acid base week 2
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
levelling effect
all strong acids and bases are equally strong in water
strongest acid and base possible in water are
H3O+ and OH-
at low pH/high [H3O+], weak acids and bases are pushed towards
their protonated forms
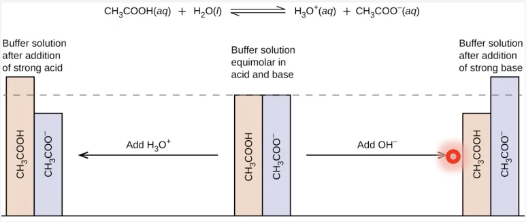
acid-base buffer
a solution that lessens the impact of addition of acid or base on pH
buffer usually consists of
conjugate acid-base pair where both species are present in appreciable quantities in solution
buffer components HA and A- are able to
consume small amounts of added OH- or H3O+ by shifting equilibrium position
buffer capacity
measure of the strength/ability to maintain pH (want high concentrations of each component and them to be closer to each other
buffer range
pKa ± 1
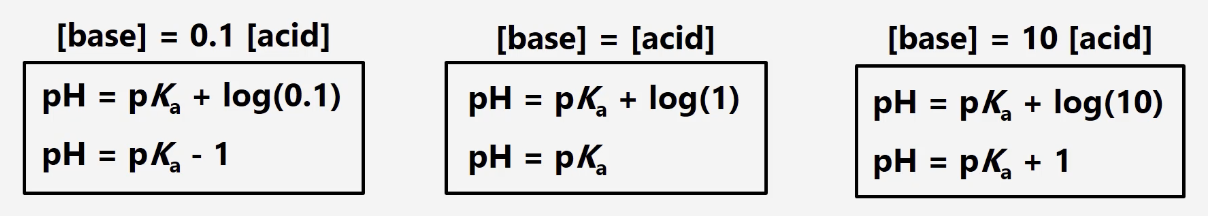
pKa of the weak acid should be close to
desired pH
preparing buffers
choose conjugate acid/base pair, calculate ratio of buffer component concs, determine buffer conc and volume of stock solutions, mix solution and correct pH if needed
acid-base titration
determine concentration of an acid/base with a solution of base/acid of known concentration, utilizing a visible indicator
acid-base indicator
weak organic acid whose color differs from that of its conjugate base
ratio of [Hin]/[In-] depends on
[H3O+] i.e. changing pH makes one conc up one conc down, changing color
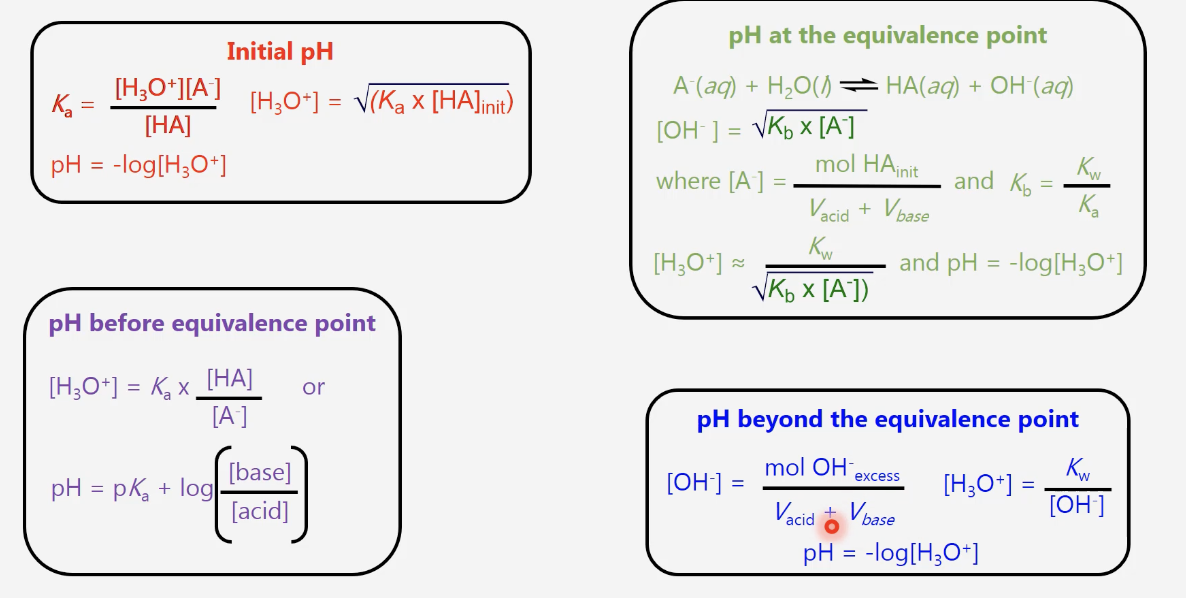
strong acid-strong base titration case
initial pH is low, increases gradually until base and acid are equimolar and the pH rises very rapidly at this equivalence point (pH = 7 for this case), pH afterward depends on excess of titrant and increases slowly

weak acid-strong base titration
initial pH is higher than strong-strong, increases gradually throughout buffer region(weak acid ←→conjugate base), equivalence point pH is higher than 7 due to reaction of conjugate base with H2O