Untitled
0.0(0)
Studied by 1 person0%Unit Mastery
0%Exam Mastery
Build your Mastery score
Supplemental Materials
Card Sorting
1/64
Earn XP
Description and Tags
Last updated 8:37 PM on 9/11/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
1
New cards
What is the DNA complementary sequence to 5' TAACCG 3'?
A. 5' ATTGGC 3'
B. 5' CGGTTA 3'
A. 5' ATTGGC 3'
B. 5' CGGTTA 3'
B. 5' CGGTTA 3'
2
New cards
Molecules that are soluble in water have which type of bond?
A. polar covalent bond
B. nonpolar covalent bond
C. hydrophobic interaction
D. double bonds
A. polar covalent bond
B. nonpolar covalent bond
C. hydrophobic interaction
D. double bonds
A. polar covalent bond
3
New cards
What is true for double bonds?
A. Trans double bonds are found in nature and are associated with cardiovascular health.
B. Cis double bonds are made artificially in food processing and are associated with poor cardiovascular health.
C. Double bonds are weaker than single bonds.
D. Double bonds keep molecules straight.
E. Unsaturated fats have double bonds and are associated with good cardiovascular health.
A. Trans double bonds are found in nature and are associated with cardiovascular health.
B. Cis double bonds are made artificially in food processing and are associated with poor cardiovascular health.
C. Double bonds are weaker than single bonds.
D. Double bonds keep molecules straight.
E. Unsaturated fats have double bonds and are associated with good cardiovascular health.
E. Unsaturated fats have double bonds and are associated with good cardiovascular health.
4
New cards
Which polysaccharide has the greatest number of branches?
A. cellulose
B. chitin
C. amylose
D. amylopectin
E. glycogen
A. cellulose
B. chitin
C. amylose
D. amylopectin
E. glycogen
E. glycogen
5
New cards
All lipids
A. are made from glycerol and fatty acids.
B. contain nitrogen.
C. have low energy content.
D. are acidic when mixed with water.
E. are poorly water soluble.
A. are made from glycerol and fatty acids.
B. contain nitrogen.
C. have low energy content.
D. are acidic when mixed with water.
E. are poorly water soluble.
E. are poorly water soluble.
6
New cards
Where in the structure of a protein would glutamic acid (hydrophilic) be mostly likely?
A. on the exterior surface of the protein
B. in the interior of the protein, away from water
C. at the active site, binding oxygen
D. at the heme-binding site
E. at a site where hemoglobin binds to a negatively charged protein
A. on the exterior surface of the protein
B. in the interior of the protein, away from water
C. at the active site, binding oxygen
D. at the heme-binding site
E. at a site where hemoglobin binds to a negatively charged protein
A. on the exterior surface of the protein
7
New cards
Which is not a function of proteins?
A. help make up membranes
B. carry the code for translation from the nucleus to the ribosome
C. bind to hormones (hormone receptor)
D. can be hormones
E. speed chemical reactions
A. help make up membranes
B. carry the code for translation from the nucleus to the ribosome
C. bind to hormones (hormone receptor)
D. can be hormones
E. speed chemical reactions
B. carry the code for translation from the nucleus to the ribosome
8
New cards
How does RNA differ from DNA?
A. DNA encodes hereditary information; RNA does not.
B. DNA forms duplexes; RNA does not.
C. DNA contains thymine; RNA contains uracil.
D. DNA contains five bases; RNA contains four.
E. all of the above
A. DNA encodes hereditary information; RNA does not.
B. DNA forms duplexes; RNA does not.
C. DNA contains thymine; RNA contains uracil.
D. DNA contains five bases; RNA contains four.
E. all of the above
C. DNA contains thymine; RNA contains uracil.
9
New cards
What is incorrect about the following statement? "The tRNA sequence reads ATGCCATGG..."
A. tRNA is made of amino acids.
B. tRNA has U in place of T.
C. tRNA is not linear.
D. A and C
E. None of the choices is correct.
A. tRNA is made of amino acids.
B. tRNA has U in place of T.
C. tRNA is not linear.
D. A and C
E. None of the choices is correct.
B. tRNA has U in place of T.
10
New cards
If you located a single-stranded piece of nucleic acid in a cell, what would it be made of?
A. nucleotides
B. amino acids
C. fatty acids
D. sugars
E. glycerol
A. nucleotides
B. amino acids
C. fatty acids
D. sugars
E. glycerol
A. nucleotides
11
New cards
If you were given a sample of a polysaccharide and told that the only subunit was glucose, what would you have?
A. glycogen
B. starch
C. cellulose
D. amylopectin
E. You cannot know from the information given.
A. glycogen
B. starch
C. cellulose
D. amylopectin
E. You cannot know from the information given.
E. You cannot know from the information given.
12
New cards
Which sugar is most important for making RNA?
A. glucose
B. ribose
C. fructose
D. glyceraldehyde
E. sucrose
A. glucose
B. ribose
C. fructose
D. glyceraldehyde
E. sucrose
B. ribose
13
New cards
Which falls outside the lipids?
A. estrogen
B. cholesterol
C. glucose
D. triacylglyceride
E. trans fat
A. estrogen
B. cholesterol
C. glucose
D. triacylglyceride
E. trans fat
C. glucose
14
New cards
For which of the following could genomics be used?
A. studying disease
B. conservation work
C. predicting future medical problems
D. evolutionary studies
E. all of the above
A. studying disease
B. conservation work
C. predicting future medical problems
D. evolutionary studies
E. all of the above
E. all of the above
15
New cards
Which provides the most compact energy storage?
A. proteins
B. carbohydrates
C. lipids
D. nucleic acids
E. All provide about the same amount of energy stored per unit volume.
A. proteins
B. carbohydrates
C. lipids
D. nucleic acids
E. All provide about the same amount of energy stored per unit volume.
C. lipids
16
New cards
For what is water most important when digesting food?
A. dehydration reactions
B. temperature reduction
C. solubility
D. hydrolysis reactions
E. none of the above
A. dehydration reactions
B. temperature reduction
C. solubility
D. hydrolysis reactions
E. none of the above
D. hydrolysis reactions
17
New cards
If a DNA sample were composed of 20% adenine, what would be the percentage of cytosine?
A. 20%
B. 30%
C. 40%
D. 60%
E. 80%
A. 20%
B. 30%
C. 40%
D. 60%
E. 80%
B. 30%
18
New cards
When observing the synthesis of a biological macromolecule, more of what should appear?
A. water
B. amino acids
C. alcohol
D. ions
E. fatty acids
A. water
B. amino acids
C. alcohol
D. ions
E. fatty acids
A. water
19
New cards
From what are polysaccharides made?
A. monosaccharides
B. glucose
C. disaccharides
D. sucrose
E. none of the above
A. monosaccharides
B. glucose
C. disaccharides
D. sucrose
E. none of the above
A. monosaccharides
20
New cards
If you were asked to chose a lipid subunit, which would you choose?
A. fatty acid
B. steroid
C. cholesterol
D. unsaturated side chain
E. none of the above
A. fatty acid
B. steroid
C. cholesterol
D. unsaturated side chain
E. none of the above
A. fatty acid
21
New cards
Which is least important for metabolism?
A. receptor protein
B. contractile protein
C. enzymatic protein
D. hormonal protein
E. structural protein
A. receptor protein
B. contractile protein
C. enzymatic protein
D. hormonal protein
E. structural protein
E. structural protein
22
New cards
Which level of protein structure is most immediately encoded in DNA?
A. primary
B. secondary
C. tertiary
D. quaternary
E. There is no difference between the four answers.
A. primary
B. secondary
C. tertiary
D. quaternary
E. There is no difference between the four answers.
A. primary
23
New cards
isomers
Compounds with the same formula but different structures.
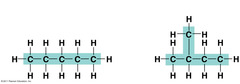
24
New cards
structural isomers
differ in the covalent arrangements of their atoms
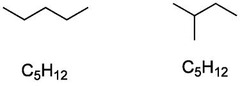
25
New cards
cis-trans isomers
carbons have covalent bonds to the same atoms, but these atoms differ in their spatial arrangements due to the inflexibility of double bonds
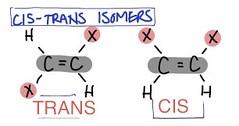
26
New cards
trans isomers
the two x's are on opposite sides
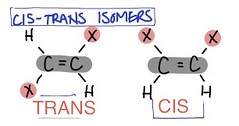
27
New cards
Enatiomers
isomers that are mirror images of each other
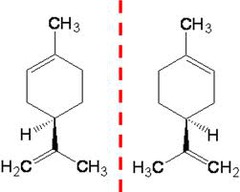
28
New cards
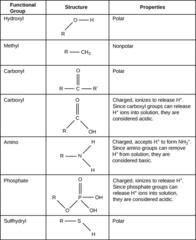
functional groups
the components of organic molecules that are most commonly involved in chemical reactions
29
New cards
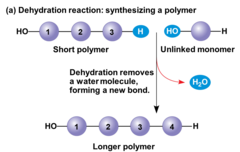
dehydration reaction
A chemical reaction in which two molecules become covalently bonded to each other with the removal of a water molecule.
30
New cards
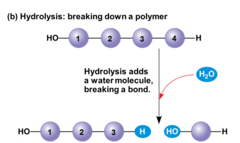
hydrolysis
Breaking down complex molecules by the chemical addition of water
31
New cards
glycosidic linkage
A covalent bond formed between two monosaccharides by a dehydration reaction.
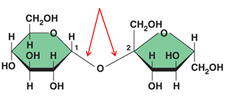
32
New cards
glycogen
An extensively branched glucose storage polysaccharide found in the liver and muscle of animals; the animal equivalent of starch.

33
New cards
cellulose
Carbohydrate component of plant cell walls.
34
New cards
chitin
A structural polysaccharide, consisting of amino sugar monomers, found in many fungal cell walls and in the exoskeletons of all arthropods.
35
New cards
triacylglycerol
three fatty acids linked to one glycerol molecule
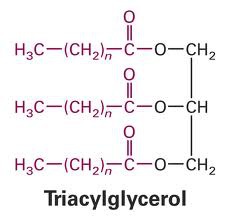
36
New cards
saturated fatty acid
a long-chain hydrocarbon with single covalent bonds in the carbon chain; the number of hydrogen atoms attached to the carbon skeleton is maximized
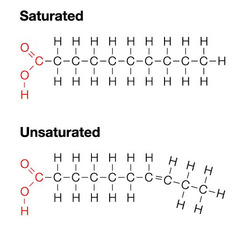
37
New cards
unsaturated fatty acid
A fatty acid possessing one or more double bonds between the carbons in the hydrocarbon tail. Such bonding reduces the number of hydrogen atoms attached to the carbon skeleton.
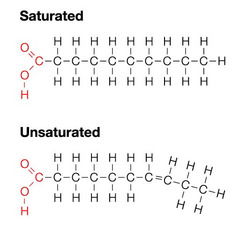
38
New cards
trans fats
An unsaturated fat, formed artificially during hydrogenation of oils, containing one or more trans double bonds.
39
New cards
Phospholipids
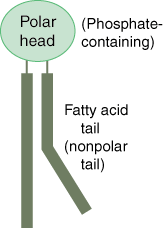
A molecule that is a constituent of the inner bilayer of biological membranes, having a polar, hydrophilic head and a nonpolar, hydrophobic tail.
40
New cards
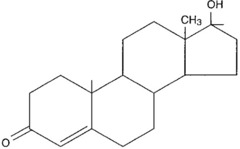
steroids
lipids characterized by a carbon skeleton consisting of four fused rings
41
New cards
Cholesterol
A type of fat made by the body from saturated fat; a minor part of fat in foods.
42
New cards
enzymatic proteins
carry out metabolic reactions directly

43
New cards
storage proteins
storage of amino acids
44
New cards
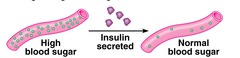
hormonal proteins
coordination of an organism's activities
45
New cards
contractile and motor proteins
movement
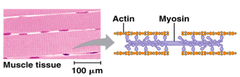
46
New cards
defensive proteins
protection against disease
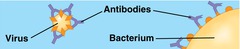
47
New cards
transport proteins
allow passage of hydrophilic substances across the membrane
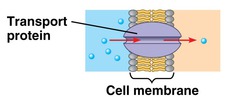
48
New cards
receptor proteins
response of cell to chemical stimuli
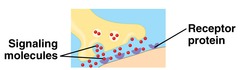
49
New cards
structural proteins
to support

50
New cards
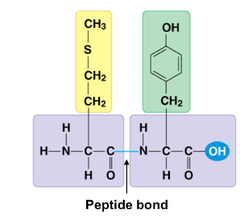
peptide bond
covalent bond formed between amino acids
51
New cards
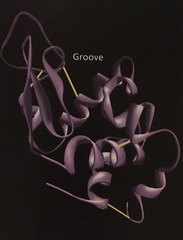
ribbon model
Shows how the single polypeptide chain folds and coils to form the functional protein
52
New cards
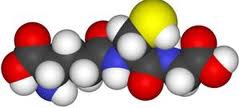
space-filling model
a model of a molecule showing the relative sizes of the atoms and their relative orientations
53
New cards
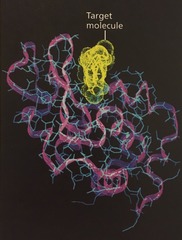
wireframe model
shows the backbone with the side chains extending from it
54
New cards
primary structure
The first level of protein structure; the specific sequence of amino acids making up a polypeptide chain.
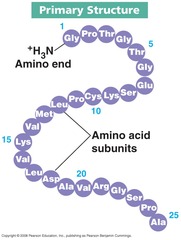
55
New cards
secondary structure
The second level of protein structure; the regular local patterns of coils or folds of a polypeptide chain.
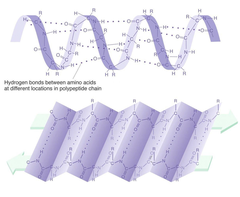
56
New cards
a helix
Something spiral in form.
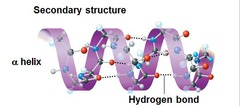
57
New cards
B pleated sheet
protein structure with two or more segments of the polypeptide chain link side by side (called B strands) connected by hydrogen bonds between parts of the two parallel segments of the polypeptide backbone.
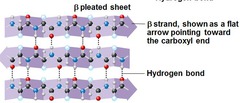
58
New cards
tertiary structure
The third level of protein structure; the overall, three-dimensional shape of a polypeptide due to interactions of the R groups of the amino acids making up the chain.
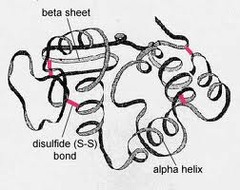
59
New cards
hydrophobic interactions
Interactions between hydrophobic molecules grouping together to avoid water
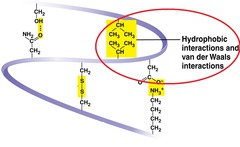
60
New cards
disulfide bridges
covalent bonds that may further reinforce the shape of a protein
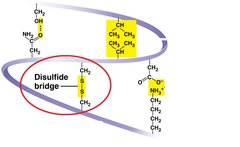
61
New cards
quaternary structure
The fourth level of protein structure; the shape resulting from the association of two or more polypeptide subunits.
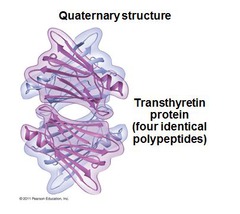
62
New cards
Denaturation
loss of normal shape of a protein due to heat or other factor
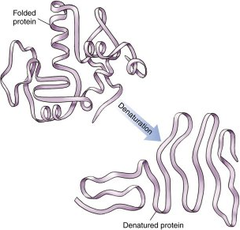
63
New cards
X-ray crystallography
A technique that depends on the diffraction of an X-ray beam by the individual atoms of a crystallized molecule to study the three-dimensional structure of the molecule.
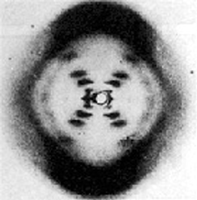
64
New cards
Pyrimidines
cytosine, thymine, uracil
65
New cards
purines
Adenine and Guanine