Organic Chemistry Chapter 19
1/80
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
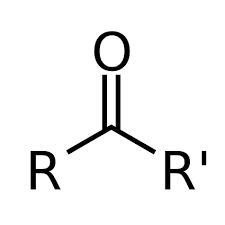
aldehyde
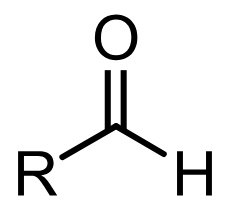
ketone
naming aldehydes ending & numbering
-e becomes -al
C=O carbon is always numbered 1
naming ketones ending & numbering
-e becomes -one
C=O carbon given lowest number (doesn’t have to be 1)
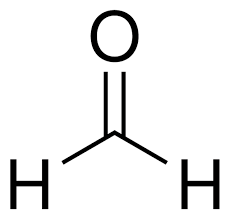
formaldehyde
acetaldehyde
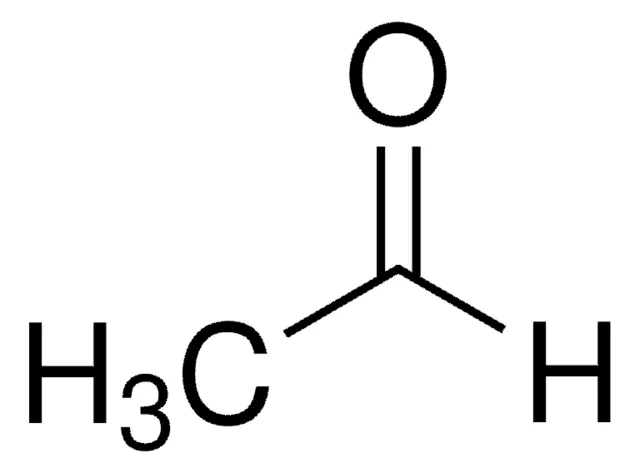
benzaldehyde
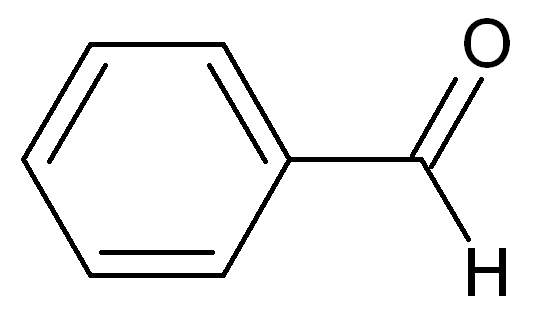
acetone
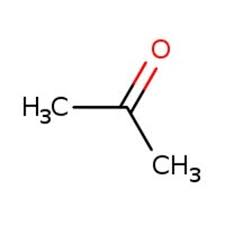
acetophenone
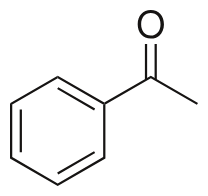
benzophenone
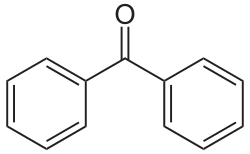
name for aldehyde next to a ring
carbaldehyde
(entire molecule is cyclo___carbaldehyde)
3 methods of preparing aldehydes
oxidation
ozonolysis
hydroboration-oxidation of terminal alkenes
preparing aldehydes oxidation what it looks like
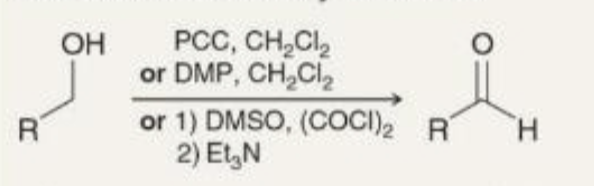
preparing aldehydes oxidation reagents
PCC, CH2Cl2
OR
DMP, CH2Cl2
OR
1)DMSO, (COCl)2
2) Et3N
preparing aldehydes ozonolysis what it looks like
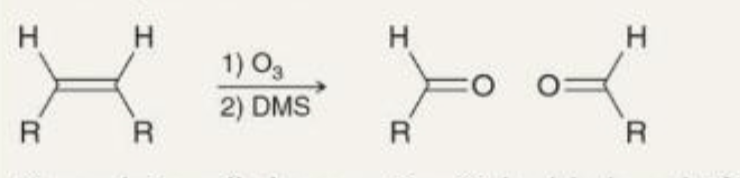
preparing aldehydes ozonolysis reagents
1)O3
2)DMS
preparing aldehydes hydroboration-oxidation what it looks like
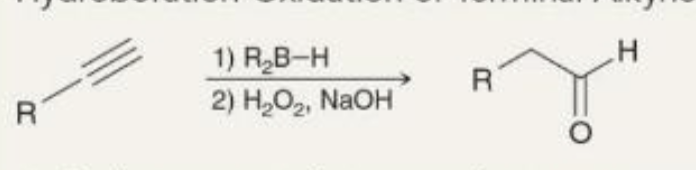
preparing aldehydes hydroboration-oxidation reagents
1) R2BH
2) H2O2, NaOH
4 methods of preparing ketones
oxidation of secondary alcohols
ozonolysis
acid-catalyzed hydration
friedel-crafts acylation
preparing ketones oxidation of secondary alcohols what it looks like
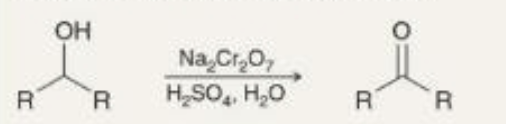
preparing ketones oxidation of secondary alcohols reagents
Na2Cr2O7
H2SO4, H2O
preparing ketones ozonolysis what it looks like
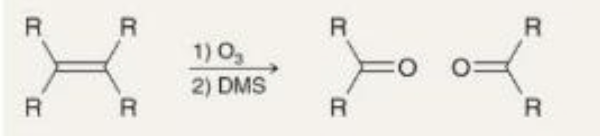
preparing ketones ozonolysis reagents
1) O3
2) DMS
preparing ketones acid-catalyzed hydration what it looks like
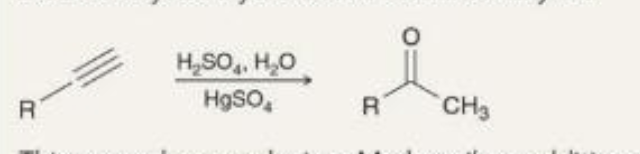
preparing ketones acid-catalyzed hydration reagents
H2SO4, H2O
HgSO4
preparing ketones friedel-crafts acylation what it looks like
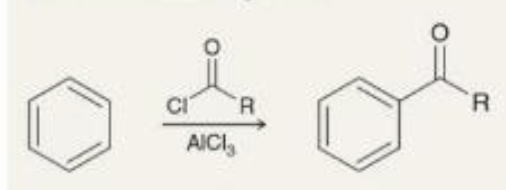
preparing ketones friedel-crafts acylation reagents
ClCOR (carbonyl w Cl & R coming off)
AlCl3
what allows nucleophilic addition reaction to occur on aldehydes/ketones
carbonyl carbon is electrophilic due to resonance and induction so can be attacked by nucleophiles
are aldehydes or ketones more reactive for nucleophilic addition reactions and why
aldehydes - less sterically hindered and larger partial positive charge on carbonyl (less stabilized)
nucleophilic addition reactions with strong nucleophiles conditions
basic
nucleophilic addition reactions basic conditions mechanism
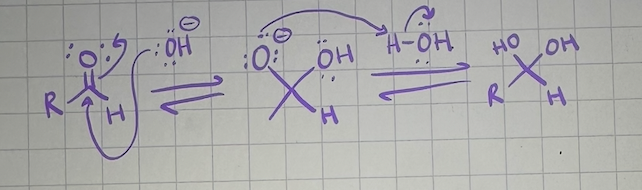
nucleophilic addition reactions with weak nucleophiles conditions
acidic
nucleophilic addition reactions acidic conditions mechanism
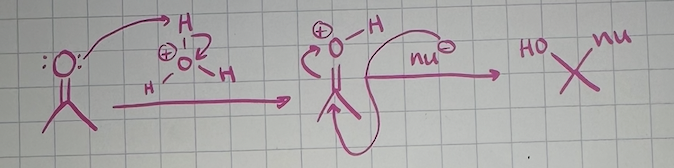
nuc addition to C=O equilibrium favors reactants when
nucleophile can function as a good leaving group
nuc addition to C=O equilibrium favors products when
nucleophile is a bad leaving group
ketone/aldehyde hydrate looks like
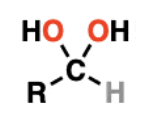
speed of ketone/aldehyde to hydrate reaction
slow unless using acidic or basic conditions
when is hydrate vs. reactants favored at equilibrium
hydrate is favored only with simple aldehydes as reagent, otherwise reactants are favored
hydrate formation under basic conditions mechanism
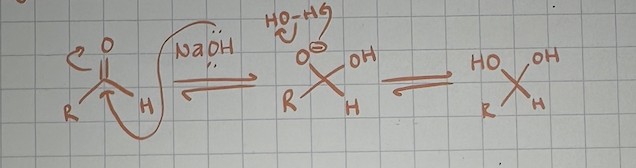
hydrate formation under acidic conditions mechanism (I put in the reverse reaction for some reason so pretend it’s flipped)
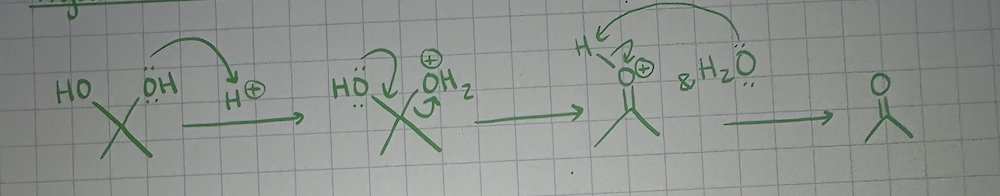
reagents for ketone/aldehyde to form hydrate
ketone/aldehyde & H2O
acetal/ketal what it looks like
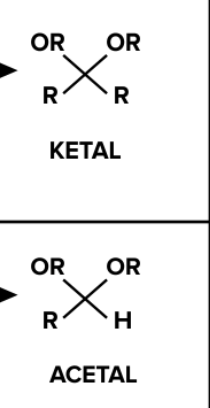
reagents for aldehyde-acetal/ketone-ketal
aldehyde/ketone and 2 alcohols
commonly used acid catalysts for acetal/ketal formation
TsOH (p-toluenesulfonic acid)
sulfuric acid
mechanism of acetal formation (acidic conditions)
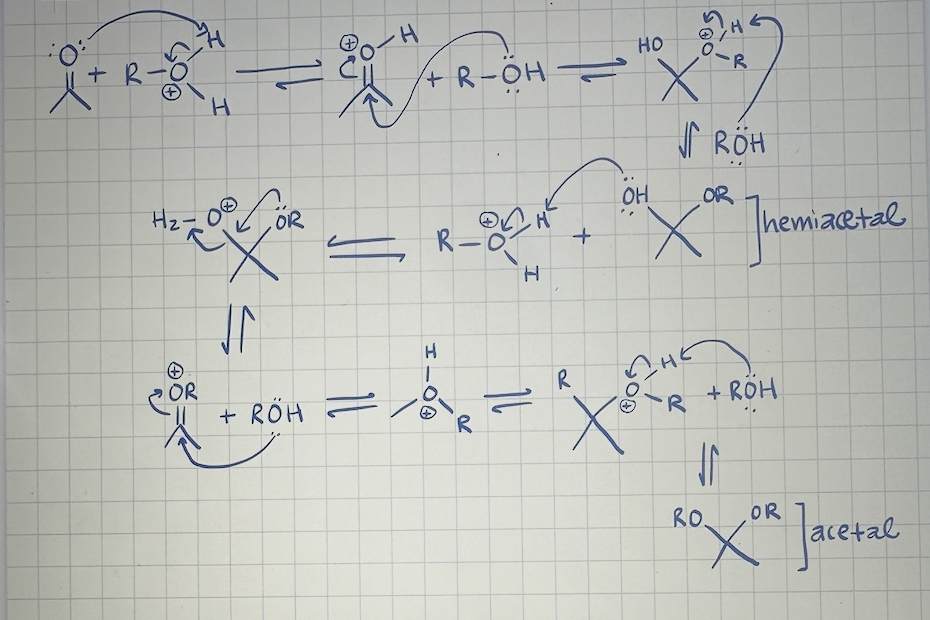
acetal formation if a diol is used instead of 2 separate alcohol molecules
forms cyclic acetal
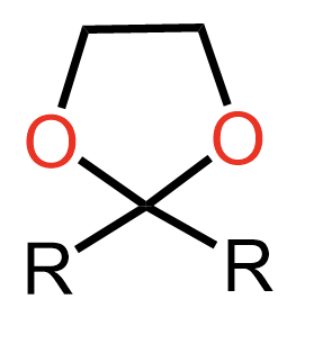
benefit of forming cyclic acetals
can usually isolate cyclic hemiacetal but not regular hemiacetal
reagents to convert acetal back to aldehyde/ketone
H2O, H+
how to favor acetal formation from aldehyde/ketone
remove H2O
common acetal synthesis use
as protecting group
acetal as protecting group example
usually would use LAH (LiAlH4) to remove carbonyl from ring, but this would also remove the ketone, which we don’t want. If an acetal is formed first, LAH can be used to remove the carbonyl, and then add H3O+ to get desired product.
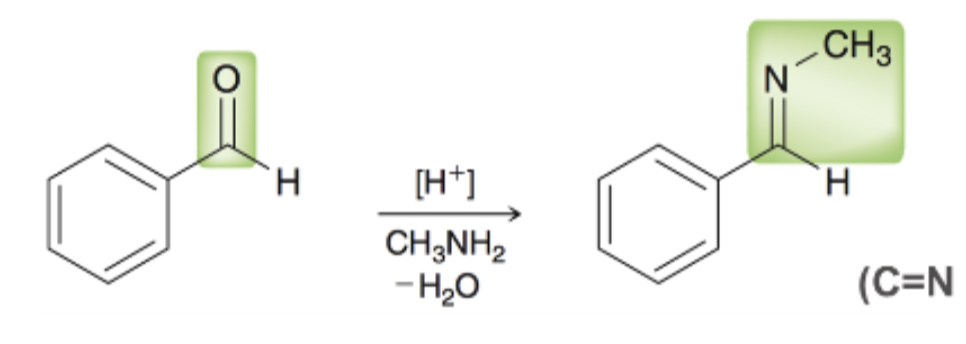
reagents to form imine
ald/ket, primary amine, acidic conditions
[H+], CH3NH2, -H2O
formation of imine what it looks like
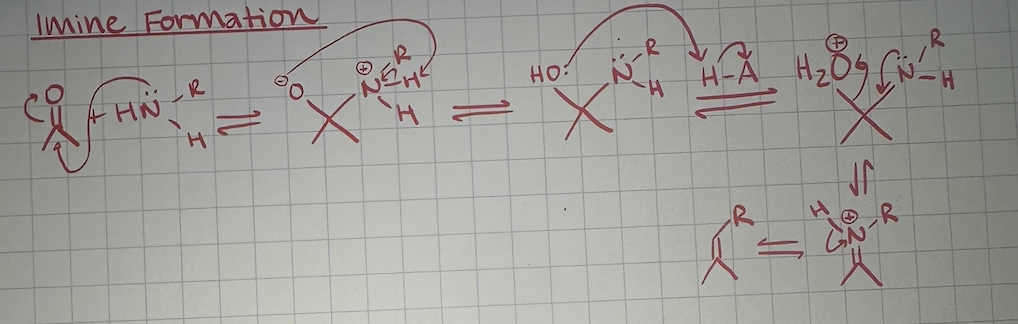
imine formation mechanism
hydrazones
imine analogs from hydrazines, middling stability
oximes
imine analogs from hydroxylamines, most stable
reagents for enamine formation
ald/ket, secondary amine, acidic conditions
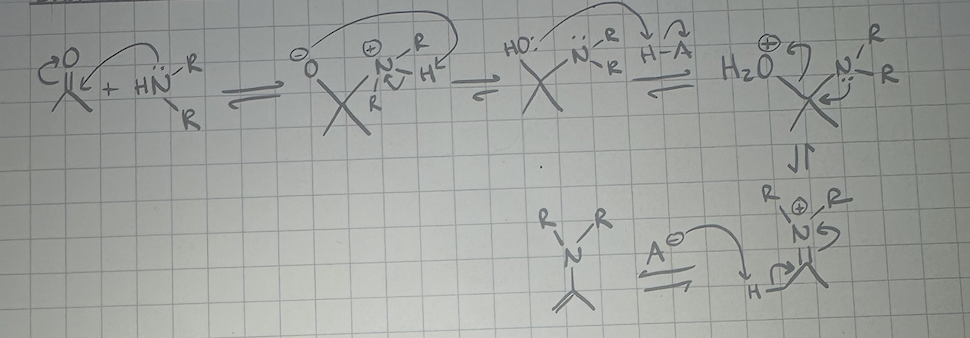
enamine formation mechanism
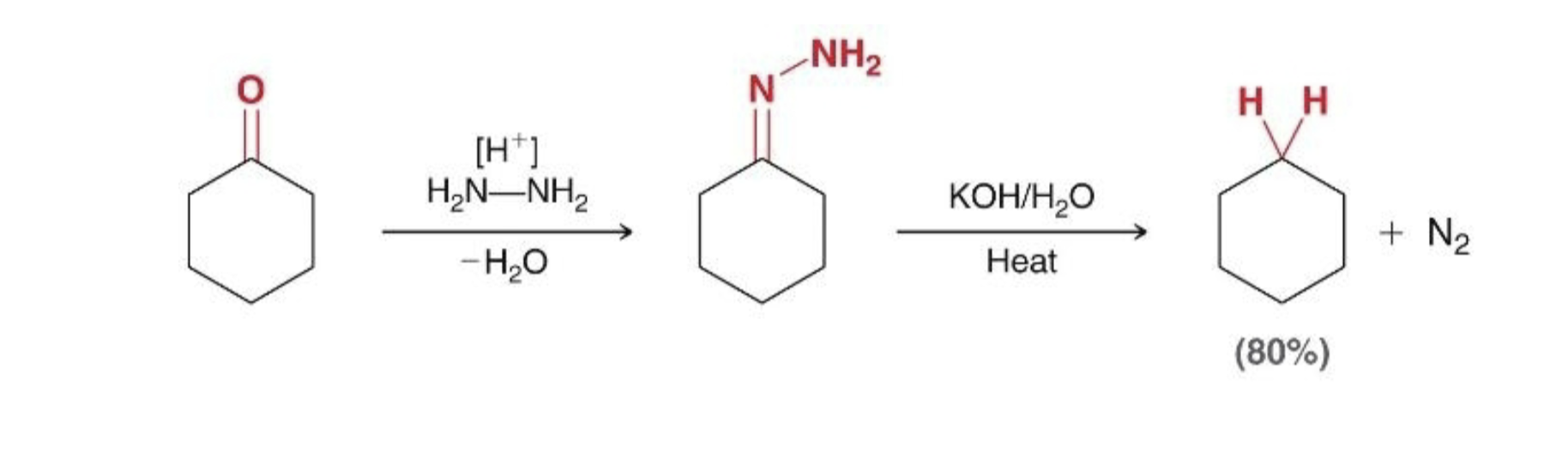
what is wolff-kirschner reduction what it does & its steps
2 step synthesis to reduce ketone to alkane
1) form hydrazine
2) elimination
reagents for wolff-kirscher reduction
first reduction: H+, NH2-NH2, subtract H2O
second reduction: KOH/H2O, heat
wolff-kirscher reduction what it looks like
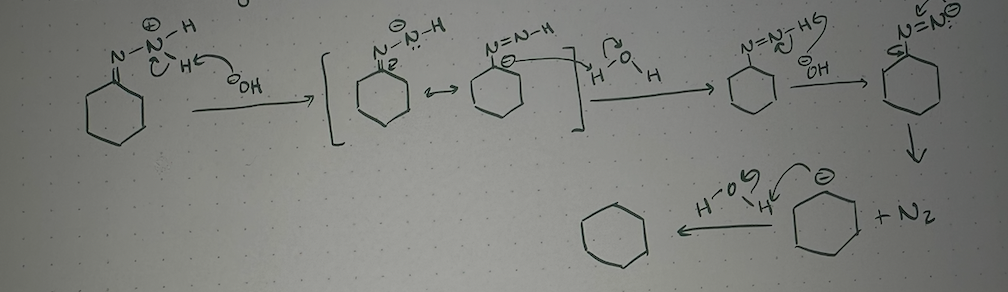
wolff-kirschner mechanism
hydrolysis of acetals what it does
acetal and aqueous acid is reduced to a ketone/aldehyde and 2 eqs of alcohol (is the reverse of acetal formation)
hydrolysis of acetals what it looks like
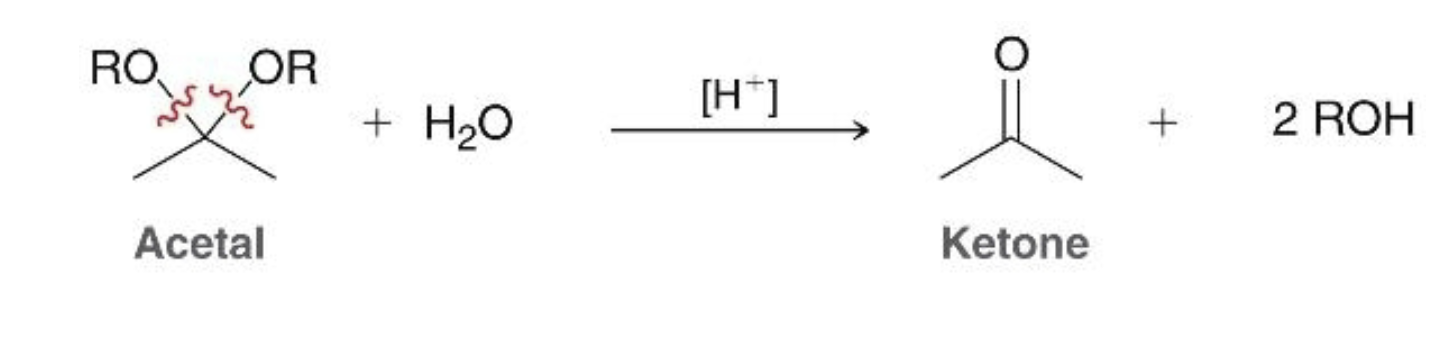
conditions required for acetal hydrolysis
acidic (no reaction will occur under basic conditions!)
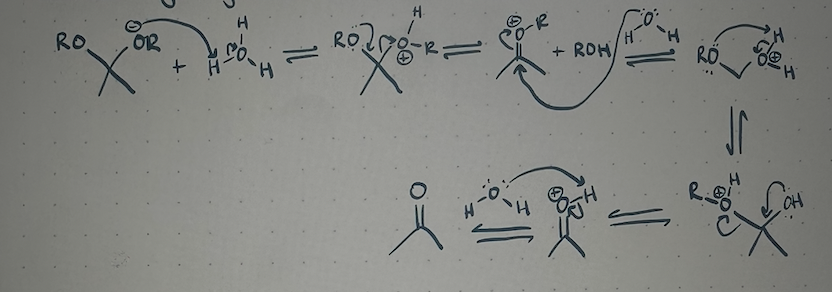
acetal hydrolysis mechanism
imine & enamine hydrolysis what it looks like
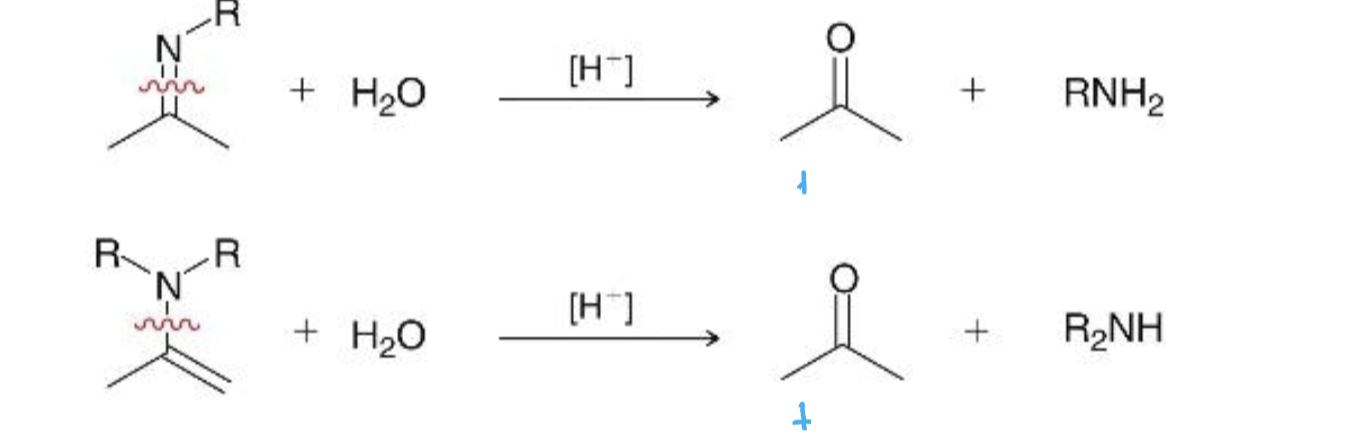
hydrolysis reagents
acetal/imine/enamine & H2O, +H
imine & enamine hydrolysis mechanism
reverse of their formation, similar to acetal hydrolysis (didn’t draw in class)
3 methods of reducing ketone/aldehyde to alkane
using raney nickel
wolff-kirschner reduction
clemmenson reduction
clemmenson reduction (ch 18 review) and conditions/limitations
get rid of carbonyl
uses Zn(Hg) & H2O
takes place in strong acidic conditions
can’t use with cmps sensitive to acid
wolff-kirscher reduction conditions/limitations
needs strong basic conditions
sensitive to bases
can’t use with cmps
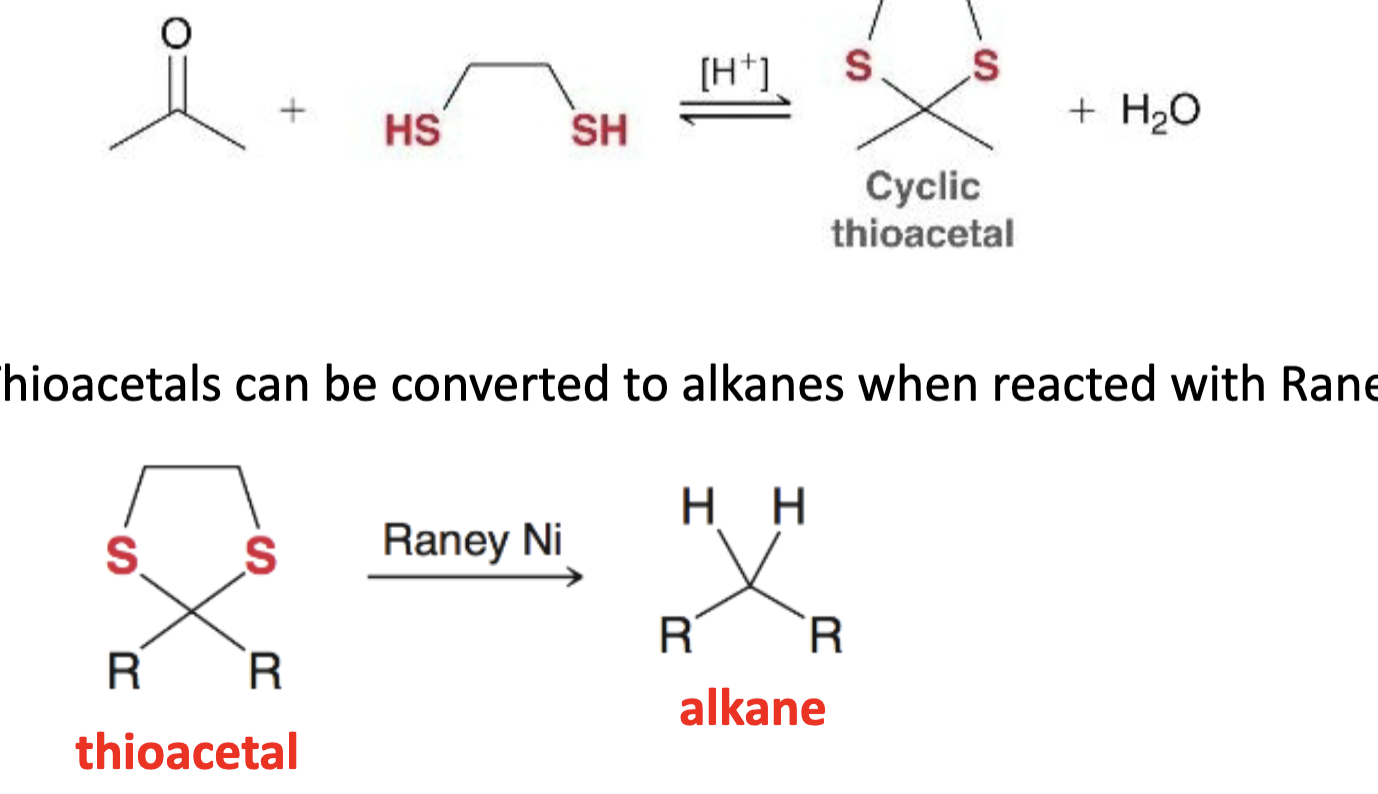
ketone/aldehyde to alkane reduction w raney nickel what it looks like
reaction of ket/ald with thiol what it looks like
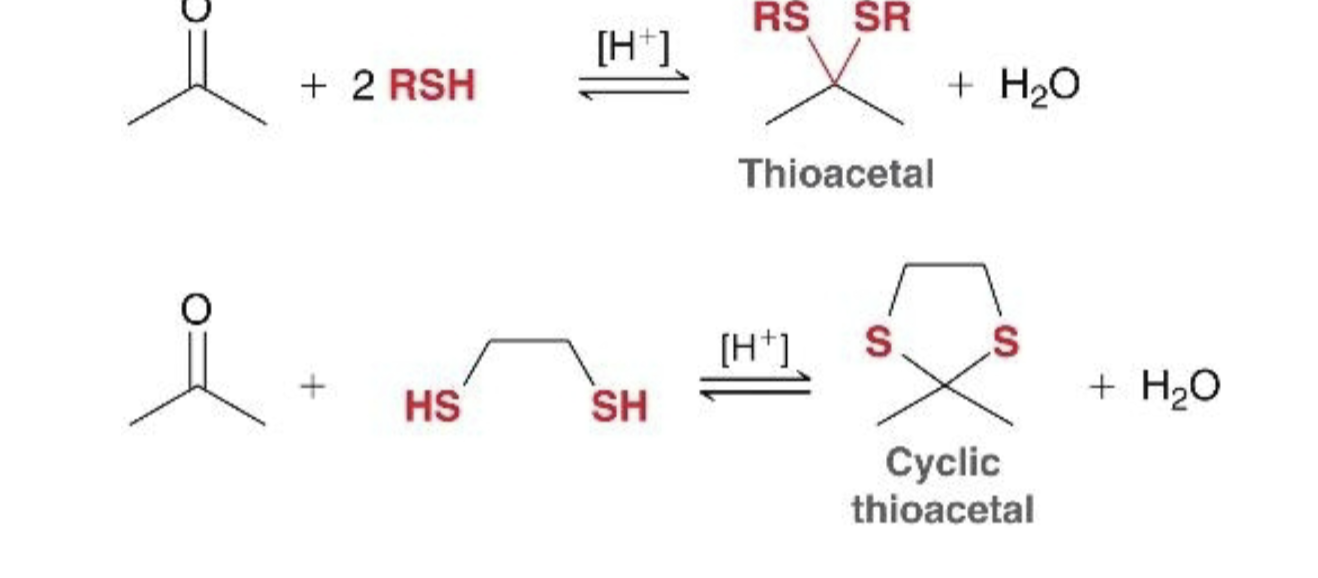
reagents for ket/ald thiol rxn
H+, ket/ald reacts with 2RSH to make thioacetal, reacts with SH(CH2)2SH to make cyclic thioacetal
reagent for cyclic thioacetal to alkane
raney nickel
reagents for ket/ald to alkane w raney nickel
1) H+, HS(CH2)2SH
2) raney nickel
ket/ald to alkane w raney nickel limitations
difficult lab workup
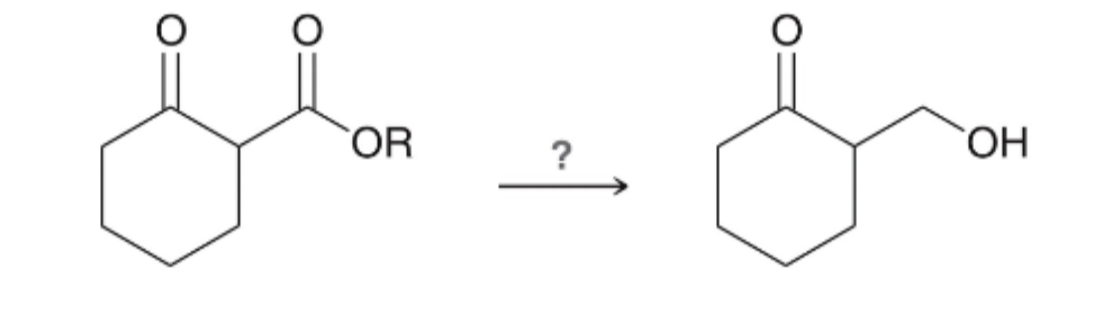
reagents for ald/ket to alcohol
hydride reagent:
1) LiAlH4
2) H2O
OR
NaBH4, MeOH
reduction of asymmetrical ket to alcohol forms…
new chiral center
ket/ald to alcohol mechanism