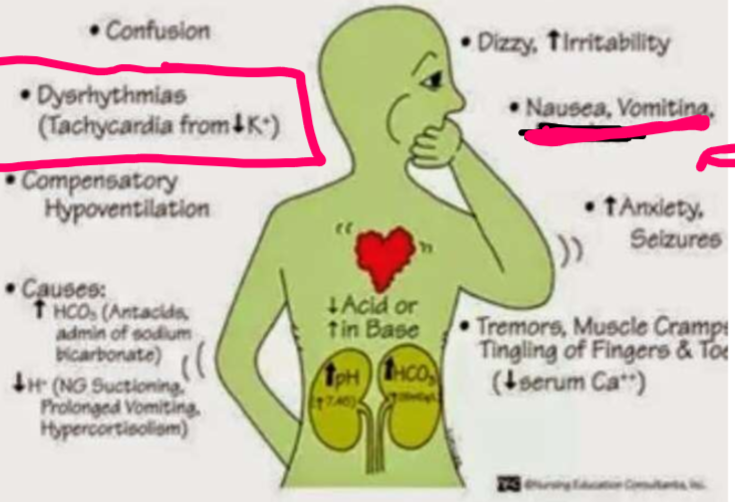
Why is metabolic alkalosis patient green?
Due to nausea and vomiting
What is big sign and symptom of metabolic alkalosis?
Dysrhythmias like tachycardia from decreased K+
What are causes of metabolic alkalosis?
Increased HCO3- due to antacids or administration of sodium bicarbonate, decreased H+ due to NG suctioning, prolonged vomiting, or hypercortisolism
An anxiety attack often causes hyperventilation leading to what?
Respiratory alkalosis
How do you interpret arterial blood gases (ABGs)?
Determine whether pH is acidic or alkalotic, determine whether CO2 is acidic or alkalotic, determine whether HCO3- is acidic or alkalotic, match pH (acidic A or alkalotic B) with other similar value (either CO2 or HCO3-), name the blood gas, determine next steps
What is compensation?
When body tries to return to normal (and maintain homeostasis)
True/False: Bicarbonate indicates metabolic problem whereas CO2 indicates respiratory problem
True
What is ABG interpretation of pH 7.22, PaCO2 55, and HCO3- 25?
pH is acidic, PaCO2 is acidic, HCO3- is neutral; so name is respiratory acidosis
What is ABG interpretation of pH 7.50, PaCO2 42, and HCO3- 33?
pH is basic, PaCO2 is neutral, HCO3- is basic; so name is metabolic acidosis
True/False: When interpreting ABGs (arterial blood gases), start with pH every time to determine acid-base imbalance
True, then determine if PaCO2 and HCO3- are acidic, basic, or neutral to determine and name problem