1. // S1.1 Models of the particulate nature of matter
1/74
Earn XP
Description and Tags
chem from lynn
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
Mixtures
Components may be elements or compounds.
Are not chemically bonded together and not in a fixed ratio.
Retain their individual properties.
Homogeneous: all the components of a mixture are in the same phase. e.g. Air
Heterogeneous: the components of a mixture are in different phases.
Exothermic
Reaction releases energy
Endothermic
Eeaction takes up/ requires energy
electron affinity
mininum energy released when 1 mole of e- is added to 1 mol of atoms (g)
ionization energy
minumun energy needed to remove 1 mol of e- from 1 mol of atoms (g)
ionic radious
radious of an ion, this is different from atomic radious due to change in amout of electrons, positive ions usually get smaller, negative bigger
Absorbance
to take light in and not reflect or refract it
Precision
a measure of how close a series of measurements are to one another

accuracy
how close a measurement is to the true value

analog measurements
analog measurements
manual measuring, measurement where you Estimate what the device says.
- uncertainty is half of the smallest increment
digital devices
round +/- 1 to smallest increment (e.g. analytical scale, digi thermostat)
uncertainty
an estimate of how much a measured or calculated value differs from a true value
absolute uncertainty
uncertainty associated with a measurement
Percentage uncertainty
The uncertainty given as a percentage of the measurement taken (absolute uncertainty/ measurment x 100)
structural formula
a formula that shows the arrangement of atoms in the molecule of a compound
deposition
gas to solid
Sublimination
solid to gas
Vaporization
Liquid to gas
condensation
Gas to liquid
melting
solid to liquid
freezing
liquid to solid
solid
defined shape and volume, low Ek
liquid
flexible shape that adjusts to container, fixed volume, middle Ek
gas
moves freely and adjusts to volume of container, high Ek
Kinetic Molecular Theory
All matter is made up of particles that are constantly moving.
The energy of the particles depends upon the absolute temperature.
A change of state may occur when the energy of the particles is changed.
The average amount of empty space between particles generally increases as matter moves from the solid to the liquid and gas phases.
There are forces of attraction between particles which become stronger as the particles move closer together.
solvation
seperation method: dissolving a mixture in a soluvent
-based on solubility, less soluble substance stays undissolved longer
Filtration
seperation method: a solid can be saparated from a liquid using a filter (filter paper) which lets the liquid pass through
-based on different particle size
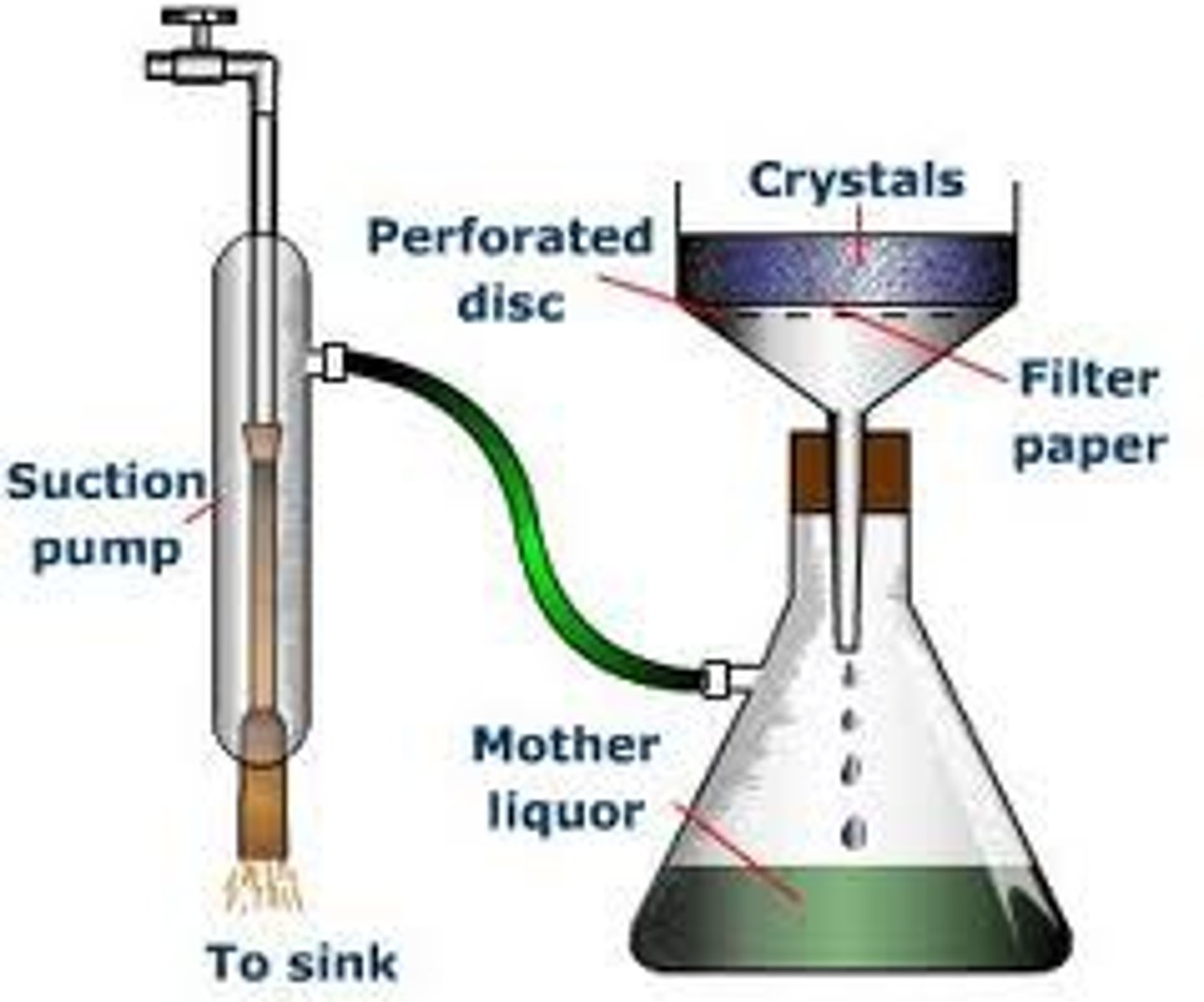
Suction filtration
seperation method: (very tiny ) solid can be saparated from a liquid using a filter (filter paper) which lets the liquid pass through
-based on different particle size
Melting point determination
Checking a substances purity
Pure substances: fixed sharp melting point, value can be compared with the literature value.
Impure substances: Lower melting point and tend to melt over a wider temperature range.
Sample is placed in a small melting point tube and the melting point is determined either by using commercial melting point apparatus or by attaching it to a thermometer in an oil bath that is gradually heated.
decantation
seperation method: insoluable solids can be seperated from liquid by letting it sink to the bottom and then taking out liquid
-based on different particle size and density
recystalization/drying
seperation method: by heating a solution to the boiling point of the soluvent, this then evaporates until only solid ( salt) is left
-based on different B.p.
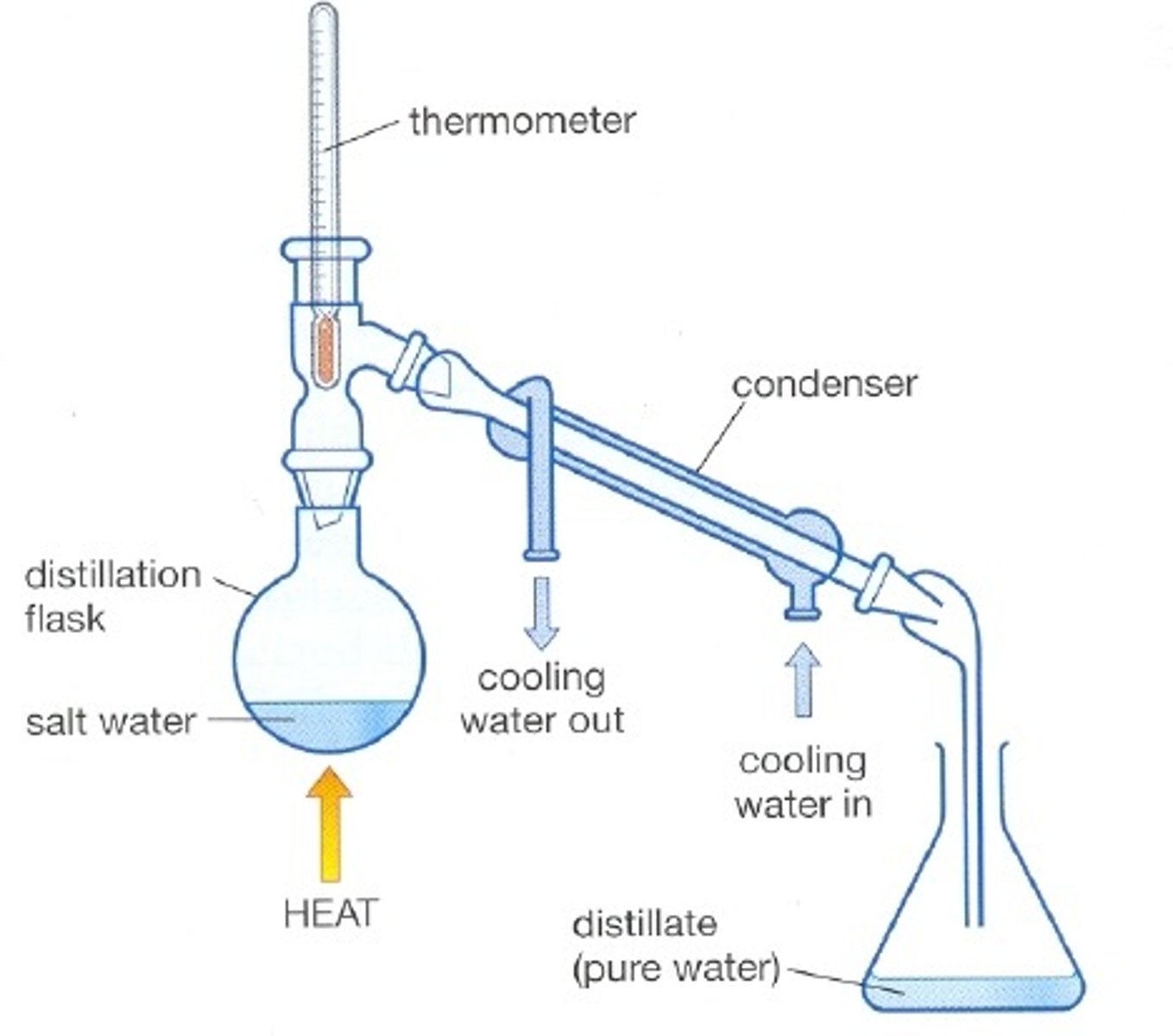
distilation
seperation method: a mixture of two liquids can be seperated by heating the micture until the B.P of one liquid is reaches, this will evaporate out of the mixture and is caugth using a conderncer
-based on different B.P
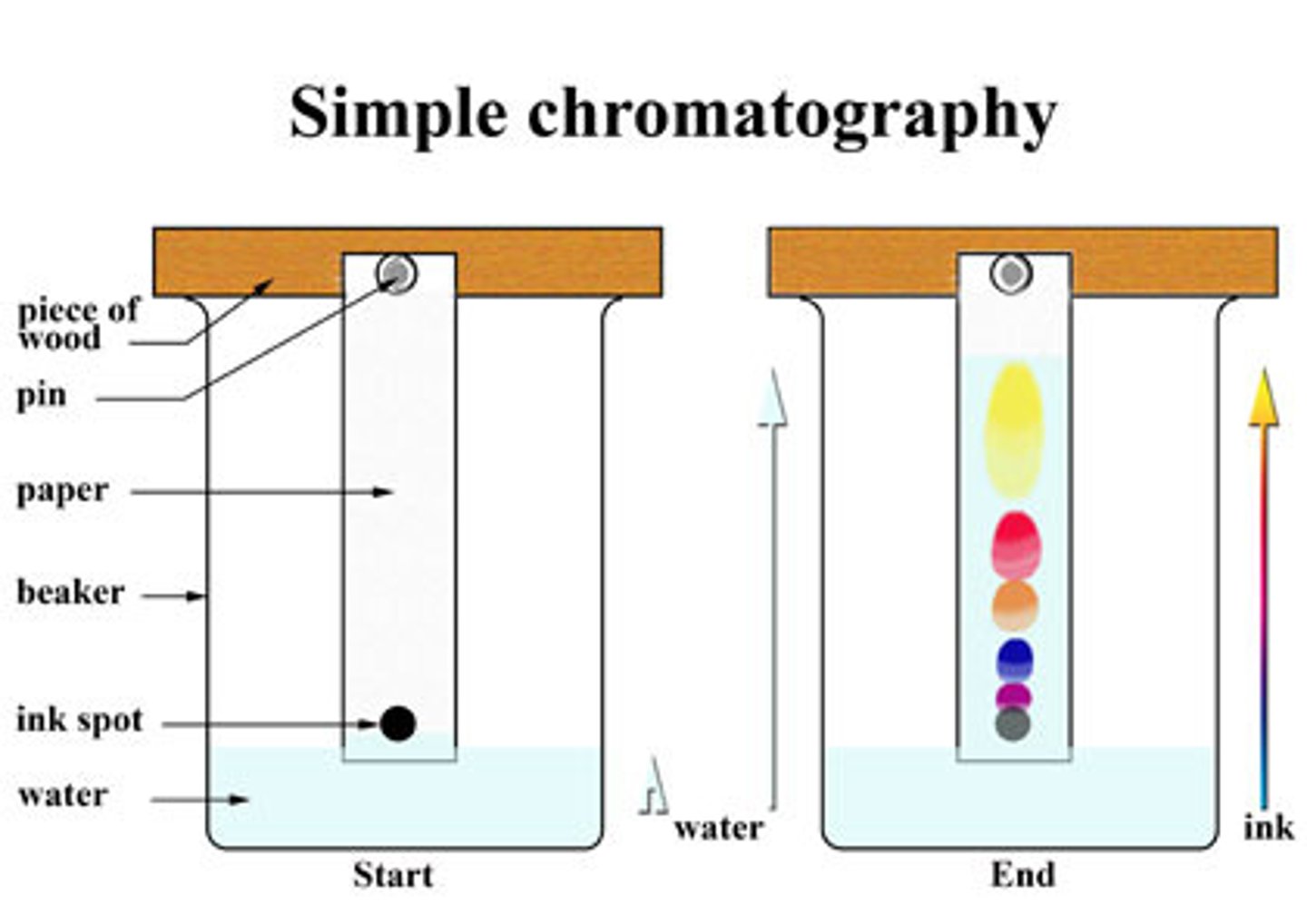
Chromatography
seperation method: Ink or other liquid mixtures can be seperated using water a paper, by letting the water suck into a paper stained with ink, the ink paticles are seperated and carried by the water. the smallest particles go the furthest and the others stay back
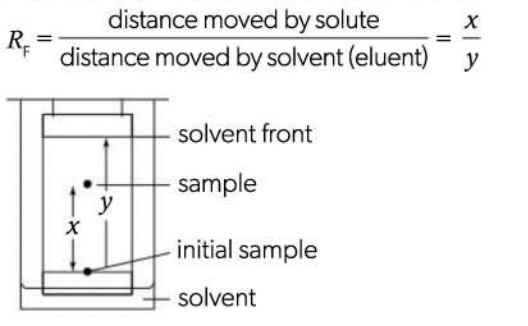
Retardation factor (RF)
Substances can be identified by their RF values. lf two substances have similar RF values in one solvent, the paper can be turned 90° and a different solvent used (two-way chromatography)
extraction (solid)
seperation method: by dissolving a mixture of a soluable and insoluable solid, these solids can be seperated as the soluable solid will dissolve into water, the insoluable solid can be filterd out
-based on different soluablitliys
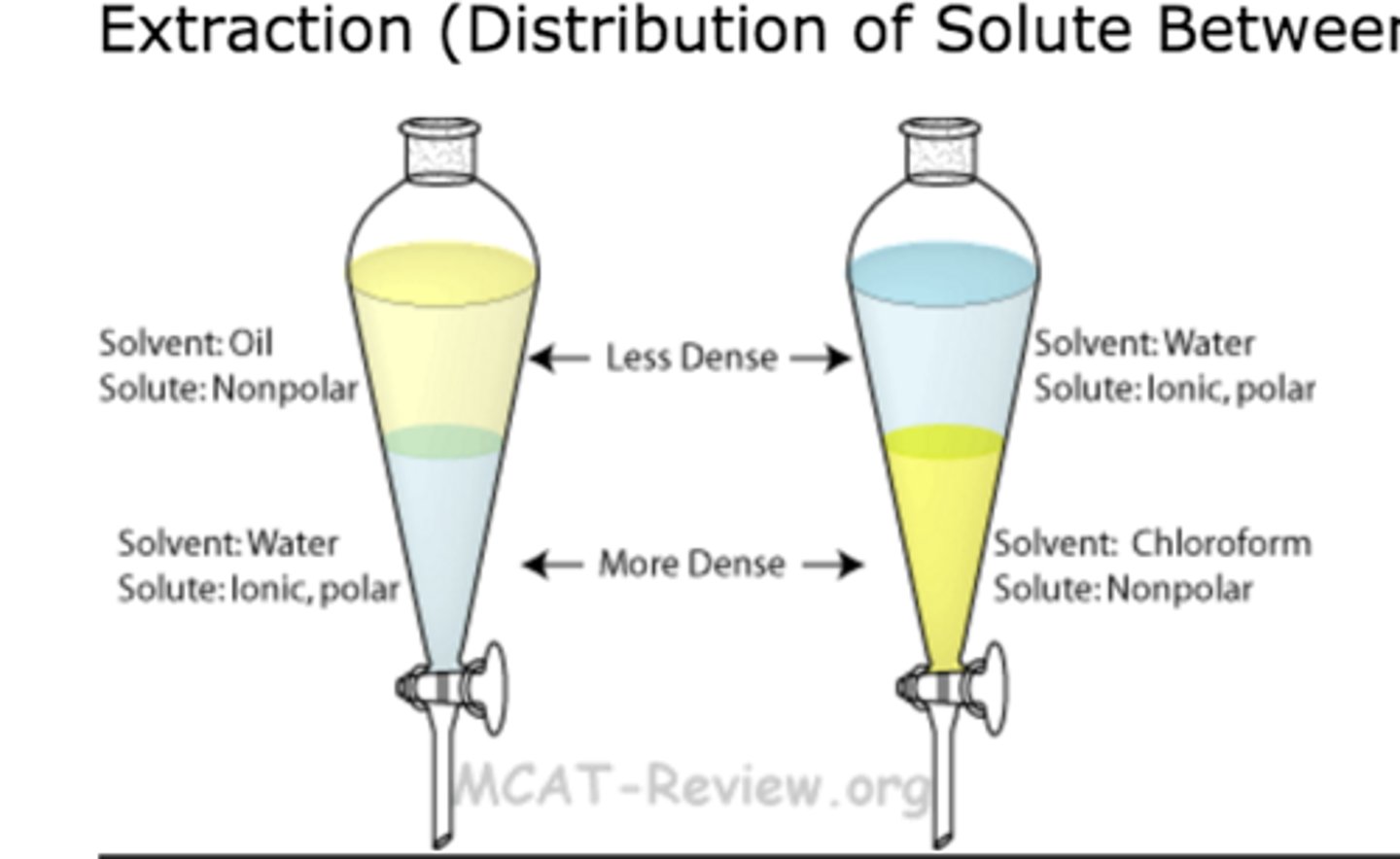
extraction (liquid)
seperation method: two immensible liquids can be seperated using a seperatory funnel
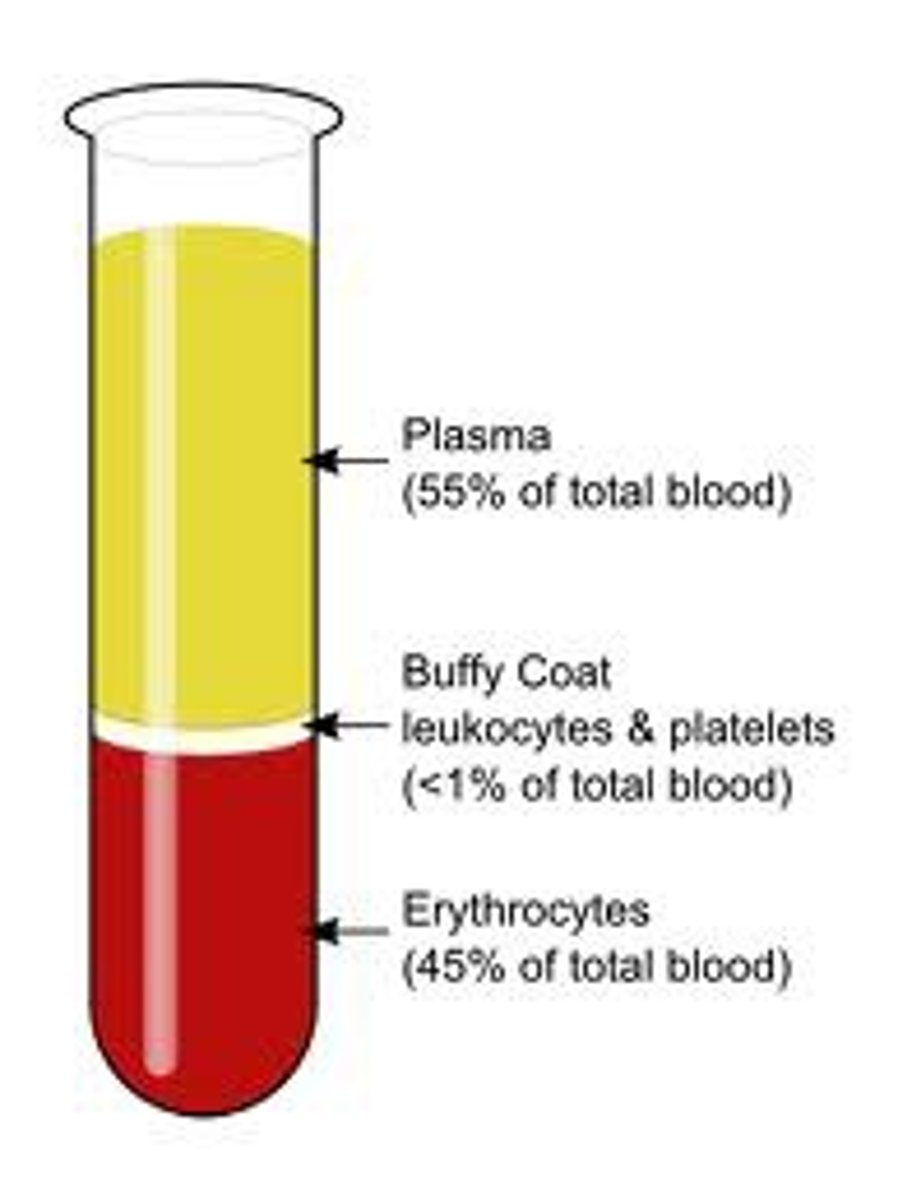
centrifugation
seperation method: small particles can be seperated from liquids using centrifugation. (e.g. red blood cells from plasma)
-based on different densitys
pure substance
A substance made of only one kind of matter and having definite properties.
element
only one type of atom
compound
a substance made of two or more atoms combined in specific ratio
mixture
two or more pure substances mixed with individual properties, seperatable with physical methods
homogenous
uniform in composition
heterogenous
non uniform compositions, different through out
colloids
mixtures that look homogenous but are heterogenous
isoelectrons
atoms with the same electron configuration
(e.g. K+ und Cl-)
diatomic
A molecule consisting of two atoms in nature
halide
ion of halogen
reduction
gain of electrons, (oxidation agent)
oxidation
loss of electrons, (reducing agent)
replacement/ displacement reaction
Where are compound breaks apart and forms a new compound plus a free reactant
In such a reaction, the element and compound combine. An element separates from the compound and combine with the element to give a new compound as a product. The element is replaced in the compound. For that reason it is also called the replacement reaction
Halogens commonly go through this process; when helogen with a samller NC and one with a bigger NC come close the bigger one gets oxidixed/ replaced by the smaller one which gets reduced (oxidation agent)
metalloid
an element that has properties of both metals and nonmetals
one-half the distance between the nuclei of two atoms of the same element when the atoms are joined
nuclear charge
number of protons in the nucleus
shielding effect
Refers to the protection of valence electrons by core electrons in inner energy levels
(trend down the period)
electromagnetic spectrum
the complete range of electromagnetic waves placed in order of increasing frequency
the specific energies an electron in an atom or other system can have (kinda same as shells is bohrs model)
absorb
to take in
photon
the smallest particle of light
quantize
electrons can only exist at specific energy levels, separated by specific intervals, so the values
Quantized means that it is measured in discrete values. For example, energy levels of electrons are quantized so there are specific values that an energy level can have, such as n=1,2,etc. it can't just be any number.
Cation
Positively charged ion
Anion
Negatively charged ion
elastic collision
A collision in which colliding objects bounce off without lasting deformation or the generation of heat.
solution
A mixture that forms when one substance dissolves another.
solute
A substance that is dissolved in a solution.
solvent
the substance in which the solute dissolves
Molarity
(concentration) — The number of moles of solute per liter of solution
Standard solution
a solution of known concentration
mass percentage
the number of grams of solute in each 100 g of solution
phenolphthalein indicator
Acid - Colorless, alkali( basic) - pink