RX 422: Nuclear Pharmacy
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
69 Terms
Radiation
Energy emitted from a source that is transmitted through space or a material medium.
Four types of ionizing radiation
- alpha
- beta
- positron
- gamma
Radioactive Decay
Atom with an unstable nucleus emits radiation to produce a more stable nucleus.
Types of Radioactive Decay
- Alpha Decay
- Beta-minus Decay
- Positron Decay (Beta-plus Decay)
- Gamma Decay
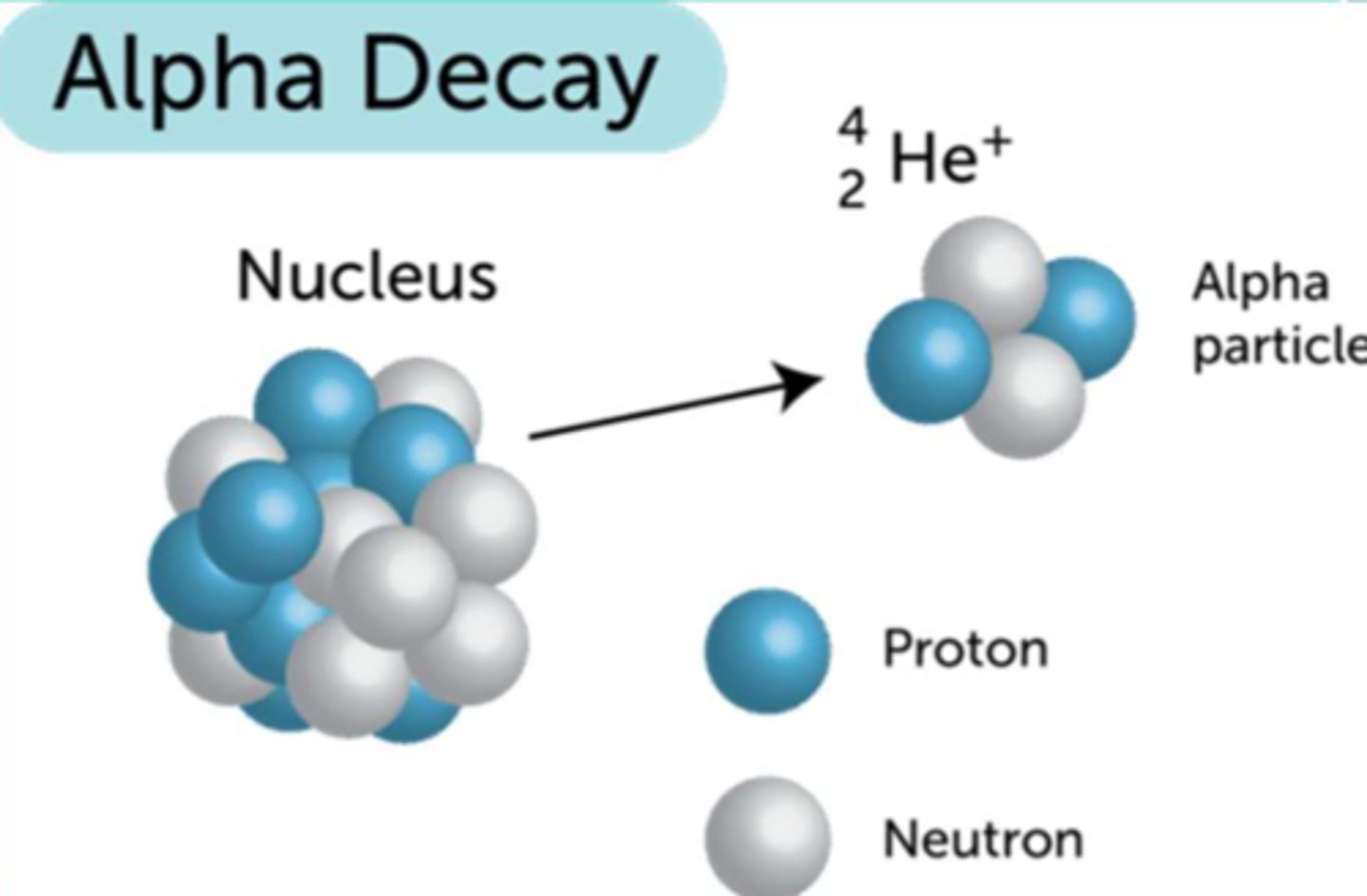
Alpha Decay Notes
- Particulate radiation
- Composed of 2 protons and 2 neutrons
- Very large mass, very energetic
- Range in tissue is less than a tenth of a mm, destructive
- NOT useful for diagnostic purposes (good for therapeutic)
Alpha Decay (image)
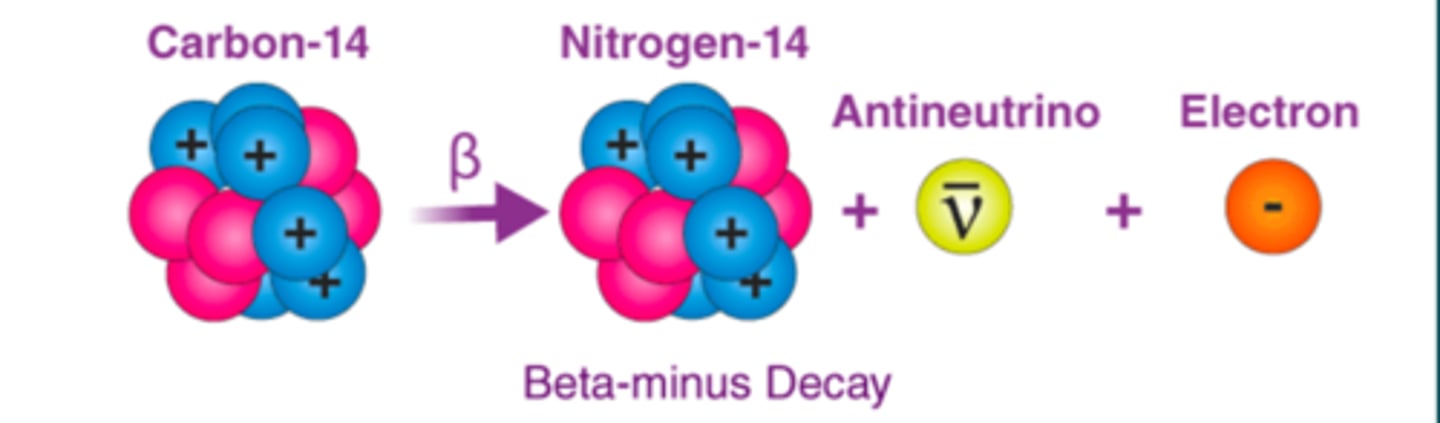
Beta-minus Decay Notes
- Occurs in unstable nuclei with excess neutrons (n0->p+)
- Emission of a (-) electron and antineutrino
- The electron has a smaller mass than alpha
- range in tissue is mms. but moves with higher velocity than alphas
- pure beta emitters can be useful in radiation therapy0+
Beta-minus Decay (image)
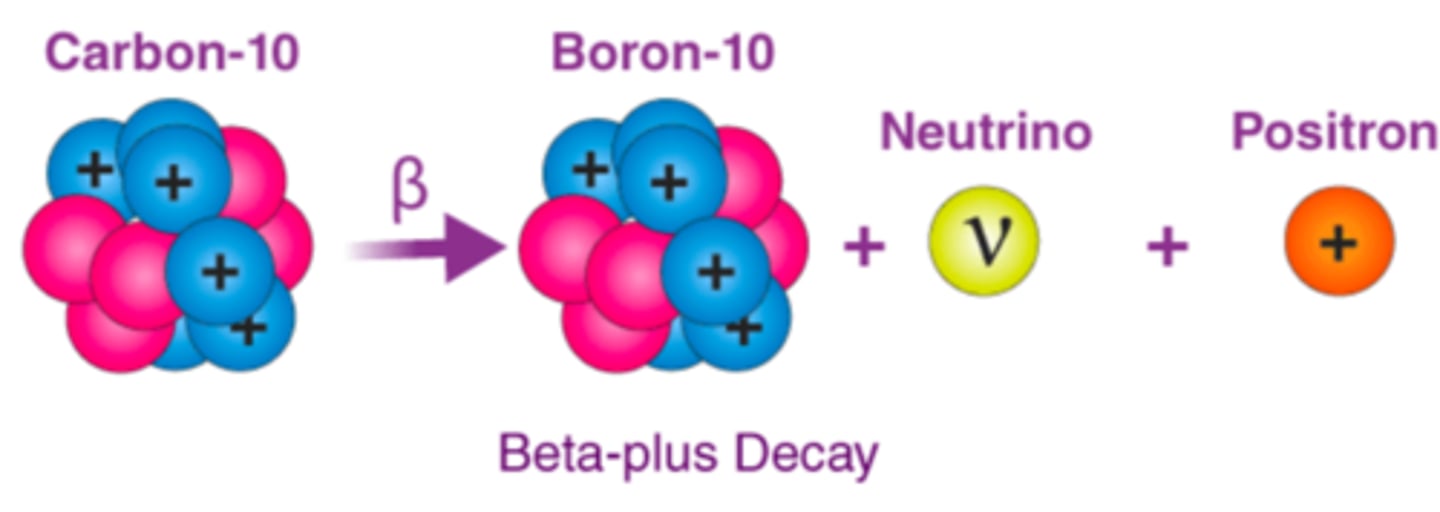
Positron Decay (Beta-plus Decay)
- Occurs in unstable nuclei with excess protons (p+->n0)
- Emission of a (+)positron and a neutrino
- Positron only travels a short distance before it combines with an electron = annihilation
- Annihilation process forms two gamma photons emitted at 180 angle to each other
Positron Decay (Beta-plus Decay) (image)
Gamma Decay
- Non-particulate radiation; no mass or charge
- Emission as packets or bundles of energy = photons
- ability to penetrate large thicknesses of material
- Overall damage is relatively low because it travels fast
- Ideal for diagnostic purposes
dps
disintegrations per second (dps)
Bq
Becquerel
Ci
Curies
Units for measurement of Radioactivity
- disintegrations per second (dps)
- Becquerel (Bq): often used as MBq or GBq
- Curies (Ci): often used as mCi
Radioactive material undergoes 1 decay event per second, its activity is ___ Bq
1 Bq
1 Bq = ___ dps
1 dps
1 Ci =___ GBq
37 GBq
1 Ci = ___ Bq
3.7 x 10^10 Bq
1 Ci = ___ dps
3.7 x 10^10 dps
Half-life (t1/2)
time during which one half of the radioactive atoms emit their characteristic radiation
t/12 equation
t1/2 = (ln2)/λ
Radioactive Decay is what kind of reaction?
1st order reaction (derived from differential rate law)
Decay rate is proportional to what?
number of atoms that are present
Radioactive Decay equation
D = λN
where
D = disintegration rate
λ = decay constant
N = number of atoms
Radioactive Decay determines what?
determines decrease in the number of radioactive nuclei per unit of time
Decay Equation
A(t) = Aoe^-λt
where
A(t) = total activity
Ao = initial activity
λ = decay constant (particular to each isotope)
t = elapsed time
Radioactive Decay: Decay Equation Definition
Spontaneous transformation of one radioisotope into one or more different isotopes accompanied by a decrease in radioactivity compared to the parent material.
Half-life
This spontaneous transformation of one radioisotope into one or more different isotopes accompanied by a decrease in radioactivity compared to the parent material over a defined period of time
Decay Factor
rate at which the variable's value diminishes through time
Each radioisotope has its own what?
decay factor
Decay factors can be used in combination to achieve what?
a particular elapsed time
Decay factors assist in calculating what?
radioactive decay without the use of a scientific calculator
Naturally Occurring vs. Artificially Produced Radionuclides
- All elements can have radioactive isotopes. If enough neutrons are added to an atom, it becomes unstable and decays.
- All of the radioactive isotopes undergo radioactive decay and each have a unique half-life (and unique decay constant).
- Most all naturally occurring radionuclides have very long half-lives ( ~millions to billions of years).
- Man-made radionuclides or artificial radionuclides are produced in a particle accelerator (cyclotron) by bombarding a stable nucleus with particles, making the nucleus unstable.
- Most importantly of these artificial radioisotopes, is Tc-99m, which is widely used in nuclear medicine studies across the globe.
Naturally Occurring Radioactivity
- Occurs in nature as members of a decay chain
- Long half-life parent radionuclide decays to start a series of radionuclides that decays until a stable end product is produced
- All the parent isotopes have extremely long half-lives and undergo multi-step decay series with a wide variety half-lives in the decay chain
Examples of naturally occurring radioactivity
Lantern mantles (thorium nitrate), granite countertops (veins can contain naturally occurring radioactive elements), vaseline glass (uranium), fiestaware plates (uranium oxide glaze)
Artificially Produced Radionuclides
- All radioisotopes for medical imaging and therapy purposes are artificially produced
- When we artificially create isotopes we perform a nuclear transmutation
--- Convert a stable nucleus into an unstable nucleus
- Force something into the nucleus to create instability -> requires a lot of energy
--- cyclotrons (particle accelerators), nuclear reactors
Radioisotopes in Nuclear Medicine: Half-Life
Radiopharmaceuticals should have a relatively short effective half-life -long enough to examine metabolic processes, but short enough to minimize exposure.
Radioisotopes in Nuclear Medicine: Energy
Decays with relatively low energy (if possible) and can be detected by a camera easily. Usually, gamma or beta. Alpha and Beta may be useful for therapy due to the effective damage to abnormal cells.
Radioisotopes in Nuclear Medicine: Chemistry
Radioisotopes that can easily attach to a drug to image different areas/organs of interest or for use as therapy.
Radioisotopes in Nuclear Medicine: Availability
The radiopharmaceutical should be easily produced and readily available to nuclear medicine facilities.
Radioisotopes in Nuclear Medicine: Tc-99m
- has the most desirable properties and is most commonly used in nuclear medicine.
- Half-life: 6 hours
- Gamma radiation
- Versatile chemistry
- Readily available through nuclear pharmacies
Radioisotopes used in Nuclear Medicine: Tc-99m
- Chemical form: Sodium Pertechnetate
- Half-life: 6 hours
- Uses: Kit preparation, MUGA, GI Bleed, Thyroid
- Source: Generator produced
- (Mo-99 generator -> reactor produced)
Radioisotopes used in Nuclear Medicine: Tl-201
- Chemical form: Thallous Chloride
- Half-life: 73.1 hours
- Uses: Myocardial Perfusion
- Source: Cyclotron produced
Radioisotopes used in Nuclear Medicine: In-111
- Chemical form: Indium chloride, Indium oxine, Indium DTPA
- Half-life: 2.83 days
- Uses: Mab labeling, WBC labeling, cisternography
- Source: Cyclotron produced
Radioisotopes used in Nuclear Medicine: Ga-68
- Chemical form: Gallium chloride
- Half-life: 68 minutes
- Uses: Tumor and infection imaging
- Source: Generator produced
- (Ge-68 generator -> reactor produced)
Radioisotopes used in Nuclear Medicine: I-131
- Chemical form: Sodium iodide
- Half-life: 8 days
- Uses: Thyroid uptake and therapy/ablation
- Source: Reactor produced
Radioisotopes used in Nuclear Medicine: Xe-133
- Chemical form: Xenon (gas)
- Half-life: 5.25 days
- Uses: Pulmonary perfusion
- Source: Reactor produced
Radioisotopes used in Nuclear Medicine: I-123
- Chemical form: Sodium iodide
- Half-life: 13 hours
- Uses: Thyroid uptake and imaging
- Source: Cyclotron produced
Radioisotopes used in Nuclear Medicine: F-18
- Chemical form:
- Half-life: 110 minutes
- Uses: complexed with tracers for cancer imaging
- Source: Cyclotron produced
Mo-99 and Tc-99m
- Tc-99m is one of the most widely used radioisotopes in nuclear medicine.
- The parent isotope, Mo-99, is produced in a nuclear reactor via nuclear fission of Uranium 235.
- Targets are irradiated with neutrons to produce Mo-99.
- Chemical processing takes place to separate the desirable Mo-99 from the target.
- The molybdate solution is loaded onto a column for use in a Tc-99m generator.
Mo-99/Tc-99m Generator Overview
- Generators are shipped to nuclear pharmacies
- Tc-99m pertechnetate (TcO4) is produced on the alumina column as the Mo-99 decays
- Normal saline is used to 'wash' the pertechnetate off of the column
- The resulting sodium pertechnetate (NaTcO4) solution is called an elution
- Elutions of sodium pertechnetate are used for dispensing doses and to prepare various radiopharmaceutical kits
Chemistry of Mo-99/Tc-99m Generators
- Sterile normal saline (Na+Cl-) used for eluting the generator -> resulting solution is called sodium pertechnetate (NaTcO4 )
- Mo-99 is strongly bound to the alumina column as anions.
- Both Tc-99m (TcO4-) and Mo-99 (MoO42-) are anions with Mo-99 having the strongest fixation'
- After Transient Equilibrium is reached, the daughter isotope appears to decay with the half-life of the parent isotope (effective half-life)
- The amount of Tc-99m on a generator decreases about 20% each day
- Maximum build up on each generator occurs ~24 hours after each elution
The molybdate solution is of high specific activity (1000Ci/g), which allows for many advantages such as...
- Small amounts of alumina
- Small elution volume and higher concentrations
- Small column size
- Easier shielding
- Small amounts of radionuclidic impurities
Chemical Reactions of Radiolabeling: Tc-99m Sodium Pertechnetate
- When Tc-99m is eluted off of the generator it exists as [Tc+7O4] -, oxidation state of +7
- Most imaging agents contain Tc-99m in the +5, +3, +1 or +7 oxidation state
Chemical Reactions of Radiolabeling: Reduction of the pertechnetate ion [Tc+7O4] -
- Lower the oxidation state so it can bind to another chelating molecule
- The most common reducing agent in most drug kits is stannous chloride dihydrate, SnCl2 • 2H2O
RUBY-FILL Rubidium Rb-82 Generator
- Elution/Injection system for delivery of Rubidium Rb-82 chloride
- Rb-82 has a 75 second half-life
- Elution injected directly into the patient
- Indicated for PET myocardial perfusion imaging
- Generator has a 60-day expiration
- Located within cardiac clinics and administered by CMNT
SPECT
Single Photon Emission Computed Tomography
PET
Positron Emission Tomography
SPECT vs. PET Imaging
- Single Photon Emission Computed Tomography
--- contains gamma camera detectors that detect gamma rays from radiopharmaceuticals
--- produce computer generated 3D images
--- Most common: Tc-99m
- Positron Emission Tomography --- contains cameras that detect positrons
--- offers better contrast and spatial resolution
--- Most common: F-18
- Both are attached to a tracer to image functionality in the body
The most common SPECT nuclear medicine imaging scan is what?
a myocardial perfusion scan.
There are 2 main Tc-99m labeled drugs that can be used for imaging. What are they?
- Tc-99m Sestamibi (Cardiolite)
- Tc-99m Tetrofosmin(Myoview)
Physicians may order stress/rest myocardial perfusion imaging
- To assess overall function of heart muscles
- To assess any damage following heart attacks
- To diagnose symptoms of CAD
- To assess extent of coronary artery stenosis
- Once diagnosis is made, proper treatment and therapy can be prescribed by the physician
Mo-99; Half-life = 65.9 hrs; Decay constant = 0.105/hr; If you start with 100mCi of Mo-99, how much will be available after 66 hours?
A. 25mCi
B. 44mCi
C. 50mCi
D. 100mCi
C. 50mCi
Tc-99m; Half-life = 6.01 hrs; Decay Constant = 0.1153/hr; If you start with 100mCi of Tc-99m, how much will be available after 10 hours?
A. 50.1mCi
B. 65.9mCi
C. 100mCi
D. 31.6mCi
D. 31.6mCi
Mo-99; Half-life = 65.9 hrs; Calculate the decay constant of Mo-99:
A. 0.1153/hr
B. 0.0105/hr
C. 0.0095/hr
D. 0.0862/hr
B. 0.0105/hr
Tc-99m; Half-life = 6.01 hrs; Calculate the decay constant of Tc-99m:
A. 0.1153/hr
B. 0.0105/hr
C. 0.0095/hr
D. 0.0862/hr
A. 0.1153/hr
The pharmacy technician dispensing on 1st run, is drawing up a dose for a customer. He needs 30mCi of Tc-99m sestamibi calibrated for 0930. The current time is 0230. What amount does he need to draw now, so that he draws enough for the calibration time?
A. 67.2 mCi
B. 35.2 mCi
C. 60.0 mCi
D. 100mCi
A. 67.2 mCi
Which of the following characteristic(s) make a radioisotope desirable for use in the medical field?
A. Half-life
B. Energy type
C. Chemistry
D. All of the above
D. All of the above