Assessment Ch 20: heart and neck vessels
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
Structure and surface landmarks
Cardiovascular system: consist of the heart (a muscular pump) and the blood vessels
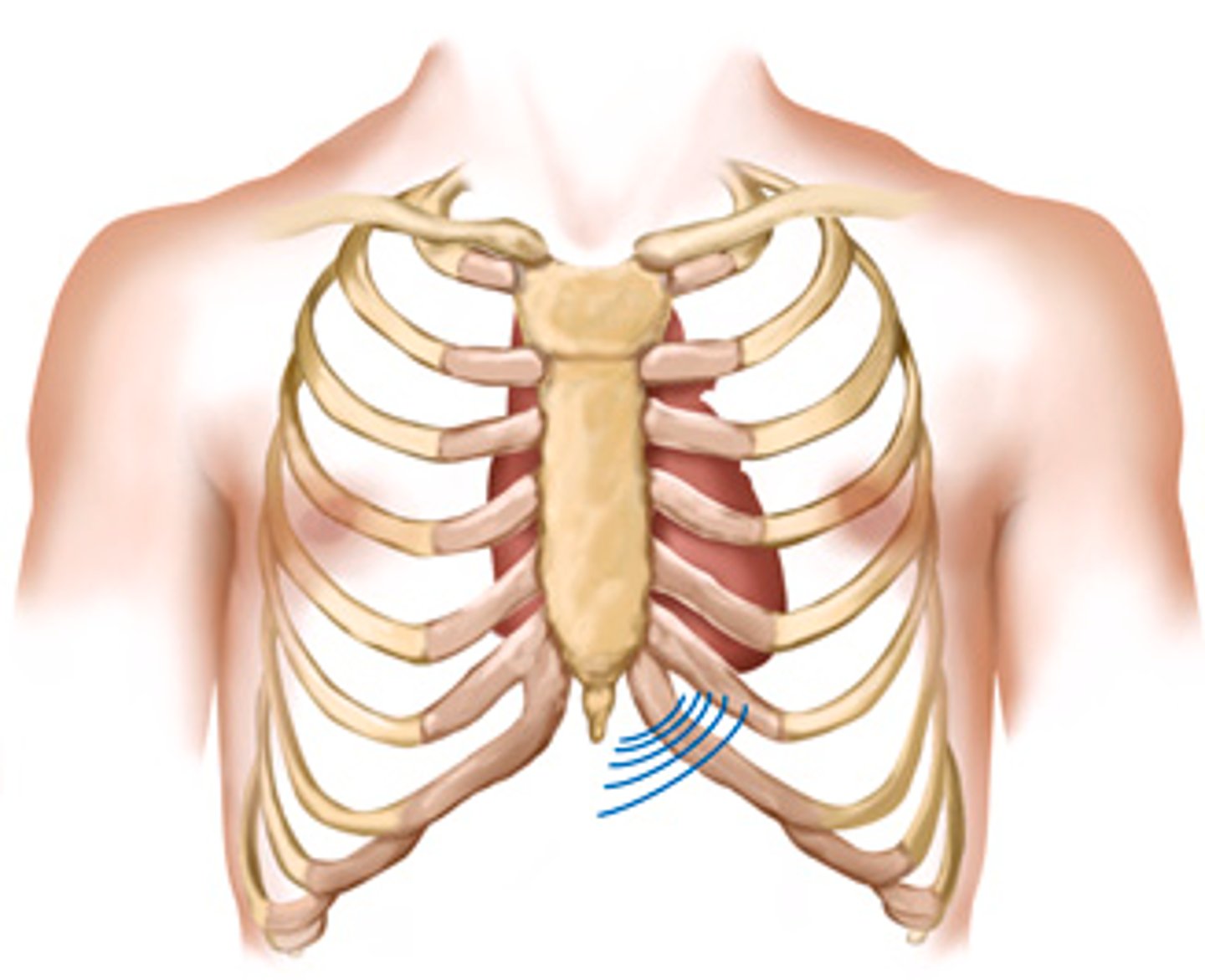
Precordium -> area on the anterior chest directly overlying the heart and great vessels
Great vessels -> the major arteries and veins connected to the heart.
(all located between the lungs in the middle third of the thoracic cage (mediastinum)
extends from 2nd to 5th intercostal space from the right border of the sternum to the left midclavicular line
Structure and surface landmarks ( pt2)
-Heart is rotated so right side is anterior, left side mostly posterior
Heart 4 chamber ->
-right ventricle behind the sternum and greatest area of anterior cardiac surface.
-left ventricle lies behind the right ventricle and forms the apex and slender area of the left border
-the right atrium lies to the right and above the right ventricle and forms the right border.
-left atrium is located posteriorly, with only a small portion, the left atrial appendage, showing anteriorly.
Structure and surface landmarks ( pt 3 blood vessel arrangement
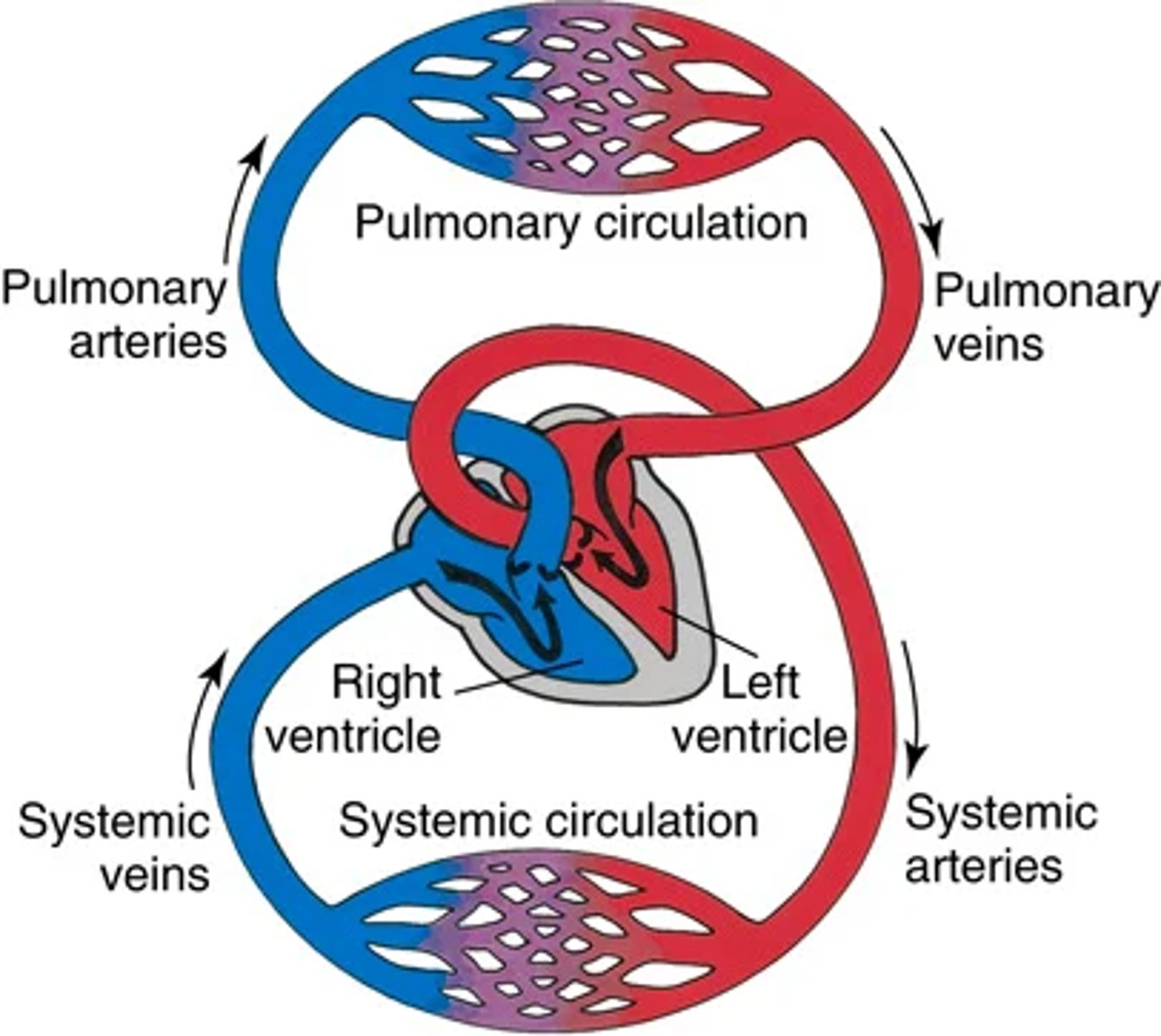
2 continuous loops: Pulmonary circulation and systemic circulation
-Upon contraction blood pumps into both loops simultaneously
top of heart = base
bottom of heart = apex (down to left)
contraction - > apex beats against the chest wall producing an apical pulse
(palpable at the 5th intercostal, 7-9 inches from the mid sternal line)
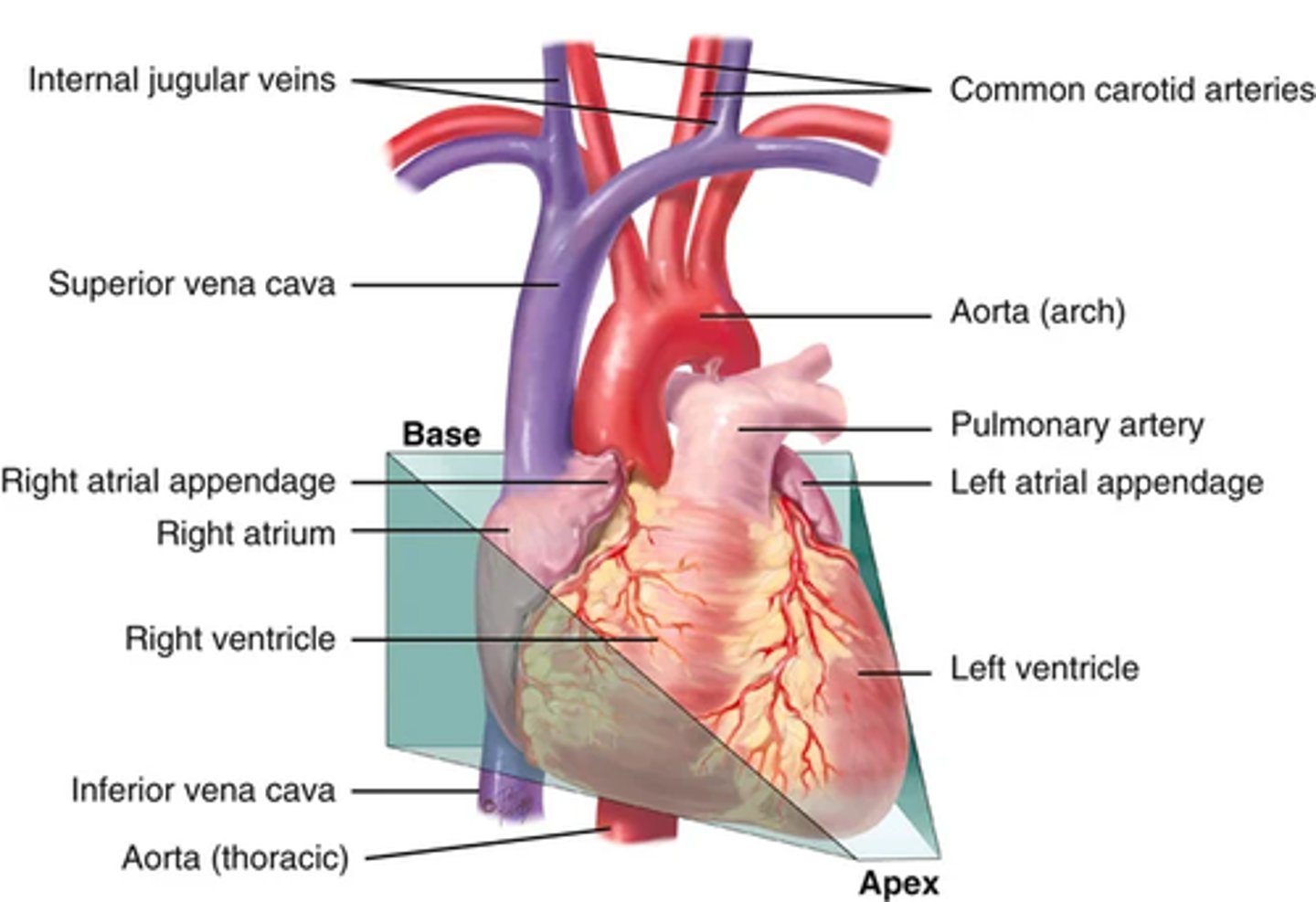
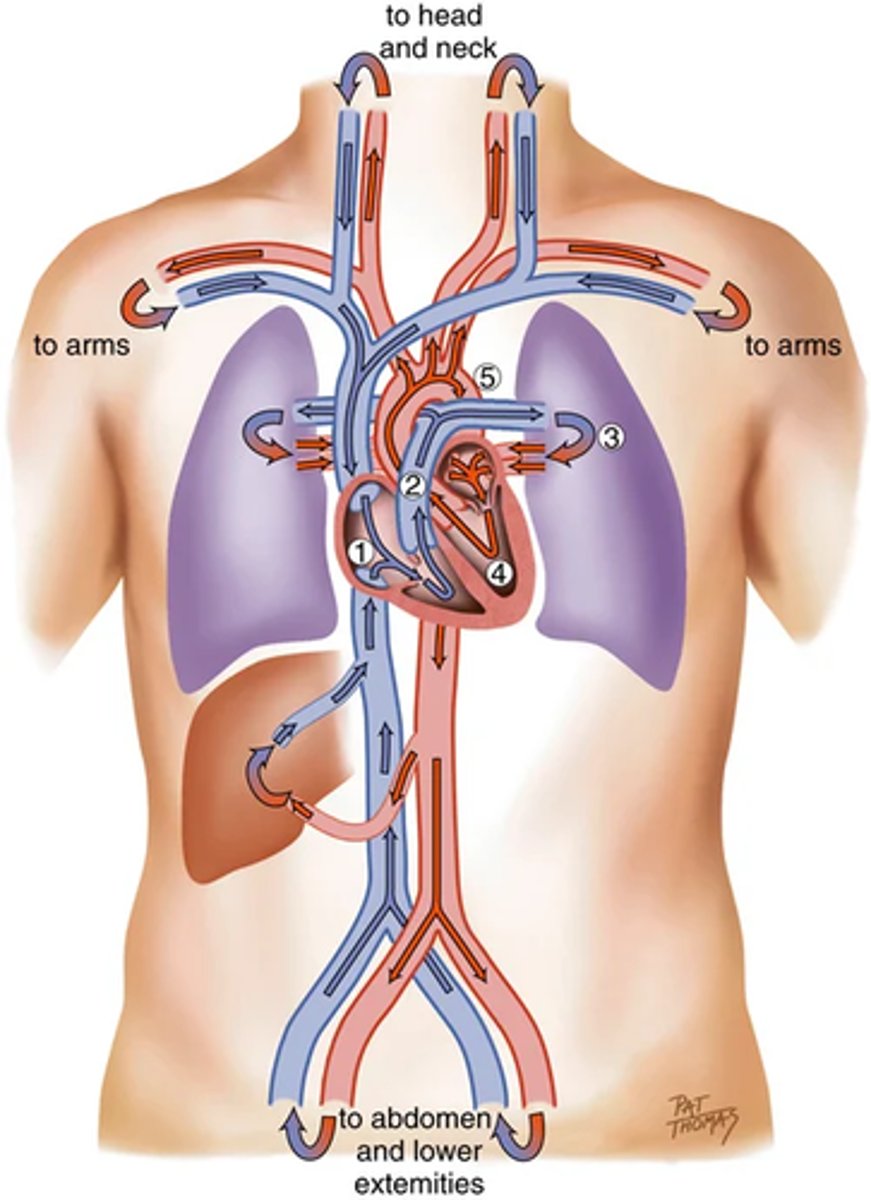
Surface structures and landmarks pt 4 (great vessels)
The great vessels lie bunched above the base of the heart.
The superior and inferior vena cava return unoxygenated venous blood to the right side of the heart.
The pulmonary artery leaves the right ventricle, bifurcates, and carries the venous blood to the lungs.
. The pulmonary veins return the freshly oxygenated blood to the left side of the heart, and the aorta carries it out to the body.
The aorta ascends from the left ventricle, arches back at the level of the sternal angle, and descends behind the heart.
Heart walls, Chambers, and valves (heart walls and chambers)
Heart wall layers :
pericardium -> tough, fibrous, double-walled sac that surrounds and protects the heart
(2 layers; with few mm of pericardial fluid in-between for friction free movement of heart muscles )
Pericardium is adherent to great vessels, esophagus, sternum, and pleurae (anchored to diaphragm)
Myocardium -> muscular wall of the heart that does the pumping
endocardium -> thin layer of endothelial tissue that lines the inner surface of the heart layers and valves
heart is actually 2 pumps with 2 sides separated by the septum; each having an atrium and a ventricle
(atrium is thinner walled and holds blood while the ventricles are thicker and pump blood)
Heart walls, Chambers, and valves (valves)
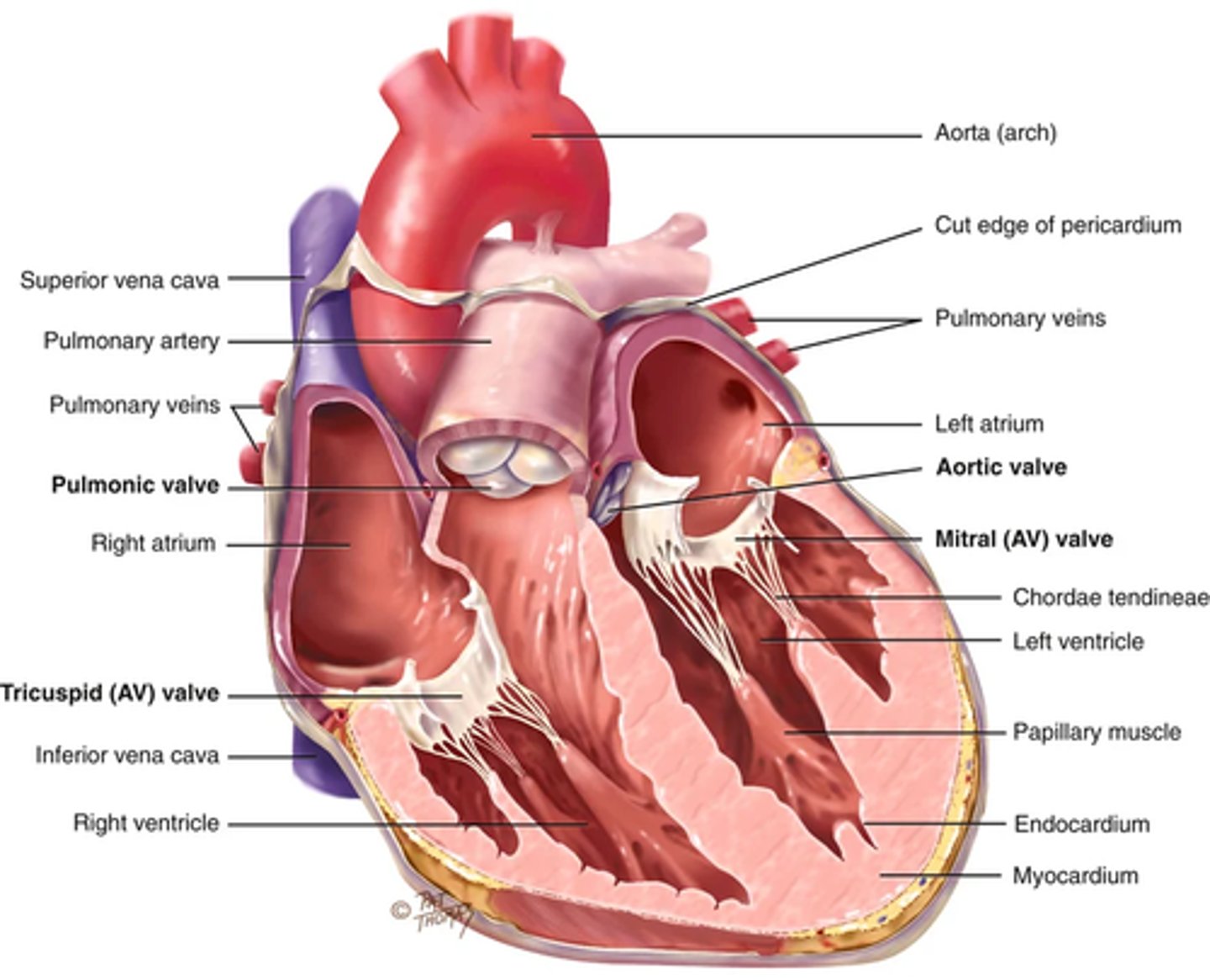
The purpose of the valves are to separate the 4 chambers and prevent blood from back flowing (unidirectional valves that open and close passively in response to pressure from blood)
2 atrioventricular (Av) valves that separate the a an v:
1) right AV valve - tricuspid
2) Left AV valve - Bicuspid or mitral valve
Valves thin leaflets are anchored by the collagenous fibers; Chordae tendinea; to the papillary muscles imbedded into the ventricle floor
AV valves open during hearts filling phase (DIASTOLE) and allow the ventricles to fill with blood
During the pumping phase (SYSTOLE) the AV valves close to precent regurgitation of blood back into the atria (papillary muscles contract so the leaflets close and make a perfect seal)
Semilunar valves (SL) -> between the ventricles and arteries (each valve has 3 cusps that look like half moons
1) Pulmonic valve in the right side of heart
2) aortic valve in the left side
(both open during pumping (systole)
SIDE NOTE ABOUT THE RIGHT SIDE OF THE HEART
There are no valves between the vena cava and the right atrium or between the pulmonary veins and the left atrium.
-abnormally high pressure in the left side of the heart gives a person symptoms of pulmonary venous congestion
-abnormally high pressure in the right side of the heart shows in the distended neck veins and abdomen.
Direction of blood flow
1. From liver to RA through inferior vena cava.
Superior vena cava drains venous blood from the head and upper extremities.
From RA venous blood travels through tricuspid valve to RV.
2. From RV venous blood flows through pulmonic valve to pulmonary artery.
Pulmonary artery delivers unoxygenated blood to lungs.
3. Lungs oxygenate blood.
Pulmonary veins return fresh blood to LA.
4. From LA arterial blood travels through mitral valve to LV.
LV ejects blood through aortic valve into aorta.
5. Aorta delivers oxygenated blood to bod
images on the heart filling and pressure changes
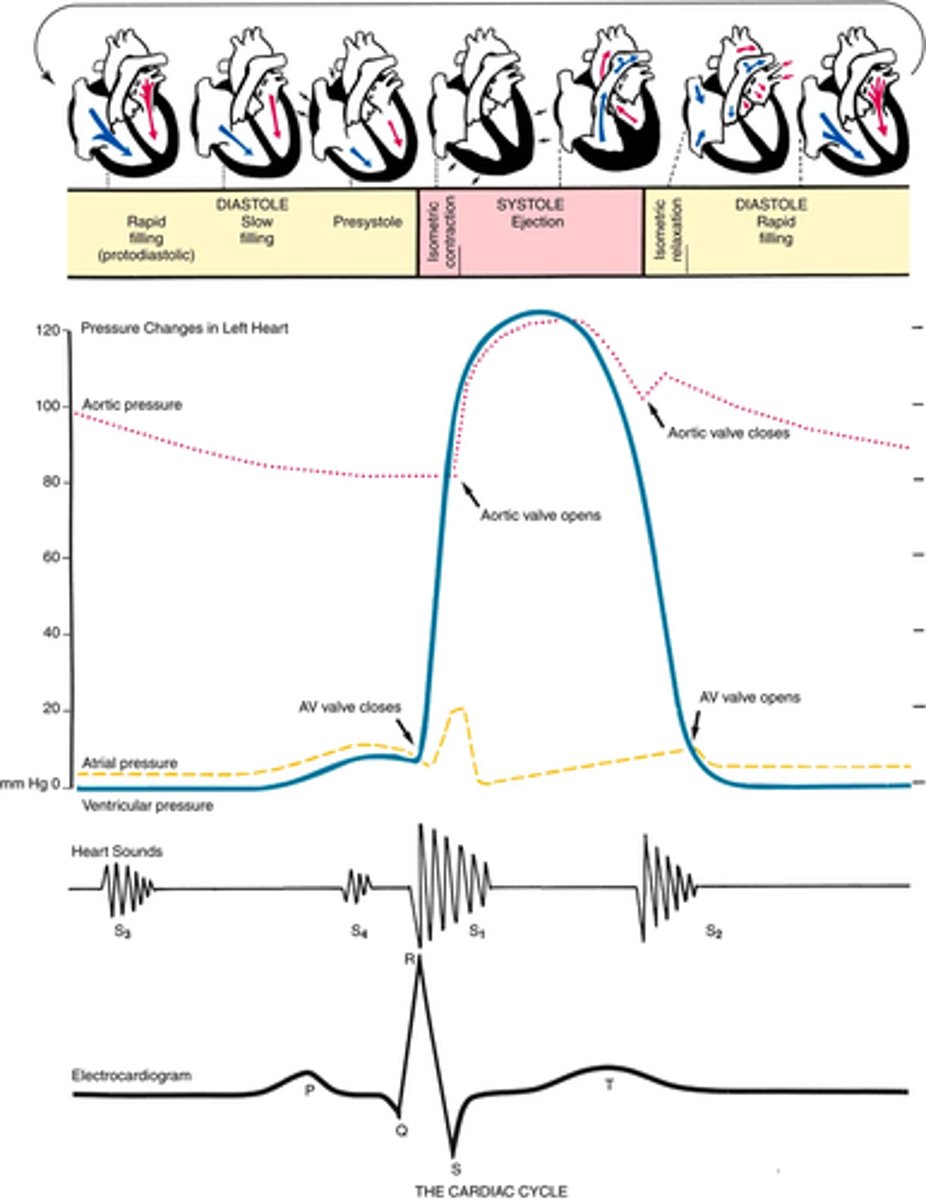
Cardiac cycle (phase 1 diastole )
Diastole -> ventricles are relaxed, and the AV valves (i.e., the tricuspid and mitral) are open
Atrial pressure is higher than ventricular so we have a passive floor of blood from A -> V
1st passive filling phase = early or pre-diastolic filling
towards end of diastole -> atria contract and push the last amount of blood (about 25% of stroke volume) into the ventricles -> THIS ACTIVE FILLING PHASE = PRESYSTOLE OR ATRIAL SYSTOLE
(atrial systole occurs during ventricular diastole)
Cardiac cycle (phase 2 systole)
blood now pumped into the ventricles -> Ventricular pressure is now higher than atrial pressure (MITRAL AND TRICUSPID SWING SHUT)
Closure of the AV valves = first heart sound S1
(prevents regurgitation of blood back into atria)
For a brief moment all valves are shut and the ventricular walls contract -> ISOMETRIC CONTRACTION -> builds pressure in the ventricles
once ventricular pressure > aortic pressure -> ventricular contents are ejected
(As blood is ejected the pressure in the ventricles fall, once ventricular pressure is less than aortic the aortic valve closes)
CLOSURE OF AORTIC/PULMONIC SEMILUNAR VALVE = S2 SECOND HEART SOUND AND END OF SYSTOLE
Cardiac cycle (phase 3 diastole again and events in the right/left side)
All 4 valves are closed and the ventricles relax
(isometric or isovolumetric relaxation)
While this is occurring the atria are filling with blood from the lungs (atrial pressure now higher than the relaxing ventricular and the AV valves open)
The left and right side of the heart mirror each other in movements
Only diff? -> Right sided pressure is less than the left sided pressure since less energy is needed to pump blood through pulmonary circulation
Also... events occur slightly later in the right side of the heart because of the route of myocardial depolarization -->
Bc of this there are actually 2 distinct components of S1 (we usually hear them together, but sometimes you can hear them separately)
In S1 -> the first component M1 (the mitral component) closes just before the T1 (tricuspid component)
In S2 -> The aortic closure (A2) occurs slightly before the pulmonic closure (p2)
The heart sounds: Normal heart sounds
First heart sounds (S1) -> occurs with closure of the AV valves and signals the start of systole
Mitral component (M1) slightly precedes the tricuspid component (T1) (sounds usually fused as 1)
(S1 can be heard all over the precordium but is usually loudest at the apex)
Second heart sound (s2) -> occurs with closure of semilunar valve and signals the end of systole
Aortic component of 2nd sound (A2) slightly precedes the pulmonic component (P2)
(S2 can be heard all over but is usually loudest at the base)
The heart sounds : Effect of respiration
Volume of left and right ventricular systole is about equal , but can be affected by respiration
(moRe to the Right heart,
Less to the Left )
During inspiration -> intrathoracic pressure is decreased -> more blood is pushed into the vena cava -> increasing venous return to he right side of the heart -> increased right ventricular strike volume
(This prolongs right ventricular systole and delays pulmonic valve closure)
On the left side - greater amount of blood is sequestered in the lungs during inspiration -> momentarily decreases the amount returned to the left side of the heart -> decreases left ventricular stroke volume
(decreased volume -> shorten left ventricular systole -> aortic valve closes a bit earlier )
When the aortic valve closes significantly earlier than the pulmonic valve we heart the 2 components separately (SPLIT S2)
heart sounds extra heart sounds: Third (s3) and fourth (s4) heart sounds
Third heart sound (S3): Diastole is usually a silent event; w/ some conditions ventricular filling creates vibrations that can be heard over the chest -> these vibrations = S3 heart sounds
(This occurs when the ventricles are resistant to filling during early rapid filling phase (protodiastole) -> This occurs immediately after s2 when the AV valves are open and atrial blood first pours into the ventricle)
S4 occurs at the end of diastole at pre-systole when the ventricles are resistant to filling
(the atria contracts and push blood into a non compliant ventricle -> S4 heart sounds that occur just before S1)
Extra heart sounds: Murmurs
Blood circulating through chambers and valves usually makes no noise
Noise (Murmurs) can occur from conditions that create turbulent blood flow and collision currents
(Murmurs are like a pile of stones in stream that create a noisy waterflow)
Murmur -> gentle, blowing, swooshing sound that can be heard on the chest wall.
Murmur causing conditions:
1. Velocity of blood increases (flow murmur) (e.g., in exercise, thyrotoxicosis)
2. Viscosity of blood decreases (e.g., in anemia)
3. Structural defects in the valves (a stenotic or narrowed valve, an incompetent or regurgitant valve) or unusual openings occur in the chambers (dilated chamber, septal defect)
Characteristics of all heart sounds (descriptors)
1. Frequency (pitch)—Heart sounds are described as high pitched or low pitched, although these terms are relative because all are low-frequency sounds, and you need a good stethoscope to hear them.
2. Intensity (loudness)—Loud or soft
3. Duration—Very short for heart sounds; silent periods are longer
4. Timing—Systole or diastole
heart conduction
Heart can contract by itself (independent of any body signals)
-contracts in response to an electrical current conveyed by a conduction system
Specialized cells in the Sinoatrial node (SA) (pacemaker) near the superior vena cava initiate an electrical impulse
Current then flows to AV node low in the atrial septum -> slight delay so atria has time to contract before ventricles -> impulse travels to bundle of HIS -> left and right bundle branches -> then through the ventricles
(signal stimulates heart to work)
On ECG: the haves are labels PQRST
P wave—Depolarization of the atria
PR interval—From the beginning of the P wave to the beginning of the QRS complex (the time necessary for atrial depolarization plus time for the impulse to travel through the AV node to the ventricles)
QRS complex—Depolarization of the ventricles
T wave—Repolarization of the ventricles
Pumping ability of the heart
Normal heart pumps 4-6L per minute throughout the body
Cardiac output = the volume of blood in each systole (called the stroke volume) times the number of beats per minute (heart rate). (SVxHR)
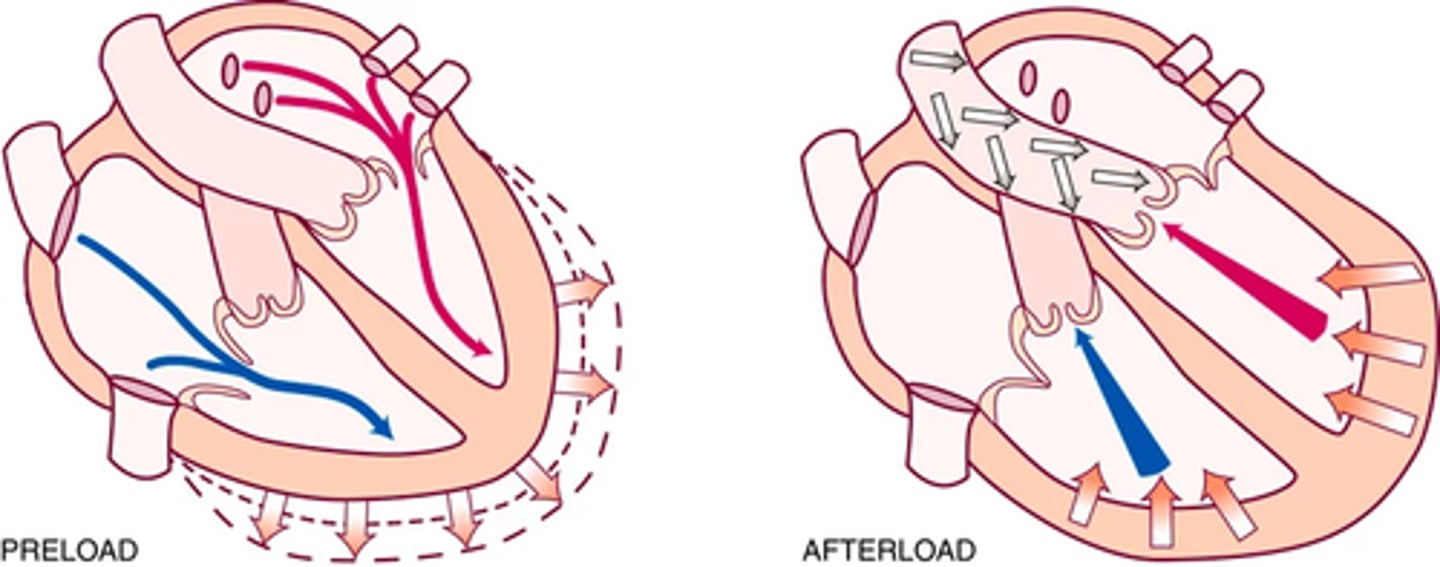
Pre-load volume -> the venous return that builds during diastole; the length to which the ventricular muscle is stretched at the end of diastole just before contraction
(volume of blood retuned to the ventricles -> bundles are stretched beyond their normal resting state to accommodate -> force of this stretch = pre load)
(THE STRONGER THE STRETCH THE STRONGER THE CONTRACTION WITH INCREASED STROKE VOLUME )
Afterload -> the opposing pressure the ventricles must generate to open the aortic valve against the higher aortic pressure
(ventricle must pump against the aortic pressure)
(Ventricular end diastolic pressure is 5-10mmHg where as the aorta is 70-80mmHg; ventricle muscles must tense (Isovolumetric contraction to build and overcome this pressure)
The neck vessels : carotid artery pulse
- A pressure wave generated by each systole pumping blood into the aorta.
Carotid artery = central artery
(Its timing closely coincides with ventricular systole)
Carotid artery -> located in the groove between the trachea and the sternomastoid muscle, medial to and alongside that muscle.
Characteristic of the wave form -> a smooth rapid upstroke, a summit that is rounded and smooth, and a downstroke that is more gradual and has a dicrotic notch caused by closure of the aortic valve (signified by d in the next flashcard image)
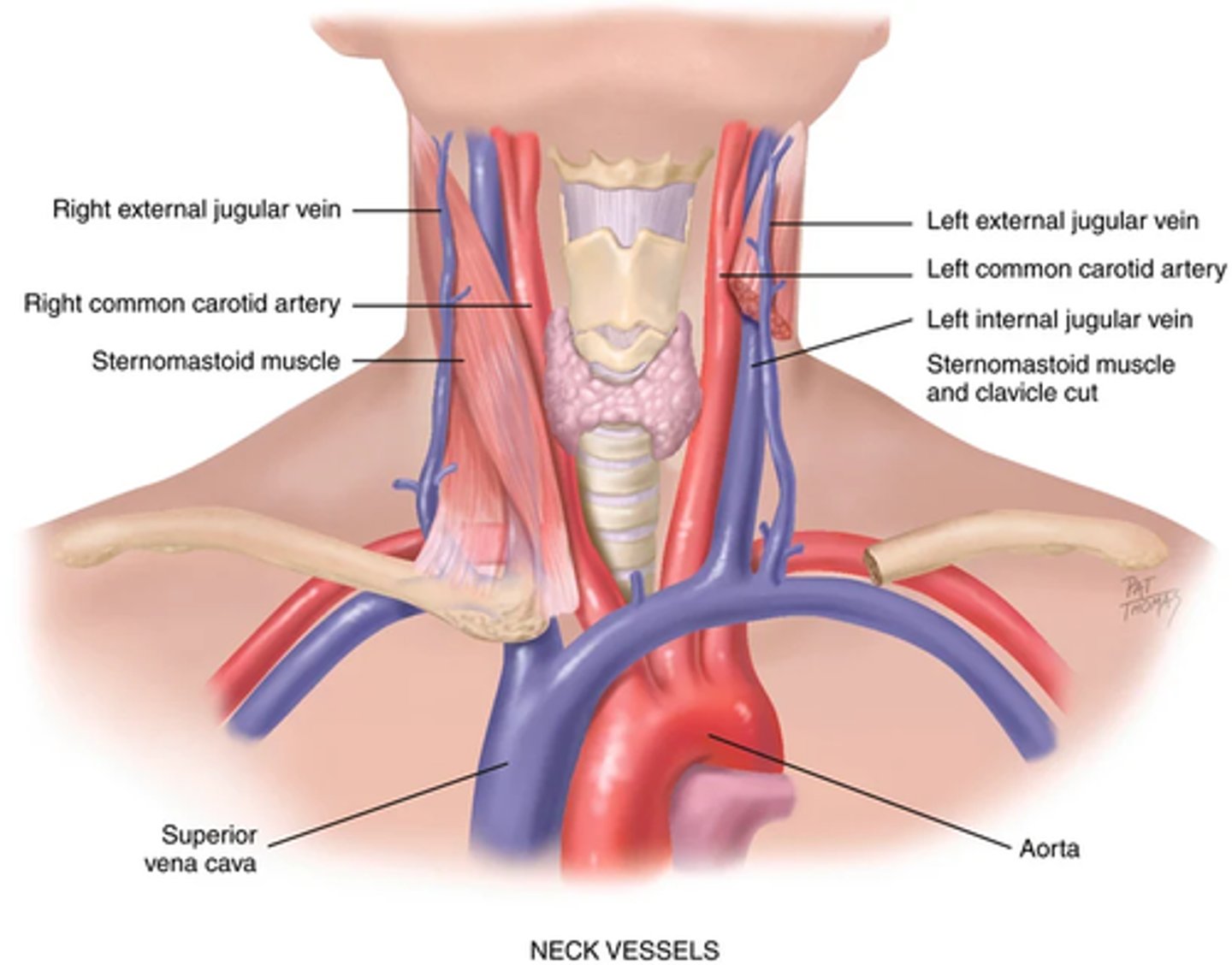
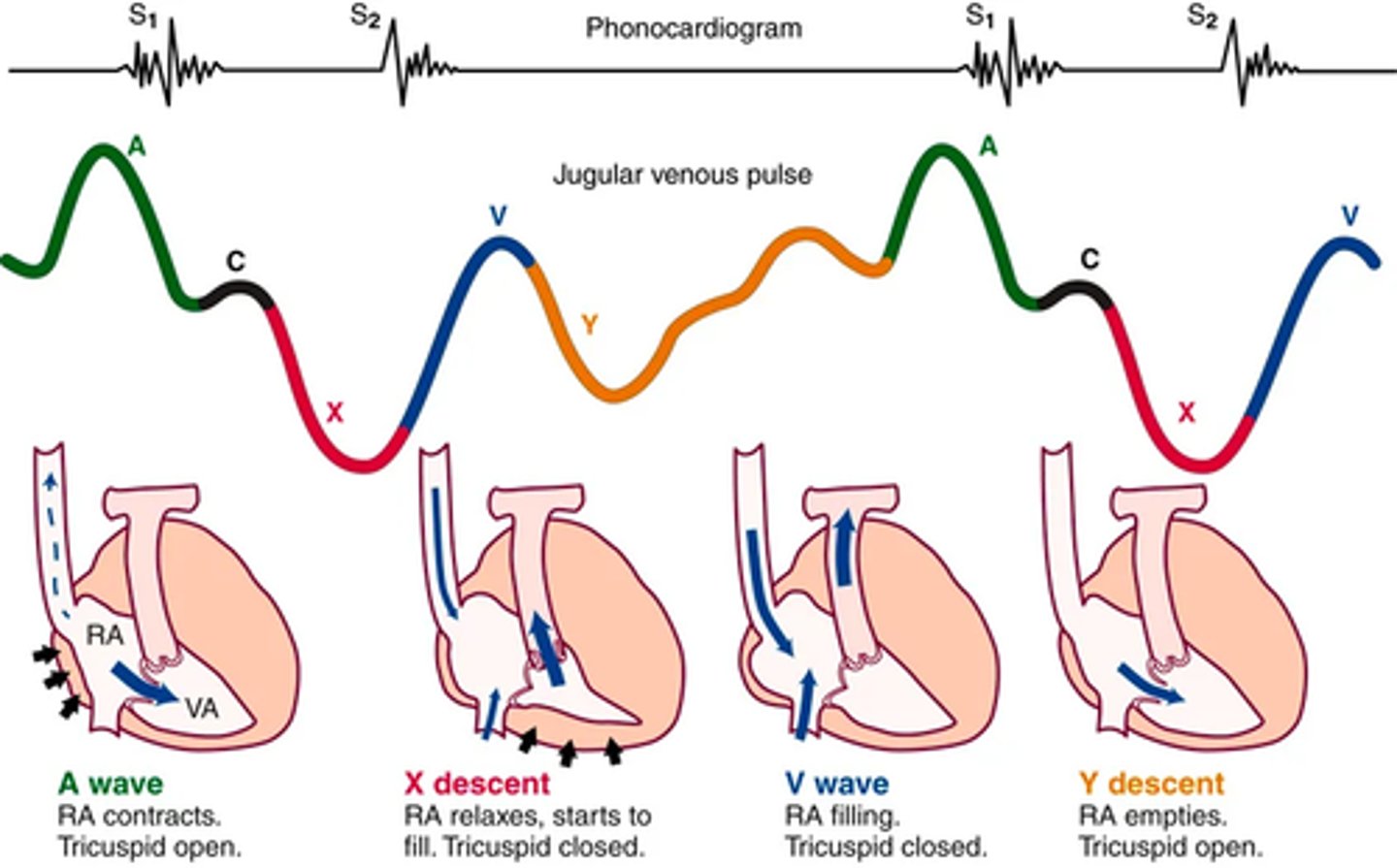
The neck vessels: Jugular venous pulse and pressure
Jugular veins -> empty unoxygenated blood directly into the superior vena cava
(no valves separate the atria and and vena cava; jugular vein gives info about right heart activity reflecting filling pressure and volume changes)
Volume/ pressure increase when right side of heart fails to pump efficiently -> revealed in jugular vein (becomes distended)
Internal jugular -> lies deep and medial to the sternomastoid muscle. (not usually visible)
External jugular -> vein is more superficial; it lies lateral to the sternomastoid muscle, above the clavicle.
The neck vessels: Jugular venous pulse and pressure pt 2
Jugular pulse is diff than the arterial pulse
(venous pulse results from a backwash, waveform caused by events upstream)
5 components:
A wave -> reflects atrial contraction because some blood flows backward to the vena cava during right atrial contraction
C wave -> or ventricular contraction, is backflow from the bulging upward of the tricuspid valve when it closes at the beginning of ventricular systole
X Descent -> shows atrial relaxation when the right ventricle contracts during systole and pulls the bottom of the atria downward.
V wave -> occurs with passive atrial filling because of the increasing volume in the right atria and increased pressure.
Y descent -> reflects passive ventricular filling when the tricuspid valve opens and blood flows from the RA to the RV.
Developmental competence: pregnancy
The CV system adapts to ensure adequate blood supply to the uterus and placenta, to deliver oxygen and nutrients to the fetus, and to allow the mother to function normally during this altered state.
2nd trimester -> plasma volume increases 40% and red cell mass by 30%. The increased circulating volume contributes to an increase in stroke volume.
(HR also increases by 15% to 25% during pregnancy and returns to baseline within the first 10 postpartum days)
the increases in stroke volume and HR increase the CO by 30% to 45%.
Despite the increased cardiac output, arterial BP decreases in pregnancy as a result of peripheral vasodilation. The BP drops to its lowest point during the 2nd trimester and rises after that
Developmental Competence: Aging Adult
difficult to isolate the "aging process" of the CV system per se because it is so closely interrelated with lifestyle, habits, and diseases.
lifestyle modifies the development of CV disease; smoking, diet, alcohol use, exercise patterns, and stress have an immense influence
Lifestyle also affects the aging process; cardiac changes once thought to be caused by aging are partially the result of the sedentary lifestyle accompanying aging
The aging adult: hemodynamic changes with aging pt 1 (systolic BP and arteriosclerosis)
increase in systolic BP, termed isolated systolic hypertension
Caused by -> thickening and stiffening of the large arteries, which in turn are caused by collagen and calcium deposits in vessel walls and loss of elastic fibers.
Stiffening aka Arteriosclerosis -> an increase in pulse wave velocity because the less compliant arteries cannot store the volume ejected.
No changes in heart size, but left ventricular wall thickness increases.
(adaptive mechanism to accommodate the vascular stiffening that creates an increased workload on the heart.)
The aging adult: hemodynamic changes with aging pt 2 (Diastolic Bp and cardiac output augmentation)
Diastolic BP may decrease after the 5th decade.
(combine with rising systolic -> greater difference in pulse pressure)
There is a decreased ability of the heart to augment cardiac output with exercise. shown by a decreased maximum heart rate with exercise and diminished sympathetic response.
Non-cardiac factors leading to a decrease in maximum work performance with aging -> decrease in skeletal muscle performance, increase in muscle fatigue, increased sense of dyspnea
(aerobic exercise conditioning modifies many of the aging changes in CV function.)
The aging adult: Dysrhythmias
presence of supraventricular and ventricular dysrhythmias increases with age.
Ectopic beats common in old heads; usually asymptomatic in healthy old heads
(may compromise cardiac output and BP when disease is present.)
Tachydysrhythmias may not be tolerated as well in older people.
(myocardium is thicker and less compliant, and early diastolic filling is impaired at rest)
-> may not tolerate a tachycardia as well because of shortened diastole.
tachydysrhythmias may further compromise a vital organ whose function has already been affected by aging or disease.
The aging adult: Electrocardiograph
Age related ECG changes due to histologic changes in Conduction system:
-Prolonged P-R interval (first-degree AV block) and prolonged Q-T interval, but the QRS interval is unchanged.
-Left axis deviation from age-related mild LV hypertrophy and fibrosis in left bundle branch.
-Increased incidence of bundle branch block.
The changes don't seem severe, but its a fact that CVD (cardiovascular disease)increases with age
CVD -> leading cause of death in those ages 65 years and older
lifestyle habits (smoking, chronic alcohol use, obesity, lack of exercise, diet) play a significant role in the acquisition of heart disease.
increasing the physical activity of older adults—even at a moderate level—shows a reduced risk of death from CVDs and respiratory illnesses.
Genetics and environment
Cardiovascular disease (CVD) remains the number one cause of death worldwide.
(1990-2019 global CVD prevalence doubled to 523 million people)
burden of CVD—in monetary cost, as well as death and disability—is staggering, especially in low- and middle-income countries
CVD risk is determined by a complex interplay between genetic and lifestyle factors.
Genetic risk of just coronary artery disease (CAD) is influenced by more than 160 loci in the genome.
Loci contribute to -> lipid metabolism, blood pressure (BP) regulation, inflammation, and vascular remodeling
Despite genetics, lifestyle has a huge influence on CVD risk
Among persons with a high genetic risk, adopting a favorable lifestyle is associated with a 46% lower risk of CAD events than is an unfavorable lifestyle.
4 major life style health factors -> no current smoking, no obesity (i.e., body mass index < 30 kg/m2), physical activity at least once per week, and a healthy diet
Genetics and environment pt 2
adopting a healthy lifestyle is associated with a significant drop in the risk of CAD events.
2019 Guideline on Primary Prevention of CVD, the American College of Cardiologists and American Heart Association recommend that all adults ages 40 to 75 should undergo 10-year atherosclerotic cardiovascular disease (ASCVD) risk estimation.
Risk calculator includes -> factors in age; sex; race; total and high-density lipoprotein serum cholesterol; systolic blood pressure; and personal history of diabetes, smoking, and treatment for hypertension.
Contributing risk factors -> family history of premature ASCVD, metabolic syndrome, chronic kidney disease, chronic inflammatory conditions, high-risk race/ethnicity, and other concerning findings from the lipid profile
(as well as premature menopause and history of pregnancy related cardiovascular complications)
Genetics and environment: hypertension
Untreated hypertension -> direct damage to the arterial system
(hypertension also contributes to CAD; it accelerates the process of atherosclerosis; it increases the workload on the heart, and it increases the oxygen demand on the heart already compromised by atherosclerosis.)
Risk by race -> highest in African Americans, 58.3% and 57.6% of non-Hispanic Black men and women; 51% and 40.5% among non-Hispanic White men and women; 51% and 42.1% among Asian men and women; and 50.6% and 40.8% among Hispanic men and women
Black Americans develop high BP earlier in life, and their average BPs are much higher than in White Americans. This results in a greater rate of stroke, death from heart disease, and end-stage kidney disease for Black Americans.
(because of social detriments of health)
Genetics and environment: Smoking and tobacco use
Smoking -> increased oxygen demand on the heart and causes concomitant decresae in oxygen supply, by an activation of platelets and fibrinogen, and by an adverse change in the lipid profile.
(Tobacco products, whether cigarette smoking or use of e-cigarettes, also increase heart rate and blood pressure)
use of tobacco products has declined, tobacco use remains a significant health concern for select groups. For example, e-cigarette usage is increasing, especially among adolescents. Prevalence of e-cigarette use among adolescents in 2019 was up to 27.4%, compared with just 1.5% in 2011.
Genetics and environment: elevated cholesterol, type 2 diabetes, and nutrition/diet
High levels of low-density lipoprotein (LDL, or the "bad" cholesterol) add to the lipid core of plaque formation in coronary and carotid arteries, which results in MI and stroke.
(persons with intermediate or greater 10 year ASCVD risk scores; people age 40-75 with diabetes; people ages 20-75 with LDL of 190mg/Dl are considered for satin therapy)
Diabetes management is broad, including a heart-healthy diet that promotes glycemic control, adequate physical activity, and weight management.
(administration of metformin, an oral diabetes medication that improves insulin resistance, may further reduce ASCVD risk)
Healthful diets are rich in vegetables, fruits, legumes, nuts, whole grains, and fish and contain limited refined carbohydrates, processed meats, sugary drinks, saturated fats, trans fats, cholesterol, and sodium.
Genetics and environment: Exercise, obesity, and sex/gender differences in CVD
At least 150 minutes of moderate intensity aerobic exercise can reduce ASCVD risk
Overweight (BMI = 25 to 29.9 kg/m2) and obesity (BMI ≥30 kg/m2) both contribute to ASCVD risk
women tend to have smaller coronary arteries than men.
Gender differences are more complex, involving physiologic differences, as well as factors such as ethnicity, culture, and socioeconomic status.
Genetics and environment: risk
health care providers tend to perceive women as being at lower risk for CAD than men, which is not true.
(Smoking, hypertension, and diabetes have a more prominent effect on ASCVD risk in women than men; also higher risk from prior pregnancy complications such as hypertension, diabetes, and preeclampsia)
Women may also have slightly different risk factors (e.g., increased risk of chronic inflammatory conditions
(Black women = highest risk for MI; multiple factors such as increased prevalence of CAD risk equivalents (e.g., diabetes) and a lower likelihood of being treated and educated according to evidence-based guidelines.)
Genetics and environment: Presentation and diagnosis
Women report different symptoms of acute coronary syndrome (e.g., shortness of breath, nausea, and fatigue in addition to chest discomfort
Women also tend to have blockages in smaller coronary arteries (versus larger/main coronary arteries in men), which contributes to differences in presentation.
Differences in symptomatology contribute to a delay in women seeking care, and health care providers may not as readily recognize the symptoms of MI, both of which contribute to delays in diagnosis and treatment
Genetics and environment: treatment, Outcomes, and SGM
Treatment -> Women may be treated for MI differently than men; women are less likely to undergo invasive testing and treatment once MI is suspected and are less likely to be treated with evidence-based medical therapy after an MI (e.g., appropriate antiplatelet therapy, statins, and blood pressure management)
outcomes -> Even considering that women who have MI are likely to be older than men, there are still concerning differences in outcome.
(after MI, women have higher mortality, bleeding risk, new-onset heart failure, and reduced quality of life.)
SGM -> Transgender and gender-nonconforming (TGNC) people have a higher prevalence of cardiovascular disease.
(social detriments?)
Subjective data: pt 1) Chest pain
1. Chest pain. Any chest pain or tightness
Onset -> how long? When? first time?
Location-> Where? radiates?
Character -> crushing, stabbing, burning, heavy etc (chest pain can have gastro, musculo, or pulmonary cause)
Rationale -> Angina, an important cardiac symptom, occurs when the heart's own blood supply cannot keep up with metabolic demand.
(usually clenched fist sign is characteristic of angina)
Pain brough on by activity? What type? rest? after eating? during sex?
Any associated symptoms: sweating, ashen gray or pale skin, heart skips beat, shortness of breath, nausea or vomiting, racing of heart?
rationale -> Diaphoresis, cold sweats, pallor, grayness. Palpitations, dyspnea, nausea, tachycardia, fatigue.
Pain made worse by moving arms, breathing, lying flat, palpating?
Rationale -> Differentiate pain of cardiac versus noncardiac origin. Pain with breathing (pleuritic), affected by position, reproduced with palpation, or unrelated to exertion is less likely cardiac in origin.
Subjective data pt 2 : dyspnea
2) . Dyspnea. Any shortness of breath?
-What activity leads to dyspnea?
-Onset? unexpected onset?
-Duration? constant or comes and goes
-affected by position? lying down?
Rationale -> Dyspnea on exertion (DOE)—Quantify exactly (e.g., DOE after walking two level blocks). Paroxysmal, constant, intermittent?
-awaken from sleep at night?
(Paroxysmal nocturnal dyspnea (PND) occurs with heart failure; Lying down increases volume of intrathoracic blood, and the weakened heart cannot accommodate the increased load.)
-Does SOB interfere with ADLS?
Subjective data pt 3 ( 3. orthopnea and 4. cough)
3) Orthopnea? how many pillows do you use when sleeping?
Rationale -> need to be more upright to breath; note the # of pillows used
4) cough?
-Duration? how long have you had it ?
-Frequency? is it related to time of day?
-Type? Dry, hacking, barky, hoarse, congested?
Do you cough up mucus?
-Color? odor? blood tinged? Consistency
Rationale -> Sputum production, mucoid or purulent. Hemoptysis is often a pulmonary disorder but also occurs with mitral stenosis.
-Associated with activity? Position, anxiety, talking?
-Does activity make it better or worse?
-Relieved by rest or medication?
Subjective data pt 4 (5) fatigue 6) Cyanosis or pallor)
5) fatigue? tire easily? can't keep up with kids
Rationale -> Unusual fatigue is a top prodromal MI symptom for women
-Onset? When start? Sudden or gradual? recent changes in energy?
-fatigue related to time of day?
Rationale -> Fatigue from decreased cardiac output is worse in the evening, whereas fatigue from anxiety or depression occurs all day or is worse in the morning.
6) Cyanosis or pallor?
Rationale -> Cyanosis or pallor occurs with MI or low cardiac output states as a result of decreased tissue perfusion.
Subjective data pt 5 (7) edema 8) Nocturia))
7) Edema? any swelling of feet and legs
-Onset? when first notice? recent changes?
Rationale ->Edema is dependent when caused by heart failure.
-What time of day does the swelling occur?
tighter shoes at end of day?
Rationale -> Cardiac edema is worse at evening and better in morning after elevating legs all night.
-How much swelling? both legs equally?
Rationale -> Cardiac edema is bilateral; unilateral swelling has a local vein cause.
-Swelling decrease with rest, elevation, or sleep?
-Any associated SOB? does it occur before or after swelling?
8) Nocturia? do you awaken at night with urge to urinate? how long this occur? any recent changes?
Rationale ->
Nocturia—Recumbency at night promotes fluid resorption and excretion; this occurs with heart failure in the person who is ambulatory during the day.
Subjective data pt 6 (9) past cardiac history and 10) family cardiac history)
9) past cardiac history?
-history of: hypertension, elevated cholesterol or triglycerides, heart murmur, congenital heart disease, rheumatic fever or unexplained joint pains as child, recurrent tonsilitis or anemia?
-Ever have heart disease? when was this treated?
-Last ECG, stress ECG, serum cholesterol, other test?
10) Family cardiac history?
family history of hypertension, high cholesterol, obesity, diabetes, sudden cardiac death, CAD, or stroke at a younger age?
Subjective data pt 7 (11) Patient centered care)
11. Patient-centered care (cardiac risk factors
Nutrition: Please describe your usual daily diet. (Note if this diet is representative of the basic food groups, the amount of calories, cholesterol, and any additives such as salt.) What is your usual weight? Has there been any recent change?
Smoking: Do you smoke cigarettes or other tobacco? At what age did you start? How many packs per day? For how many years have you smoked this amount?
Alcohol: How much alcohol do you usually drink each week, or each day? When was your last drink? How many drinks during that episode?
Exercise: What is your usual amount of exercise each day or week? What type of exercise (state type or sport)? If a sport, what is your usual amount
Drugs: Do you take any antihypertensives, beta-blockers, calcium channel blockers, digoxin, diuretics, aspirin/anticoagulants, over-the-counter or street drugs?
Rationale -> risk factors for CAD -
Collect data regarding elevated cholesterol, elevated BP, blood sugar levels above 100 mg/dL or known DM, obesity, cigarette smoking, low activity level, and length of any hormone replacement therapy for postmenopausal women.
Guidelines now recommend against starting low-dose aspirin for primary prevention of atherosclerotic CVD, except in select, high-risk individuals age 40 to 59 years when the risk of MI is greater than the risk of GI bleeding
Additional history for pregnancy
1. Have you had any high BP during this or earlier pregnancies?
• What was your usual BP level before pregnancy? How has your BP been monitored during the pregnancy?
• If high BP, what treatment has been started?
• Any associated symptoms: weight gain; protein in urine; swelling in feet, legs, or face?
2. Have you had any faintness or dizziness with this pregnancy?
Additional history for the aging adult
1. Do you have any known heart or lung disease: hypertension, CAD, chronic emphysema, or bronchitis?
• What efforts to treat this have been started?
• Usual symptoms changed recently? Does your illness interfere with activities of daily living?
2. Do you take any medications for a heart or BP condition? Aware of side effects? Have you recently stopped taking your medication? Why?
3. Environment: Does your home have any stairs? How often do you need to climb them? Does this have any effect on activities of daily living?
Objective data pt 1) Palpate the carotid artery
-Located central to the heart
-Palpate each carotid artery medial to the sternomastoid muscle in the neck (no excessive pressure on carotid sinus area higher in neck)
-excessive vagal stimulation here could slow down the heart rate, especially in older adults
(only palpate 1 at a time)
-Feel the contour and amplitude of the pulse; normally smooth contour with a brisk upstroke and a slower downstroke with moderate strength
ABNORMAL -> Carotid sinus hypersensitivity is the condition in which pressure over the carotid sinus leads to a decreased heart rate, decreased BP, and cerebral ischemia with syncope.
-Diminished carotid pulse feels small and weak (decreased stroke volume as in cardiogenic shock).

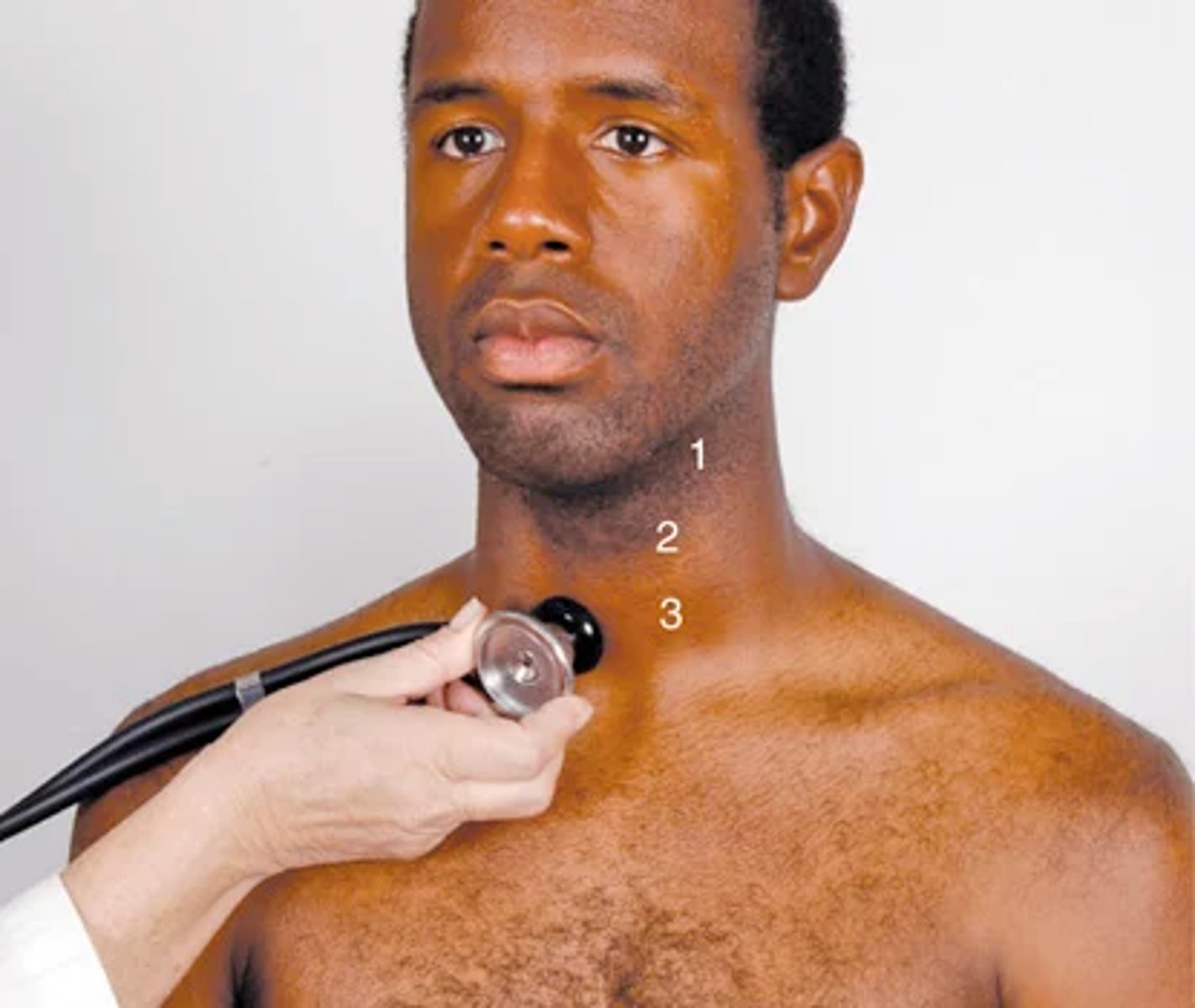
Objective data pt 2) Auscultate the carotid artery
older people who show signs of CVD -> auscultate each carotid artery for the presence of a bruit
(blowing swishing sound indicating blood flow turbulence)
Abnormal -> indicates turbulence from a local vascular cause and is a marker for atherosclerotic disease.; only moderately correlates with carotid artery stenosis
keep the neck in neutral position, lightly apply the bell over the carotid at 3 levels
(1) the angle of the jaw, (2) the midcervical area, and (3) the base of the neck
-Do not compress artery or you could create an artificial bruit or compromise circulation
-Ask the person to take a breath, exhale, and hold it briefly while you listen so tracheal breath sounds do not mask or mimic a carotid artery bruit
(can often hear normal heart sounds in the neck do not confuse with bruit)
ABNORMAL -> carotid bruit is audible when the lumen is occluded by 1⁄2 to 2⁄3; sound increases as atherosclerosis worsens until 2/3 occlusion
-After that, bruit loudness decreases. When the lumen is completely occluded, the bruit disappears
A murmur sounds much the same but is caused by a cardiac disorder.
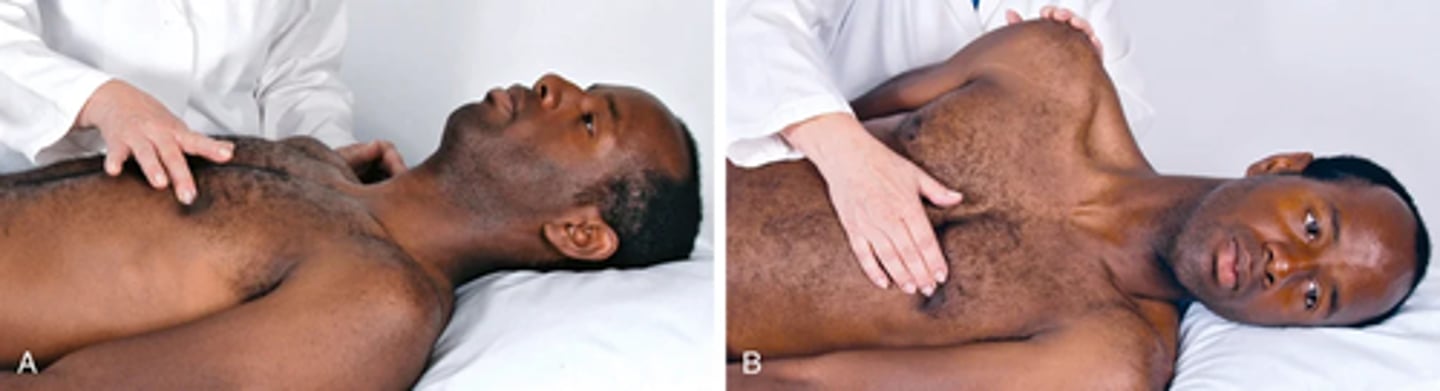
Objective data pt 3) inspect the jugular venous pulse
can assess central venous pressure (CVP) and thus judge the heart's efficiency as a pump and the intravascular volume status
stand on right side since veins on that side go right to heart -> can see the top of the external jugular vein distention overlying the sternomastoid muscle or the pulsation of the internal jugular vein in the sternal notch
Position the person supine anywhere from a 30- to a 45-degree angle, wherever you can best see the top of the vein or pulsations.
(higher the venous pressure the higher the position needs to be)
Remove the pillow to avoid flexing the neck; the head should be in the same plane as the trunk.
(make sure head is turned to the opposite side as examination with strong tangential lighting)
Note the external jugular veins overlying the sternomastoid muscle. In some people the veins are not visible at all
(As the person is raised to a sitting position, these external jugulars flatten and disappear, usually at 45 degrees.)
look for pulsations of the internal jugular veins in the area of the suprasternal notch or around the origin of the sternomastoid muscle around the clavicle
You must be able to distinguish internal jugular vein pulsation from that of the carotid artery.
ABNORMAL ->
Unilateral distention of external jugular veins is caused by local cause (kinking or aneurysm).
Full distended external jugular veins above 45 degrees signify increased CVP as with heart failure.
Objective data pt 4) Characteristics of jugular versus carotid pulse
1) location
internal Jugular -> Lower, more lateral, under or behind the sternomastoid muscle
Carotid -> Higher and medial to this muscle
2) Quality
Internal jugular -> Undulant and diffuse; two visible waves per cycle
Carotid -> Brisk and localized; one wave per cycle
3) Respiration
Internal jugular -> Varies with respiration; its level descends during inspiration when intrathoracic pressure is decreased
Carotid -> Does not vary
4) Palpable
Internal jugular pulse -> No
Carotid pulse -> Yes
5) Pressure
Internal jugular -> Light pressure at the base of the neck easily obliterates
Carotid pulse -> No change
6) Position of person
Internal jugular -> Level of pulse drops and disappears as person is brought to sitting position
Carotid pulse -> Unaffected
Objective data pt 5) The precordium; inspecting the anterior chest
Inspect the anterior chest -> Arrange tangential lighting to accentuate any flicker of movement.
Pulsations. You may or may not see the apical impulse, the pulsation created as the left ventricle rotates against the chest wall during systole.
(when visible it will be seen in the 4th-5th intercostal space or inside the midclavicualr line; easier to see with thinner chest walls)
ABNORMAL -> A heave or lift is a sustained forceful thrusting of the ventricle during systole. It occurs with ventricular hypertrophy as a result of increased workload.
(right ventricular heave is seen at the sternal border; a left ventricular heave is seen at the apex)
Objective data pt 6) the precordium; Palpate the apical impulse
Localize the apical impulse precisely by using one finger pad -> ask person to exhale and hold it to help examiner locate pulse
(may have to have person roll midway to left to find it)
You feel it best at the end of expiration when the heart is closest to the chest wall; then it moves quickly away from your finger.
Note: Location—The apical impulse should occupy only one interspace, the 4th or 5th, and be at or medial to the midclavicular line
-Size—Normally 1 × 2 cm, best measured in left lateral position
-Amplitude—Normally a short, gentle tap
-Duration—Short; normally occupies only first half of systole
ABNORMAL -> Cardiac enlargement:
Left ventricular dilation (volume overload) displaces impulse down and to left and increases size more than one space. A diameter of ≥4 cm is likely a dilated hear
(occurs with heart failure and cardiomyopathy)
A sustained impulse with increased force and duration but no change in location occurs with left ventricular hypertrophy and no dilation (pressure overload)
The apical impulse is palpable in the supine position in 25% to 40% of adults and in the left lateral position in 50% to 73% of adults
(difficult with obesity or thick chest walls)
With high cardiac output states (anxiety, fever, hyperthyroidism, anemia), the apical impulse increases in amplitude and duration.
Abnormal -> Not palpable with pulmonary emphysema because of overriding lungs.
Objective data pt 7) Palpate across the precordium
Using the palmar aspects of your four fingers, gently palpate the apex, the left sternal border, and the base, searching for any other pulsation
(usually none occur; if any note the timing)
ABNORMAL -> A thrill is a palpable vibration. It feels like the throat of a purring cat. The thrill signifies turbulent blood flow and directs you to locate the origin of loud murmurs.
absence of a thrill does not rule out the presence of a murmur.
Accentuated first and second heart sounds and extra heart sounds also may cause abnormal pulsations.
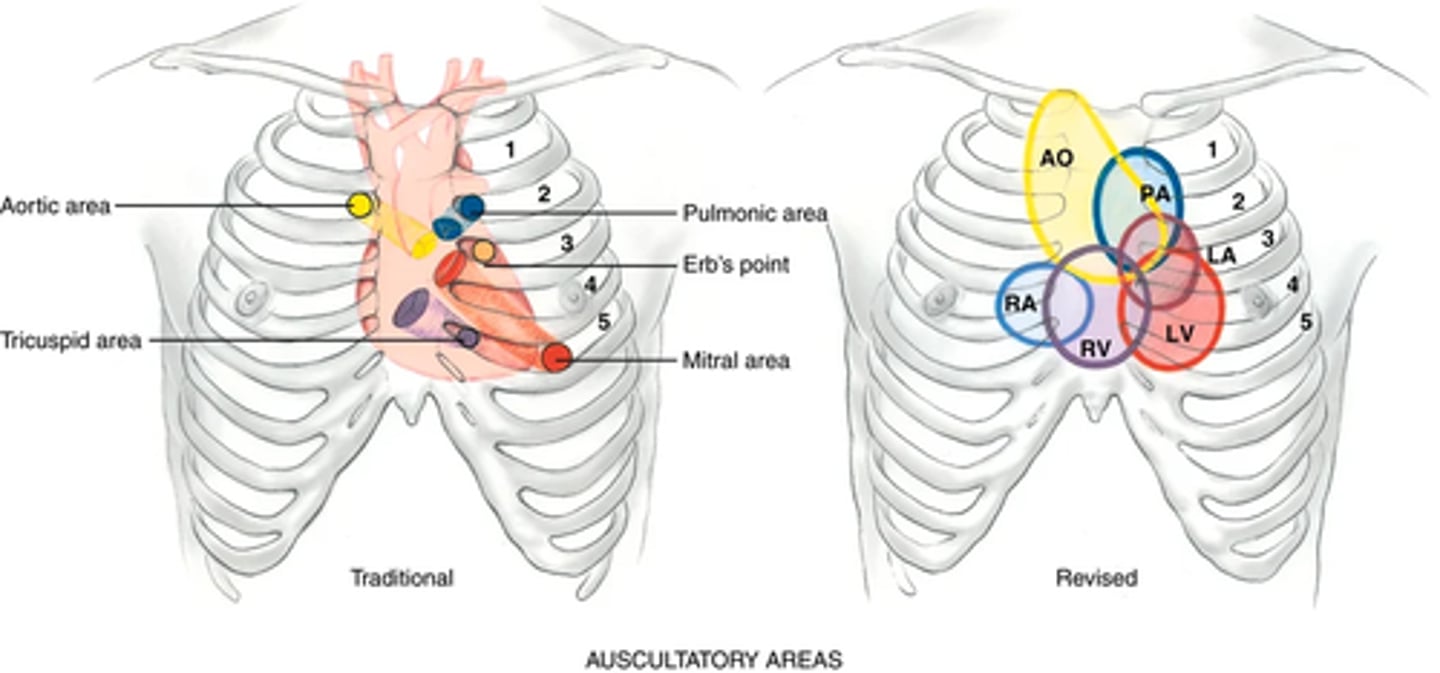
Objective data pt 8) Auscultation pt 1
Identify the auscultatory areas where you will listen. These include the four traditional valve "areas" (not over actual areas just where the sounds are best heard)
Valve areas ->
• Second right interspace—Aortic valve area
• Second left interspace—Pulmonic valve area
• Left lower sternal border—Tricuspid valve area
• Fifth interspace at around left midclavicular line—Mitral valve area
Do not limit your auscultation to only four locations. Sounds produced by the valves may be heard all over the precordium.
(earn to inch your stethoscope in a rough Z pattern, from the base of the heart across and down and over to the apex.)
Recall the characteristics of a good stethoscope
(Clean the endpieces with an alcohol wipe; you will use both endpieces.)
Make sure that your earpieces fit snugly and are aimed forward, toward your nose, to avoid air leak
(completely quiet room)
Objective data pt 8) Auscultation pt 2
Before you begin, alert the person: "I always listen to the heart in a number of places on the chest. (does not mean anything is wrong)
After you place the stethoscope, try closing your eyes briefly to tune out any distractions. Concentrate and listen selectively to one sound at a time. (there will be at minimum 2 heart sounds in less than 1 second, maybe 3 or 4)
Cannot process everything at once ; Begin with the diaphragm endpiece and use the following routine:
(1) note the rate and rhythm
(2) identify S1 and S2
(3) assess S1 and S2 separately
(4) listen for extra heart sounds
(5) listen for murmurs.
Objective data pt 8) Auscultation pt 3 (NOTE RATE AND RYTHM)
Note the rate and rhythm: rate ranges normally from 50 to 95 beats/min
-The rhythm should be regular, although sinus arrhythmia occurs normally in young adults and children.
(rhythm varies with the person's breathing, increasing at the peak of inspiration and slowing with expiration.)
ABNORMAL->
Premature beat—An isolated beat is early, or a pattern occurs in which every third or fourth beat sounds early.
Irregularly irregular—No pattern to the sounds; beats come rapidly and at random intervals as in atrial fibrillation.
Objective data pt 8) Auscultation pt 4 NOTE RATE AND RHYTHM
When you notice any irregularity, check for a pulse deficit by auscultating the apical beat while simultaneously palpating the radial pulse.
Count a serial measurement (one after the other) of apical beat and radial pulse.
Normally every beat you hear at the apex should perfuse to the periphery and be palpable.
The two counts should be identical. When different, subtract the radial rate from the apical, and record the remainder as the pulse deficit.
ABNORMAL -> A pulse deficit signals a weak contraction of the ventricles; it occurs with atrial fibrillation, premature beats, and heart failure.
Objective data pt 8) Auscultation pt 5 (IDENTIFY S1 AND S2)
Identify S1 and S2. This is important because S1 is the start of systole and thus serves as the reference point for the timing of all other cardiac sounds.
must learn to distinguish systole from diastole before you can attach meaning to all other sounds.
you can identify S1 instantly because you hear a pair of sounds close together (lub-dup) and S1 is the first of the pair.
(works except in the case of tachydysrhythmia; rates over 100 bpm)
Diastolic filling time shortened and beats are too close to distinguish
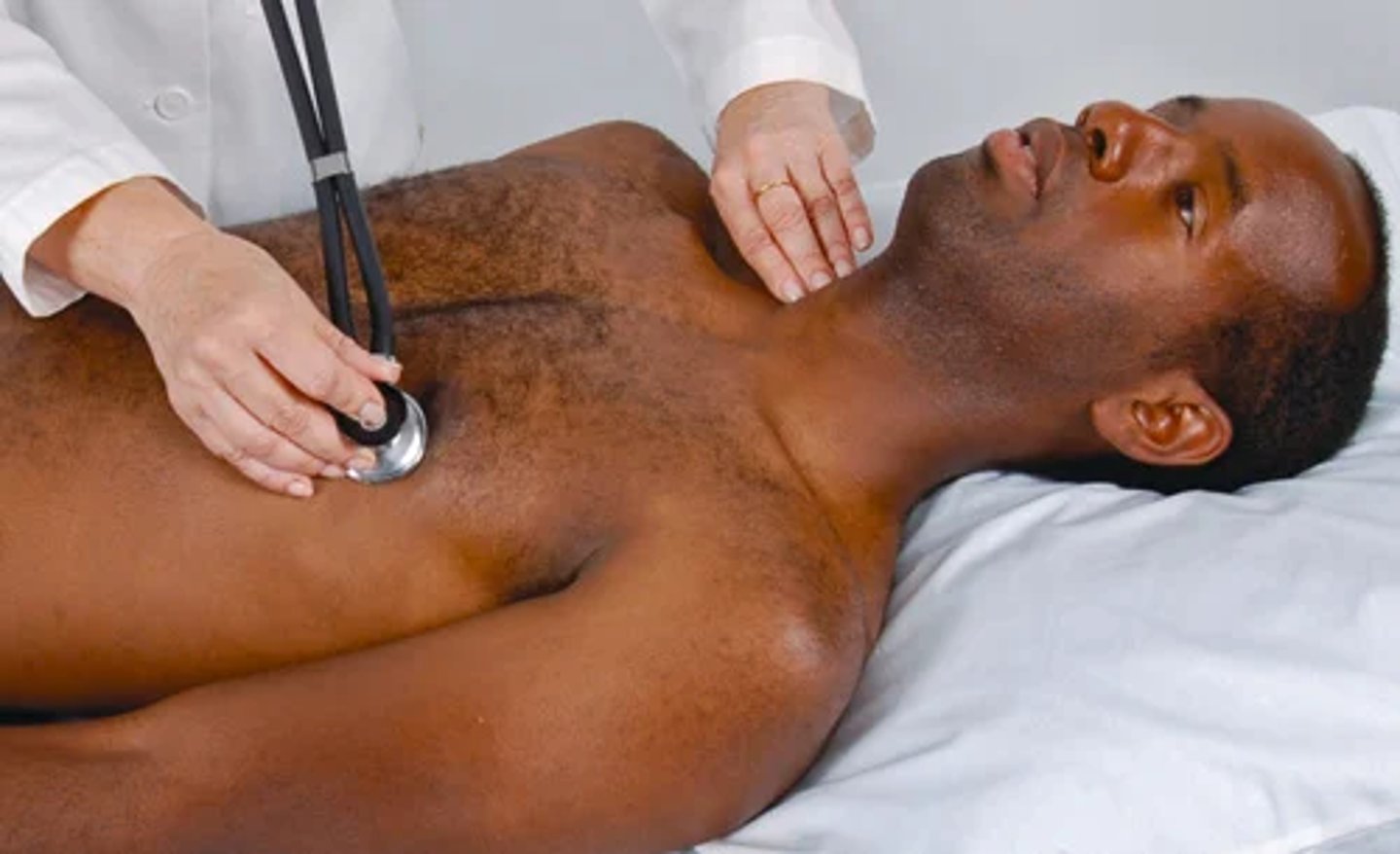
Objective data pt 8) Auscultation pt 5 more on distinguishing S1 and S2
• S1 is louder than S2 at the apex; S2 is louder than S1 at the base.
• S1 coincides with the carotid artery pulse. Feel the carotid gently as you auscultate at the apex; the sound you hear as you feel each pulse is S1 (Fig. 20.20).
• S1 coincides with the R wave (the upstroke of the QRS complex) if the person is on an ECG monitor.
Listen to S1 and S2 Separately. Note whether each heart sound is normal, accentuated, diminished, or split. Inch your diaphragm across the chest as you do this.
Objective data pt 8) Auscultation pt 6 First heart sounds (S1)
First Heart Sound (S1) -> Caused by closure of the AV valves, S1 signals the beginning of systole.
(loudest at the apex)
(Sometimes the two sounds are equally loud at the apex because S1 is lower pitched than S2.)
ABNORMAL ->
-Causes of accentuated or diminished S1
-Both heart sounds are diminished with conditions that place an increased amount of tissue between the heart and your stethoscope: emphysema (hyperinflated lungs), obesity, pericardial fluid.
-Can hear S1 with diaphragm in any position and equally well on inspiration and expiration
(A split S1 is normal, but it occurs rarely.)
Split S1 -> means that you are hearing the mitral and tricuspid components separately.
Audible in tricuspid valve area, the left lower sternal border
(split is very rapid <0.03 seconds apart )
Objective data pt 8) Auscultation pt 7 Second heart sound S2
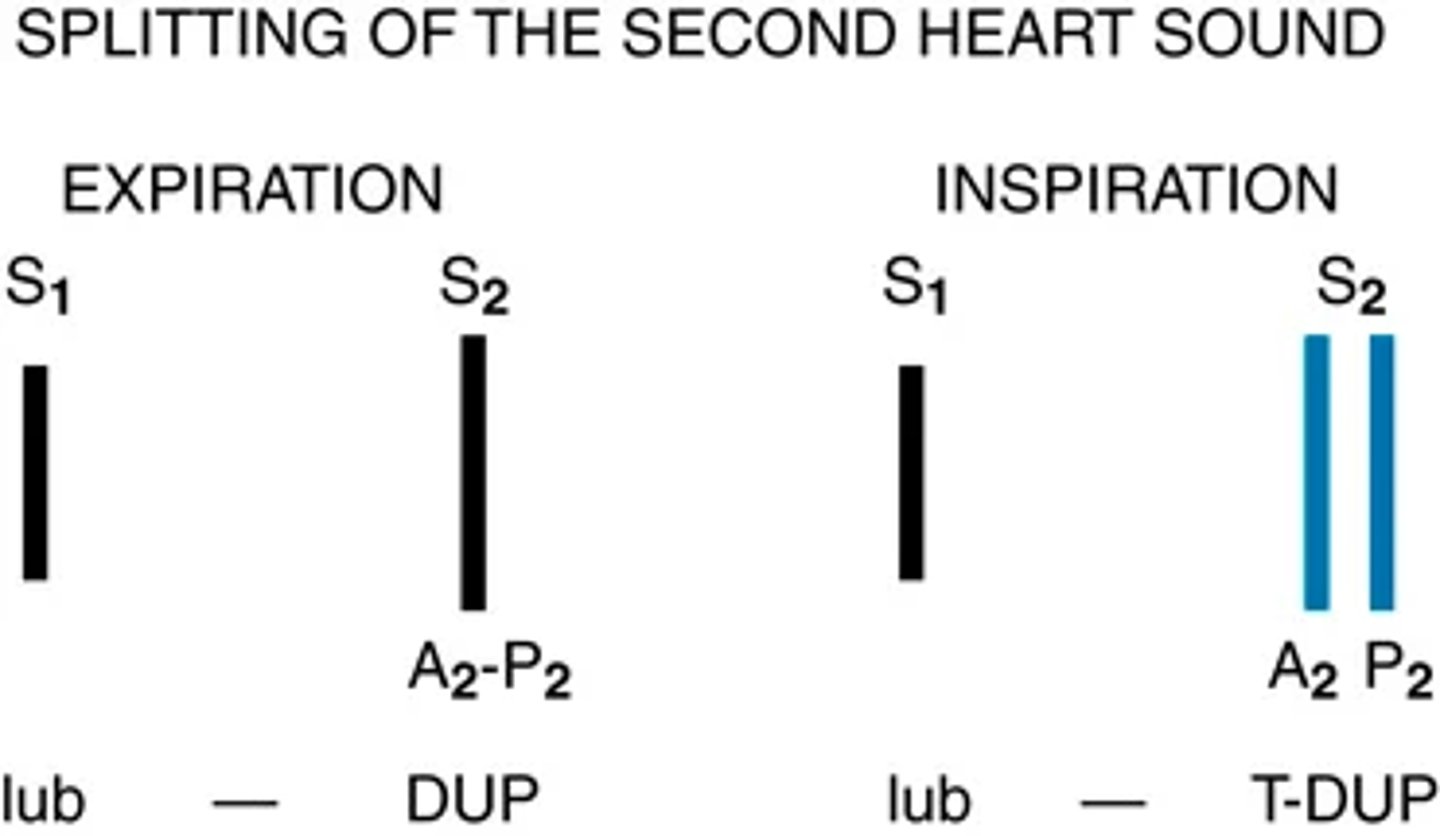
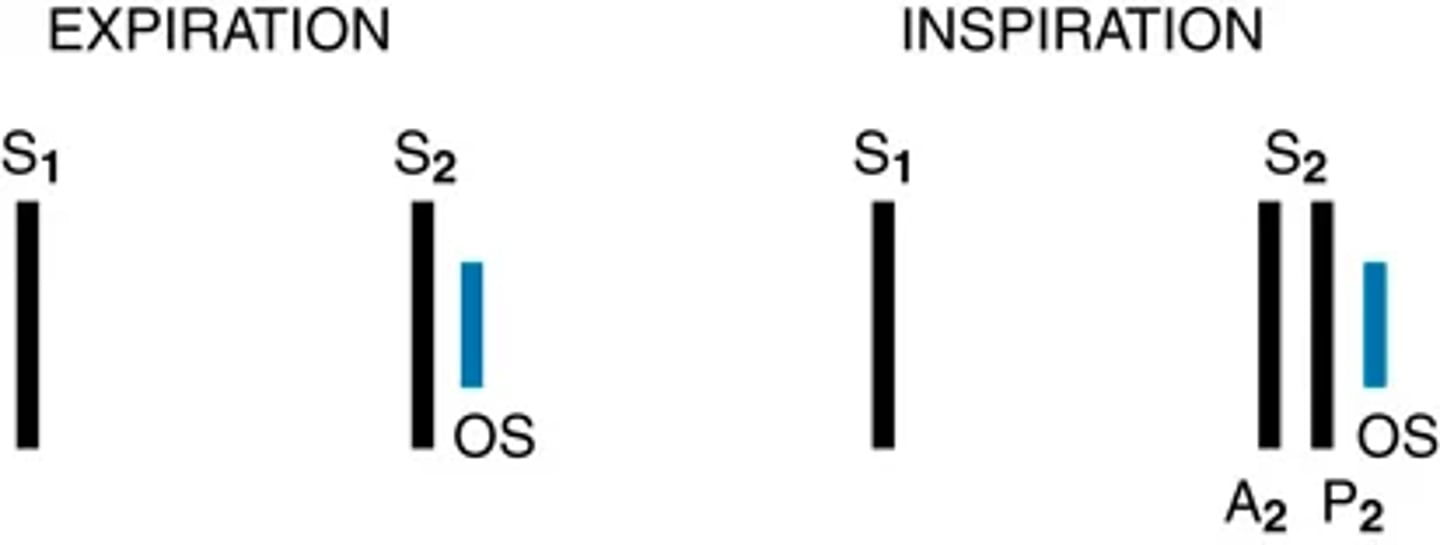
Splitting of S2. A split S2 is a normal phenomenon that occurs toward the end of inspiration in some people.
(closure of the aortic and pulmonic valves is nearly synchronous.)
Because of the effects of respiration on the heart described earlier, inspiration separates the timing of the two valves' closure, and the aortic valve closes 0.06 second before the pulmonic valve.
Instead of hearing DUP we hear a split sound T-DUP
During expiration, synchrony returns and the aortic and pulmonic components fuse together
A split S2 is heard only in the pulmonic valve area, the second left interspace.
Objective data pt 8) Auscultation pt 9 more on S2 split
When you first hear the split S2, do not be tempted to ask the person to hold their breath so you can concentrate on the sounds.
(equalizes pressure and causes split to go away)
Instead, concentrate on the split as you watch the person's chest rise up and down with breathing
(split usually occurs about every 4th heartbeat; fading with inhalation and fading with exhale)
Fixed split -> is unaffected by respiration; the split is always there.
Paradoxical split -> the opposite of what you would expect; the sounds fuse on inspiration and split on expiration
Objective data pt 8) Auscultation pt 10 Focus on systole and then diastole
Focus on systole and then on diastole (listen for extra heart sounds)
Listen with the diaphragm; then switch to the bell, covering all auscultatory area
(usually silent periods with no heart sound)
-During systole the midsystolic click (which is associated with mitral valve prolapse) is the most common extra sound
-The third and fourth heart sounds occur in diastole; either may be normal or abnormal
ABNORMAL -> A pathologic S3 (ventricular gallop) occurs with heart failure and volume overload; a pathologic S4 (atrial gallop) occurs with CAD
Objective data pt 8) Auscultation pt 11 (listen for murmurs)
Listen for Murmurs. A murmur is a blowing, swooshing sound that occurs with turbulent blood flow in the heart or great vessels.
Except for the innocent murmurs described, murmurs are abnormal.
(if you hear a murmur indicate the characteristics on the next few cards)
(ABNORMAL -> Murmurs may be caused by congenital and acquired valvular defects.)
Objective data pt 8) Auscultation pt 12 Characteristics of murmurs (loudness)
Loudness-> Describe the intensity in terms of 6 "grades." For example, record a grade 2 murmur as "2/6."
Grade 1—Barely audible; heard only in a quiet room and then with difficulty
Grade 2—Clearly audible but faint
Grade 3—Moderately loud; easy to hear
Grade 4—Loud; associated with a thrill palpable on the chest wall
Grade 5—Very loud; heard with one corner of the stethoscope lifted off the chest wall; associated thrill
Grade 6—Loudest; still heard with entire stethoscope lifted just off the chest wall; associated thrill
Objective data pt 8) Auscultation pt 12 Characteristics of murmurs (Pitch, pattern quality, location, radiation)
Pitch -> Describe the pitch as high, medium, or low. The pitch depends on the pressure and rate of blood flow producing the murmur.
pattern -> The intensity may follow a pattern during the cardiac phase, growing louder (crescendo), tapering off (decrescendo) or increasing to a peak, and then decreasing (crescendo-decrescendo or diamond shaped).
quality -> Describe the quality as musical, blowing, harsh, or rumbling.
(murmur of mitral stenosis is low-pitched and rumbling, whereas that of aortic stenosis is harsh)
Location -> Describe the area of maximum intensity (where it is best heard) ( note the valve area or intercostal spaces)
(The murmur of aortic stenosis may radiate to the neck)
Radiation -> The murmur may be transmitted downstream in the direction of blood flow; may be heard in another place on the precordium, the neck, the back, or the axilla.
Objective data pt 8) Auscultation pt 12 Characteristics of murmurs (posture)
Posture-> Some murmurs disappear or are enhanced by a change in position.
Some murmurs are common in healthy adolescents and children (innocent or functional)
- Innocent indicates having no valvular or other pathologic cause
-functional is caused by increased blood flow in the heart (e.g., in anemia, fever, pregnancy, hyperthyroidism).
contractile force of the heart is greater in children.
increased blood flow velocity -> increased velocity+ smaller chest measurement -> audible murmur.
Objective data pt 8) Auscultation pt 13 - the innocent murmurs
Sound -> generally soft (grade 2), midsystolic, short, crescendo-decrescendo, and with a vibratory or musical quality ("vooot" sound like fiddle strings).
location -> 2nd or 3rd left intercostal space and disappears with sitting, and the young person has no associated signs of cardiac dysfunction.
distinguish innocent murmurs from pathologic ones (ECG/Echo gives more accurate diagnosis)
Objective data pt 8) Auscultation pt 14 characteristics of murmurs (changing position)
Change Position -> After auscultating in the supine position, roll the person toward the left side.
Use bell, listen at apex for diastolic filling sounds (s3 or s4)
(S3 and S4 + the murmurs of mitral stenosis may only be heard on the left side)
Ask person to sit up, lean forward slightly, and exhale; listen with diaphragm firmly @ base, right, and left sides
Check for soft high pitched, early diastolic murmur of aortic or pulmonic regurgitation
(soft diastolic murmur of aortic regurgitation may be heard only when the person is leaning forward in the sitting position.)
Objective data pt 8) Auscultation pt 14 characteristics of murmurs (changing position) -standing to squatting
Standing to Squatting -> A screening measure to detect hypertrophic cardiomyopathy in children, adolescents, and young adults is to change position, which changes the venous return to the heart.
Systolic murmur? -> listen to the murmur while the person changes from standing to squatting, and then squatting to standing. (note sound changes)
The murmur of hypertrophic cardiomyopathy (HCM) grows softer with standing-to-squatting, and it grows louder with squatting-to-standing.
HCM = inherited thickening of the myocardium affecting 1 in 500 people
Objective data: Procedures for advanced practices -> Estimate the jugular venous pressure
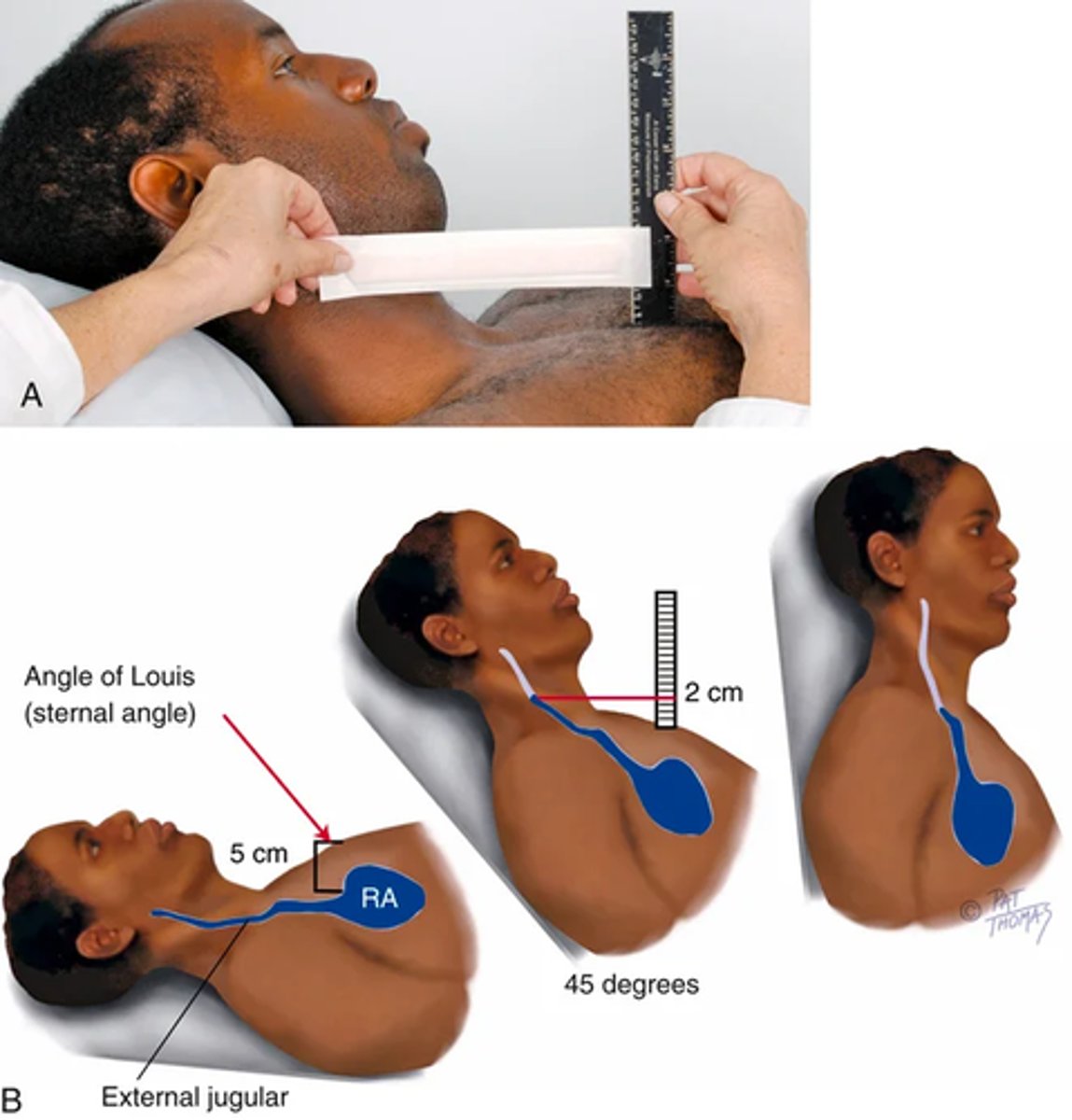
Estimate the jugular venous pressure;
- Think of the jugular veins as a CVP manometer attached directly to the right atrium.
-You can "read" the CVP at the highest level of pulsations.
-Use the angle of Louis (sternal angle) as an arbitrary reference point, and compare it with the highest level of the distended vein or venous pulsation
Hold ruler on sternal angle -> align a straight edge on the rule like a T-square and adjust the level of the horizontal straight edge to the level of pulsation
(read the level of intersect on the vertical ruler; more accurate during expiration; a normal jugular venous pulsation is less than 3 cm above the sternal angle)
state the person's position (e.g., "internal jugular vein pulsations 2 cm above sternal angle when elevated 30 degrees").
try to estimate a CVP for the patient with ascites or edema; when elevated it suggest heart or lung disease; if normal the problem may be something like liver disease
Objective data: Procedures for advanced practices -> Estimate the jugular venous pressure pt 2
If you cannot find the internal jugular veins, use the external jugular veins and note the point where they look collapsed.
Objective data: Procedures for advanced practices -> abdominojugular test (hepatojugular reflex)
If venous pressure is elevated or you suspect heart failure (Abdominojugular test aka Hepatojugular reflex)
1) Position the person supine, have them breath quietly with mouth open
2) Hold your right hand over the midabdomen and watch the level of jugular pulsation as you gradually push in with your hand.
3) . Exert firm sustained pressure for 10 seconds. This displaces venous blood out of the splanchnic vessels and adds its volume to the venous system.
4) If the heart is able to pump this additional volume (i.e., if no elevated CVP is present), the jugular veins will rise for a few seconds and then recede back to the previous level.
5) If heart failure is present, the jugular veins will elevate more than 4 cm and stay elevated as long as you push (a positive test).
Objective data: the aging adult
gradual rise in SBP is common with aging;
DBP stays fairly constant with a resulting widening of pulse pressure
orthostatic hypotension -> sudden drop in BP when rising to sit or stand.
(take precaution with auscultation and palpating carotid -> vagus reflex slowing HR)
W/ aging -> increases in anteroposterior diameter (diff. to palpate apical pulse and hear the splitting of S2)
(can have S4 with older adults with no known cardiac dz, murmurs common, occur in over 50%)
premature ectopic beats common, does not necessarily indicate underlying dz
(when in doubt ECG)
-ECG records for only one isolated minute in time and may need to be supplemented by a test of 24-hour
health promotion and Pt teaching
Appropriate aspirin therapy -> 40-59 yrs old, higher risk for ASCVD, might be best to take daily low dose aspirin (newer treatments go against HA and stroke prevention)
BP control -> BP check every 6 months to make sure it isn't rising, make life style changes
Cholesterol control -> screen every year for men beginning at age 35 years and for women at age 45 years. An elevation in one type of cholesterol can damage the blood vessels that feed your heart. (LDL)
Smoking cessation -> willing to quit. This is probably the biggest thing you can do to protect your heart. I will give you advice on how to quit and make certain you are connected with our nurse telephone quit lines before you leave.
Small life style changes -> 1) heart healthy diet, 2) physical activity (150 min per week), 3) keep weight in a healthy rage
Abnormal findings (Chest pain diagnoses): Cardiovascular (ischemic )
Angina pectoris: stable (no change in pain pattern within last 60 days)
description: Pressurelike pain (e.g., tightness, squeezing, burning, heaviness that lasts 3-5 minutes precipitated by activity
(resolves with rest / nitroglycerin)
Location: Generalized substernal or retrosternal: can radiate to teeth, jaw, neck, one or both arms or shoulders; or there may be no pain and only associated symptoms
Symptoms: Diaphoresis, nausea, vomiting, dyspnea, fatigue
Prinzmetal or variant angina
Description: Pressurelike discomfort often occurring at rest, unrelated to physical or emotional stress
Location: Generalized substernal or
retrosternal: can radiate to teeth, jaw, neck, one or both arms or shoulders; or there may be no pain and only associated symptoms
Possible symptoms: Palpitations, syncope, or feelings of syncope
Abnormal findings (Chest pain diagnoses): Cardiovascular (ischemic ) pt 2
Acute coronary syndrome (ACS) (unstable angina, myocardial infarction)
Description: Heaviness; viselike, squeezing, crushing, tightness; vague, burning, constricting, or pressure; poorly localized pain lasting 20-30 minutes to hours and does not resolve with rest or nitroglycerin
location: Generalized substernal or retrosternal: can radiate to teeth, jaw, neck, one or both arms or shoulders; or there may be no pain and only associated symptoms
symtpoms: Indigestion-like feeling, nausea, vomiting, dizziness, flushing, perspiration, palpitations, dyspnea, fatigue
Abnormal findings(Chest pain diagnoses): Cardiovascular (Non-ischemic)
Pericarditis
Description: Sudden sharp and stabbing pain relieved often by sitting or leaning forward and worsens by lying down or with inspiration
Location: Substernal, which can radiate to trapezius muscle region
Symptoms: Dry cough, muscle and joint aches, fever
Mitral valve prolapse
Description: Sharp pain not associated with activity
Location: Chest pain without radiation
Symptoms: Fatigue, light-headedness, dyspnea, irregular heartbeat, palpitations, exercise intolerance
Aortic dissection
Description: Sudden severe pain with change in location and/or tearing sensation lasting for hours
Location: Anterior chest pain with radiation to the neck, jaw, or intrascapular region of the back
symptoms: Mental status changes, limb pain and weakness, dyspnea
Pulmonary hypertension (secondary)
Description: Cardiac-like chest pain with exertion
location: Chest region
Dyspnea, lower-extremity edema, fatigue
Abnormal findings (Chest pain diagnoses): Pulmonary
Pulmonary embolism
Description: Sharp, stabbing pain worsening with deep breaths
Location: Pain can be experienced in chest, back, shoulder, or upper abdomen
Symptoms: Dyspnea, hemoptysis, cough
pneumonia
Description: Sharp or stabbing pain associated with cough; may be worse with deep breathing
Location: Mostly generalized to one side of chest but can have upper abdominal pain
symptoms: Cough, fever, dyspnea, chills, sputum, myalgia, malaise
Pneumothorax
Description: Acute/sudden and sharp
Location: Lateral region of the chest but can have referred pain to shoulder
Symptoms: Acute dyspnea, cough
Abnormal findings (Chest pain diagnoses): gastrointestinal
Gastroesophageal reflux
Description: May be angina-like; however, usually burning sensation with eating large meals reproduced by lying down and relieved by sitting up
Location: Retrosternal region
symptoms: Cough, regurgitation of food, abdominal pain, sour/bitter taste
Esophageal spasm
description: Crushing chest pain
Location: substernal
Symptoms: Dysphagia, sensation of object in throat or esophagus
Cholecystitis:
Descriptions: Sudden onset of pain that crescendos and can last for up to 20 minutes, usually after eating a fatty meal
Location: Epigastrium or right upper abdomen that can radiate to right intrascapular region, shoulder, or back
Symptoms: Nausea, vomiting, anorexia, fever
Pancreatitis
Description: Sudden dull, boring, steady pain unrelieved by lying supine; leaning forward or the fetal position may ease pain
Location: Epigastrium or periumbilical pain radiating to back
Symptoms: Nausea, vomiting, anorexia, and sometimes diarrhea
Abnormal findings (Chest pain diagnoses): Dermatologic, musculoskeletal/neurologic
Herpes zoster
description: Unilateral, burning, bore like pain Location: Chest region in dermatome distribution
Associated symptoms: Tingling, itching, burning
Costochondritis:
Description: Sharp, pleuritic-type pain worsens with deep breathing, palpation, or movement
Location: Area from 2nd through 5th intercostal spaces; can radiate to arm, depending on where initial inflammation occurs
Associated symptoms: Chest tightness, warmth at area of pain
Chest wall muscle strain
Description: Sharp pain with moving, stretching, or pushing movements of the arms; palpation of area reproduces the pain
Location: Area around the strained muscle, sternum, or ribs
Associated symptoms: Muscle spasm, crepitation, swelling, loss of strength
Abnormal findings (Chest pain diagnoses) : Psychogenic
Depression
description: Heaviness
Location: Chest region
associated: Fatigue, restlessness, withdrawal, weight gain or loss, depressed mood
Anxiety
Description: Sharp pain
location: Chest region
Associated: Palpitations, dizziness, sweating, shaking, restlessness, fatigue, irritability, feeling of fear or panic
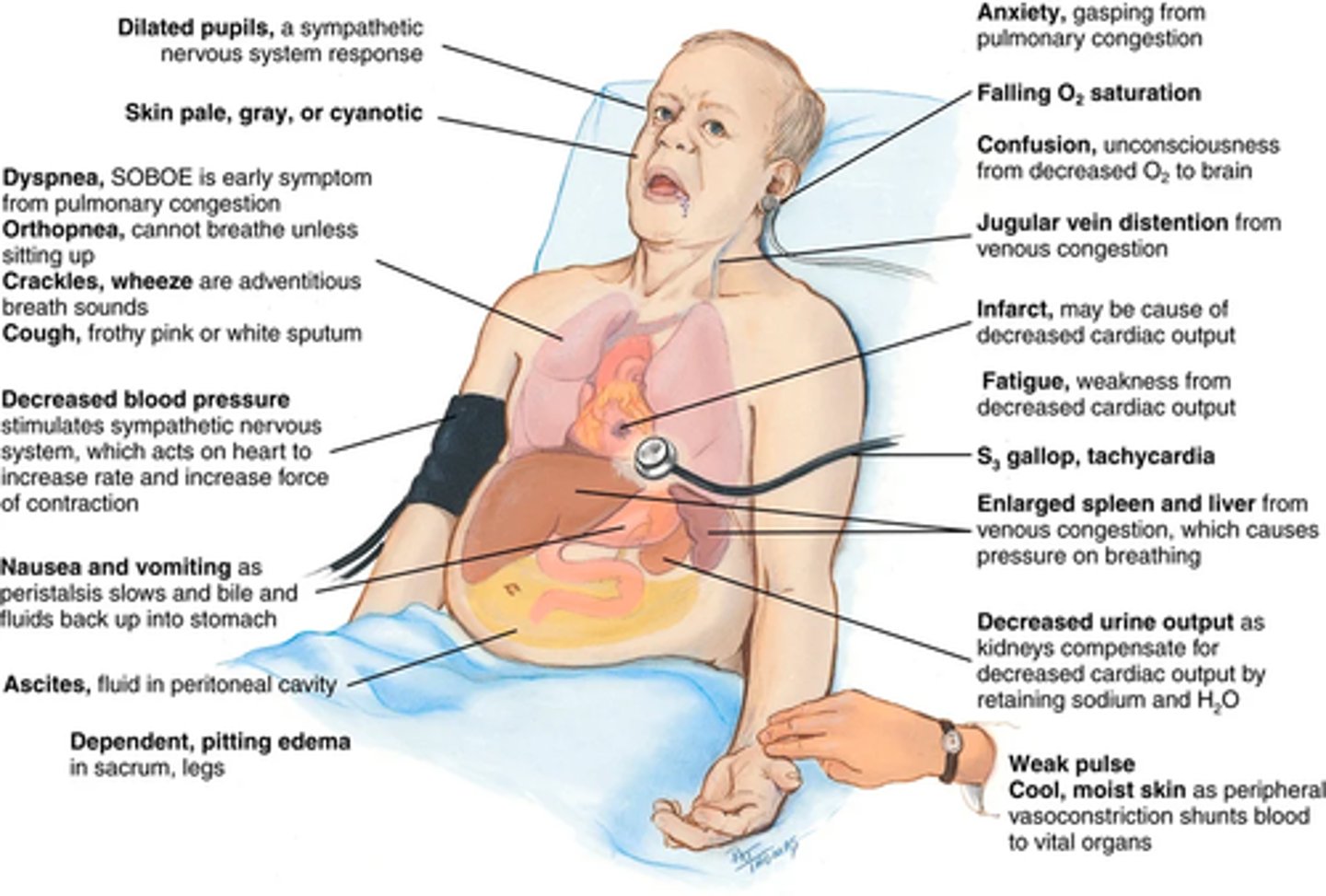
Clinical portrait of heart failure:
Decreased cardiac output -> occurs when the heart fails as a pump and the circulation becomes backed up and congested.
Signs and symptoms come from:
(1) the heart's inability to pump enough blood to meet the metabolic demands of the body; (2) the kidney's compensatory mechanisms of abnormal retention of sodium and water to compensate for the decreased cardiac output.
(increased blood volume and venous return -> more congestion)
Onset: (1) acute, as following a myocardial infarction when the heart's contracting ability has been directly damaged
(2) chronic, as with hypertension, when the ventricles must pump against chronically increased pressure
Heart failure may involve systolic dysfunction -> heart cannot contract properly, resulting in a low ejection fraction (the stroke volume divided by the end-diastolic volume, normally 50% to 70%). AKA Heart failure with reduced ejection fraction
Diastolic dysfunction: failure of the heart to relax fully between heartbeats; here the heart muscle wall is stiff and does not fill properly; there is low cardiac output but a normal ejection fraction (also known as heart failure with preserved ejection fraction, or HFpEF).
(EF normal at rest, but may not raise properly with the stress of exercise)
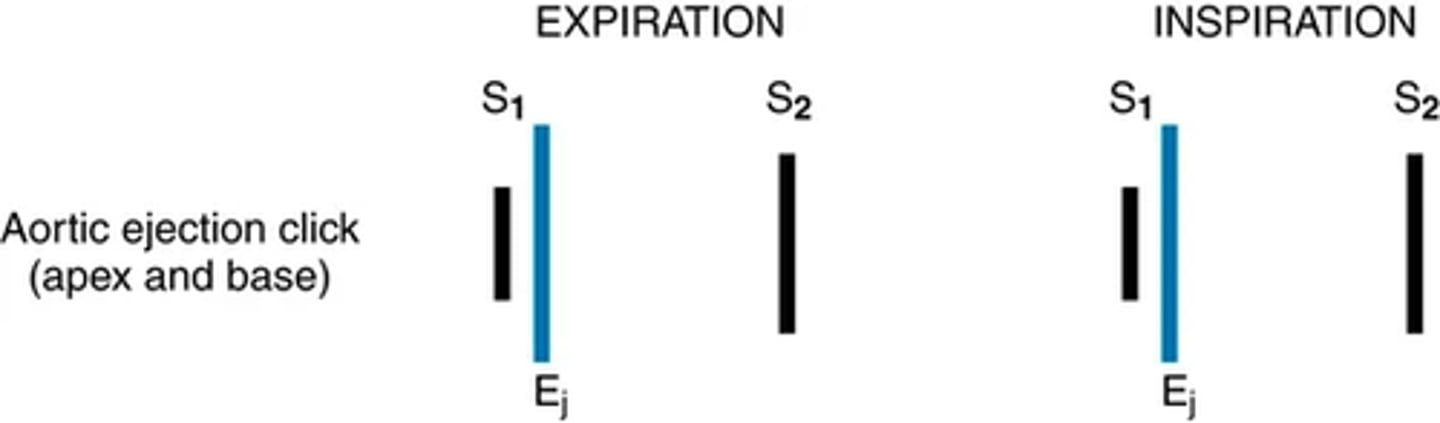
Systolic extra sounds:
Early systolic: ejection click (Aortic prosthetic valve sounds)
Mid/late systolic: Mid systolic (mitral click)
Aortic ejection click (apex and base): expiration and inspiration image
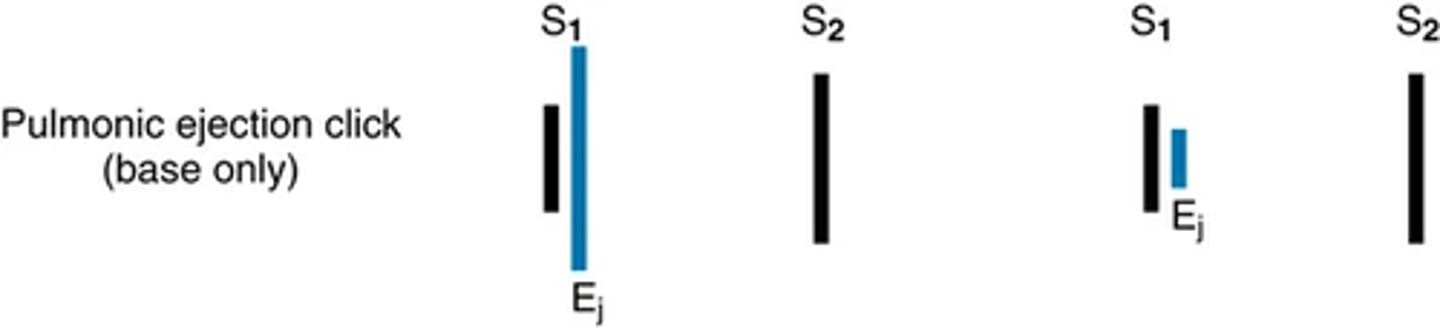
Systolic extra sounds: Pulmonic ejection clock (base only)
systolic extra sounds: Ejection click
-occurs early in systole at the start of ejection, results from the opening of the semilunar valves
(usually opens silently, but in the presence of stenosis their opening makes a short and high pitched sound with a click quality; better with diaphragm)
The aortic ejection click is heard at the 2nd right interspace and apex and may be loudest at the apex.
(intensity unchanged with respiration)
The pulmonic ejection click is best heard in the 2nd left interspace ( often grows softer with inspiration.)
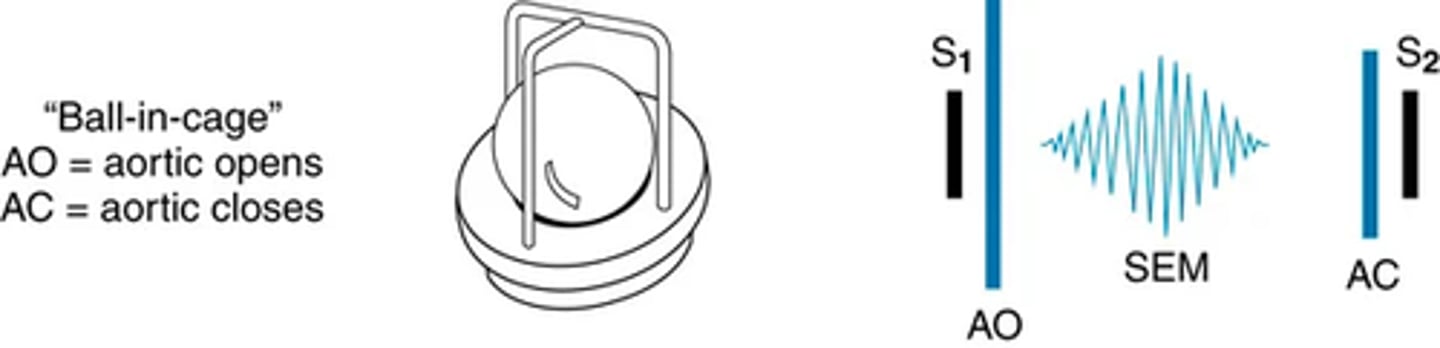
Systolic extra sounds: Aortic prosthetic valve sound
As a sequela of modern technologic intervention for heart problems, some people now have iatrogenically induced heart sounds.
The opening of a mechanical aortic ball-in-cage prosthesis produces an early systolic sound. This sound is less intense with a tilting disk prosthesis and is absent with a bioprosthetic valve
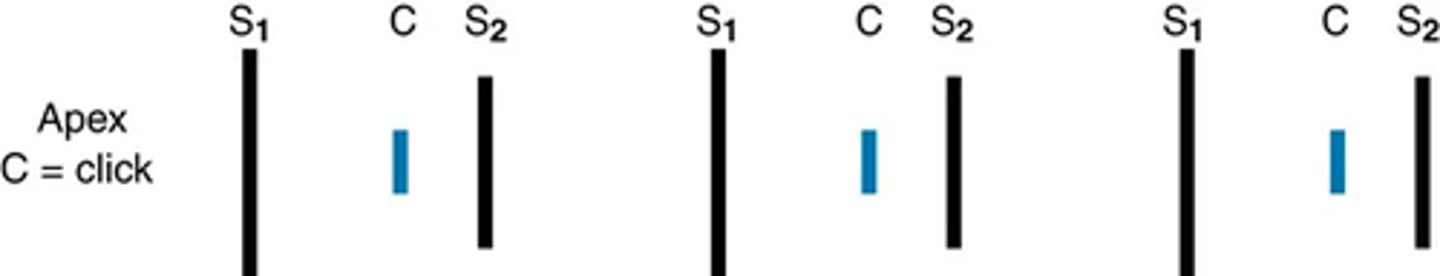
systolic extra sounds: midsystolic click
(Although it is systolic, this is not an ejection click) -> associated with mitral valve prolapse, in which the mitral valve leaflets not only close with contraction but balloon back up into the left atrium
During ballooning, sudden tensing of the valve leaflets and chordae tendineae create click
sound occurs in mid- to late systole and is short and high pitched with a click quality. It is best heard with the diaphragm, at the apex, but also may be heard at the left lower sternal border.
The click usually is followed by a systolic murmur
Click and murmur change with posture; when squatting the click may move closer to S2 and murmur may sound louder and delayed
(Valsalva maneuver does the same)
Diastolic extra sounds:
Early diastole:Opening snap
Mitral prosthetic valve sound
Mid-diastole:Third heart sound
Summation sound (S3 + S4)
Late diastole:Fourth heart sound
Pacemaker-induced sound
Diastolic extra sounds: Opening snap
Opening of Av valve usually silent, but with stenosis -> increasingly higher atrial pressure is required to open the valve. The deformed valve opens with a noise: the opening snap.
(sharp and high pitched with snapping quality)
It sounds after S2 and is best heard with the diaphragm at the 3rd or 4th left interspace at the sternal border, less well at the apex.
The opening snap usually is not an isolated sound. As a sign of mitral stenosis, the opening snap usually ushers in the low-pitched diastolic rumbling murmur of that condition.
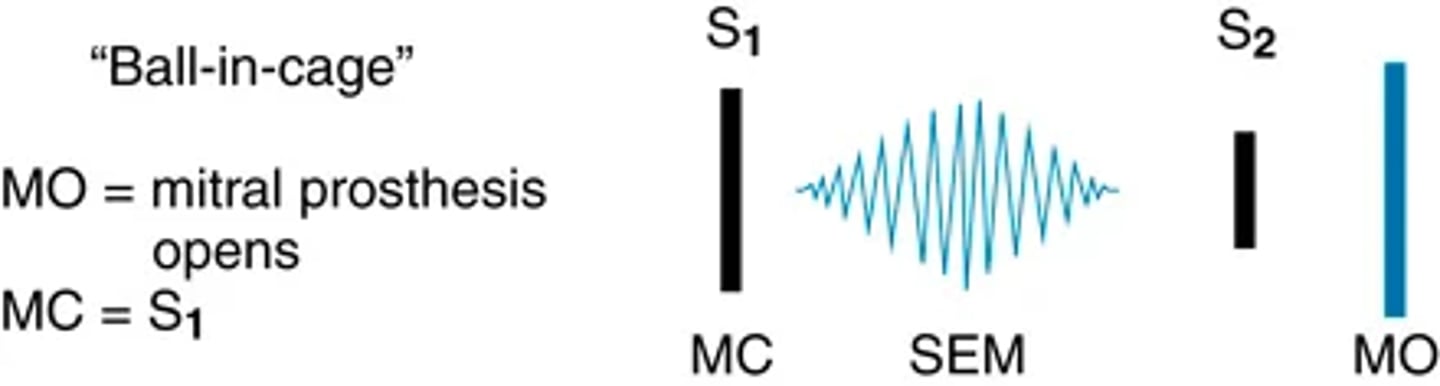
Diastolic extra sounds: Mitral prosthetic valve sound
An iatrogenic sound, the opening of a ball-in-cage mitral prosthesis gives an early diastolic sound: an opening click just after S2. It is loud, heard over the whole precordium, and loudest at the apex and left lower sternal border.
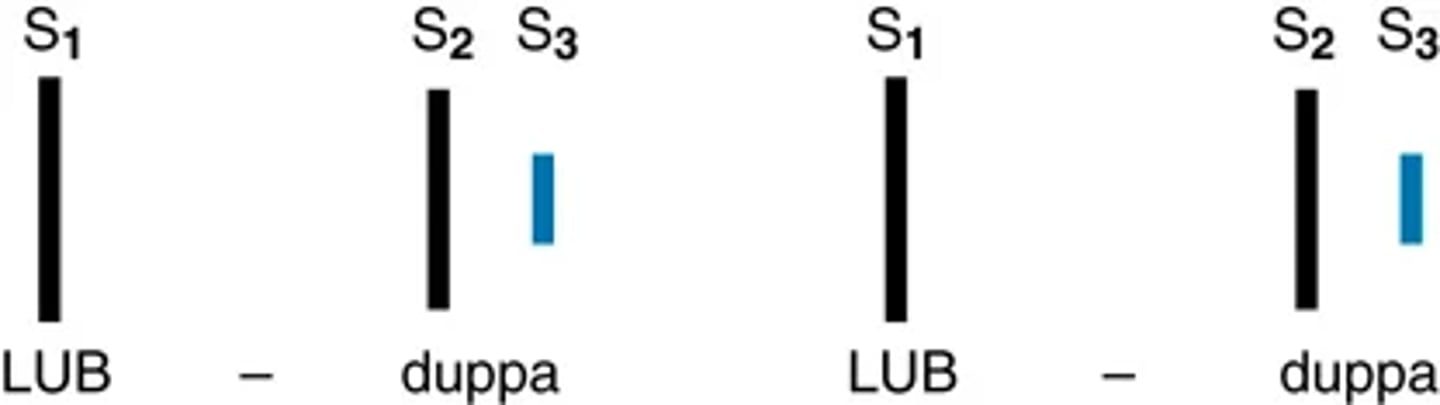
Diastolic extra heart sounds: Third heart sounds pt 1
The S3 is a ventricular filling sound. It occurs in early diastole during the rapid filling phase.
Your hearing quickly accommodates to the S3; thus it is best heard when you listen initially. It sounds after S2 but later than an opening snap would be
It is a dull, soft sound; and it is low pitched, like "distant thunder." It is heard best in a quiet room, at the apex, with the bell held lightly (just enough to form a seal), and with the person in the left lateral position.
Diastolic extra heart sounds: Third heart sounds pt 2
The S3 can be confused with a split S2. Use these guidelines to distinguish the S3:
• Location—The S3 is heard at the apex or left lower sternal border; the split S2 at the base.
• Respiratory variation—The S3 does not vary in timing with respirations; the split S2 does.
• Pitch—The S3 is lower pitched; the pitch of the split S2 stays the same.
The S3 may be normal (physiologic) or abnormal (pathologic). The physiologic S3 is heard frequently in children and young adults; it occasionally may persist after 40 years, especially in women. The normal S3 usually disappears when the person sits up.
Diastolic extra heart sounds: Third heart sounds pt 3 pathological and physiological
The S3 may be normal (physiologic) or abnormal (pathologic). The physiologic S3 is heard frequently in children and young adults; it occasionally may persist after 40 years, especially in women. The normal S3 usually disappears when the person sits up.
In adults the S3 is usually abnormal. The pathologic S3 is also called a ventricular gallop or an S3 gallop, and it persists when sitting up. The S3 indicates decreased compliance of the ventricles, as in heart failure. It may be the earliest sign of heart failure.
(Sound may originate from either the left or right ventricle; left-sided S3 is heard at the apex in the left lateral position, and a right-sided S3 is heard at the left lower sternal border with the person supine and is louder in inspiration.)
Diastolic extra heart sounds: Third heart sounds pt 4
The S3 also occurs with conditions of volume overload such as mitral regurgitation and aortic or tricuspid regurgitation. The S3 is also found in high cardiac output states in the absence of heart disease such as hyperthyroidism, anemia, and pregnancy. When the primary condition is corrected, the gallop disappears.
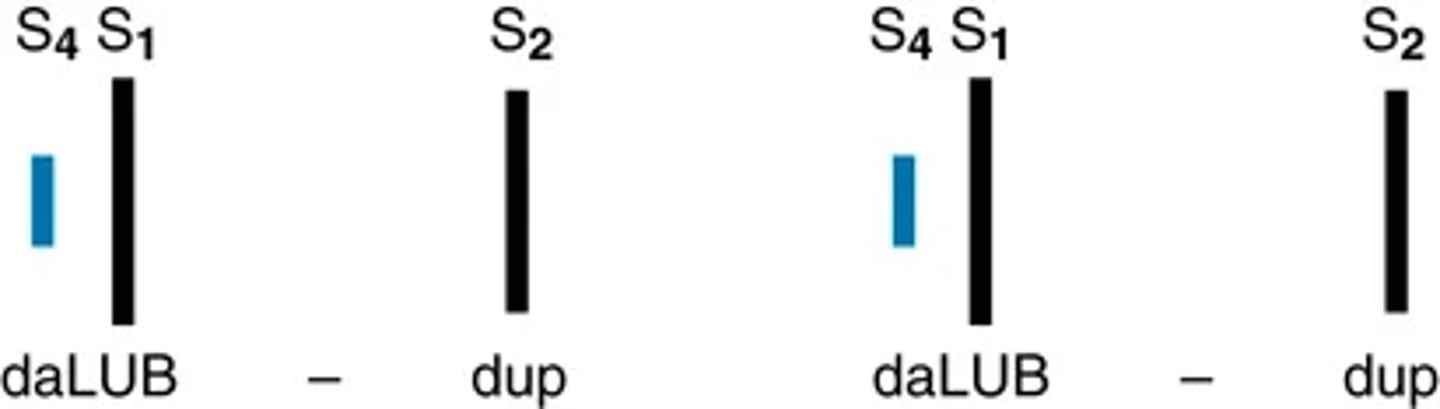
Diastolic extra heart sounds: 4th heart sound
The S4 is a ventricular filling sound. It occurs when the atria contract late in diastole. It is heard immediately before S1. This is a very soft sound of very low pitch.
(heard best with bell in left lateral position)
Physiological s4 -> may occur in adults older than 40 or 50 years with no evidence of cardiovascular disease, especially after exercise
Pathological s4 -> atrial gallop or an S4 gallop. It occurs with decreased compliance of the ventricle (e.g., coronary artery disease, cardiomyopathy) and systolic overload (afterload), including outflow obstruction to the ventricle (aortic stenosis) and systemic hypertension.
A left-sided S4 occurs with these conditions. It is heard best at the apex, in the left lateral position.
A right-sided S4 is less common. It is heard at the left lower sternal border and may increase with inspiration. It occurs with pulmonary stenosis or pulmonary hypertension.
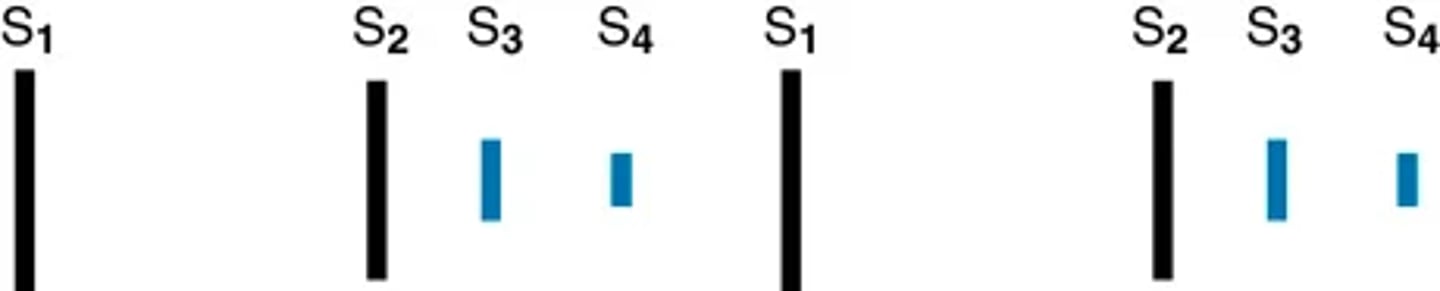
Diastolic extra heart sounds: summation sound and extra cardiac sounds
When both the pathologic S3 and S4 are present, a quadruple rhythm is heard.
(cardiac stress -> tachycardic response)
During rapid rates the diastolic filling time shortens, and the S3 and S4 move closer together.
(sound superimposed in mid-diastole, one loud, prolonged, summated sound, often louder than either S1 or S2.)
IMAGE IS OF EXTRA CARDIAC SOUNDS
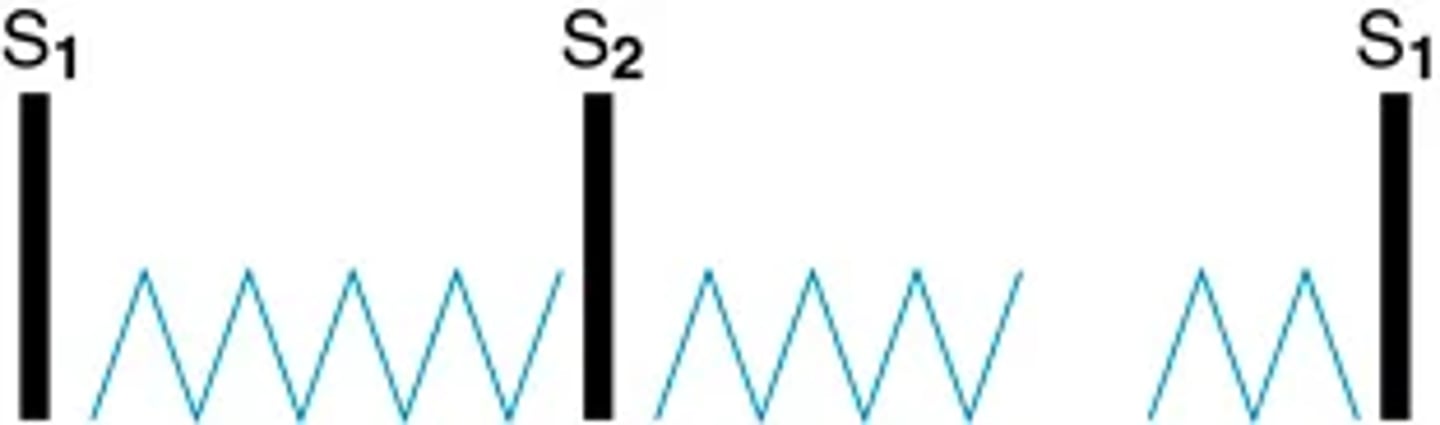
Diastolic extra heart sounds: Pericardial friction rub
Inflammation of the pericardium gives rise to a friction rub. The sound is high pitched and scratchy, like sandpaper being rubbed.
(person sitting up, lean forward, hold breath, and listen with diaphragm)
A friction rub can be heard any place on the precordium but usually is best heard at the apex and left lower sternal border, places where the pericardium comes in close contact with the chest wall.
(diastolic and systolic timing)
(common the 1st feet after a myocardial infraction and may only last a few hours)
Abnormal pulsations on the precordium: Base Thrill
A thrill in the 2nd and 3rd right interspaces occurs with severe aortic stenosis and systemic hypertension.
A thrill in the 2nd and 3rd left interspaces occurs with pulmonic stenosis and pulmonic hypertension.
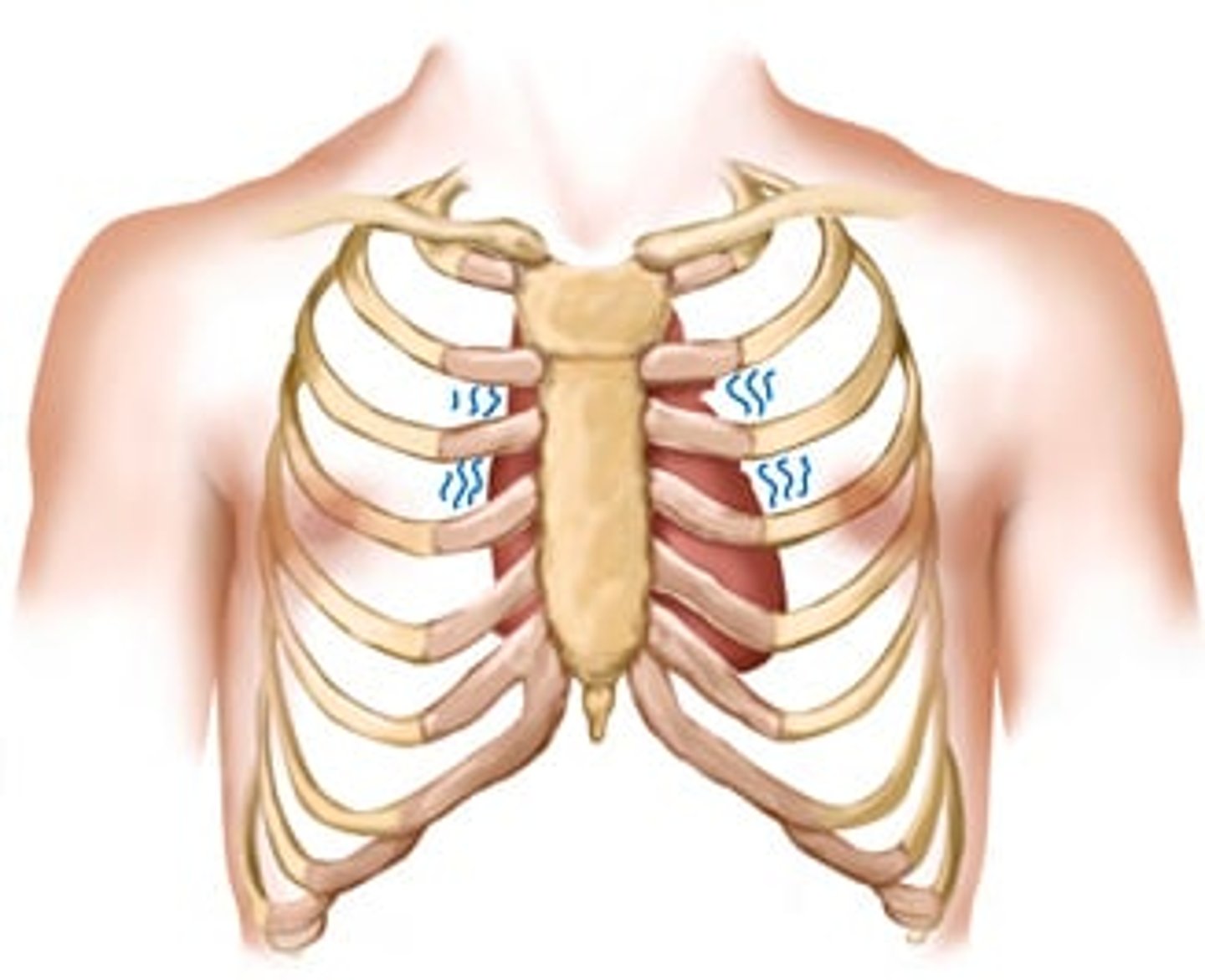
Abnormal pulsations on the precordium: Left sternal border lift (heave)
A lift (heave) occurs with right ventricular hypertrophy, as found in pulmonic valve disease, pulmonic hypertension, and chronic lung disease.
diffuse lifting impulse during systole at the left lower sternal border
may be associated with retraction at the apex because the left ventricle is rotated posteriorly by the enlarged right ventricle.